Finite Element Analysis in Setting of Fillings of V-Shaped Tooth Defects Made with Glass-Ionomer Cement and Flowable Composite
Abstract
:1. Introduction
2. Materials and Methods
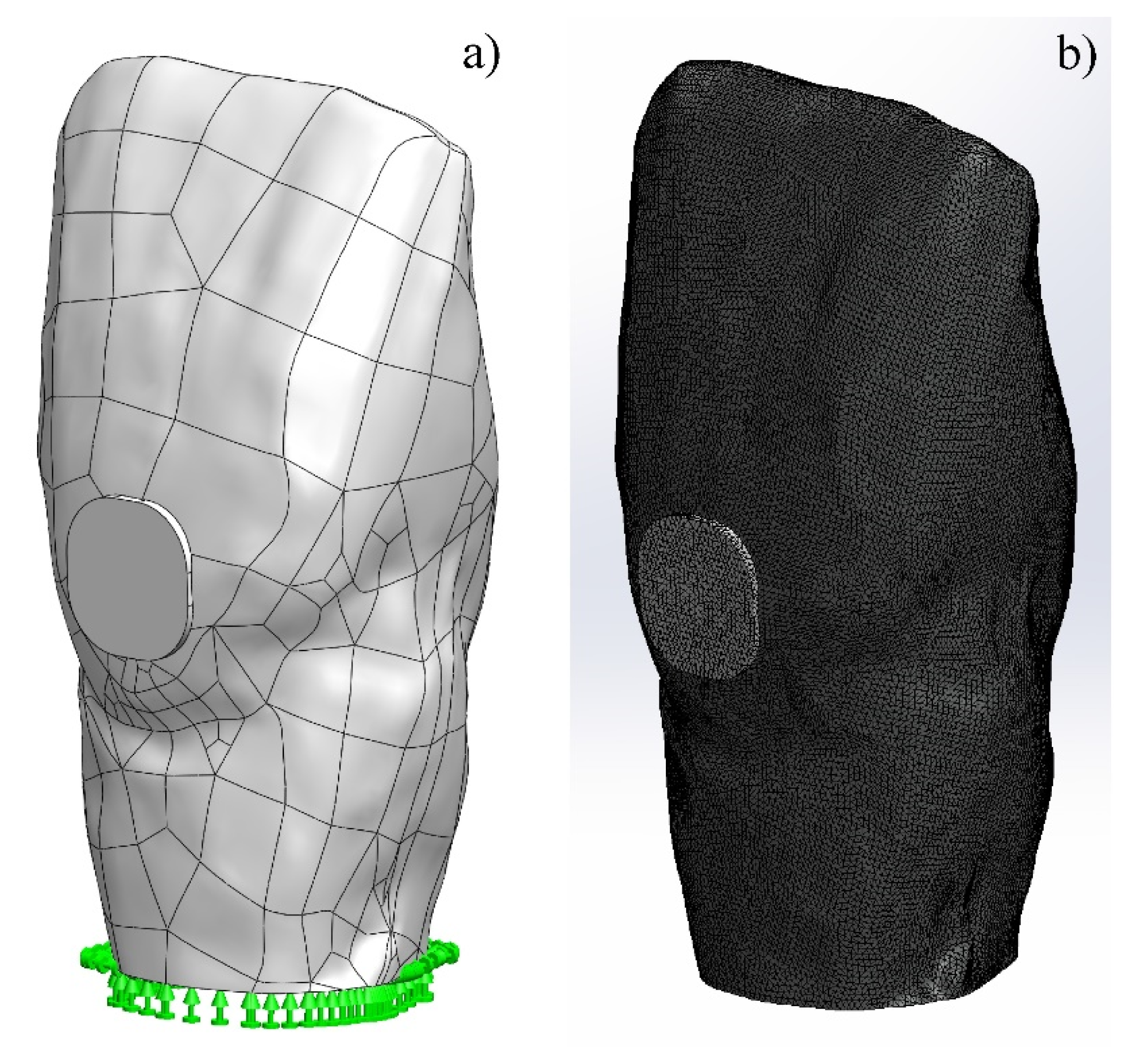
2.1. Numerical Modelling by FEA
2.2. In-Vitro Investigation of Micro-Leakage
3. Results
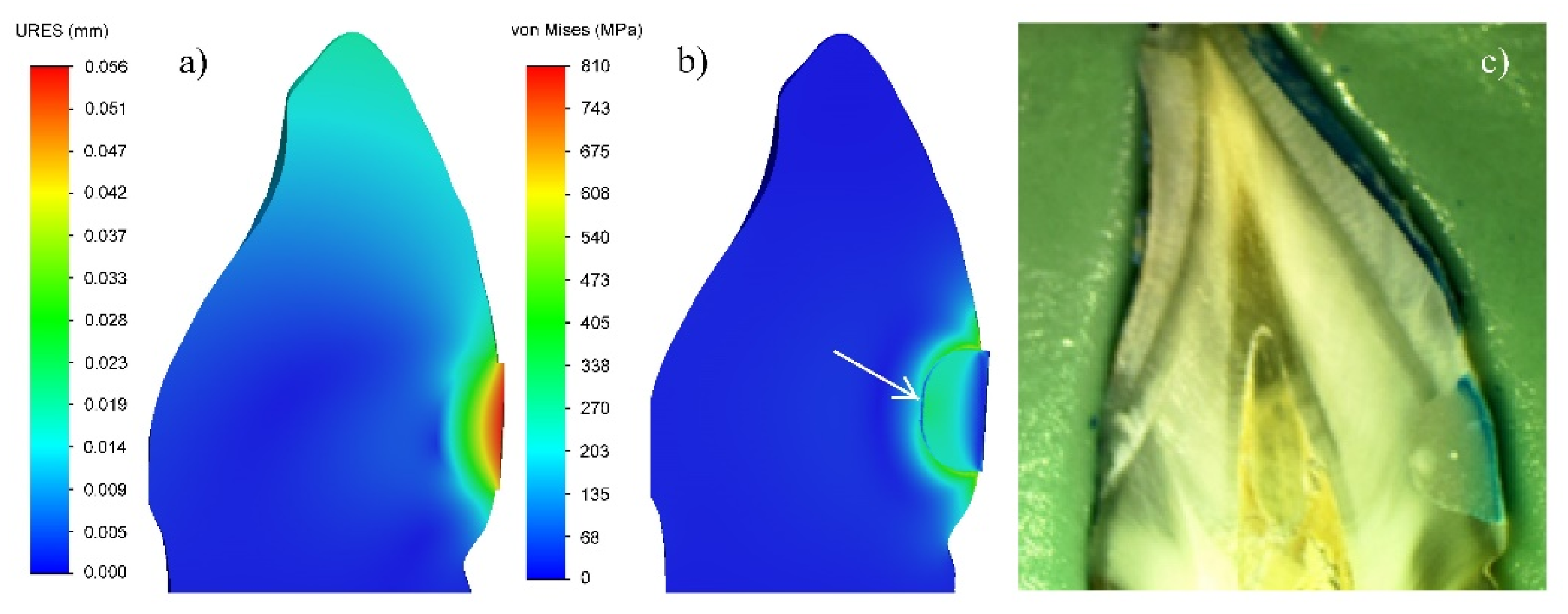
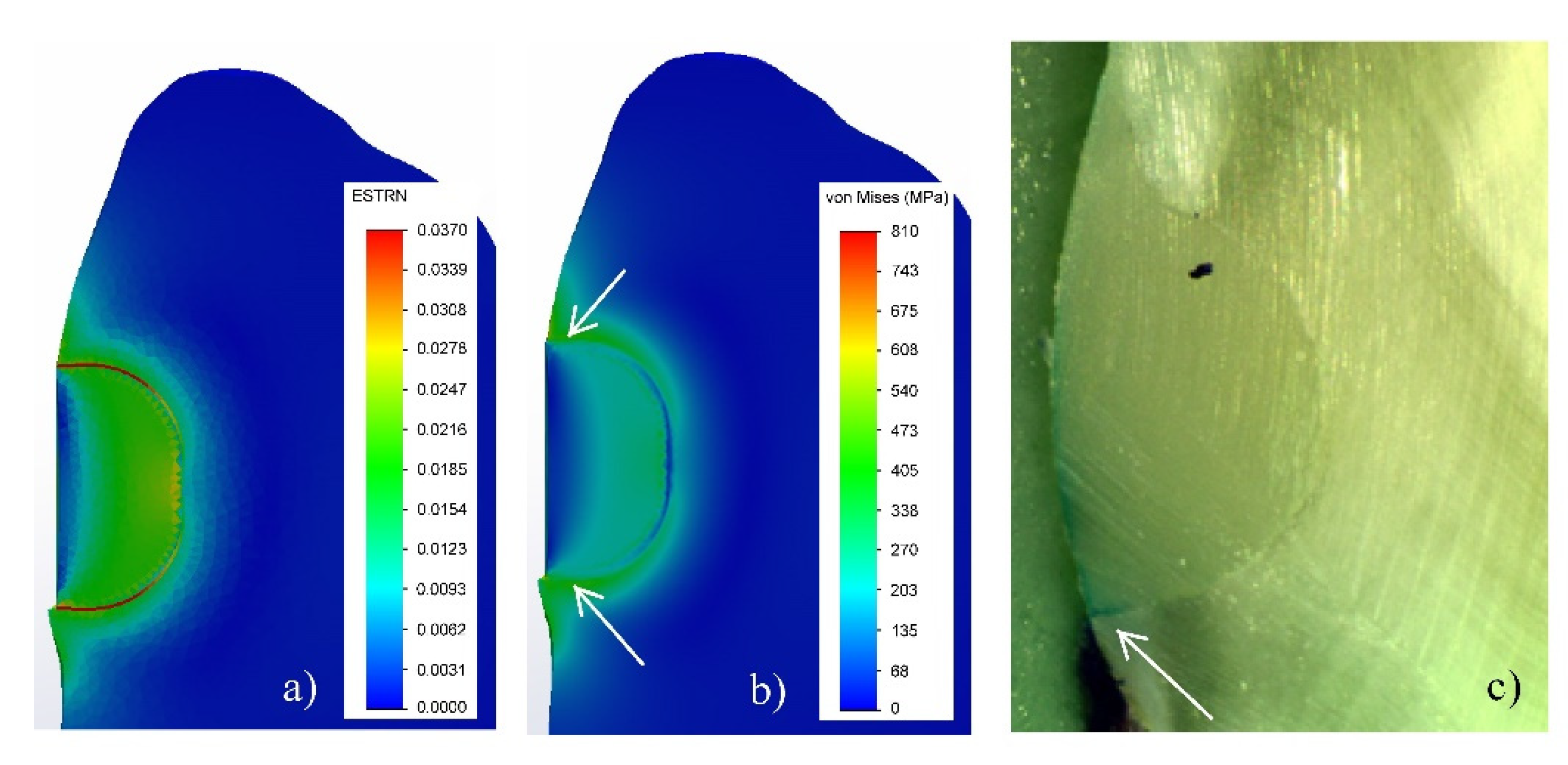
3.1. Numerical Modelling by FEA
3.2. In-Vitro Investigation of Micro-Leakage
4. Discussion
4.1. V-Shaped Defects Filled with GIC
4.2. V-Shaped Defects Filled with FPC
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ichim, I.P.; Schmidlin, P.R.; Li, Q.; Kieser, J.A.; Swain, M.V. Restoration of non-carious cervical lesions: Part II. Restorative material selection to minimize fracture. Dent. Mater. 2007, 23, 1562–1569. [Google Scholar] [CrossRef] [PubMed]
- Kemp-Scholte, C.M.; Davidson, C.L. Complete marginal seal of Class V resin composite restorations effected by increased flexibility. J. Dent. Res. 1990, 69, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Kemp-Scholte, C.M.; Davidson, C.L. Marginal integrity related to bond strength and strain capacity of composite resin restorative systems. J. Prosthet. Dent. 1990, 64, 658–664. [Google Scholar] [CrossRef]
- Li, Q.; Jepsen, S.; Albers, H.K.; Eberhard, J. Flowable materials as an intermediate layer could improve the marginal and internal adaptation of composite restorations in Class-V-cavities. Dent. Mater. 2006, 22, 250–257. [Google Scholar] [CrossRef] [PubMed]
- OäNAL, B.A.N.U.; Pamir, T. The two-year clinical performance of esthetic restorative materials in noncarious cervical lesions. J. Am. Dent. Assoc. 2005, 136, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Ichim, I.; Li, Q.; Loughran, J.; Swain, M.V.; Kieser, J. Restoration of non-carious cervical lesions: Part I. Modelling of restorative fracture. Dent. Mater. 2007, 23, 1553–1561. [Google Scholar] [CrossRef]
- Van Dijken, J.W. Retention of a resin-modified glass ionomer adhesive in non-carious cervical lesions. A 6-year follow-up. J. Dent. 2005, 33, 541–547. [Google Scholar] [CrossRef]
- Swift, E.J.; Pawlus, M.A.; Vargas, M.A. Shear bond strengths of resin-modified glass-ionomer restorative materials. Oper. Dent. 1995, 20, 138. [Google Scholar]
- Tyas, M.J.; Burrow, M.F. Clinical evaluation of a resin-modified glass ionomer adhesive system: Results at five years. Oper. Dent. 2002, 27, 438–441. [Google Scholar]
- Peumans, M.; Kanumilli, P.; De Munck, J.; Van Landuyt, K.; Lambrechts, P.; Van Meerbeek, B. Clinical effectiveness of contemporary adhesives: A systematic review of current clinical trials. Dent. Mater. 2005, 21, 864–881. [Google Scholar] [CrossRef]
- Chee, B.; Rickman, L.J.; Satterthwaite, J.D. Adhesives for the restoration of non-carious cervical lesions: A systematic review. J. Dent. 2012, 40, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Tuncer, D.; Yazici, A.R.; Özgünaltay, G.; Dayangac, B. Clinical evaluation of different adhesives used in the restoration of non-carious cervical lesions: 24-month results. Aust. Dent. J. 2013, 58, 94–100. [Google Scholar] [CrossRef] [PubMed]
- May, S.; Cieplik, F.; Hiller, K.A.; Buchalla, W.; Federlin, M.; Schmalz, G. Flowable composites for restoration of non-carious cervical lesions: Three-year results. Dent. Mater. 2017, 33, e136–e145. [Google Scholar] [CrossRef] [PubMed]
- Dikova, T.; Dzhendov, D.; Simov, M.; Katreva-Bozukova, I.; Angelova, S.; Pavlova, D.; Abadzhiev, M.; Tonchev, T. Modern trends in the development of the technologies for production of dental constructions. J. IMAB Annu. Proc. Sci. Pap. 2015, 21, 974–981. [Google Scholar] [CrossRef] [Green Version]
- Dikova, T.; Vasilev, T. Bending fracture of Co-Cr dental bridges, produced by additive technologies: Simulation analysis and test. Eng. Fract. Mech. 2019, 218, 106583. [Google Scholar] [CrossRef]
- Dikova, T.; Vasilev, T.; Dolgov, N. Failure of ceramic coatings on cast and selective laser melted Co-Cr dental alloys under tensile test: Experiment and finite element analysis. Eng. Fail. Anal. 2019, 105, 1045–1054. [Google Scholar] [CrossRef]
- AL-Gharrawi, H.A.; Saeed, M.W. Static stress analysis for three different types of composite materials experimentally and numerically. Int. J. Sci. Eng. Res. 2016, 7, 498–504. [Google Scholar]
- Miletic, V.; Peric, D.; Milosevic, M.; Manojlovic, D.; Mitrovic, N. Local deformation fields and marginal integrity of sculptable bulk-fill, low-shrinkage and conventional composites. Dent. Mater. 2016, 32, 1441–1451. [Google Scholar] [CrossRef]
- Rees, J.S.; Jacobsen, P.H. The effect of cuspal flexure on a buccal Class V restoration: A finite element study. J. Dent. 1998, 26, 361–367. [Google Scholar] [CrossRef]
- Tanaka, M.; Naito, M.; Yokota, M.; Kohno, M. Finite element analysis of the possible mechanism of cervical lesion formation by occlusal force. J. Oral Rehabil. 2003, 30, 60–67. [Google Scholar] [CrossRef]
- Papadogiannis, D.Y.; Lakes, R.S.; Papadogiannis, Y.; Palaghias, G.; Helvatjoglu-Antoniades, M. The effect of temperature on the viscoelastic properties of nano-hybrid composites. Dent. Mater. 2008, 24, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Aleksandrova, V. Biomechanical Problems in Direct Restoration of Class I and II Carious Defects in Patients with Bruxism [In Bulgarian]. Ph.D. Thesis, Medical University of Plovdiv, Plovdiv, Bulgaria, 2018. [Google Scholar]
- Petrovic, B.; Markovic, D.; Kojic, S.; Peric, T.; Dubourg, G.; Drijaca, M.; Stojanovic, G. Characterization of glass ionomer cements stored in various solution. Mater. Technol. 2019, 53, 285–293. [Google Scholar] [CrossRef]
- GC Fuji VIII GP; GC Corporation Japan; GC EUROPE N.V.; GC EEO-Bulgaria. Available online: https://cdn.gceurope.com/v1/PID/fuji8gp/leaflet/LFL_Fuji_VIII_GP_bg.pdf (accessed on 13 February 2020).
- Scientific Documentation Adhese® Universal, Ivoclar Vivadent AG, Liechtenstein, April. 2015, p. 58. Available online: https://dspconnect.s3.amazonaws.com/Ivoclar/Adhesive/AdheseUniversalScientificDocumentation.pdf (accessed on 20 March 2020).
- Technical Report, Estelite® Flow Quick and Estelite® Flow Quick High Flow, Tokuyama Dental Corporation Inc.: Japan. p. 23. Available online: http://www.tokuyama-dental.com/tdc/pdf/technicalreport/EFQ_and_EFQ_HF_TechnicalReport.pdf (accessed on 13 February 2020).
- Safty, S. Elastic and Viscoelastic Properties of Resin Composites at the Macroscopic and Nano Scales. Ph.D. Thesis, University of Manchester, Manchester, UK, 2012. [Google Scholar]
- Hirayama, S.; Iwai, H.; Tanimoto, Y. Mechanical evaluation of five flowable resin composites by the dynamic micro-indentation method. J. Dent. Biomech. 2014, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Anastasova, R.; Dikova, T.; Panov, V. In Vitro study of dental composite roughness and microleakage of repaired obturations by various techniques. J. IMAB Annu. Proc. Sci. Pap. 2019, 25, 2419–2425. [Google Scholar] [CrossRef]
- Anusavice, K.J.; Shen, C.; Rawls, H.R. Phillips’ Science of Dental Materials, 12th ed.; Elsevier Health Sciences, Elsevier Saunders: St. Louis, MO, USA, 2012; pp. 275–307. [Google Scholar]
- Van Meerbeek, B.; Willems, G.; Celis, J.P.; Roos, J.R.; Braem, M.; Lambrechts, P.; Vanherle, G. Assessment by nano-indentation of the hardness and elasticity of the resin-dentin bonding area. J. Dent. Res. 1993, 72, 1434–1442. [Google Scholar] [CrossRef]
- Senawongse, P.; Pongprueksa, P.; Tagami, J. The effect of the elastic modulus of low-viscosity resins on the microleakage of Class V resin composite restorations under occlusal loading. Dent. Mater. J. 2010, 29, 324–329. [Google Scholar] [CrossRef] [Green Version]




| Material/Tissue Type | Modulus of Elasticity, GPa | Poisson’s Ratio | Shrinkage during Setting, % | Adhesion Strength to Dentin, MPa |
|---|---|---|---|---|
| Dentin | 19 | 0.31 | - | - |
| GIC: FUJI VIII GP | 8.32 | 0.27 | 3 | 5.8 |
| Adhesive: Adhese Universal | 4.85 | 0.30 | - | 35 |
| FPC: Estelite Flow Quick - High Flow | 9.9 | 0.30 | 3 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dikova, T.; Vasilev, T.; Hristova, V.; Panov, V. Finite Element Analysis in Setting of Fillings of V-Shaped Tooth Defects Made with Glass-Ionomer Cement and Flowable Composite. Processes 2020, 8, 363. https://doi.org/10.3390/pr8030363
Dikova T, Vasilev T, Hristova V, Panov V. Finite Element Analysis in Setting of Fillings of V-Shaped Tooth Defects Made with Glass-Ionomer Cement and Flowable Composite. Processes. 2020; 8(3):363. https://doi.org/10.3390/pr8030363
Chicago/Turabian StyleDikova, Tsanka, Tihomir Vasilev, Vesela Hristova, and Vladimir Panov. 2020. "Finite Element Analysis in Setting of Fillings of V-Shaped Tooth Defects Made with Glass-Ionomer Cement and Flowable Composite" Processes 8, no. 3: 363. https://doi.org/10.3390/pr8030363
APA StyleDikova, T., Vasilev, T., Hristova, V., & Panov, V. (2020). Finite Element Analysis in Setting of Fillings of V-Shaped Tooth Defects Made with Glass-Ionomer Cement and Flowable Composite. Processes, 8(3), 363. https://doi.org/10.3390/pr8030363





