Using Sensory Evaluation to Determine the Highest Acceptable Concentration of Mango Seed Extract as Antibacterial and Antioxidant Agent in Fresh-Cut Mango
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Characterization
2.2. Preparation of Mango Seed Extracts
2.3. Identification and Quantification of Major Phenolic Compounds in Mango Seed Extract
2.4. Total Phenolic Content of Mango Seed Extract
2.5. Total Flavonoid Content of Mango Seed Extract
2.6. Antioxidant Activity of Mango Seed Extract by DPPH Radical Scavenging Assay
2.7. 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) Radical Savenging Activity of Mango Seed Extract
2.8. Antibacterial Activity
2.9. Sensory Evaluation of Fresh-Cut Mango Treated with Mango Seed Extract
2.10. Phenolic Content and Antioxidant Activity of Fresh-Cut Mango Treated with Mango Seed Extract
2.11. Bacterial Load Reduction in Fresh-Cut Mango Treated with Mango Seed Extract
2.12. Statistical Analysis
3. Results and Discussions
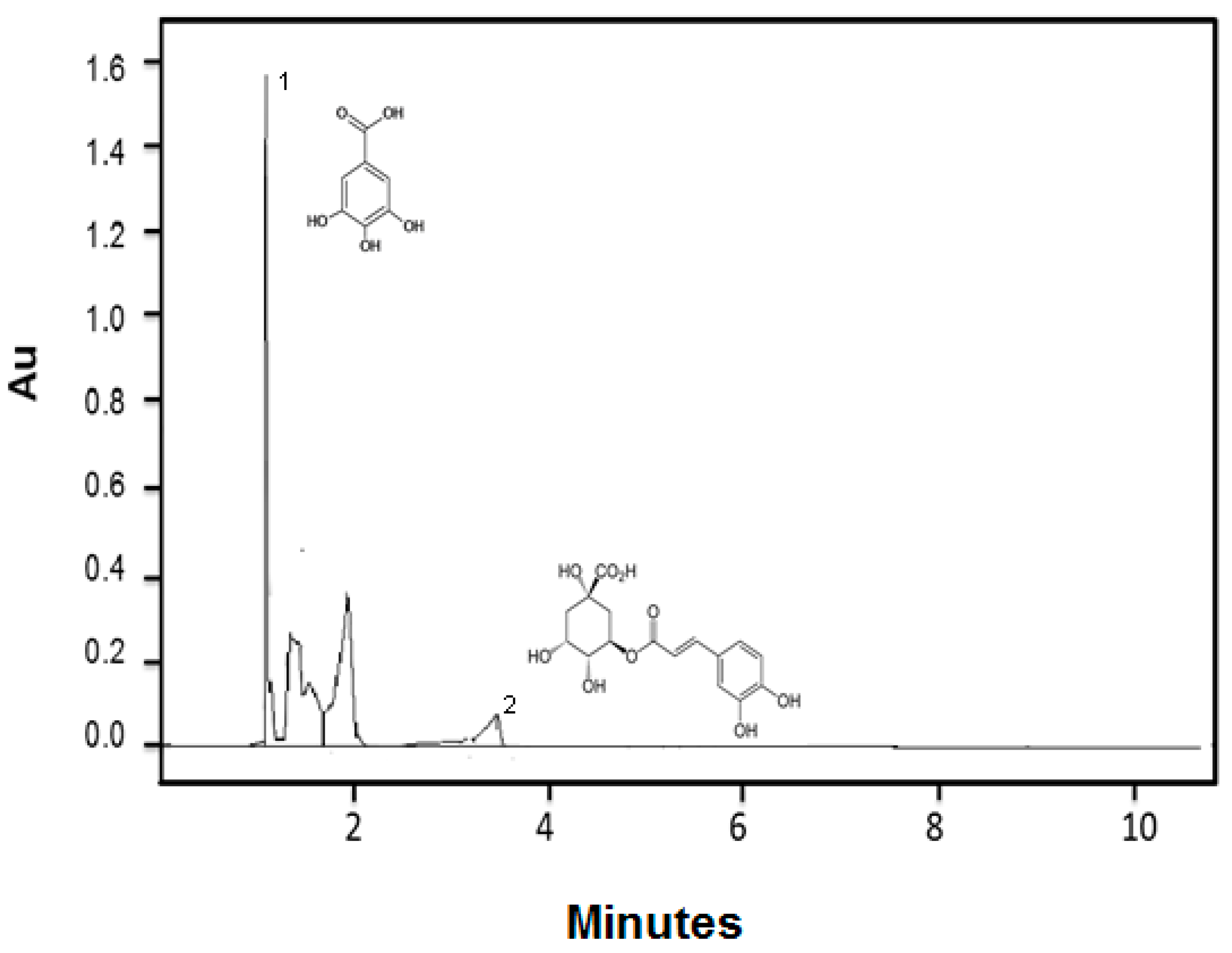
3.1. Phenolic Compounds, Antioxidant and Antimicrobial Activity of Mango Seed Extract
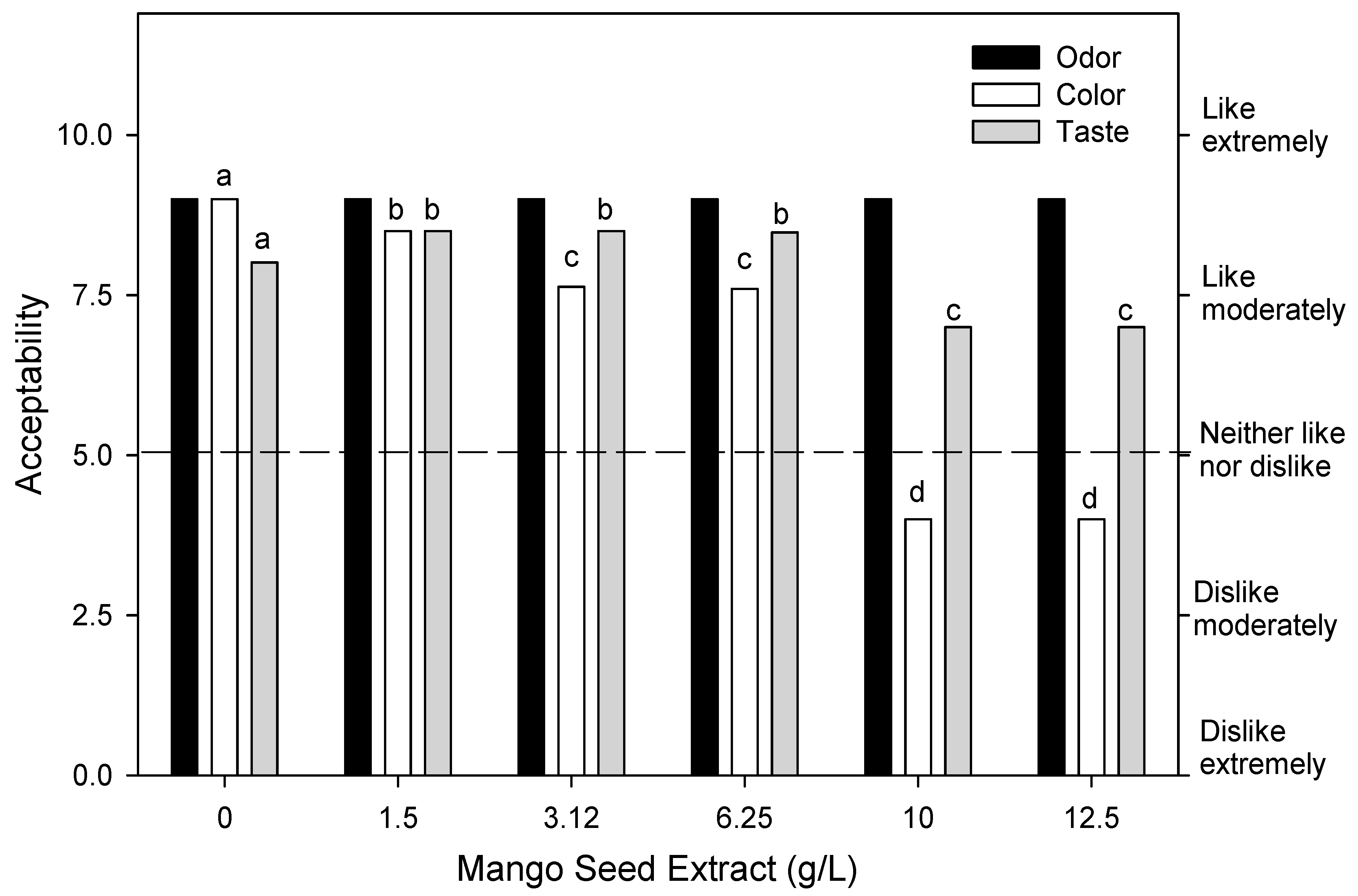
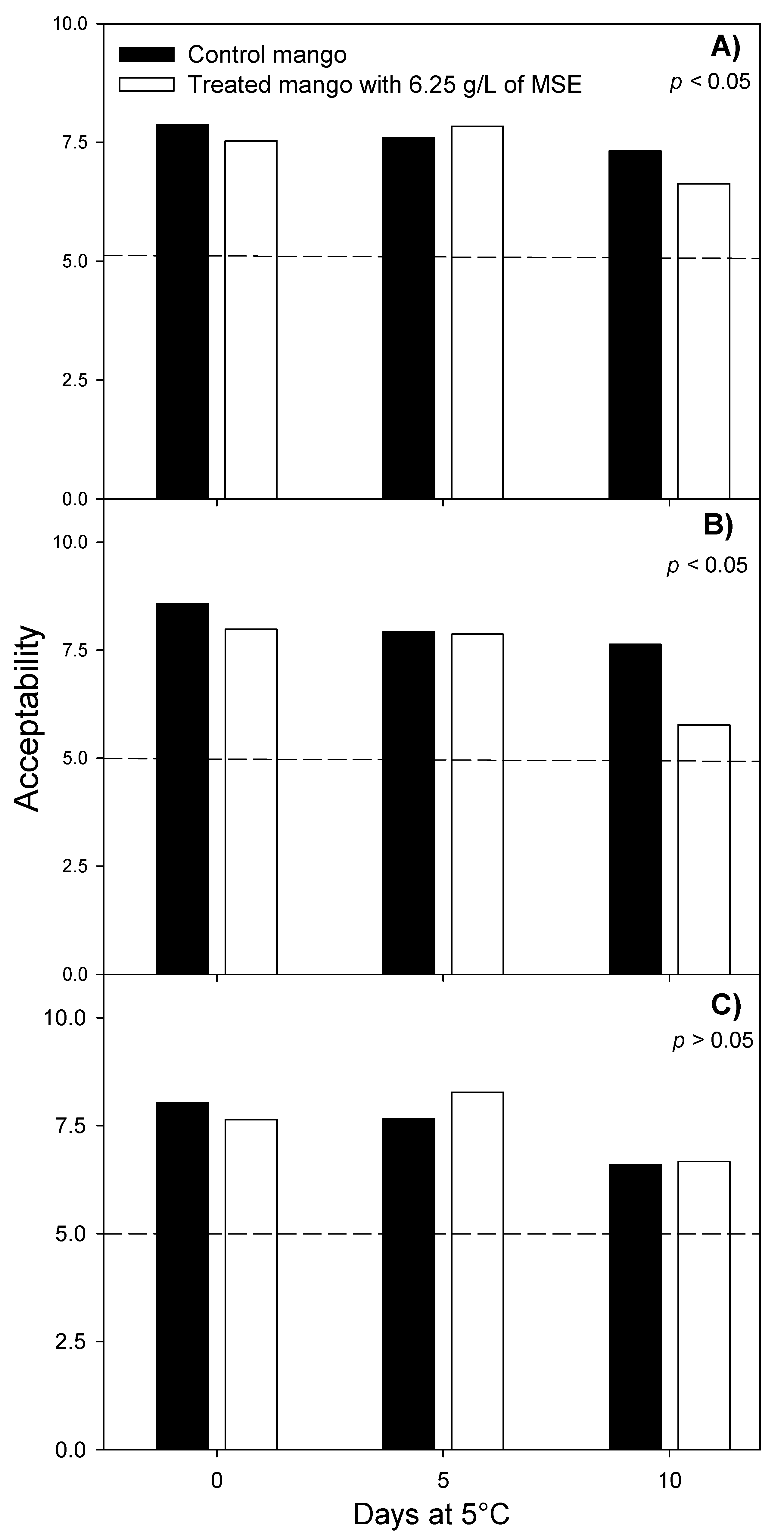
3.2. Sensory Evaluation of Fresh-Cut Mango Treated with Mango Seed Extract
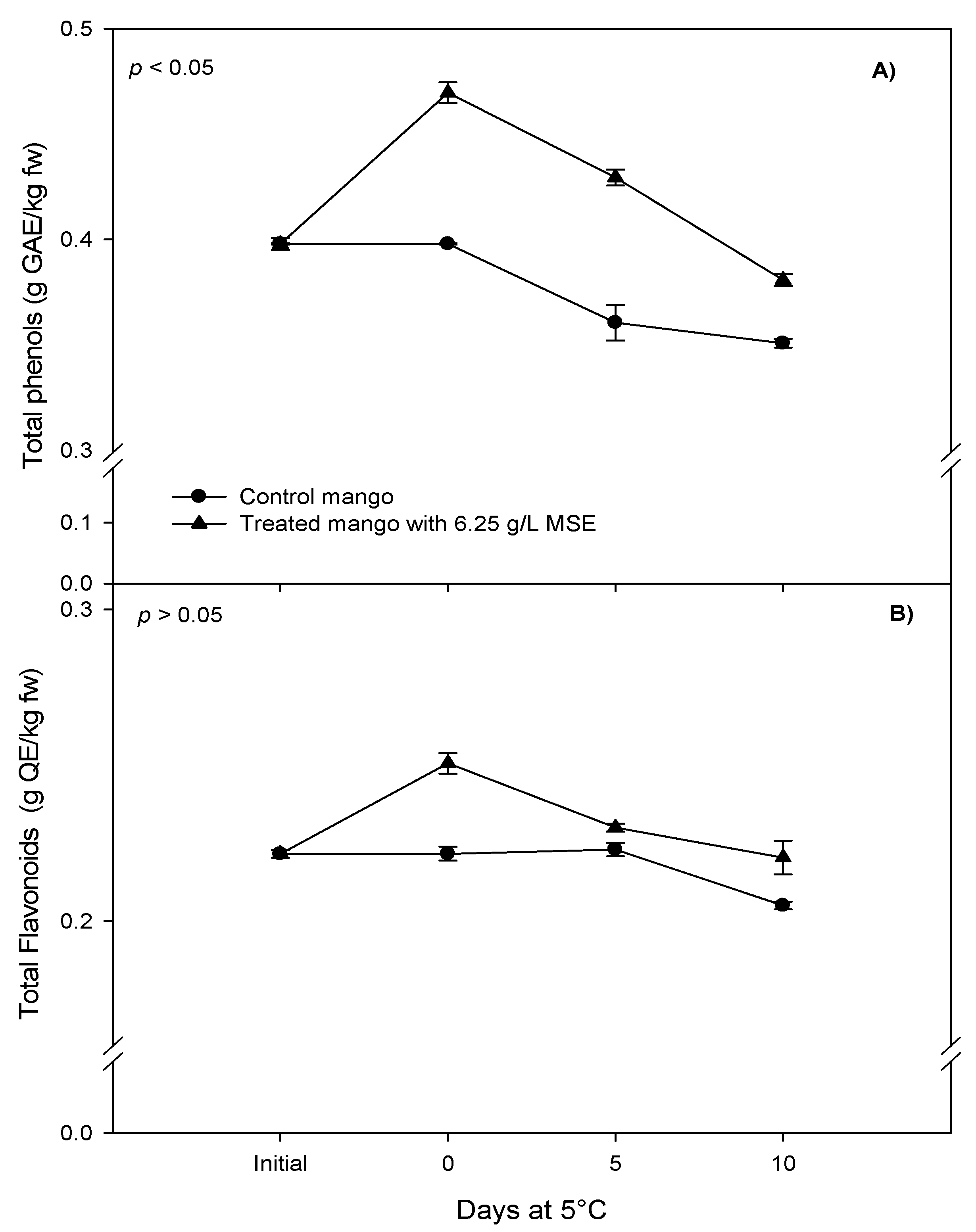
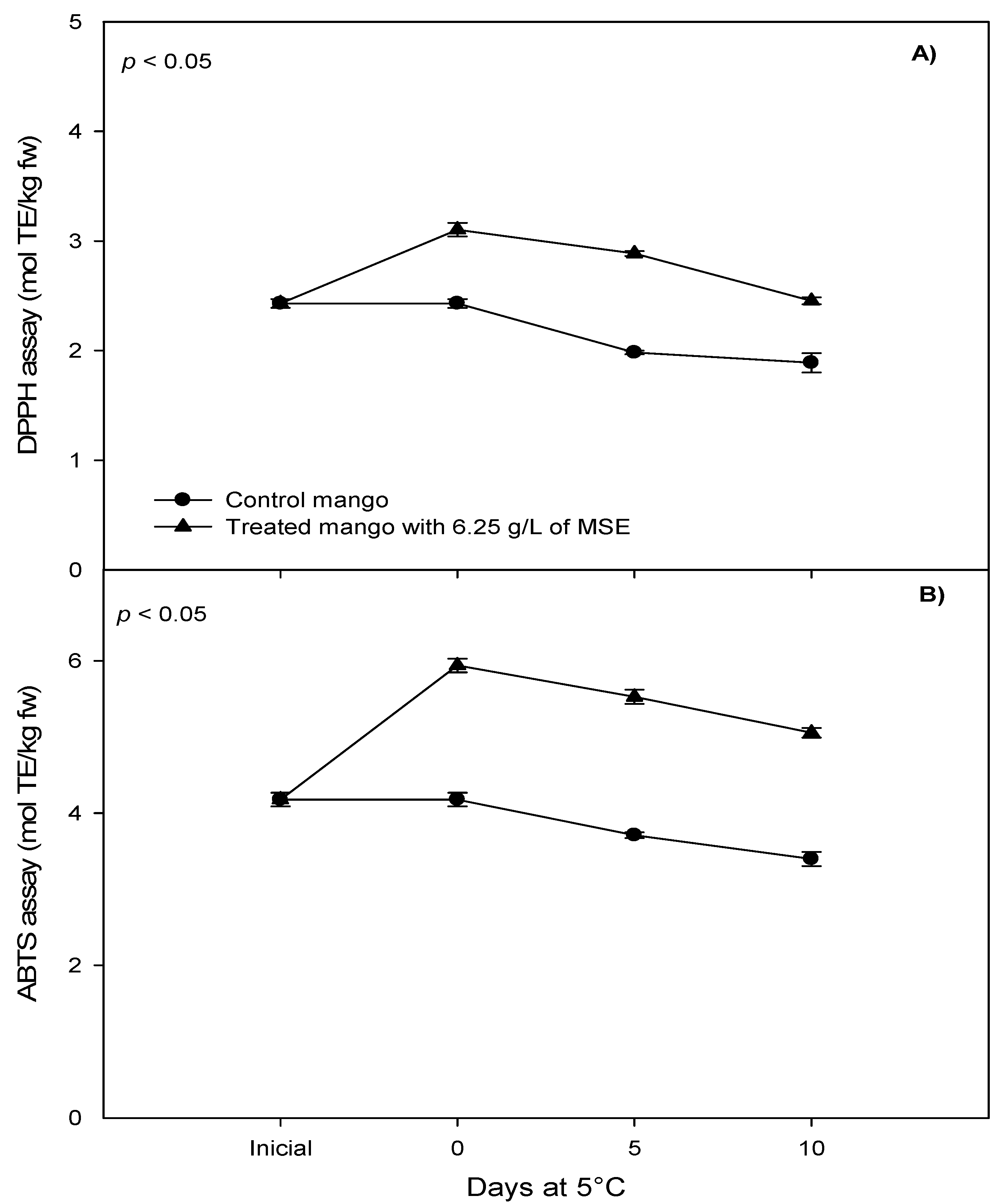
3.3. Phenolic Compounds and Antioxidant Capacity of Fresh-Cut Mango Treated with Mango Seed Extract
3.4. Bacterial Load of Fresh-Cut Mango Treated with Mango Seed Extract
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ayala-Zavala, J.; Vega-Vega, V.; Rosas-Domínguez, C.; Palafox-Carlos, H.; Villa-Rodriguez, J.; Siddiqui, M.W.; Dávila-Aviña, J.; González-Aguilar, G. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Okino Delgado, C.H.; Fleuri, L.F. Orange and mango by-products: Agro-industrial waste as source of bioactive compounds and botanical versus commercial description—A review. Food Rev. Int. 2016, 32, 1–14. [Google Scholar] [CrossRef]
- Dorta, E.; Lobo, M.G.; Gonzalez, M. Reutilization of mango byproducts: Study of the effect of extraction solvent and temperature on their antioxidant properties. J. Food Sci. 2012, 77, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Vega-Vega, V.; Silva-Espinoza, B.A.; Cruz-Valenzuela, M.R.; Bernal-Mercado, A.T.; Gonzalez-Aguilar, G.A.; Ruíz-Cruz, S.; Moctezuma, E.; Siddiqui, M.W.; Ayala-Zavala, J.F. Antimicrobial and antioxidant properties of byproduct extracts of mango fruit. J. Appl. Bot. Food Qual. 2013, 86, 205–211. [Google Scholar]
- Dorta, E.; González, M.; Lobo, M.G.; Sánchez-Moreno, C.; de Ancos, B. Screening of phenolic compounds in by-product extracts from mangoes (Mangifera indica L.) by HPLC-ESI-QTOF-MS and multivariate analysis for use as a food ingredient. Food Res. Int. 2014, 57, 51–60. [Google Scholar] [CrossRef]
- Ashoush, I.; Gadallah, M. Utilization of mango peels and seed kernels powders as sources of phytochemicals in biscuit. World J. Dairy Food Sci. 2011, 6, 35–42. [Google Scholar]
- Abdalla, A.E.; Darwish, S.M.; Ayad, E.H.; El-Hamahmy, R.M. Egyptian mango by-product 2: Antioxidant and antimicrobial activities of extract and oil from mango seed kernel. Food Chem. 2007, 103, 1141–1152. [Google Scholar] [CrossRef]
- Vega-Vega, V.; Silva-Espinoza, B.A.; Cruz-Valenzuela, M.R.; Bernal-Mercado, A.T.; González-Aguilar, G.A.; Vargas-Arispuro, I.; Corrales-Maldonado, C.G.; Ayala-Zavala, J.F. Antioxidant enrichment and antimicrobial protection of fresh-cut mango applying bioactive extracts from their seed by-products. Food Nutr. Sci. 2013, 4, 197–203. [Google Scholar] [CrossRef]
- Ma, W.; Guo, A.; Zhang, Y.; Wang, H.; Liu, Y.; Li, H. A review on astringency and bitterness perception of tannins in wine. Trends Food Sci. Technol. 2014, 40, 6–19. [Google Scholar] [CrossRef]
- Giza, B.; Scott, T.; Verhagen, J. Method of Inducing Sweetness by Gallic Acid and Its Applications. U.S. Patent 20020068123 A1, 6 June 2002. [Google Scholar]
- Ayala-Zavala, J.F.; Silva-Espinoza, B.A.; Cruz-Valenzuela, M.R.; Vega-Vega, V.V. Obtaining Hydroalcoholic Extract Rich in Gallic Acid Used as Antimicrobial Agent and Antioxidant, Involves Mashing Agroindustrial Waste of Mango Seeds and Then Performing Alkaline and Acidic Hydrolysis to Produce Monomers of Gallic Acid. M.X. Patent 2013002761-A1, 31 August 2014. [Google Scholar]
- Ayala-Zavala, J.F.; González-Aguilar, G.A. Optimizing the use of garlic oil as antimicrobial agent on fresh-cut tomato through a controlled release system. J. Food Sci. 2010, 75, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.; Barry-Ryan, C.; Bourke, P. The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int. J. Food Microbiol. 2008, 124, 91–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozarda, O.; Demirkoz, A.B.; Özdemir, M. Sensory characteristics and antioxidant capacity of red raspberry extract as a preservative in fruity flavoured beverages. J. Food Sci. Technol. 2015, 52, 6687–6694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- AOAC. Official Methods of Analysis. Method 942.15 Acidity (Titratable) of Fruit Products. In Association of Official Agricultural Chemistry, 15th, ed.; AOAC: Washington, DC, USA, 1990. [Google Scholar]
- Fratianni, F.; Coppola, R.; Nazzaro, F. Phenolic composition and antimicrobial and antiquorum sensing activity of an ethanolic extract of peels from the apple cultivar annurca. J. Med. Food 2011, 14, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enology Vitic. 1965, 16, 144–158. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Villa-Rodríguez, J.A.; Molina-Corral, F.J.; Ayala-Zavala, J.F.; Olivas, G.I.; González-Aguilar, G.A. Effect of maturity stage on the content of fatty acids and antioxidant activity of ‘hass’ avocado. Food Res. Int. 2011, 44, 1231–1237. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved abts radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Ortega-Ramirez, L.A.; Silva-Espinoza, B.A.; Vargas-Arispuro, I.; Gonzalez-Aguilar, G.A.; Cruz-Valenzuela, M.R.; Nazzaro, F.; Ayala-Zavala, J.F. Combination of cymbopogon citratus and allium cepa essential oils increased antibacterial activity in leafy vegetables. J. Sci. Food Agric. 2017, 97. [Google Scholar] [CrossRef] [PubMed]
- Kabuki, T.; Nakajima, H.; Arai, M.; Ueda, S.; Kuwabara, Y.; Dosako, S. Characterization of novel antimicrobial compounds from mango (mangifera indica L.) kernel seeds. Food Chem. 2000, 71, 61–66. [Google Scholar] [CrossRef]
- Abdalla, A.E.; Darwish, S.M.; Ayad, E.H.; El-Hamahmy, R.M. Egyptian mango by-product 1. Compositional quality of mango seed kernel. Food Chem. 2007, 103, 1134–1140. [Google Scholar] [CrossRef]
- Simoes, M.; Bennett, R.N.; Rosa, E.A. Understanding antimicrobial activities of phytochemicals against multidrug resistant bacteria and biofilms. Nat. Prod. Rep. 2009, 26, 746–757. [Google Scholar] [CrossRef] [PubMed]
- Negi, P.S. Plant extracts for the control of bacterial growth: Efficacy, stability and safety issues for food application. Int. J. Food Microbiol. 2012, 156, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simoes, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound phenolics in foods, a review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Soong, Y.-Y.; Barlow, P.J. Antioxidant activity and phenolic content of selected fruit seeds. Food Chem. 2004, 88, 411–417. [Google Scholar] [CrossRef]
- Martín-Diana, A.B.; Rico, D.; Barry-Ryan, C. Green tea extract as a natural antioxidant to extend the shelf-life of fresh-cut lettuce. Innov. Food Sci. Emerg. Technol. 2008, 9, 593–603. [Google Scholar] [CrossRef] [Green Version]
- Ajila, C.; Aalami, M.; Leelavathi, K.; Rao, U. Mango peel powder: A potential source of antioxidant and dietary fiber in macaroni preparations. Innov. Food Sci. Emerg. Technol. 2010, 11, 219–224. [Google Scholar] [CrossRef]
- Ortiz, L.; Dorta, E.; Lobo, M.G.; González-Mendoza, L.A.; Díaz, C.; González, M. Use of banana (Musa acuminata Colla AAA) peel extract as an antioxidant source in orange juices. Plant Foods Hum. Nutr. 2016, 72, 60–66. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Values |
|---|---|
| Total soluble solids (°Brix) | 10.43 ± 0.13 * |
| pH | 3.82 ± 0.07 |
| Titrable acidity (%) | 0.80 ± 0.004 |
| Pulp color | |
| L | 72.15 ± 0.75 |
| Hue angle | 85.82 ± 0.21 |
| Chroma | 62.24 ± 0.42 |
| Bacteria | MSE Treatment (g/L) | Lag (h) | μmax (CFU/h) | Ymax (log CFU) | R2 |
|---|---|---|---|---|---|
| E.coli | 0 | 2.5 a,* | 0.41 a | 10.50 a | 0.97 |
| 8 | 3.0 a | 0.22 ab | 9.70 b | 0.94 | |
| 16 | 8.4 b | 0.13 b | 6.40 c | 0.98 | |
| S. Typhimurium | 0 | 3.0 a | 0.35 a | 9.97 a | 0.98 |
| 8 | 5.0 b | 0.28 a | 9.32 b | 0.98 | |
| 16 | 10.5 c | 0.12 b | 6.09 c | 0.97 | |
| L. monocytogenes | 0 | 0.0 a | 0.26 a | 9.50 a | 0.98 |
| 8 | 11.0 b | 0.34 a | 8.90 b | 0.98 | |
| 16 | 24.0 c | 0.00 b | 0.00 c | ND | |
| S. aureus | 0 | 2.5 a | 0.48 a | 14.07 a | 0.98 |
| 8 | 0.0 b | 0.40 a | 12.86 b | 0.99 | |
| 16 | 12.5 c | 0.21 b | 6.11 c | 0.98 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernal-Mercado, A.T.; Ayala-Zavala, J.F.; Cruz-Valenzuela, M.R.; Gonzalez-Aguilar, G.A.; Nazzaro, F.; Fratianni, F.; Miranda, M.R.A.d.; Silva-Espinoza, B.A. Using Sensory Evaluation to Determine the Highest Acceptable Concentration of Mango Seed Extract as Antibacterial and Antioxidant Agent in Fresh-Cut Mango. Foods 2018, 7, 120. https://doi.org/10.3390/foods7080120
Bernal-Mercado AT, Ayala-Zavala JF, Cruz-Valenzuela MR, Gonzalez-Aguilar GA, Nazzaro F, Fratianni F, Miranda MRAd, Silva-Espinoza BA. Using Sensory Evaluation to Determine the Highest Acceptable Concentration of Mango Seed Extract as Antibacterial and Antioxidant Agent in Fresh-Cut Mango. Foods. 2018; 7(8):120. https://doi.org/10.3390/foods7080120
Chicago/Turabian StyleBernal-Mercado, Ariadna Thalia, Jesus Fernando Ayala-Zavala, Manuel Reynaldo Cruz-Valenzuela, Gustavo A. Gonzalez-Aguilar, Filomena Nazzaro, Florinda Fratianni, Maria Raquel Alcantara de Miranda, and Brenda A. Silva-Espinoza. 2018. "Using Sensory Evaluation to Determine the Highest Acceptable Concentration of Mango Seed Extract as Antibacterial and Antioxidant Agent in Fresh-Cut Mango" Foods 7, no. 8: 120. https://doi.org/10.3390/foods7080120





