Light to Hydrogen: Photocatalytic Hydrogen Generation from Water with Molecularly-Defined Iron Complexes
Abstract
:1. Introduction
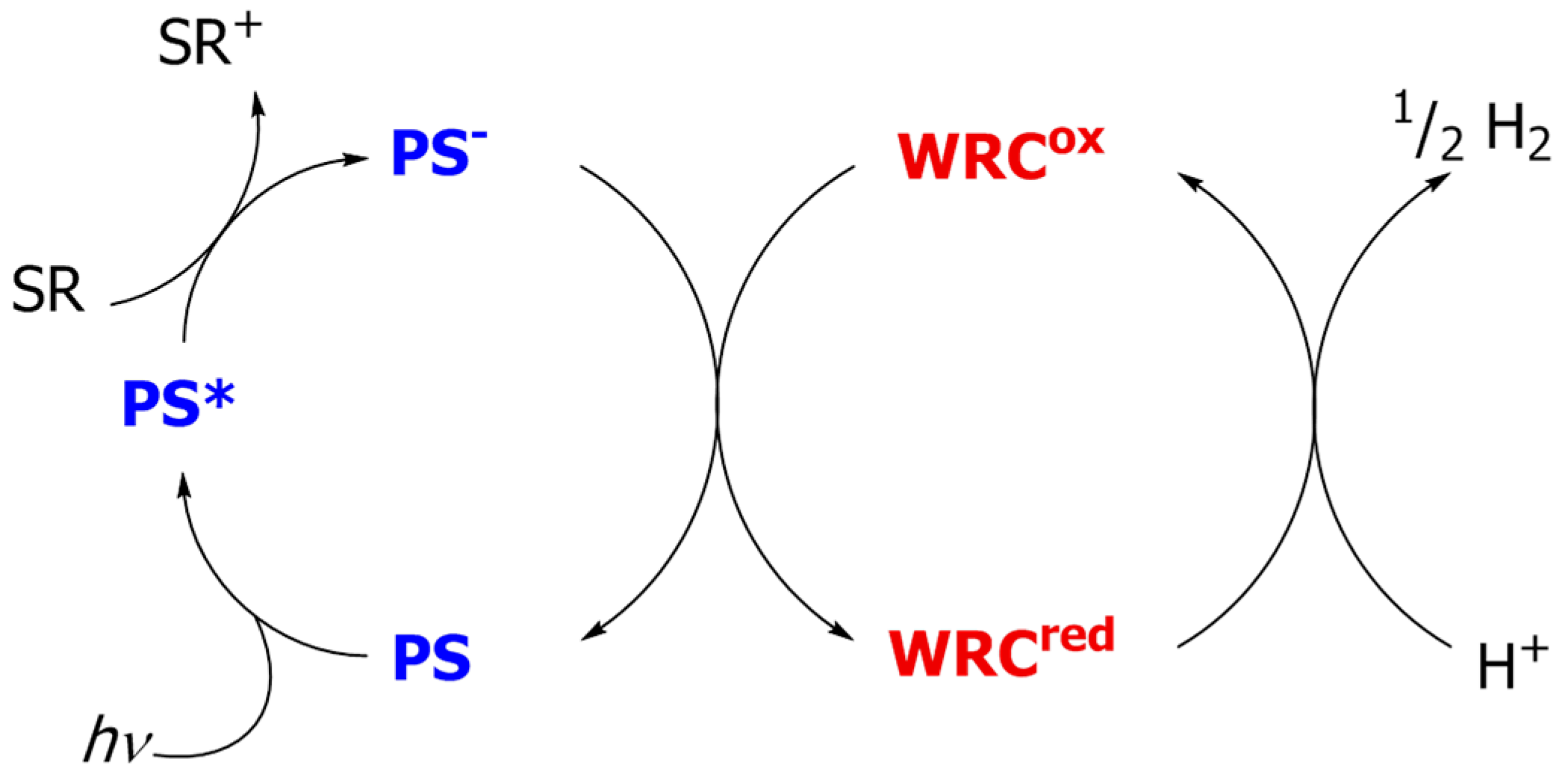
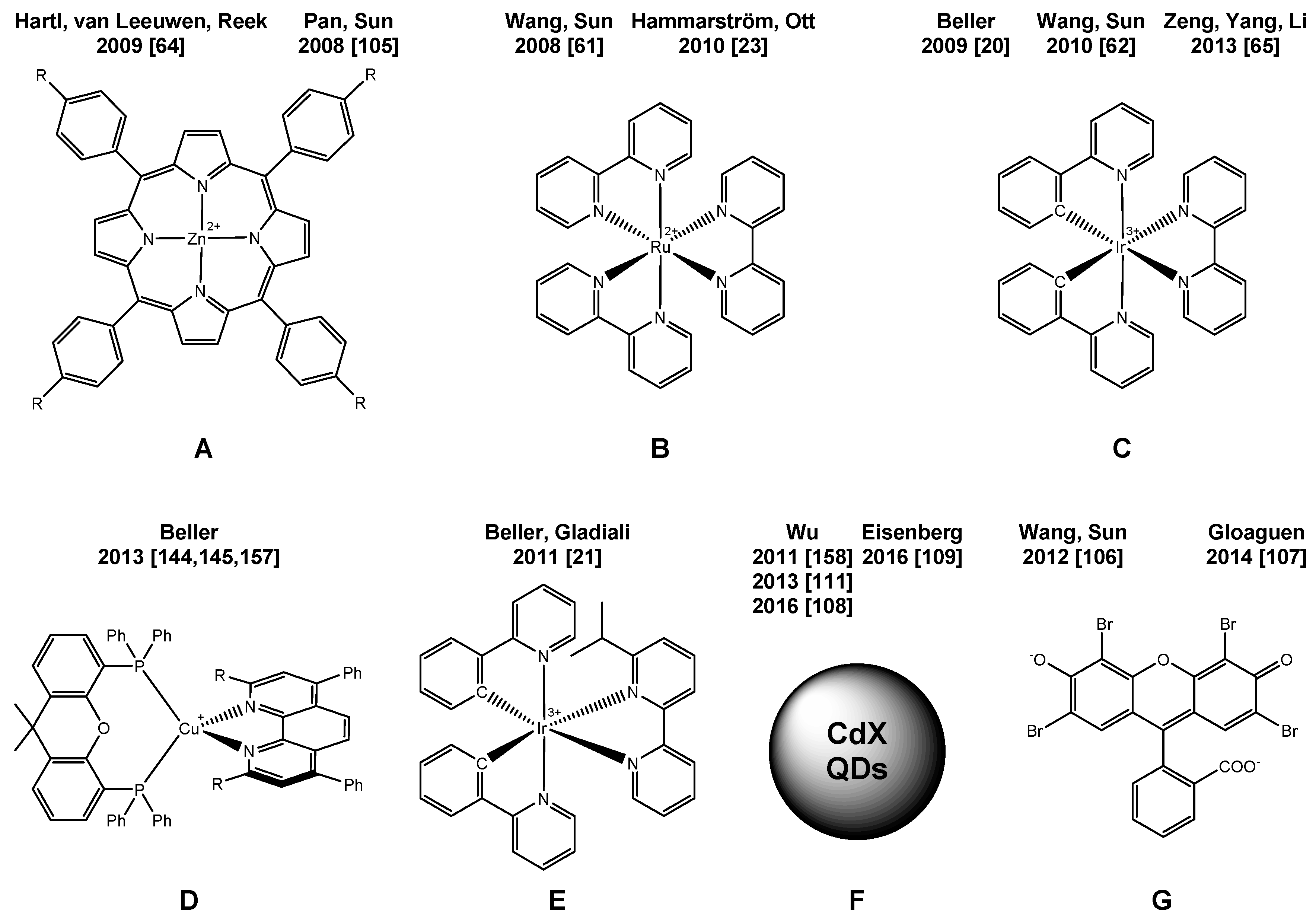
2. Overview of Molecularly-Defined Iron WRCs and the Respective Photosensitizers
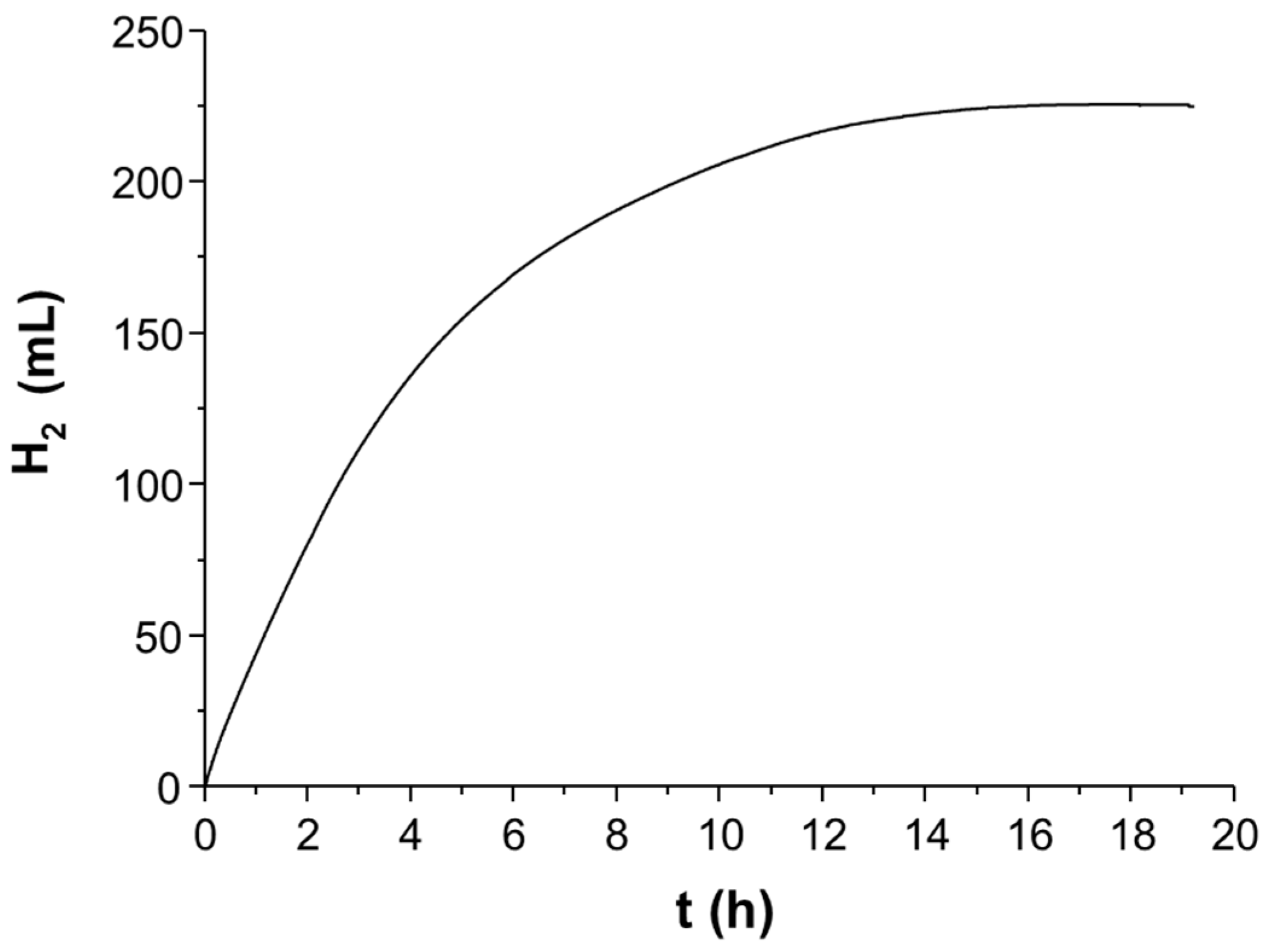
3. Light to Hydrogen: Development and Improvement of an Iron Carbonyl-Based Catalytic System
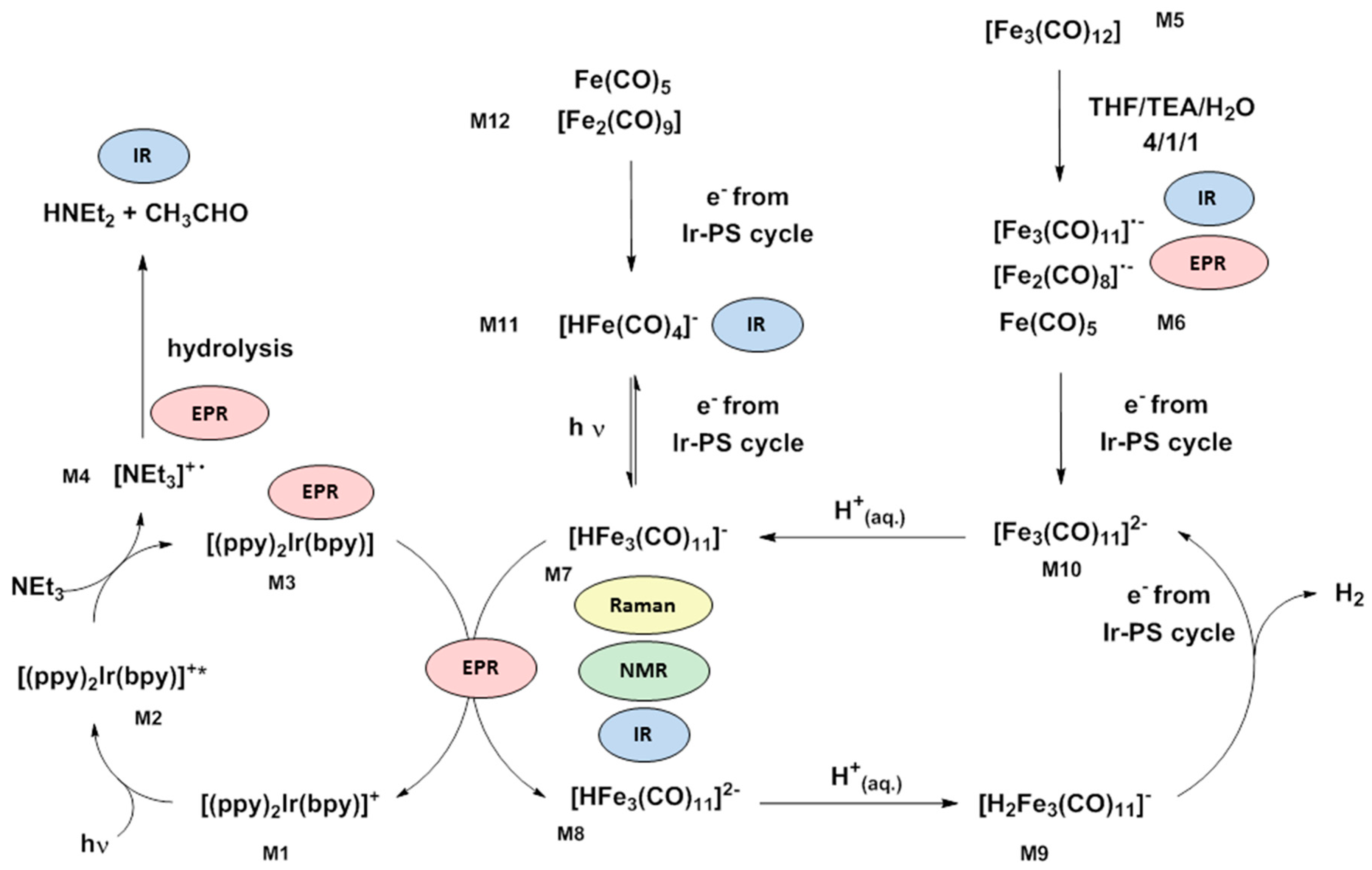
4. Improving Mechanistic Understanding by an Approach of Combined Analytical Methods
5. Summary and Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Armaroli, N.; Balzani, V. The Future of Energy Supply: Challenges and Opportunities. Angew. Chem. Int. Ed. 2007, 46, 52–66. [Google Scholar] [CrossRef] [PubMed]
- Schiermeier, Q.; Tollefson, J.; Scully, T.; Witze, A.; Morton, O. Electricity without Carbon. Nature 2008, 454, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, M.Z. Review of solutions to global warming, air pollution, and energy security. Energy Environ. Sci. 2009, 2, 148–173. [Google Scholar] [CrossRef]
- Armaroli, N.; Balzani, V. Energy for a Sustainable World, 1st ed.; Wiley-VCH: Weinheim, Germany, 2011. [Google Scholar]
- Arakawa, H.; Aresta, M.; Armor, J.N.; Barteau, M.A.; Beckman, E.J.; Bell, A.T.; Bercaw, J.E.; Creutz, C.; Dinjus, E.; Dixon, D.A.; et al. Catalysis Research of Relevance to Carbon Management: Progress, Challenges, and Opportunities. Chem. Rev. 2001, 101, 953–996. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.S.; Nocera, D.G. Powering the planet: Chemical challenges in solar energy utilization. Proc. Natl. Acad. Sci. USA 2006, 103, 15729–15735. [Google Scholar] [CrossRef] [PubMed]
- Centi, G.; Perathoner, S. Towards Solar Fuels from Water and CO2. ChemSusChem 2010, 3, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Balzani, V.; Credi, A.; Venturi, M. Photochemical Conversion of Solar Energy. ChemSusChem 2008, 1, 26–58. [Google Scholar] [CrossRef] [PubMed]
- Lubitz, W.; Reijerse, E.J.; Messinger, J. Solar water-splitting into H2 and O2: Design principles of photosystem II and hydrogenases. Energy Environ. Sci. 2008, 1, 15–31. [Google Scholar] [CrossRef]
- Armaroli, N.; Balzani, V. The Hydrogen Issue. ChemSusChem 2011, 4, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Esswein, A.J.; Nocera, D.G. Hydrogen Production by Molecular Photocatalysis. Chem. Rev. 2007, 107, 4022–4047. [Google Scholar] [CrossRef] [PubMed]
- Osterloh, F.E. Inorganic Materials as Catalysts for Photochemical Splitting of Water. Chem. Mater. 2008, 20, 35–54. [Google Scholar] [CrossRef]
- Nocera, D.G. Personalized Energy: The Home as a Solar Power Station and Solar Gas Station. ChemSusChem 2009, 2, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Kudo, A.; Miseki, Y. Heterogeneous photocatalyst materials for water splitting. Chem. Soc. Rev. 2009, 38, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Yerga, R.M.N.; Galván, M.C.Á.; del Valle, F.; de la Mano, J.A.V.; Fierro, J.L.G. Water Splitting on Semiconductor Catalysts under Visible-Light Irradiation. ChemSusChem 2009, 2, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shen, S.; Guo, L.; Mao, S.S. Semiconductor-based Photocatalytic Hydrogen Generation. Chem. Rev. 2010, 110, 6503–6570. [Google Scholar] [CrossRef] [PubMed]
- Schulz, M.; Karnahl, M.; Schwalbe, M.; Vos, J.G. The role of the bridging ligand in photocatalytic supramolecular assemblies for the reduction of protons and carbon dioxide. Coord. Chem. Rev. 2012, 256, 1682–1705. [Google Scholar] [CrossRef]
- Blankenship, R.E.; Tiede, D.M.; Barber, J.; Brudvig, G.W.; Fleming, G.; Ghirardi, M.; Gunner, M.R.; Junge, W.; Kramer, D.M.; Melis, A.; et al. Comparing Photosynthetic and Photovoltaic Efficiencies and Recognizing the Potential for Improvement. Science 2011, 332, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Bensaid, S.; Centi, G.; Garrone, E.; Perathoner, S.; Saracco, G. Towards Artificial Leaves for Solar Hydrogen and Fuels from Carbon Dioxide. ChemSusChem 2012, 5, 500–521. [Google Scholar] [CrossRef] [PubMed]
- Gärtner, F.; Sundararaju, B.; Surkus, A.-E.; Boddien, A.; Loges, B.; Junge, H.; Dixneuf, P.H.; Beller, M. Light-Driven Hydrogen Generation: Efficient Iron-Based Water Reduction Catalysts. Angew. Chem. Int. Ed. 2009, 48, 9962–9965. [Google Scholar] [CrossRef] [PubMed]
- Gärtner, F.; Cozzula, D.; Losse, S.; Boddien, A.; Anilkumar, G.; Junge, H.; Schulz, T.; Marquet, N.; Spannenberg, A.; Gladiali, S.; et al. Synthesis, Characterisation and Application of Iridium(III) Photosensitisers for Catalytic Water Reduction. Chem. Eur. J. 2011, 17, 6998–7006. [Google Scholar] [CrossRef] [PubMed]
- Gärtner, F.; Denurra, S.; Losse, S.; Neubauer, A.; Boddien, A.; Anilkumar, G.; Spannenberg, A.; Junge, H.; Lochbrunner, S.; Blug, M.; et al. Synthesis and Characterization of New Iridium Photosensitizers for Catalytic Hydrogen Generation from Water. Chem. Eur. J. 2012, 18, 3220–3225. [Google Scholar] [CrossRef] [PubMed]
- Streich, D.; Astuti, Y.; Orlandi, M.; Schwartz, L.; Lomoth, R.; Hammarström, L.; Ott, S. High-Turnover Photochemical Hydrogen Production Catalyzed by a Model Complex of the [FeFe]-Hydrogenase Active Site. Chem. Eur. J. 2010, 16, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wang, M.; Li, C.; Li, X.; Dong, J.; Sun, L. Photochemical H2 production with noble-metal-free molecular devices comprising a porphyrin photosensitizer and a cobaloxime catalyst. Chem. Commun. 2010, 46, 8806–8808. [Google Scholar] [CrossRef] [PubMed]
- Lazarides, T.; Delor, M.; Sazanovich, I.V.; McCormick, T.M.; Georgakaki, I.; Charalambidis, G.; Weinstein, J.A.; Coutsolelos, A.G. Photocatalytic hydrogen production from a noble metal free system based on a water soluble porphyrin derivative and a cobaloxime catalyst. Chem. Commun. 2014, 50, 521–523. [Google Scholar] [CrossRef] [PubMed]
- Luca, O.R.; Blakemore, J.D.; Konezny, S.J.; Praetorius, J.M.; Schmeier, T.J.; Hunsinger, G.B.; Batista, V.S.; Brudvig, G.W.; Hazary, N.; Crabtree, R.H. Organometallic Ni Pincer Complexes for the Electrocatalytic Production of Hydrogen. Inorg. Chem. 2012, 51, 8704–8709. [Google Scholar] [CrossRef] [PubMed]
- Sala, X.; Romero, I.; Rodriguez, M.; Escriche, L.; Llobet, A. Molecular Catalysts that Oxidize Water to Dioxygen. Angew. Chem. Int. Ed. 2009, 48, 2842–2852. [Google Scholar] [CrossRef] [PubMed]
- Limburg, B.; Bouwman, E.; Bonnet, S. Molecular water oxidation catalysts based on transition metals and their decomposition pathways. Coord. Chem. Rev. 2012, 256, 1451–1467. [Google Scholar] [CrossRef]
- Young, K.J.; Martini, L.A.; Milot, R.L.; Snoeberger, R.C., III; Batista, V.S.; Schmuttenmaer, C.A.; Crabtree, R.H.; Brudvig, G.W. Light-driven water oxidation for solar fuels. Coord. Chem. Rev. 2012, 256, 2503–2520. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Eisenberg, R. Catalysts made of earth-abundant elements (Co, Ni, Fe) for water splitting: Recent progress and future challenges. Energy Environ. Sci. 2012, 5, 6012–6021. [Google Scholar] [CrossRef]
- Eckenhoff, W.T.; Eisenberg, R. Molecular systems for light driven hydrogen production. Dalton Trans. 2012, 41, 13004–13021. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chen, L.; Sun, L. Recent progress in electrochemical hydrogen production with earth-abundant metal complexes as catalysts. Energy Environ. Sci. 2012, 5, 6763–6778. [Google Scholar] [CrossRef]
- Wang, M.; Sun, L. Hydrogen Production by Noble-Metal-Free Molecular Catalysts and Related Nanomaterials. ChemSusChem 2010, 3, 551–554. [Google Scholar] [CrossRef] [PubMed]
- Losse, S.; Vos, J.G.; Rau, S. Catalytic hydrogen production at cobalt centres. Coord. Chem. Rev. 2010, 254, 2492–2504. [Google Scholar] [CrossRef]
- Artero, V.; Chavarot-Kerlidou, M.; Fontecave, M. Splitting Water with Cobalt. Angew. Chem. Int. Ed. 2011, 50, 7238–7266. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, C.; Guttentag, M.; Spingler, B.; Alberto, R. 3d Element Complexes of Pentadentate Bipyridine-Pyridine-Based Ligand Scaffolds: Structures and Photocatalytic Activities. Inorg. Chem. 2013, 52, 6055–6061. [Google Scholar] [CrossRef] [PubMed]
- Mc Laughlin, M.P.; Mc Cormick, T.M.; Eisenberg, R.; Holland, P.L. A stable molecular nickel catalyst for the homogeneous photogeneration of hydrogen in aqueous solution. Chem. Commun. 2011, 47, 7989–7991. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; McNamara, W.R.; Eum, M.E.; Holland, P.L.; Eisenberg, R. A Nickel Thiolate Catalyst for the Long-Lived Photocatalytic Production of Hydrogen in a Noble-Metal-Free System. Angew. Chem. Int. Ed. 2012, 51, 1667–1670. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Han, Z.; Brennessel, W.W.; Holland, P.L.; Eisenberg, R. Nickel Complexes for Robust Light-Driven and Electrocatalytic Hydrogen Production from Water. ACS Catal. 2015, 5, 1397–1406. [Google Scholar] [CrossRef]
- Han, Z.; Shen, L.; Brennessel, W.W.; Holland, P.L.; Eisenberg, R. Nickel Pyridinethiolate Complexes as Catalysts for the Light-Driven Production of Hydrogen from Aqueous Solutions in Noble-Metal-Free Systems. J. Am. Chem. Soc. 2013, 135, 14659–14669. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.D.; Newell, R.H.; McNevin, M.J.; Muckermann, J.T.; DuBois, M.R.; DuBois, D.L. Hydrogen Oxidation and Production Using Nickel-Based Molecular Catalysts with Positioned Proton Relays. J. Am. Chem. Soc. 2006, 128, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Helm, M.L.; Stewart, M.P.; Bullock, R.M.; DuBois, M.R.; DuBois, D.L. A Synthetic Nickel Electrocatalyst with a Turnover Frequency Above 100,000 s−1 for H2 Production. Science 2011, 333, 863–866. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Cao, S.; Fu, W.-F. A stable dual-functional system of visible-light-driven Ni(II) reduction to a nickel nanoparticle catalyst and robust in situ hydrogen production. Chem. Commun. 2013, 49, 11251–11253. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J.; Cao, S.; Qin, B.; Zhang, C.; Li, T.-T.; Fu, W.-F. Photoreduction of Iron(III) to Iron(0) Nanoparticles for Simultaneous Hydrogen Evolution in Aqueous Solution. ChemSusChem 2014, 7, 1924–1933. [Google Scholar] [CrossRef] [PubMed]
- Brooks, A.C.; Basore, K.; Bernhard, S. Organocatalytic photoreduction of Zn(II) to zinc metal. Chem Commun. 2014, 50, 5196–5199. [Google Scholar] [CrossRef] [PubMed]
- Junge, H.; Codolà, Z.; Kammer, A.; Rockstroh, N.; Karnahl, M.; Luo, S.-P.; Pohl, M.-M.; Radnik, J.; Gatla, S.; Wohlrab, S.; et al. Copper-based water reduction catalysts for efficient light-drivenhydrogen generation. J. Mol. Catal. A Chem. 2014, 395, 449–456. [Google Scholar] [CrossRef]
- Fontecilla-Camps, J.C.; Volbeda, A.; Cavazza, C.; Nicolet, Y. Structure/Function Relationships of [NiFe]- and [FeFe]-Hydrogenases. Chem. Rev. 2007, 107, 4273–4303. [Google Scholar] [CrossRef] [PubMed]
- Vignais, P.M.; Billoud, B. Occurrence, Classification, and Biological Function of Hydrogenases: An Overview. Chem. Rev. 2007, 107, 4206–4272. [Google Scholar] [CrossRef] [PubMed]
- Lubitz, W.; Ogata, H.; Rüdiger, O.; Reijerse, E. Hydrogenases. Chem. Rev. 2014, 114, 4081–4148. [Google Scholar] [CrossRef] [PubMed]
- Frey, M. Hydrogenases: Hydrogen-Activating Enzymes. ChemBioChem 2002, 3, 153–160. [Google Scholar] [CrossRef]
- Silakov, A.; Wenk, B.; Reijerse, E.; Lubitz, W. 14N HYSCORE investigation of the H-cluster of [FeFe] hydrogenase: Evidence for a nitrogen in the dithiol bridge. Phys. Chem. Chem. Phys. 2009, 11, 6592–6599. [Google Scholar] [CrossRef] [PubMed]
- Erdem, Ö.F.; Schwartz, L.; Stein, M.; Silakov, A.; Kaur-Ghumaan, S.; Huang, P.; Ott, S.; Reijerse, E.J.; Lubitz, W. A Model of the [FeFe] Hydrogenase Active Site with a Biologically Relevant Azadithiolate Bridge: A Spectroscopic and Theoretical Investigation. Angew. Chem. Int. Ed. 2011, 50, 1439–1443. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Wang, X.; Hoffmann, C.; DuBois, D.L.; Bullock, R.M. Heterolytic Cleavage of Hydrogen by an Iron Hydrogenase Model: An Fe–H···H–N Dihydrogen Bond Characterized by Neutron Diffraction. Angew. Chem. Int. Ed. 2014, 53, 5300–5304. [Google Scholar] [CrossRef] [PubMed]
- Zell, T.; Ben-David, Y.; Milstein, D. Unprecedented Iron-Catalyzed Ester Hydrogenation. Mild, Selective, and Efficient Hydrogenation of Trifluoroacetic Esters to Alcohols Catalyzed by an Iron Pincer Complex. Angew. Chem. Int. Ed. 2014, 53, 4685–4689. [Google Scholar] [CrossRef] [PubMed]
- Langer, R.; Iron, M.A.; Konstantinovski, L.; Diskin-Posner, Y.; Leitus, G.; Ben-David, Y.; Milstein, D. Iron Borohydride Pincer Complexes for the Efficient Hydrogenation of Ketones under Mild, Base-Free Conditions: Synthesis and Mechanistic Insight. Chem. Eur. J. 2012, 18, 7196–7209. [Google Scholar] [CrossRef] [PubMed]
- Alberico, E.; Sponholz, P.; Cordes, C.; Nielsen, M.; Drexler, H.-D.; Baumann, W.; Junge, H.; Beller, M. Selective Hydrogen Production from Methanol with a Defined Iron Pincer Catalyst under Mild Conditions. Angew. Chem. Int. Ed. 2013, 52, 14162–14166. [Google Scholar] [CrossRef] [PubMed]
- Werkmeister, S.; Junge, K.; Wendt, B.; Alberico, E.; Jiao, H.; Baumann, W.; Junge, H.; Gallou, F.; Beller, M. Hydrogenation of Esters to Alcohols with a Well-Defined Iron Complex. Angew. Chem. Int. Ed. 2014, 53, 8722–8726. [Google Scholar] [CrossRef] [PubMed]
- Bornschein, C.; Werkmeister, S.; Wendt, B.; Jiao, H.; Alberico, E.; Baumann, W.; Junge, H.; Junge, K.; Beller, M. Mild and selective hydrogenation of aromatic and aliphatic (di)nitiles with a well-defined iron pincer complex. Nat. Commun. 2014, 5, 4111. [Google Scholar] [CrossRef] [PubMed]
- Lagaditis, P.O.; Sues, P.E.; Sonnenberg, J.F.; Wan, K.Y.; Lough, A.J.; Morris, R.H. Iron(II) Complexes Containing Unsymmetrical P–N–P′ Pincer Ligands for the Catalytic Asymmetric Hydrogenation of Ketones and Imines. J. Am. Chem. Soc. 2014, 136, 1367–1380. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Lagaditis, P.O.; Förster, M.; Bielinsky, B.A.; Hazari, N.; Holthausen, M.C.; Joones, W.D.; Schneider, S. Well-Defined Iron Catalysts for the Acceptorless Reversible Dehydrogenation-Hydrogenation of Alcohols and Ketones. ACS Catal. 2014, 4, 3994–4003. [Google Scholar] [CrossRef]
- Na, Y.; Wang, M.; Pan, J.; Zhang, P.; Åkermark, B.; Sun, L. Visible Light-Driven Electron Transfer and Hydrogen Generation Catalyzed by Bioinspired [2Fe2S] Complexes. Inorg. Chem. 2008, 47, 2805–2810. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wang, M.; Na, Y.; Li, X.; Jiang, Y.; Sun, L. Homogeneous photocatalytic production of hydrogen from water by a bioinspired [Fe2S2] catalyst with high turnover numbers. Dalton Trans. 2010, 39, 1204–1206. [Google Scholar] [CrossRef] [PubMed]
- Lomoth, R.; Ott, S. Introducing a dark reaction to photochemistry: Photocatalytic hydrogen from [FeFe] hydrogenase active site model complexes. Dalton Trans. 2009, 9952–9959. [Google Scholar] [CrossRef] [PubMed]
- Kluwer, A.M.; Kapre, R.; Hartl, F.; Lutz, M.; Spek, A.L.; Brouwer, A.M.; van Leeuwen, P.W.N.M.; Reek, J.N.H. Self-assembled biomimetic [2Fe2S]-hydrogenase-based photocatalyst for molecular hydrogen evolution. Proc. Natl. Acad. Sci. USA 2009, 106, 10460–10465. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Zeng, Y.; Chen, J.; Li, Y.-Y.; Yang, G.; Li, Y. Exceptional Dendrimer-Based Mimics of Diiron Hydrogenase for the Photochemical Production of Hydrogen. Angew. Chem. Int. Ed. 2013, 52, 5631–5635. [Google Scholar] [CrossRef] [PubMed]
- Pullen, S.; Fei, H.; Orthaber, A.; Cohen, S.M.; Ott, S. Enhanced Photochemical Hydrogen Production by a Molecular Diiron Catalyst Incorporated into a Metal−Organic Framework. J. Am. Chem. Soc. 2013, 135, 16997–17003. [Google Scholar] [CrossRef] [PubMed]
- Onoda, A.; Kihara, Y.; Fukumoto, K.; Sano, Y.; Hayashi, T. Photoinduced Hydrogen Evolution Catalyzed by a Synthetic Diiron Dithiolate Complex Embedded within a Protein Matrix. ACS Catal. 2014, 4, 2645–2648. [Google Scholar] [CrossRef]
- Lehn, J.-M.; Sauvage, J.-P. Chemical storage of light energy. Catalytic generation of hydrogen by visible light or sunlight. Irradiation of neutral aqueous solutions. Nouv. J. Chim. 1977, 1, 449–451. [Google Scholar]
- Kalyanasundaram, K.; Kiwi, J.; Grätzel, M. Hydrogen Evolution from Water by Visible Light, a Homogeneous Three Component Test System for Redox Catalysis. Helv. Chim. Acta 1978, 61, 2720–2730. [Google Scholar] [CrossRef]
- Kiwi, J.; Grätzel, M. Dynamics of Light-Induced Redox Processes in Microemulsion Systems. J. Am. Chem. Soc. 1978, 100, 6314–6320. [Google Scholar] [CrossRef]
- Moradpour, A.; Amouyal, E.; Keller, P.; Kagan, H. Hydrogen production by visible light irradiation of aqueous solutions of tris(2,2′-bipyridine)ruthenium(2+). Nouv. J. Chim. 1978, 2, 547–549. [Google Scholar]
- Kirch, M.; Lehn, J.-M.; Sauvage, J.-P. Hydrogen Generation by Visible Light Irradiation of Aqueous Solutions of Metal Complexes. An Approach to the Photochemical Conversion and Storage of Solar Energy. Helv. Chim. Acta 1979, 62, 1345–1384. [Google Scholar] [CrossRef]
- Abbotto, A.; Manfredi, N. Electron-rich heteroaromatic conjugated polypyridine ruthenium sensitizers for dye-sensitized solar cells. Dalton Trans. 2011, 40, 12421–12438. [Google Scholar] [CrossRef] [PubMed]
- Vougioukalakis, G.C.; Philippopoulos, A.I.; Stergiopoulos, T.; Falaras, P. Contributions to the development of ruthenium-based sensitizers for dye-sensitized solar cells. Coord. Chem. Rev. 2011, 255, 2602–2621. [Google Scholar] [CrossRef]
- DiSalle, B.F.; Bernhard, S. Orchestrated Photocatalytic Water Reduction Using Surface-Adsorbing Iridium Photosensitizers. J. Am. Chem. Soc. 2011, 133, 11819–11821. [Google Scholar] [CrossRef] [PubMed]
- Gärtner, F.; Boddien, A.; Barsch, E.; Fumino, K.; Losse, S.; Junge, H.; Hollmann, D.; Brückner, A.; Ludwig, R.; Beller, M. Photocatalytic Hydrogen Generation from Water with Iron Carbonyl Phosphine Complexes: Improved Water Reduction Catalysts and Mechanistic Insights. Chem. Eur. J. 2011, 17, 6425–6436. [Google Scholar] [CrossRef] [PubMed]
- Du, P.W.; Knowles, K.; Eisenberg, R. A Homogeneous System for the Photogeneration of Hydrogen from Water Based on a Platinum(II) Terpyridyl Acetylide Chromophore and a Molecular Cobalt Catalyst. J. Am. Chem. Soc. 2008, 130, 12576–12577. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, R.; Masaoka, S.; Sakai, K. Photo-hydrogen-evolving activity of chloro(terpyridine)platinum(II): A single-component molecular photocatalyst. Dalton Trans. 2009, 6127–6133. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-G.; Wang, F.; Wang, H.-Y.; Tung, C.-H.; Wu, L.-Z. Electron transfer and hydrogen generation from a molecular dyad: Platinum(II) alkynyl complex anchored to [FeFe] hydrogenase subsite mimic. Dalton Trans. 2012, 41, 2420–2426. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.N.; Liu, J.H.; Li, C. Photochemical hydrogen evolution catalyzed by trimetallic [Re–Fe] complexes. Inorg. Chem. Commun. 2012, 16, 81–85. [Google Scholar] [CrossRef]
- Probst, B.; Kolano, C.; Hamm, P.; Alberto, R. An Efficient Homogeneous Intermolecular Rhenium-Based Photocatalytic System for the Production of H2. Inorg. Chem. 2009, 48, 1836–1843. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Koike, K.; Morimoto, T.; Inumaru, H.; Ishitani, O. Photochemistry and photocatalysis of rhenium(I) diimine complexes. Adv. Inorg. Chem. 2011, 63, 137–186. [Google Scholar]
- Wang, H.-Y.; Wang, W.-G.; Si, G.; Wang, F.; Tung, C.-H.; Wu, L.-Z. Photocatalytic Hydrogen Evolution from Rhenium(I) Complexes to [FeFe] Hydrogenase Mimics in Aqueous SDS Micellar Systems: A Biomimetic Pathway. Langmuir 2010, 26, 9766–9771. [Google Scholar] [CrossRef] [PubMed]
- Probst, B.; Guttentag, M.; Rodenberg, A.; Hamm, P.; Alberto, R. Photocatalytic H2 Production from Water with Rhenium and Cobalt Complexes. Inorg. Chem. 2011, 50, 3404–3412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Probst, B.; Rodenberg, A.; Guttentag, M.; Hamm, P.; Alberto, R. A Highly Stable Rhenium-Cobalt System for Photocatalytic H2 Production: Unraveling the Performance-Limiting Steps. Inorg. Chem. 2010, 49, 6453–6460. [Google Scholar] [CrossRef] [PubMed]
- Harlang, T.C.B.; Liu, Y.; Gordivska, O.; Fredin, L.A.; Ponseca, C.S., Jr.; Huang, P.; Chabera, P.; Kjaer, K.S.; Mateos, H.; Uhlig, J.; et al. Iron sensitizer converts light to electrons with 92% yield. Nat. Chem. 2015, 7, 883–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poddutoori, P.; Co, D.T.; Samuel, A.P.S.; Kim, C.H.; Vagnini, M.T.; Wasielewski, M.R. Photoinitiated multistep charge separation in ferrocene–zinc porphyrin–diiron hydrogenase model complex triads. Energy Environ. Sci. 2011, 4, 2441–2450. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Onji, T.; Ohara, H.; Ikeda, N.; Harada, A. Photoinduced Hydrogen-Evolution System with an Antibody-Porphyrin Complex as a Photosensitizer. Bull. Chem. Soc. Jpn. 2009, 82, 1341–1346. [Google Scholar] [CrossRef]
- Zorlu, Y.; Dumoulin, F.; Durmus, M.; Ahsen, V. Comparative studies of photophysical and photochemical properties of solketal substituted platinum(II) and zinc(II) phthalocyanine sets. Tetrahedron 2010, 66, 3248–3258. [Google Scholar] [CrossRef]
- Amao, Y.; Aoki, K. Artificial Photosynthesis System Using Mg Chlorophyll-a Conjugated Nanocrystalline TiO2 Film Electrode via the Axial Imidazole-4-Acetic Acid Ligand. J. Biobased Mater. Bioenergy 2008, 2, 51–56. [Google Scholar] [CrossRef]
- Amao, Y.; Hirakawa, T. Hydrolysis of a mixture of saccharides by cellulase from Aspergillus niger and its application for visible-light-induced hydrogen gas production system using Mg chlorophyll-a and platinum nanoparticles. Int. J. Hydrogen Energy 2010, 35, 6624–6628. [Google Scholar] [CrossRef]
- Amao, Y.; Hirakawa, T.; Himeshima, N. Photoinduced biohydrogen production from saccharide mixture with the photosensitization of Mg chlorophyll a from green plant. Catal. Commun. 2008, 9, 131–134. [Google Scholar] [CrossRef]
- Amao, Y.; Maki, Y.; Fuchino, Y. Photoinduced Hydrogen Production with Artificial Photosynthesis System Based on Carotenoid-Chlorophyll Conjugated Micelles. J. Phys. Chem. C 2009, 113, 16811–16815. [Google Scholar] [CrossRef]
- Tomonou, Y.; Amao, Y. Visible and near-IR light induced biohydrogen production using the system containing Mg chlorophyll-a from Spirulina and colloidal platinum. Biometals 2003, 16, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Gimbert-Suriñach, C.; Albero, J.; Stoll, T.; Fortage, J.; Collomb, M.-N.; Deronzier, A.; Palomares, E.; Llobet, A. Efficient and Limiting Reactions in Aqueous Light-Induced Hydrogen Evolution Systems using Molecular Catalysts and Quantum Dots. J. Am. Chem. Soc. 2014, 136, 7655–7661. [Google Scholar] [CrossRef] [PubMed]
- Martindale, B.C.M.; Hutton, G.A.M.; Caputo, C.A.; Reisner, E. Solar Hydrogen Production Using Carbon Quantum Dots and a Molecular Nickel Catalyst. J. Am. Chem. Soc. 2015, 137, 6018–6025. [Google Scholar] [CrossRef] [PubMed]
- McCormick, T.M.; Calitree, B.D.; Orchard, A.; Kraut, N.D.; Bright, F.V.; Detty, M.R.; Eisenberg, R. Reductive Side of Water Splitting in Artificial Photosynthesis: New Homogeneous Photosystems of Great Activity and Mechanistic Insight. J. Am. Chem. Soc. 2010, 132, 15480–15483. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Wang, M.; Zhang, P.; Yang, S.; Liu, J.; Li, X.; Sun, L. Promoting Effect of Electrostatic Interaction between a Cobalt Catalyst and a Xanthene Dye on Visible-Light-Driven Electron Transfer and Hydrogen Production. J. Phys Chem. C 2011, 115, 15089–15096. [Google Scholar] [CrossRef]
- Mori, K.; Kakudo, H.; Yamashita, H. Creation of Nickel-Based Active Species within a Macroreticular Acidic Resin: A Noble-Metal-Free Heterogeneous Catalyst for Visible-Light-Driven H2 Evolution from Water. ACS Catal. 2014, 4, 4129–4135. [Google Scholar] [CrossRef]
- Yong, Y.; Wang, M.; Xue, L.; Zhang, F.; Chen, L.; Ahlquist, M.S.G.; Sun, L. Nickel Complex with Internal Bases as Efficient Molecular Catalyst for Photochemical H2 Production. ChemSusChem 2014, 7, 2889–2897. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.-H.; Wang, J.-Y.; Hu, M.-Q.; Ma, C.-B.; Wen, H.-M.; Song, X.-W.; Chen, C.-N. Efficient photo-driven hydrogen evolution by binuclear nickel catalysts of different coordination in noble-metal-free systems. Dalton Trans. 2013, 42, 8684–8691. [Google Scholar] [CrossRef] [PubMed]
- Lazarides, T.; McCormick, T.; Du, P.; Luo, G.; Lindley, B.; Eisenberg, R. Making Hydrogen from Water Using a Homogeneous System Without Noble Metals. J. Am. Chem. Soc. 2009, 131, 9192–9194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wang, M.; Dong, J.; Li, X.; Wang, F.; Wu, L.; Sun, L. Photocatalytic Hydrogen Production from Water by Noble-Metal-Free Molecular Catalyst Systems Containing Rose Bengal and the Cobaloximes of BFx-Bridged Oxime Ligands. J. Phys Chem. C 2010, 114, 15868–15874. [Google Scholar] [CrossRef]
- Zhang, W.; Hong, J.; Zheng, J.; Huang, Z.; Zhou, J.; Xu, R. Nickel−Thiolate Complex Catalyst Assembled in One Step in Water for Solar H2 Production. J. Am. Chem. Soc. 2011, 133, 20680–20683. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, M.; Zhang, S.; Pan, J.; Na, Y.; Liu, J.; Åkermark, B.; Sun, L. Noncovalent Assembly of a Metalloporphyrin and an Iron Hydrogenase Active-Site Model: Photo-Induced Electron Transfer and Hydrogen Generation. J. Phys. Chem. B 2008, 112, 8198–8202. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, M.; Chen, L.; Wang, X.; Dong, J.; Sun, L. Photocatalytic Water Reduction and Study of the Formation of FeIFe0 Species in Diiron Catalyst Sytems. ChemSusChem 2012, 5, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Orain, C.; Quentel, F.; Gloaguen, F. Photocatalytic Hydrogen Production Using Models of the Iron–Iron Hydrogenase Active Site Dispersed in Micellar Solution. ChemSusChem 2014, 7, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Jian, J.-X.; Ye, C.; Wang, X.-Z.; Wen, M.; Li, Z.-J.; Li, X.-B.; Chen, B.; Tung, C.-H.; Wu, L.-Z. Comparison of H2 photogeneration by [FeFe]-hydrogenase mimics with CdSe QDs and Ru(bpy)3Cl2 in aqueous solution. Energy Environ. Sci. 2016, 9, 2083–2089. [Google Scholar] [CrossRef]
- Lv, H.; Ruberu, P.A.; Fleischauer, V.E.; Brennessel, W.W.; Neidig, M.L.; Eisenberg, R. Catalytic Light-Driven Generation of Hydrogen from Water by Iron Dithiolene Complexes. J. Am. Chem. Soc. 2016, 138, 11654–11663. [Google Scholar] [CrossRef] [PubMed]
- Tropmann, S.; König, B. Functionalized Vesicles with Co-Embedded CdSe Quantum Dots and [FeFe]-Hydrogenase Mimic for Light-Driven Hydrogen Production. ChemistrySelect 2016, 1, 1405–1409. [Google Scholar] [CrossRef]
- Jian, J.-X.; Liu, Q.; Li, Z.-J.; Wang, F.; Li, X.-B.; Li, C.-B.; Liu, B.; Meng, Q.-Y.; Chen, B.; Feng, K.; et al. Chitosan confinement enhances hydrogen photogeneration from a mimic of the diiron subsite of [FeFe]-hydrogenase. Nat. Commun. 2013, 4, 2695. [Google Scholar] [CrossRef] [PubMed]
- Caputo, C.A.; Wang, L.; Beranek, R.; Reisner, E. Carbon nitride–TiO2 hybrid modified with hydrogenase for visible light driven hydrogen production. Chem. Sci. 2015, 6, 5690–5694. [Google Scholar] [CrossRef]
- Edel, A.; Marnot, P.A.; Sauvage, J.P. Photochemical reduction of water via energy transfer from a copper(I) complex. Nouv. J. Chim. 1984, 8, 495–498. [Google Scholar]
- Armaroli, N.; Accorsi, G.; Holler, M.; Moudam, O.; Nierengarten, J.-F.; Zhou, Z.; Wegh, R.T.; Welter, R. Highly Luminescent CuI Complexes for Light-Emitting Electrochemical Cells. Adv. Mater. 2006, 18, 1313–1316. [Google Scholar] [CrossRef]
- Cuttell, D.G.; Kuang, S.M.; Fanwick, P.E.; McMillin, D.R.; Walton, R.A. Simple Cu(I) Complexes with Unprecedented Excited-State Lifetimes. J. Am. Chem. Soc. 2002, 124, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Harkins, S.B.; Peters, J.C. A Highly Emissive Cu2N2 Diamond Core Complex Supported by a [PNP]− Ligand. J. Am. Chem. Soc. 2005, 127, 2030–2031. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.S.; Branham, C.W.; Marquardt, B.J.; Mann, K.R. Oxygen Gas Sensing by Luminescence Quenching in Crystals of Cu(xantphos)(phen)+ Complexes. J. Am. Chem. Soc. 2010, 132, 14079–14085. [Google Scholar] [CrossRef] [PubMed]
- Kuang, S.-M.; Cuttell, D.G.; McMillin, D.R.; Fanwick, P.E.; Walton, R.A. Synthesis and Structural Characterization of Cu(I) and Ni(II) Complexes that Contain the Bis[2-(diphenylphosphino)phenyl]ether Ligand. Novel Emission Properties for the Cu(I) Species. Inorg. Chem. 2002, 41, 3313–3322. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Vante, N.; Nierengarten, J.-F.; Sauvage, J.-P. Spectral Sensitization of Large-band-gap Semiconductors (Thin Films and Ceramics) by a Carboxylated Bis(1,10-Phenanthroline)copper(I) Complex. J. Chem. Soc. Dalton Trans. 1994, 1649–1654. [Google Scholar] [CrossRef]
- Bessho, T.; Constable, E.C.; Graetzel, M.; Hernandez Redondo, A.; Housecroft, C.E.; Kylberg, W.; Nazeeruddin, M.K.; Neuburger, M.; Schaffner, S. An element of surprise—Efficient copper-functionalized dye-sensitized solar cells. Chem. Commun. 2008, 3717–3719. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, L.; Friedrichs, J.; Mydlack, M.; Baumann, T.; Nieger, M.; Bräse, S. Outstanding luminescence from neutral copper(I) complexes with pyridyl-tetrazolate and phosphine ligands. Chem. Commun. 2013, 49, 6501–6503. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.D.; Ortí, E.; Bolink, H.J.; Monti, F.; Accorsi, G.; Armaroli, N. Luminescent Ionic Transition-Metal Complexes for Light-Emitting Electrochemical Cells. Angew. Chem. Int. Ed. 2012, 51, 8178–8211. [Google Scholar] [CrossRef] [PubMed]
- Sandroni, M.; Kayanuma, M.; Planchat, A.; Szuwarski, N.; Blart, E.; Pellegrin, Y.; Daniel, C.; Boujtita, M.; Odobel, F. First application of the HETPHEN concept to new heteroleptic bis(diimine) copper(I) complexes as sensitizers in dye sensitized solar cells. Dalton Trans. 2013, 42, 10818–10827. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, J.; Dan, M.; Kawai, T. Phosphorescent properties of metal-free diphosphine ligands and effects of copper binding. Dalton Trans. 2013, 42, 16096–16101. [Google Scholar] [CrossRef] [PubMed]
- Czerwieniec, R.; Kowalski, K.; Yersin, H. Highly efficient thermally activated fluorescence of a new rigid Cu(I) complex [Cu(dmp)(phanephos)]+. Dalton Trans. 2013, 42, 9826–9830. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Wang, M.; Yang, Y.; Yao, T.; Sun, L. A Molecular Copper Catalyst for Electrochemical Water Reduction with a Large Hydrogen-Generation Rate Constant in Aqueous Solution. Angew. Chem. Int. Ed. 2014, 53, 13803–13807. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Meyer, T.J. Copper(II) Catalysis of Water Oxidation. Angew. Chem. Int. Ed. 2013, 52, 700–703. [Google Scholar] [CrossRef] [PubMed]
- Barnett, S.M.; Goldberg, K.I.; Mayer, J.M. A soluble copper–bipyridine water-oxidation electrocatalyst. Nat. Chem. 2012, 4, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, C.; Liu, S.; Wang, J.-L.; Lin, W. A Biomimetic Copper Water Oxidation Catalyst with Low Overpotential. J. Am. Chem. Soc. 2014, 136, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Coggins, M.K.; Zhang, M.-T.; Chen, Z.; Song, N.; Meyer, T.J. Single-Site Copper(II) Water Oxidation Electrocatalysis: Rate Enhancements with HPO42− as a Proton Acceptor at pH 8. Angew. Chem. Int. Ed. 2014, 53, 12226–12230. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Kang, P.; Zhang, M.-T.; Stoner, B.R.; Meyer, T.J. Cu(II)/Cu(0) electrocatalyzed CO2 and H2O splitting. Energy Environ. Sci. 2013, 6, 813–817. [Google Scholar] [CrossRef]
- Choi, H.-J.; Kang, M. Hydrogen production from methanol/water decomposition in a liquid photosystem using the anatase structure of Cu loaded TiO2. Int. J. Hydrogen Energy 2007, 32, 3841–3848. [Google Scholar] [CrossRef]
- Yoong, L.S.; Chong, F.K.; Dutta, B.K. Development of copper-doped TiO2 photocatalyst for hydrogen production under visible light. Energy 2009, 34, 1652–1661. [Google Scholar] [CrossRef]
- Xu, S.; Sun, D.D. Significant improvement of photocatalytic hydrogen generation rate over TiO2 with deposited CuO. Int. J. Hydrogen Energy 2009, 34, 6096–6104. [Google Scholar] [CrossRef]
- Sakata, Y.; Yamamoto, T.; Okazaki, T.; Imamura, H.; Tsuchiya, S. Generation of Visible Light Response on the Photocatalyst of a Copper Ion Containing TiO2. Chem. Lett. 1998, 27, 1253–1254. [Google Scholar] [CrossRef]
- Kumar, D.P.; Shankar, M.V.; Kumari, M.M.; Sadanandam, G.; Srinivas, B.; Durgakumari, V. Nano-size effects on CuO/TiO2 catalysts for highly efficient H2 production under solar light irradiation. Chem. Commun. 2013, 49, 9443–9445. [Google Scholar] [CrossRef] [PubMed]
- Paracchino, A.; Laporte, V.; Sivula, K.; Grätzel, M.; Thimsen, E. Highly active oxide photocathode for photoelectrochemical water reduction. Nat. Mater. 2011, 10, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Fornasiero, P.; Gasparotto, A.; Gombac, V.; Maccato, C.; Montini, T.; Tondello, E. The Potential of Supported Cu2O and CuO Nanosystems in Photocatalytic H2 Production. ChemSusChem 2009, 2, 230–233. [Google Scholar] [CrossRef] [PubMed]
- Somasundaram, S.; Chenthamarakshan, C.R.N.; de Tacconi, N.R.; Rajeshwar, K. Photocatalytic production of hydrogen from electrodeposited p-Cu2O film and sacrificial electron donors. Int. J. Hydrogen Energy 2007, 32, 4661–4669. [Google Scholar] [CrossRef]
- McCusker, C.E.; Castellano, F.N. Design of a Long-Lifetime, Earth-Abundant, Aqueous Compatible Cu(I) Photosensitizer Using Cooperative Steric Effects. Inorg. Chem. 2013, 52, 8114–8120. [Google Scholar] [CrossRef] [PubMed]
- Kaeser, A.; Mohankumar, M.; Mohanraj, J.; Monti, F.; Holler, M.; Cid, J.-J.; Moudam, O.; Nierengarten, I.; Karmazin-Brelot, L.; Duhayon, C.; et al. Heteroleptic Copper(I) Complexes Prepared from Phenanthroline and Bis-Phosphine Ligands. Inorg. Chem. 2013, 52, 12140–12151. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-L.; Cao, X.-F.; Wang, J.-Y.; He, L.-H.; Liu, Z.-Y.; Wen, H.-R.; Chen, Z.-N. Synthesis, Characterization, and Photophysical Properties of Heteroleptic Copper(I) Complexes with Functionalized 3‑(2′-Pyridyl)-1,2,4-triazole Chelating Ligands. Inorg. Chem. 2013, 52, 9727–9740. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Buyukcakir, O.; Mara, M.W.; Coskun, A.; Dimitrijevic, N.M.; Barin, G.; Kokhan, O.; Stickrath, A.B.; Ruppert, R.; Tiede, D.M.; et al. Highly Efficient Ultrafast Electron Injection from the Singlet MLCT Excited State of Copper(I) Diimine Complexes to TiO2 Nanoparticles. Angew. Chem. Int. Ed. 2012, 51, 12711–12715. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.-P.; Mejía, E.; Friedrich, A.; Pazidis, A.; Junge, H.; Surkus, A.-E.; Jackstell, R.; Denurra, S.; Gladiali, S.; Lochbrunner, S.; et al. Photocatalytic Water Reduction with Copper-Based Photosensitizers: A Noble-Metal-Free System. Angew. Chem. Int. Ed. 2013, 52, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Mejía, E.; Luo, S.-P.; Karnahl, M.; Friedrich, A.; Tschierlei, S.; Surkus, A.-E.; Junge, H.; Gladiali, S.; Lochbrunner, S.; Beller, M. A Noble-Metal-Free System for Photocatalytic Hydrogen Production from Water. Chem. Eur. J. 2013, 19, 15972–15978. [Google Scholar] [CrossRef] [PubMed]
- Karnahl, M.; Mejía, E.; Rockstroh, N.; Tschierlei, S.; Luo, S.-P.; Grabow, K.; Kruth, A.; Brüser, V.; Junge, H.; Lochbrunner, S.; et al. Photocatalytic Hydrogen Production with Copper Photosensitizer–Titanium Dioxide Composites. ChemCatChem 2014, 6, 82–86. [Google Scholar] [CrossRef]
- Khnayzer, R.S.; McCusker, C.E.; Olaiya, B.S.; Castellano, F.N. Robust Cuprous Phenanthroline Sensitizer for Solar Hydrogen Photocatalysis. J. Am. Chem. Soc. 2013, 52, 14068–14070. [Google Scholar] [CrossRef] [PubMed]
- Montalti, M.; Credi, A.; Prodi, L.; Gandolfi, T. Chemical Actinometry. In Handbook of Photochemistry, 3rd ed.; CRC: Boca Raton, FL, USA, 2011; pp. 601–616. [Google Scholar]
- Carneiro, Z.A.; de Moraes, J.C.B.; Rodrigues, F.P.; de Lima, R.G.; Curti, C.; da Rocha, Z.N.; Paulo, M.; Bendhack, L.M.; Tedesco, A.C.; Formiga, A.L.B.; et al. Photocytotoxic activity of a nitrosyl phthalocyanine ruthenium complex—A system capable of producing nitric oxide and singlet oxygen. J. Inorg. Biochem. 2011, 105, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Heussner, K.; Peuntinger, K.; Rockstroh, N.; Nye, L.C.; Ivanovic-Burmazovic, I.; Rau, S.; Streb, C. Solution and solid-state interactions in a supramolecular ruthenium photosensitizer–polyoxometalate aggregate. Chem. Commun. 2011, 47, 6852–6854. [Google Scholar] [CrossRef] [PubMed]
- La Ganga, G.; Puntoriero, F.; Campagna, S.; Bazzan, I.; Berardi, S.; Bonchio, M.; Sartorel, A.; Natali, M.; Scandola, F. Light-driven water oxidation with a molecular tetra-cobalt(III) cubane cluster. Faraday Discuss. 2012, 155, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.J.; Guo, Y.R.; Li, L.; Odoh, S.O.; Fu, H.G.; Zhang, H.X. Structures, spectroscopic properties and redox potentials of quaterpyridyl Ru(II) photosensitizer and its derivatives for solar energy cell: A density functional study. Phys. Chem. Chem. Phys. 2011, 13, 14481–14489. [Google Scholar] [CrossRef] [PubMed]
- Shimakoshi, H.; Nishi, M.; Tanaka, A.; Chikama, K.; Hisaeda, Y. Photocatalytic function of a polymer-supported B12 complex with a ruthenium trisbipyridine photosensitizer. Chem. Commun. 2011, 47, 6548–6550. [Google Scholar] [CrossRef] [PubMed]
- McMillin, D.R.; Kirchhoff, J.R.; Goodwin, K.V. Exciplex quenching of photo-excited copper complexes. Coord. Chem. Rev. 1985, 64, 83–92. [Google Scholar] [CrossRef]
- Riesgo, E.C.; Hu, Y.-Z.; Bouvier, F.; Thummel, R.P.; Scaltrito, D.V.; Meyer, G.J. Crowded Cu(I) Complexes Involving Benzo[h]quinoline: π-Stacking Effects and Long-Lived Excited States. Inorg. Chem. 2001, 40, 3413–3422. [Google Scholar] [CrossRef] [PubMed]
- Tschierlei, S.; Karnahl, M.; Rockstroh, N.; Junge, H.; Beller, M.; Lochbrunner, S. Substitution-Controlled Excited State Processes in Heteroleptic Copper(I) Photosensitizers Used in Hydrogen Evolving Systems. ChemPhysChem 2014, 17, 3709–3713. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-Y.; Wang, H.; Chen, N.-Y.; Lennox, A.J.J.; Friedrich, A.; Xia, L.-M.; Lochbrunner, S.; Junge, H.; Beller, M.; Zhou, S.; et al. Efficient Photocatalytic Water Reduction Using In Situ Generated Knölker’s Iron Complexes. ChemCatChem 2016, 8, 2340–2344. [Google Scholar] [CrossRef]
- Weng, F.; Wang, W.-G.; Wang, X.-J.; Wang, H.-Y.; Tung, C.-H.; Wu, L.-Z. A Highly Efficient Photocatalytic System for Hydrogen Production by a Robust Hydrogenase Mimic in an Aqueous Solution. Angew. Chem. Int. Ed. 2011, 50, 3193–3197. [Google Scholar] [CrossRef] [PubMed]
- Bokarev, S.I.; Bokareva, O.S.; Kühn, O. A theoretical perspective on charge transfer in photocatalysis. The example of Ir-based systems. Coord. Chem. Rev. 2015, 304–305, 133–145. [Google Scholar] [CrossRef]
- Hollmann, D.; Gärtner, F.; Ludwig, R.; Barsch, E.; Junge, H.; Blug, M.; Hoch, S.; Beller, M.; Brückner, A. Insights into the Mechanism of Photocatalytic Water Reduction by DFT-Supported In Situ EPR/Raman Spectroscopy. Angew. Chem. Int. Ed. 2011, 50, 10246–10250. [Google Scholar] [CrossRef] [PubMed]
- Fischer, S.; Bokareva, O.S.; Barsch, E.; Bokarev, S.I.; Kühn, O.; Ludwig, R. Mechanistic Study of Photocatalytic Hydrogen Generation with Simple Iron Carbonyls as Water Reduction Catalysts. ChemCatChem 2016, 8, 404–411. [Google Scholar] [CrossRef]
- Bokarev, S.I.; Hollmann, D.; Pazidis, A.; Neubauer, A.; Radnik, J.; Kühn, O.; Lochbrunner, S.; Junge, H.; Beller, M.; Brückner, A. Spin density distribution after electron transfer from triethylamine to an [Ir(ppy)2(bpy)]+ photosensitizer during photocatalytic water reduction. Phys. Chem. Chem. Phys. 2014, 16, 4789–4796. [Google Scholar] [CrossRef] [PubMed]
- Neubauer, A.; Grell, G.; Friedrich, A.; Bokarev, S.I.; Schwarzbach, P.; Gärtner, F.; Surkus, A.-E.; Junge, H.; Beller, M.; Kühn, O.; et al. Electron- and Energy-Transfer Processes in a Photocatalytic System Based on an Ir(III)-Photosensitizer and an Iron Catalyst. J. Phys. Chem. Lett. 2014, 5, 1355–1360. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Hartung, H.; Böttcher, H.-C.; Baumeister, U.; Böhland, U.; Reinhold, J.; Sieler, J.; Ladriere, J.; Schiebel, H.M. Unexpected reaction of [Ni(CO)4−n(R2PCl)n] (n = 1, 2; R = But, Cy, Ph) with Na2[Fe2(CO)8]. Synthesis and electronic structure of the anions [Fe2(µ-CO)(CO)6(µ-PR2)]− and their reactions with H+ and [M(PPh3)]+ (M = Cu, Ag, Au). Polyhedron 1991, 10, 2423–2435. [Google Scholar] [CrossRef]
- Fischer, S.; Hollmann, D.; Tschierlei, S.; Karnahl, M.; Rockstroh, N.; Barsch, E.; Schwarzbach, P.; Luo, S.-P.; Junge, H.; Beller, M.; et al. Death and Rebirth: Photocatalytic Hydrogen Production by a Self-Organizing Copper−Iron System. ACS Catal. 2014, 4, 1845–1849. [Google Scholar] [CrossRef]
- Lennox, A.J.J.; Fischer, S.; Jurrat, M.; Luo, S.-P.; Rockstroh, N.; Junge, H.; Ludwig, R.; Beller, M. Copper-Based Photosensitisers in Water Reduction: A More Efficient In Situ Formed System and Improved Mechanistic Understanding. Chem. Eur. J. 2016, 22, 1233–1238. [Google Scholar] [CrossRef] [PubMed]
- Tschierlei, S.; Neubauer, A.; Rockstroh, N.; Karnahl, M.; Schwarzbach, P.; Junge, H.; Beller, M.; Lochbrunner, S. Ultrafast excited state dynamics of iridium(III) complexes and their changes upon immobilisation onto titanium dioxide layers. Phys. Chem. Chem. Phys. 2016, 18, 10682–10687. [Google Scholar] [CrossRef] [PubMed]


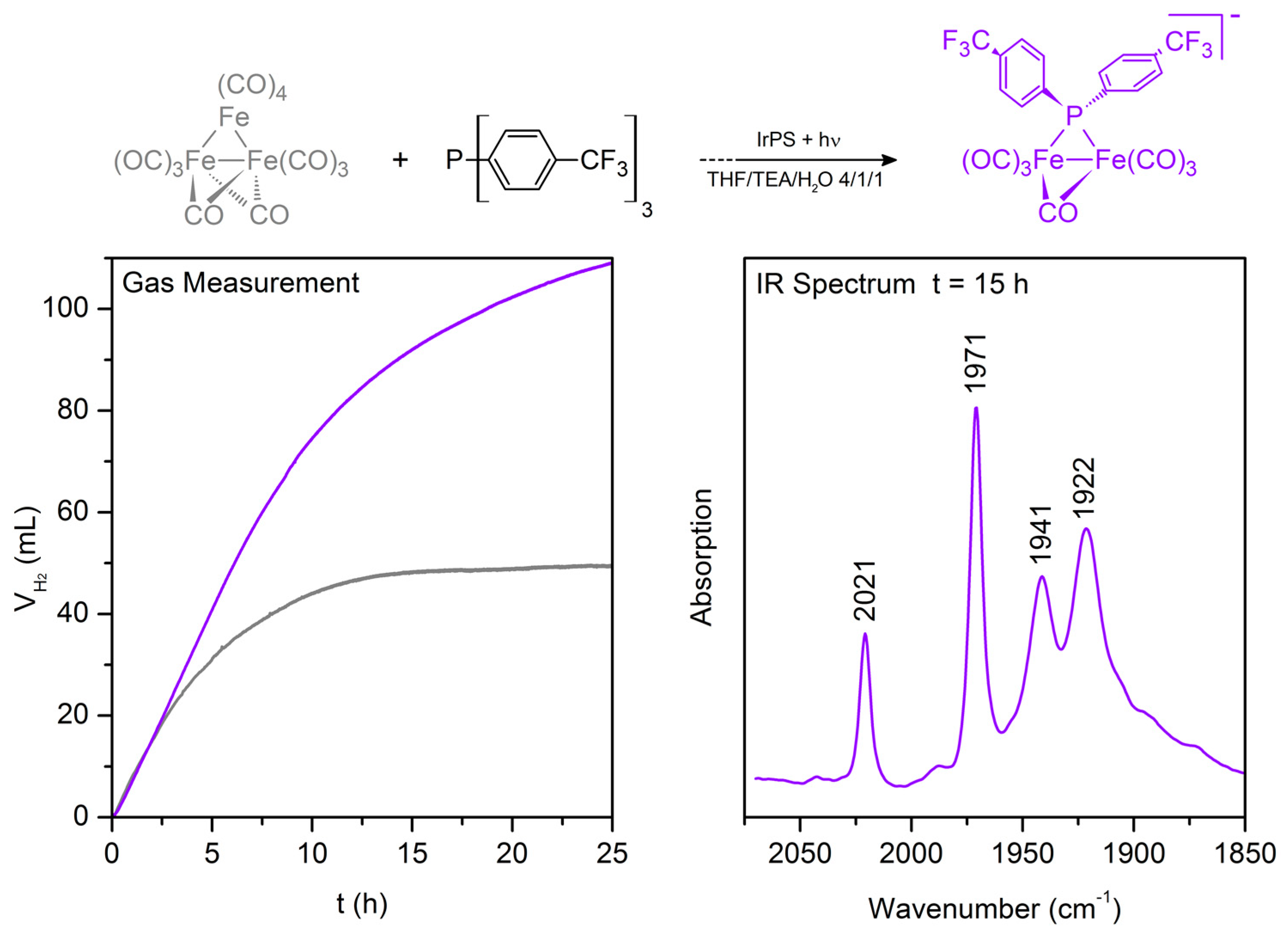
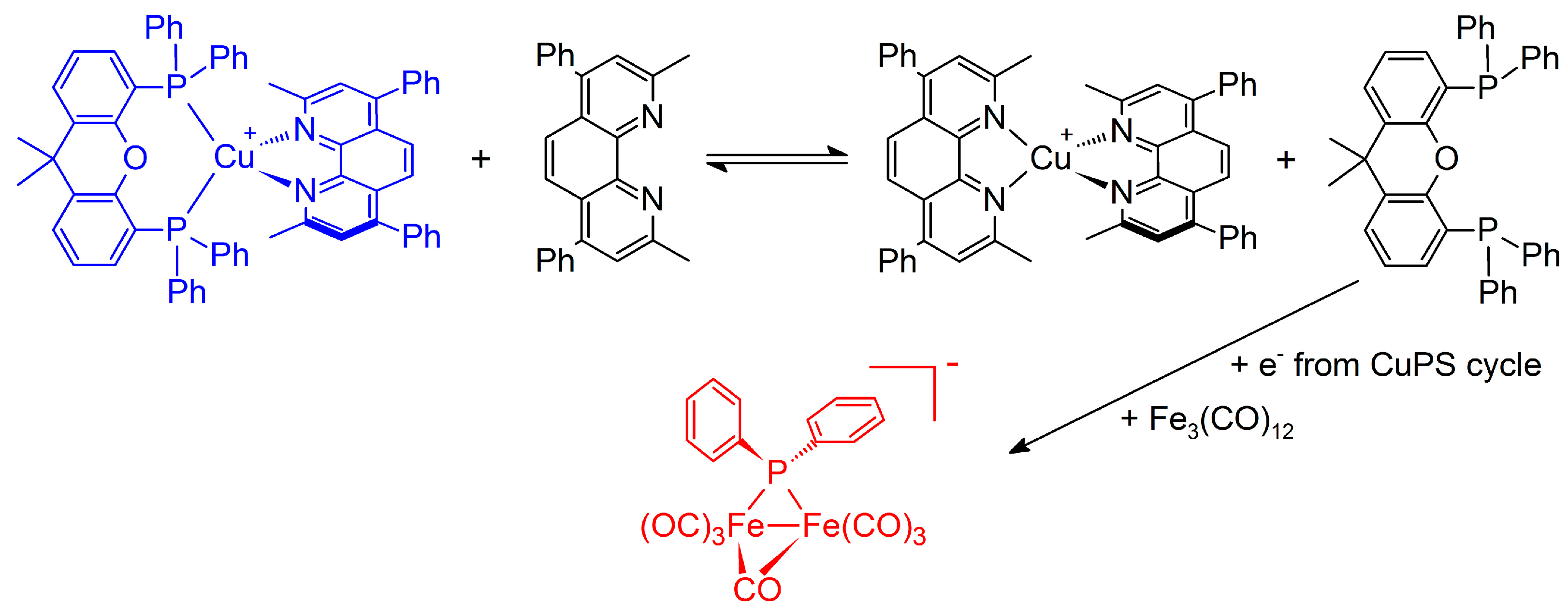
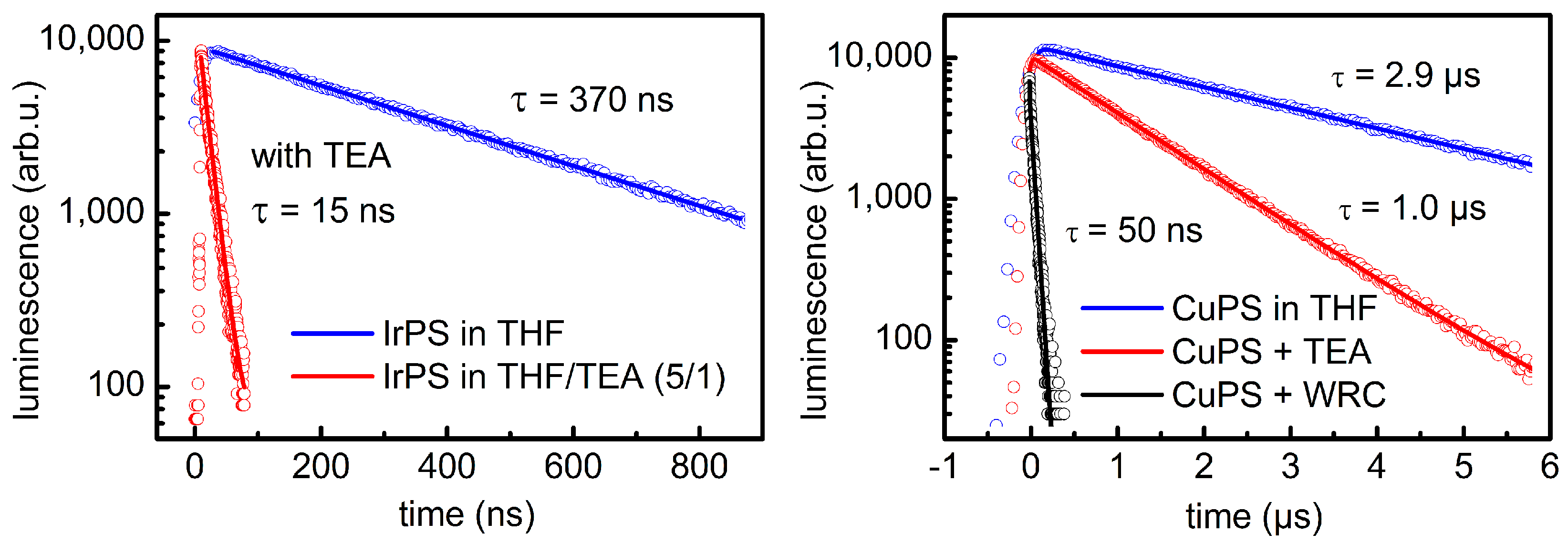
| Entry | WRC | PS | Conditions 1 | Light (tirr/h) | TONWRC | TONPS | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | 1 | A R = H | 10 mL DCM, 25 µM 1; 50 µM A; 5 mM thiosalicylic acid, 1 mM TFA | Xe, λ ≥ 400 nm (2.5 h) | 0.16 | - 2 | [105] |
| 2 | 2 | B | 5 mL ACN, 5 mL H2O, 10 µmol 2, 1 µmol B, 1 mmol ascorbic acid | Xe, λ ≥ 400 nm (3 h) | 4.3 | 86 | [61] |
| 3 | 2 | C | acetone/water (9:1 v/v), 0.05 M 2, 0.05 M C, 0.14 M TEA | Xe, λ ≥ 400 nm (8 h) | 466 | - 2 | [62] |
| 4 | 3 | B | 1.5 mL DMF/H2O (1:1), 14 µM 3, 140 µM B, 100 mM ascorbic acid (adjusted to pH 5.5 with 1 M NaOH) | 455–850 nm (2.5 h) | 200 | 20 | [23] |
| 5 | 4 | A R = H + OMe | 5 mL toluene, 5 µmol 4, 20 µmol A, 50 µmol [NiPr2EtH] [OAc] | Xe, λ ≥ 390 nm (80 min) | 5 | 4 | [64] |
| 6 | 5 | C | 10 mL THF/TEA/H2O (4:1:1 v/v/v), 18.5 µmol 5, 7.5 µmol C | Xe, λ ≥ 420 nm (output 1.5 W) (6 h) | 400 | 3035 | [20] |
| 7 | 5 | D R = Me | 10 mL THF/TEA/H2O (4:1:1 v/v/v), 5 µmol 5, 3.5 µmol D | Xe, λ ≥ 200 nm (output 1.5 W) (27 h) | 547 | 781 | [144] |
| 8 | 5 | D R = sBu | 10 mL THF/TEA/H2O (4:1:1 v/v/v), 5 µmol 5, 3.5 µmol D | Xe, λ ≥ 200 nm (output 1.5 W) (60 h) | 931 | 1330 | [145] |
| 9 | 6 | E | 20 mL THF/TEA/H2O (3:2:1, v/v/v), 6: 3.3 µmol [Fe] + 5.0 µmol L, 0.5 or 15.0 µmol E | Hg, λ = 440 nm (output 1.5 W) (20 h) | 2770 | 4550 | [21] |
| 10 | 7 | F X = Te | 10 mL H2O, 156 µM 7, 50 µM F, 85.2 mM ascorbic acid | Hg, λ ≥ 400 nm (18 h) | 505 | 79 | [158] |
| 11 | 8 | G | 10 mL EtOH/H2O (1:1, v/v), 0.1 mM 8, 1.0 mM G, 10% TEA | Xe, λ > 450 nm (15 h) | 226 | 59 | [106] |
| 12 | 9 | G | 10 mL H2O, 0.1 mM 9, 20 mM sodium dodecyl sulfate, 0.2 mM G, 10 vol % TEA | LED, λ = 455 nm (output 0.3 W) (4.5 h) | 117 | 58 | [107] |
| 13 | 10 | C | 10 mL acetone/H2O (9:1, v/v), 0.25–1.0 µM 10, 0.5 mM C, 0.6 M TEA | Xe, 400 < λ < 800 nm (8 h) | 22,200 | 44 | [65] |
| 14 | 11 | F X = Te | 10 mL H2O/MeOH (3:1, v/v), 1.0 µM 11, 0.684 µM F + 10 mg chitosan, 0.2 M ascorbic acid | LED, λ = 410 nm (60 h) | 52,800 | - 2 | [111] |
| 15 | 12 | F X = Se | 10 mL H2O, 1.0 µM 12, 5.2 µM F, 0.2 M ascorbic acid | LED, λ = 520 nm (12 h) | 26,500 | - 2 | [108] |
| 16 | 13 | D R = Me | 10 mL THF/TEA/H2O (4:3:1 v/v/v), 12 µmol 13, 12 µmol Et4NOH, 3.5 µmol D | Xe, λ ≥ 200 nm (output 1.5 W) (7 h) | 131 | 449 | [157] |
| 17 | 14 | F X = Se | 5 mL EtOH/H2O (1:1, v/v), 0.5 µM 14, 0.2 µM F (with 80 µM capping agent), 0.2 M ascorbic acid | LED, λ = 520 nm (output 0.15 W) (80 h) | 29,400 | - 2 | [109] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Junge, H.; Rockstroh, N.; Fischer, S.; Brückner, A.; Ludwig, R.; Lochbrunner, S.; Kühn, O.; Beller, M. Light to Hydrogen: Photocatalytic Hydrogen Generation from Water with Molecularly-Defined Iron Complexes. Inorganics 2017, 5, 14. https://doi.org/10.3390/inorganics5010014
Junge H, Rockstroh N, Fischer S, Brückner A, Ludwig R, Lochbrunner S, Kühn O, Beller M. Light to Hydrogen: Photocatalytic Hydrogen Generation from Water with Molecularly-Defined Iron Complexes. Inorganics. 2017; 5(1):14. https://doi.org/10.3390/inorganics5010014
Chicago/Turabian StyleJunge, Henrik, Nils Rockstroh, Steffen Fischer, Angelika Brückner, Ralf Ludwig, Stefan Lochbrunner, Oliver Kühn, and Matthias Beller. 2017. "Light to Hydrogen: Photocatalytic Hydrogen Generation from Water with Molecularly-Defined Iron Complexes" Inorganics 5, no. 1: 14. https://doi.org/10.3390/inorganics5010014







