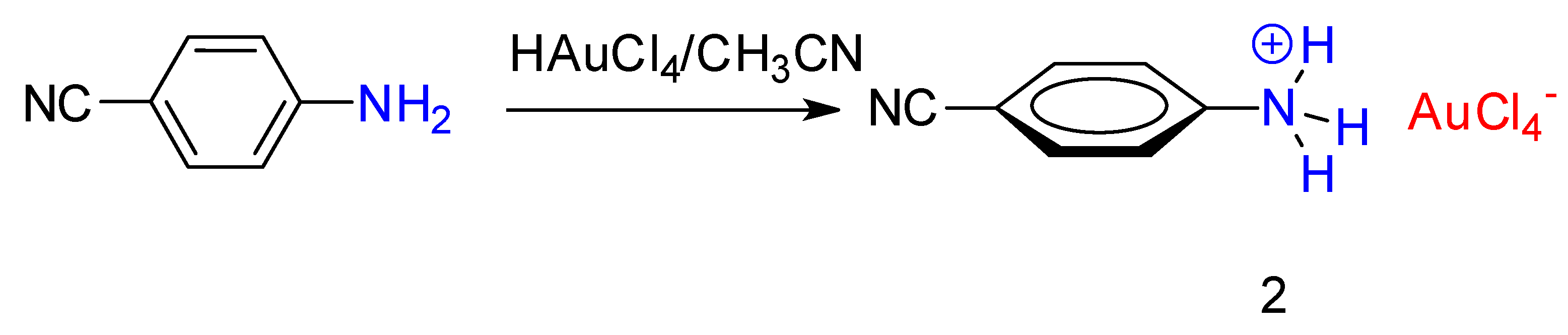
Synthesis of Diazonium Tetrachloroaurate(III) Precursors for Surface Grafting
Abstract
:1. Introduction
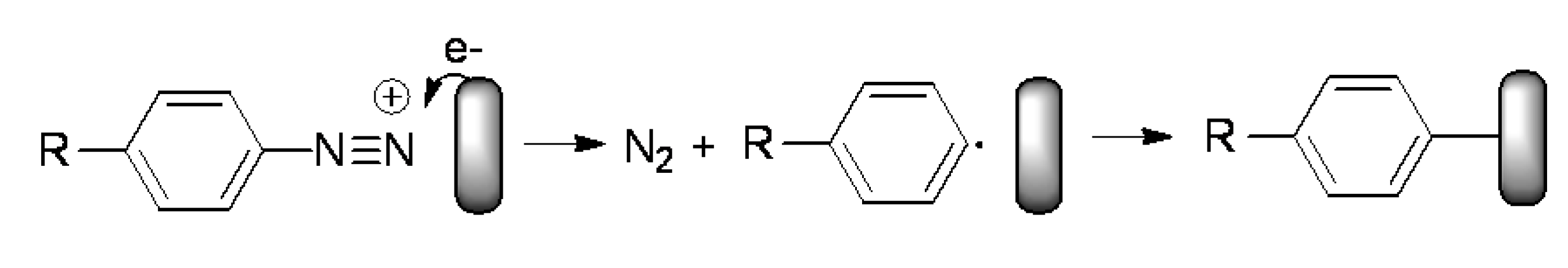
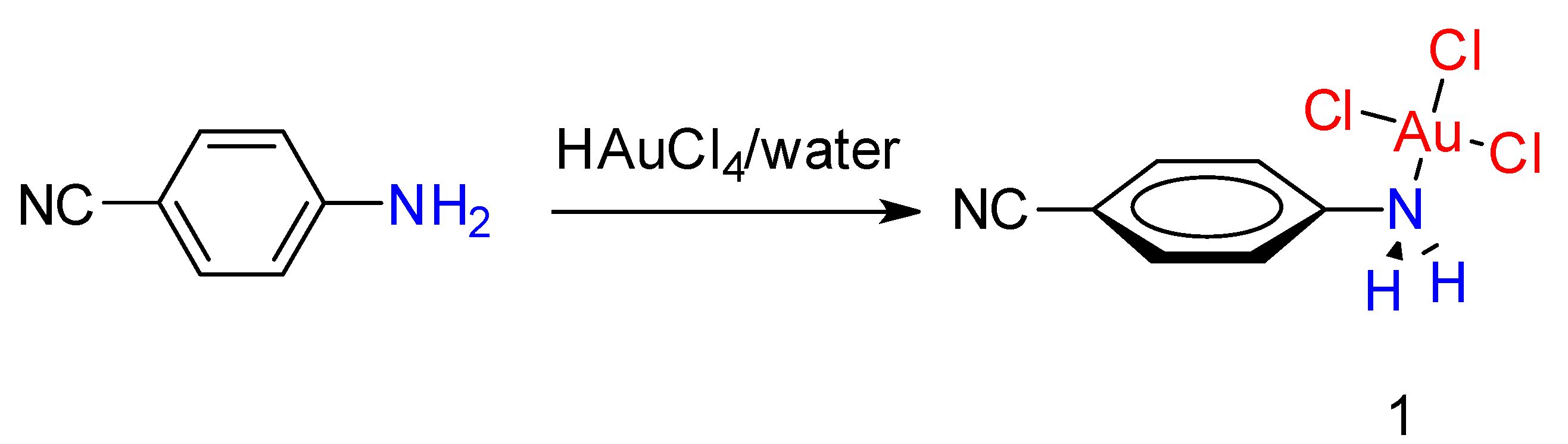
2. Results and Discussion
2.1. Synthesis and Characterization
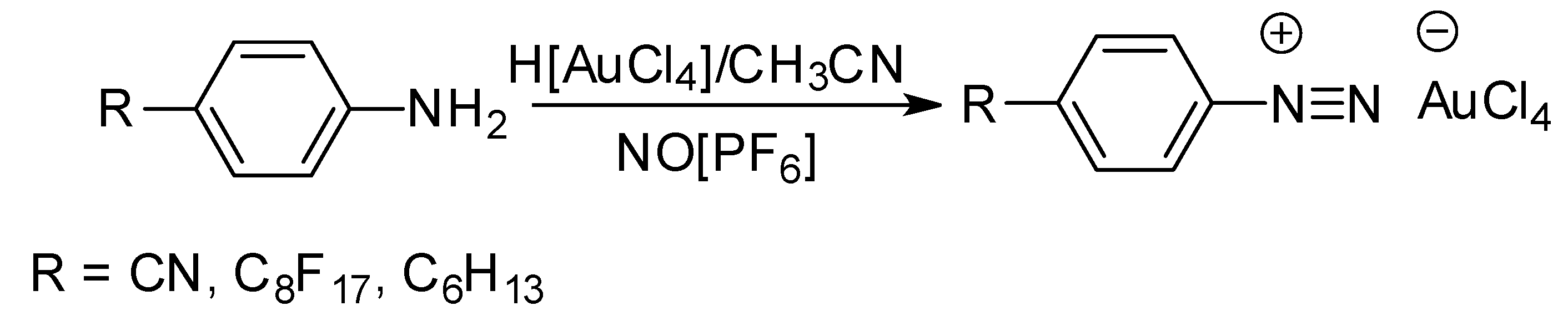

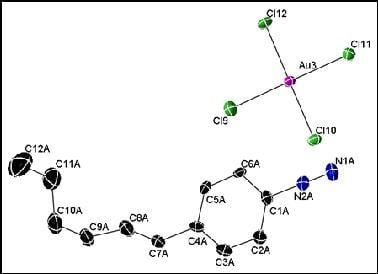
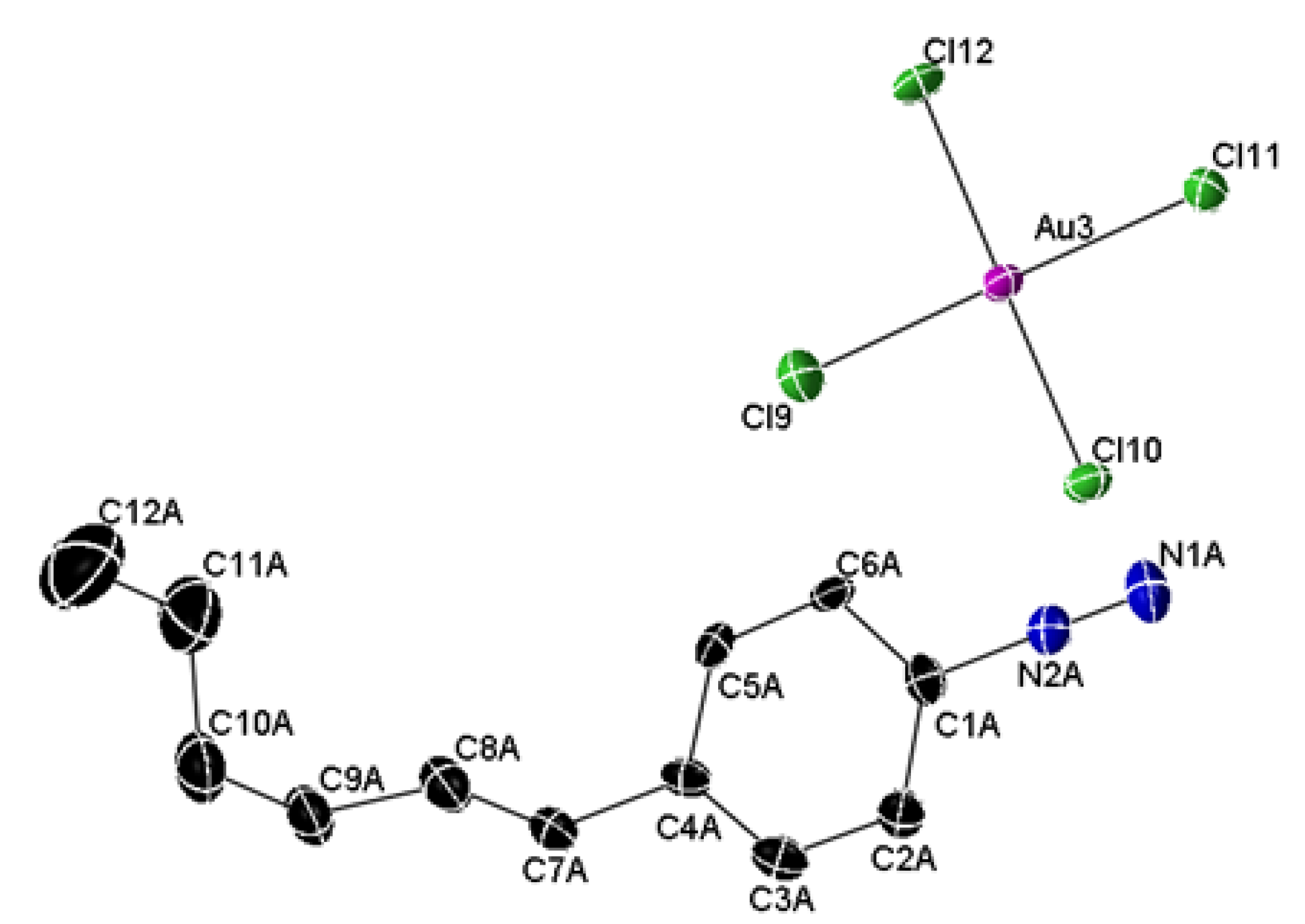
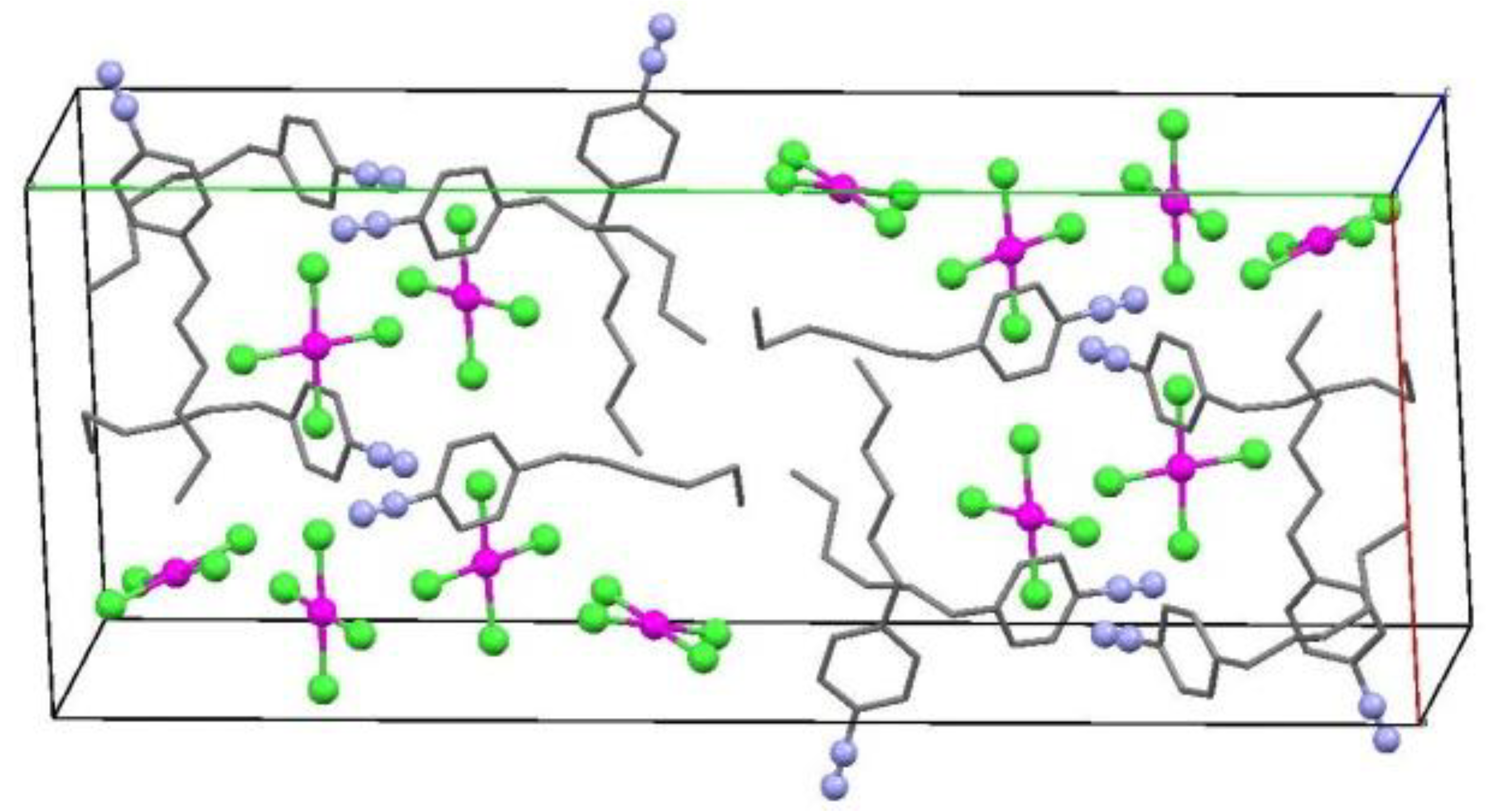
2.2. X-ray Structure
| Empirical formula | C12H17AuCl4N2 |
|---|---|
| Formula weight | 528.04 |
| Temperature, K | 173(2) |
| Wavelength, Å | 0.71073 |
| Crystal system | Monoclinic |
| Space group | P21/c |
| a (Å); ( ° ) | 15.5144(3) |
| b (Å); ( ° ) | 38.4270(8) 90.0250(10) |
| c (Å); ( ° ) | 8.7773(2) |
| Volume, Å3 | 5232.78(19) |
| Z | 12 |
| Density (cal.), Mg/m3 | 2.011 |
| Abs. coefficient, mm−1 | 21.395 |
| GOF on F2 | 1.061 |
| R1, wR2 [I > 2σ(I)] | 0.0568, 0.1530 |
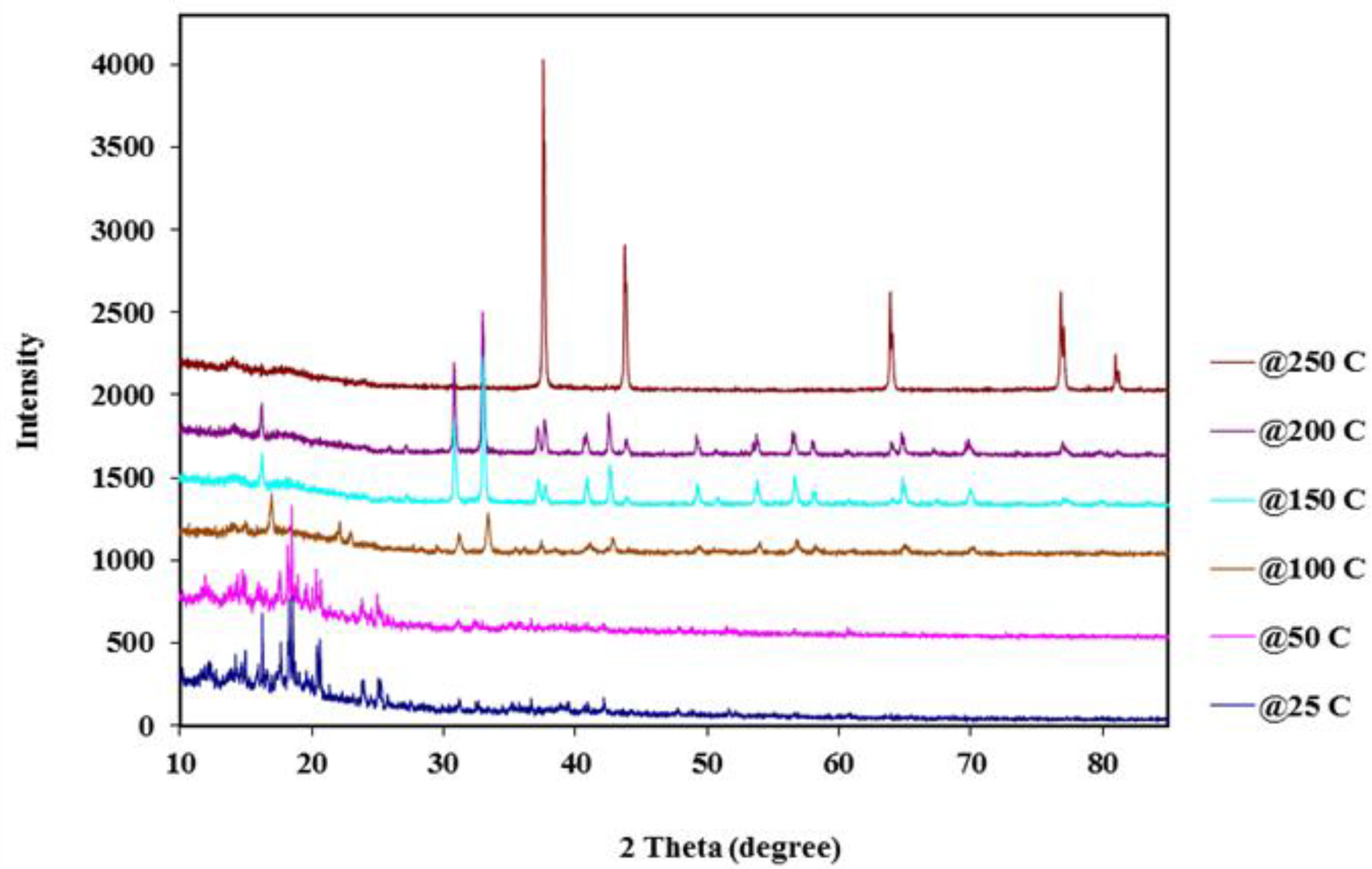
2.3. Thermal Stability and Residual Gas Analysis Studies
| Gas | Partial pressure | Gas | Partial pressure |
|---|---|---|---|
| N2 | 2.60 × 10−7 | Cl2 | 1.08 × 10−11 |
| CH3F | 9.10 × 10−1° | C6H5Cl | 6.47 × 10−12 |
| CH2Cl2 | 5.47 × 10−1° | C6H5F | 5.21 × 10−12 |
| CHCl3 | 2.90 × 10−11 | CCl4 | 4.29 × 10−12 |
| CF4 | 1.10 × 10−11 |



3. Experimental Section
3.1. General Procedures
3.2. Physical Measurements
3.3. Thermogravimetric Analysis and Residual Gas Analysis
3.4. X-ray Diffraction
3.5. Syntheses
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Chehimi, M.M. Aryl Diazonium Salts: New Coupling Agents in Polymer and Surface Science; Wiley-VCH: Weinheim, Germany, 2012. [Google Scholar]
- Sun, Z.; James, D.K.; Tour, J.M. Graphene Chemistry: Synthesis and Manipulation. J. Phys. Chem. Lett. 2011, 2, 2425–2432. [Google Scholar]
- Breton, T.; Bélanger, D. Modification of Carbon Electrode with Aryl Groups having an Aliphatic Amine by Electrochemical Reduction of in situ Generated Diazonium Cations. Langmuir 2008, 24, 8711–8718. [Google Scholar] [CrossRef] [Green Version]
- Allongue, P.; Delamar, M.; Desbat, B.; Fagebaume, O.; Hitmi, R.; Pinson, J.; Saveant, J.M. Covalent Modification of Carbon Surfaces by Aryl Radicals Generated from the Electrochemical Reduction of Diazonium Salts. J. Am. Chem. Soc. 1997, 119, 201–207. [Google Scholar] [CrossRef]
- Salmi, Z.; Lamouri, A.; Decorse, P.; Jouini, M.; Boussadi, A.; Achard, J.; Gicquel, A.; Mahouche-Chergui, S.; Carbonnier, B.; Chehimi, M.M. Grafting Polymer–Protein Bioconjugate to Boron-Doped Diamond using Aryl Diazonium Coupling Agents. Diamond Rel. Mater. 2013, 40, 60–80. [Google Scholar] [CrossRef]
- Lehr, J.; Garrett, D.J.; Paulik, M.G.; Flavel, B.S.; Brooksby, P.A.; Williamson, B.E.; Downard, A.J. Patterning of Metal, Carbon, and Semiconductor Substrates with Thin Organic Films by Microcontact Printing with Aryldiazonium Salt Inks. Anal. Chem. 2010, 82, 7027–7070. [Google Scholar] [CrossRef]
- Aswal, D.K.; Koiry, S.P.; Jousselme, B.; Gupta, S.K.; Palacin, S.; Yakhmi, J.V. Hybrid Molecule-on-Silicon Nanoelectronics: Electrochemical Processes for Grafting and Printing of Monolayers. Physica E 2009, 41, 325–344. [Google Scholar] [CrossRef]
- Gooding, J.J.; Ciampi, S. The Molecular Level Modification of Surfaces: From Self-Assembled Monolayers to Complex Molecular Assemblies. Chem. Soc. Rev. 2011, 40, 2704–2718. [Google Scholar]
- Bélanger, D.; Pinson, J. Electrografting: A Powerful Method for Surface Modification. Chem. Soc. Rev. 2011, 40, 3995–4048. [Google Scholar] [CrossRef]
- Combellas, C.; Delamar, M.; Kanoufi, F.; Pinson, J.; Podvorica, F.I. Spontaneous Grafting of Iron Surfaces by Reduction of Aryldiazonium Salts in Acidic or Neutral Aqueous Solution. Application to the Protection of Iron against Corrosion. Chem. Mater. 2005, 17, 3968–3975. [Google Scholar] [CrossRef]
- Zhu, Y.; Higginbotham, A.L.; Tour, J.M. Covalent Functionalization of Surfactant-Wrapped Graphene Nanoribbons. Chem. Mater. 2009, 21, 5284–5291. [Google Scholar] [CrossRef]
- Lomeda, J.R.; Doyle, C.D.; Kosynkin, D.V.; Hwang, W.-F.; Tour, J.M. Diazonium Functionalization of Surfactant-Wrapped Chemically Converted Graphene Sheets. J. Am. Chem. Soc. 2008, 130, 16201–16206. [Google Scholar] [CrossRef]
- Gehan, H.; Fillaud, L.; Felidj, N.; Aubard, J.; Lang, P.; Chehimi, M.M.; Mangeney, C.A. General Approach Combining Diazonium Salts and Click Chemistries for Gold Surface Functionalization by Nanoparticle Assemblies. Langmuir 2010, 26, 3975–3980. [Google Scholar]
- Mirkhalaf, F.; Paprotny, J.; Schiffrin, D.J. Synthesis of Metal Nanoparticles Stabilized by Metal-Carbon Bonds. J. Am. Chem. Soc. 2006, 128, 7400–7401. [Google Scholar] [CrossRef]
- Ghosh, D.; Chen, S.W. Palladium Nanoparticles Passivated by Metal–Carbon Covalent Linkages. J. Mater. Chem. 2008, 18, 755–762. [Google Scholar] [CrossRef]
- Ratheesh Kumar, V.K.; Gopidas, K.R. Synthesis and Characterization of Gold-Nanoparticle-Cored Dendrimers Stabilized by Metal–Carbon Bonds. Chem. Asian J. 2010, 5, 887–896. [Google Scholar] [CrossRef]
- Ratheesh Kumar, V.K.; Gopidas, K.R. Palladium Nanoparticle-Cored G1-Dendrimer Stabilized by Carbon-Pd Bonds: Synthesis, Characterization and use as Chemoselective, Room Temperature Hydrogenation Catalyst. Tetrahedron Lett. 2011, 52, 3102–3105. [Google Scholar] [CrossRef]
- Ratheesh Kumar, V.K.; Krishnakumar, S.; Gopidas, K.R. Synthesis, Characterization and Catalytic Applications of Palladium Nanoparticle-Cored Dendrimers Stabilized by Metal–Carbon Bonds. Eur. J. Org. Chem. 2012, 3447–3458. [Google Scholar]
- Laurentius, L.; Stoyanov, S.R.; Gusarov, S.; Kovalenko, A.; Du, R.; Lopinski, G.P.; McDermott, M.T. Diazonium-Derived Aryl Films on Gold Nanoparticles: Evidence for a Carbon-Gold Covalent Bond. ACS Nano 2011, 5, 4219–4227. [Google Scholar] [CrossRef]
- Adenier, A.; Bernard, M.C.; Chehimi, M.M.; Cabet-Deliry, E.; Desbat, B.; Fagebaume, O.; Pinson, J.; Podvorica, F. Covalent Modification of Iron Surfaces by Electrochemical Reduction of Aryldiazonium Salts. J. Am. Chem. Soc. 2001, 123, 4541–4549. [Google Scholar] [CrossRef]
- Nakamura, T.; Suzuki, M.; Ishihara, M.; Ohana, T.; Tanaka, A.; Koga, Y. Photochemical Modification of Diamond Films: Introduction of Perfluorooctyl Functional Groups on Their Surface. Langmuir 2004, 20, 5846–5849. [Google Scholar] [CrossRef]
- Harper, J.C.; Polsky, R.; Wheeler, D.R.; Brozik, S.M. Maleimide-Activated Aryl Diazonium Salts for Electrode Surface Functionalization with Biological and Redox Active Molecules. Langmuir 2008, 24, 2206–2211. [Google Scholar] [CrossRef]
- Corgier, B.P.; Marquette, C.A.; Blum, L.J. Diazonium-Protein Adducts for Graphite Electrode Microarrays Modification: Direct and Addressed Electrochemical Immobilization. J. Am. Chem. Soc. 2005, 127, 18328–18332. [Google Scholar] [CrossRef]
- Mévellec, V.; Roussel, S.; Tessier, L.; Chancolon, J.; Mayne-L’Hermite, M.; Deniau, G.; Viel, P.; Palacin, S. Grafting Polymers on Surfaces: A New Powerful and Versatile Diazonium Salt-Based One-Step Process in Aqueous Media. Chem. Mater. 2007, 19, 6323–6330. [Google Scholar] [CrossRef]
- Garrett, D.J.; Lehr, J.; Miskelly, G.M.; Downard, A.J. Microcontact Printing using the Spontaneous Reduction of Aryldiazonium Salts. J. Am. Chem. Soc. 2007, 129, 15456–15457. [Google Scholar] [CrossRef]
- Hossain, M.Z.; Walsh, M.A.; Hersam, M.C. Scanning Tunneling Microscopy, Spectroscopy, and Nanolithography of Epitaxial Graphene Chemically Modified with Aryl Moieties. J. Am. Chem. Soc. 2010, 132, 15399–15403. [Google Scholar] [CrossRef]
- Roglans, A.; Pla-Quintana, A.; Moreno-Manas, M. Diazonium Salts as Substrates in Palladium-Catalyzed Cross-Coupling Reactions. Chem. Rev. 2006, 106, 4622–4643. [Google Scholar] [CrossRef]
- Patai, S. The Chemistry of Diazonium and Diazo Groups. In The Chemistry of Functional Groups; John Wiley & Sons Inc: New York, NY, USA, 1978; Volume 2. [Google Scholar]
- Saunders, K.H. The Aromatic Diazo-Compounds and Their Technical Applications; Edward Arnold & Co.: London, UK, 1949. [Google Scholar]
- Zollinger, H. Diazotization of Amines and Dediazoniation of Diazonium Ions. In The Chemistry of Amino, Nitroso, Nitro and Related Groups; Patai, S., Ed.; Wiley & Sons: New York, NY, USA, 1996; pp. 636–637. [Google Scholar]
- Chamoulaud, G.; Bélanger, D. Spontaneous Derivatization of a Copper Electrode with in situ Generated Diazonium Cations in Aprotic and Aqueous Media. J. Phys. Chem. C 2007, 111, 7501–7507. [Google Scholar] [CrossRef]
- Glaser, R.; Horan, C.J. Benzenediazonium Ion. Generality, Consistency, and Preferability of the Electron Density Based Dative Bonding Model. J. Org. Chem. 1995, 60, 7518–7528. [Google Scholar] [CrossRef]
- Glaser, R.; Horan, C.J.; Lewis, M.; Zollinger, H. σ-Dative and π-Backdative Phenyl Cation–Dinitrogen Interactions and Opposing Sign Reaction Constants in Dual Substituent Parameter Relations. J. Org. Chem. 1999, 64, 902–913. [Google Scholar] [CrossRef]
- Gokel, G.W.; Cram, D.J. Molecular Complexation of Arenediazonium and Benzoyl Cations by Crown Ethers. J. Chem. Soc. Chem. Commun. 1973, 481–482. [Google Scholar] [CrossRef]
- Kuokkanen, T.; Haataja, A. Effect of Solvent on the Complexation and Thermal Stability of Benzenediazonium Tetrafluoroborate in the Presence of Crown Ethers. Acta Chem. Scand. 1993, 47, 872–876. [Google Scholar] [CrossRef]
- Morosin, B.; Lingafelter, E.C. The Crystal Structure of Tetramethylammonium Tetrachlorozincate and Tetrachlorocobaltate. Acta Crystallogr. 1959, 12, 611–612. [Google Scholar] [CrossRef]
- Doctorovich, F.; Escola, N.; Trápani, C.; Estrin, D.A.; Lebrero, M.C.G.; Turjanski, A.G. Stabilization of Aliphatic and Aromatic Diazonium Ions by Coordination: An Experimental and Theoretical Study. Organometallics 2000, 19, 3810–3817. [Google Scholar] [CrossRef]
- Mohamed, A.A. Advances in the Coordination Chemistry of Nitrogen Ligand Complexes of Coinage Metals. Coord. Chem. Rev. 2010, 254, 1918–1947. [Google Scholar] [CrossRef]
- Overton, A.T.; Mohamed, A.A. Gold Diazonium Complexes for Electrochemical Reductive Grafting. Inorg. Chem. 2012, 51, 5500–5502. [Google Scholar] [CrossRef]
- Gougoutas, J.Z.; Johnson, J. Structure and Solid-State Chemistry of 3-Carboxy-2-Naphthalenediazonium Bromide. J. Am. Chem. Soc. 1978, 100, 5816–5820. [Google Scholar] [CrossRef]
- Greenberg, B.; Okaya, Y. Crystal and Molecular Structure of 2-Diazonium-4-Phenolsulfonate Monohydrate, C6H3N2+.SO3-.OH.H2O. Acta Cryst. B 1969, 25, 2101–2108. [Google Scholar] [CrossRef]
- Mostad, A.; Rømming, C. The Crystal Structure of p-Benzenebisdiazonium Tetrachlorozincate. Acta Chem. Scand. 1968, 22, 1259–1266. [Google Scholar] [CrossRef]
- Romming, C. The Structure of Benzene Diazonium Chloride. Acta Chem. Scand. 1963, 17, 1444–1454. [Google Scholar] [CrossRef]
- Romming, C. The Crystal Structure of p-Benzenediazonium Sulphonate. Acta Chem. Scand. 1972, 26, 523–533. [Google Scholar] [CrossRef]
- Romming, C.; Tjornhom, T. The Crystal Structure of the 1:1 Complex Benzenediazonium Chloride-Acetic Acid. Acta Chem. Scand. 1968, 22, 2934–2942. [Google Scholar] [CrossRef]
- Filimonov, V.D.; Trusova, M.; Postnikov, P.; Krasnokutskaya, E.A.; Lee, Y.M.; Hwang, H.Y.; Kim, H.; Chi, K.-W. Unusually Stable, Versatile, and Pure Arenediazonium Tosylates: Their Preparation, Structures, and Synthetic Applicability. Org. Lett. 2008, 10, 3961–3964. [Google Scholar] [CrossRef]
- Pauling, L. The Nature of the Chemical Bond, 3rd ed.; Cornell University Press: Ithaca, NY, USA, 1960. [Google Scholar]
- Gremillion, A.F.; Jonassen, H.B.; O’Connor, R.J. The Thermal Stabilities and Infrared Spectra of some Metal Salt Stabilized Diazonium Salts. J. Am. Chem. Soc. 1959, 81, 6134–6138. [Google Scholar] [CrossRef]
- Savitsky, A.; Siggia, S. Analysis of Primary Aromatic Amines and Nitrite by Diazotization and Pyrolysis Gas Chromatography. Anal. Chem. 1974, 46, 149–152. [Google Scholar] [CrossRef]
- SMART, Version 4.043; Software for the CCD Detector System; Bruker Analytical X-ray Systems: Madison, WI, USA, 1995.
- SAINT, Version 4.035. Software for the CCD Detector System; Bruker Analytical X-ray Systems: Madison, WI, USA, 1995.
- Blessing, R.H. SADABS. Program for absorption corrections using Siemens CCD based on the method of Robert Blessing. Acta Cryst. A 1995, 51, 33. [Google Scholar] [CrossRef]
- Scheldrick, G.M. SHELXS-97; Program for the Solution of Crystal Structure; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- SHELXTL 5.03; Program Library for Structure Solution and Molecular Graphics; Bruker Analytical X-ray Systems: Madison, WI, USA, 1995.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Neal, S.N.; Orefuwa, S.A.; Overton, A.T.; Staples, R.J.; Mohamed, A.A. Synthesis of Diazonium Tetrachloroaurate(III) Precursors for Surface Grafting. Inorganics 2013, 1, 70-84. https://doi.org/10.3390/inorganics1010070
Neal SN, Orefuwa SA, Overton AT, Staples RJ, Mohamed AA. Synthesis of Diazonium Tetrachloroaurate(III) Precursors for Surface Grafting. Inorganics. 2013; 1(1):70-84. https://doi.org/10.3390/inorganics1010070
Chicago/Turabian StyleNeal, Sabine N., Samuel A. Orefuwa, Atiya T. Overton, Richard J. Staples, and Ahmed A. Mohamed. 2013. "Synthesis of Diazonium Tetrachloroaurate(III) Precursors for Surface Grafting" Inorganics 1, no. 1: 70-84. https://doi.org/10.3390/inorganics1010070