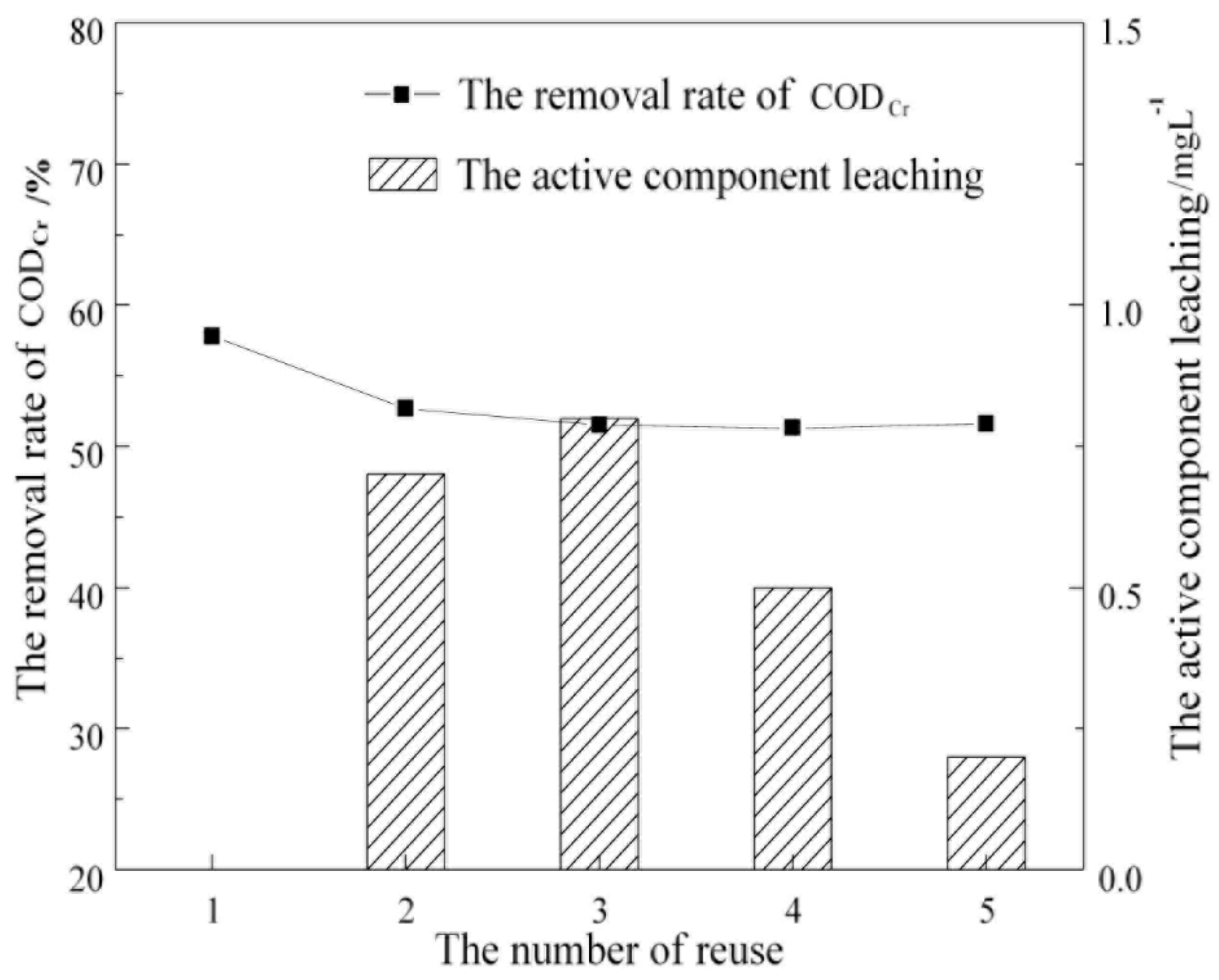
1. Introduction
Coalbed methane is an important source of natural gas that is produced during coal formation and found in coal seams in an adsorbed state [
1,
2]. Coalbed methane is generally produced through drainage and pressure lowering [
3]. In this process, coalbed methane-produced water is also produced. After undergoing water-rock and water-gas interactions, the quality and quantity of this type of water change greatly, yielding a complicated composition. It causes soil salinization and surface and ground water pollution if is discharged directly into the environment. And these pollutions cause an adverse impact on industrial and agricultural production as well as human health. Most coalbed methane reserves in China are concentrated in arid north and northwest regions. If coalbed methane-produced water in these regions can be appropriately treated and utilized for industrial purposes, irrigation, or drinking water sources, water shortage could be significantly alleviated.
As stated above, the compositions of the water produced from coalbed methane could be very complicated. Generally speaking, the major compositions are limited to the solved ions, including both cations and anions, and organics from the chemicals used in the methane extraction process, while the compositions of trace metals and isotopic chemicals are rarely reported. Specifically, the ions in the water produced from coalbed methane include Sodium (Na), bicarbonate (
), Calcium (Ca), sulfate (
), Magnesium (Mg) and other Total Dissolved Solids (TDS), depending on the geographical locations of the wells [
4].
The management and treatment of coalbed methane-produced water have been of great interest to engineers and scientists. So far, two typical options have been proposed so far. The first treatment method is disposal by reinjection. In the method, the produced water is injected to the deep underground, so that it will not bring direct side impacts to the environment and human beings on the ground. This option is only applicable for small amounts and for non-harmful water. By now, this option has been used in the San Juan Basin (New Mexico State) and the Piceance Basin (Colorado State) in the United States of America (USA) [
5]. The biggest advantage of this method is the low cost. The second treatment method is direct disposal to waterways. To achieve this, the produced water should be treated with various chemical and/or physical methods to meet the national and local regulations on water quality. Most of the Basins in the world tend to have effective methods to treat the produced water from coalbed methane [
6].
The major physical methods include reverse osmosis (RO), ion exchange (IE), microfiltration (MF), and ultrafiltration (UF) technologies. These technologies can effectively remove the ions in the produced water, so that the treated water can reach the standards for irritation and industrial applications. However, in most cases, when the organics are included in the produced water, physical methods may not be very effective. In this case, chemical methods can be used. Specifically, chemical absorption [
7] and oxidation [
8,
9,
10] were employed to treat the water produced from coalbed methane extraction. However, overuse of treatment agents, large investment in infrastructure, and complicated technological processes increase the cost of fracturing wastewater treatment [
11,
12,
13,
14]. Among the various oxidation techniques, heterogeneous Fenton method [
15] is one of the most popular one because of tis high efficiency, low cost, and easy operation. It has been widely used for the treatment of waste water from the printing and dyeing [
4,
16], pharmaceutical [
5,
17], and coal-chemical engineering [
18,
19] industries.
However, challenges still exist for the further application of the Fenton method for coalbed methane produced water.
Firstly, the generation and utilization rates of hydroxyl radical are not high [
20,
21]. The key to improve the efficiency of Fenton treatment in the process of wastewater treatment is to improve the production and utilization rates of hydroxyl radical in the system. However, based on a large number of experimental studies, the total amount of H
2O
2 required for complete mineralization of unit wastewater is much higher than the theoretical calculations, which shows that the generation and utilization rates of hydroxyl radicals are not high. More importantly than all of that, the low generation and utilization rates of hydroxyl radical have a negative impact on the Fenton reaction rate and pollutant removal efficiency.
Secondly, the strict reaction conditions easily lead to secondary pollution [
22,
23]. The Fenton reaction treatment of wastewater with high efficiency is achieved in reaction conditions that require accurate control of the reaction system pH value, and the dosage and proportion of H
2O
2 and Fe
2+, which limits the application of Fenton advanced oxidation technology to a considerable extent and increases the cost of wastewater treatment. Although hydroxylation of iron ions has a good coagulation effect, it is beneficial for the removal of pollutants. However, at the same time, it also contains Fe
3+ ions, which cause secondary pollution.
Finally, the cost of processing was too high [
24]. The reasons for the higher processing costs lie in two aspects: one is that the Fenton reagent costs more, and the other is that the limit of Fenton reaction conditions (pH, temperature, inorganic ions, etc.) increases the wastewater treatment cost.
Based on the reasons above, our research team has been working on the improvement of a Fenton reaction system to enhance the capacity and reduce the cost of coalbed methane water treatment. Fe/γ-Al
2O
3 and Fe-Co/γ-Al
2O
3 catalysts were synthesized and applied for coalbed methane water treatment in this paper. Compared to Fe/γ-Al
2O
3, Cobalt is added to Fe-Co/γ-Al
2O
3, which could potentially increase the Fenton reaction’s ability to convert a broad range of pollutants to less harmful products [
25,
26]. It is noted that Co/γ-Al
2O
3 catalyst was not synthesized for the experiment, as Fe is an indispensable part in Fenton reaction system [
27].
2. Experimental Materials and Methods
2.1. Main Reagents and Instruments
Deionized water was used in the experiment. The following chemicals were used: γ-Al2O3 (Zibo Superco Alumina Material Co. LTD, Zibo, China, 99.8%), ferrous sulfate (FeSO4•7H2O, Sigma Aldrich, St. Louis, MO, USA, >97%), cobalt sulfate (CoSO4•7H2O, Sigma Aldrich, 99.8% trace metals basis), urea (CO(NH2)2, Sigma Aldrich, >98%) sulfuric acid (H2SO4, Sigma Aldrich, >96%), hydrochloric acid (HCl, Sigma Aldrich, 37%), and 30% hydrogen peroxide (H2O2, Sigma Aldrich, 30% (w/w) in H2O). The following instruments were also used: electronic analytical balance, COD fast detector, thermostat shaker, water bath cauldron, constant-temperature drying oven, X-ray diffractometer (XRD), scanning electron microscope (SEM), surface area analyzer, X-ray fluorescence spectrometer, Fourier transform infrared spectrometer, X-ray photoelectron spectrometer, pH meter, and ICP-AES analyzer.
2.2. Catalyst Preparation
2.2.1. Homogeneous Precipitation Method
Supported catalysts are typically prepared using an ordinary impregnation method. In this method, active species enriched in or deposited in a carrier weakly and strongly interact with carriers simultaneously; as a result, active components are non-uniformly distributed. To solve this problem, researchers developed a homogeneous precipitation method by facilitating the interaction between a metal salt solution and an alkali. Thus, metal hydroxides or carbonates are precipitated in carrier particles. At low temperatures, urea [CO (NH
2)
2], as a weak base, does not hydrolyze. Above 90 °C, urea undergoes hydrolysis [Equation (1)]; in this reaction, OH
− is slowly released, thereby causing the active components to be uniformly distributed in the carrier particles.
2.2.2. Preparation of Carriers Supported on Fe-γ-Al2O3
γ-Al2O3 (2 g) was placed in a 300 mL glass conical flask, and deionized water (200 mL) was added. FeSO4•7H2O (0.596 g, 2.14 mmol) was added into the conical flask at the iron to carrier ratio of 6% (w/w) and mixed through ultrasonic vibration for 10 min. Urea (0.385 g, 6.42 mmol) was dissolved in the resulting suspension to ensure the molar ratio of urea to Fe2+ is 3:1. Afterward, the mixture was placed in a shaker with a constant temperature of 20 °C and shaken at 150 r/min for 30 min to completely dissolve the urea and evenly mix the suspension. The suspension was heated in a water bath at 95 °C and mechanically stirred at 200 r/min for 2 h to complete the precipitation. The suspension was stirred and then left untouched to promote aging for 3 h; afterward, the suspension was filtered through a 0.45 μm membrane. The precipitate was obtained and alternately washed with deionized water and ethanol several times until no sulfate ions were detected using BaCl2 solution. The product was finally formed, dried at 105 °C for 1 h, and baked in a muffle furnace at 300 °C for 4 h.
2.2.3. Preparation of the Co-Doped Fe-γ-Al2O3 Carrier
It is known that the pH values of the hydroxide precipitate of Co and Fe are similar; therefore, these elements can co-precipitate on a carrier surface. Co-doped Fe-γ-Al2O3 carrier was prepared, as in Step (1). Iron and cobalt mixture was used as a precursor for the metal salt solution. FeSO4•7H2O (0.298 g, 1.07 mmol) was added at the iron to carrier ratio of 3% (w/w), and CoSO4•7H2O (0.286 g, 1.07 mmol) was added to obtain a Fe2+ to Co2+ molar ratio of 1:1. Urea (0.385 g, 6.42 mmol) was dissolved in the resulting suspension to ensure the molar ratio of urea to (Fe2+ + Co2+) is 3:1.
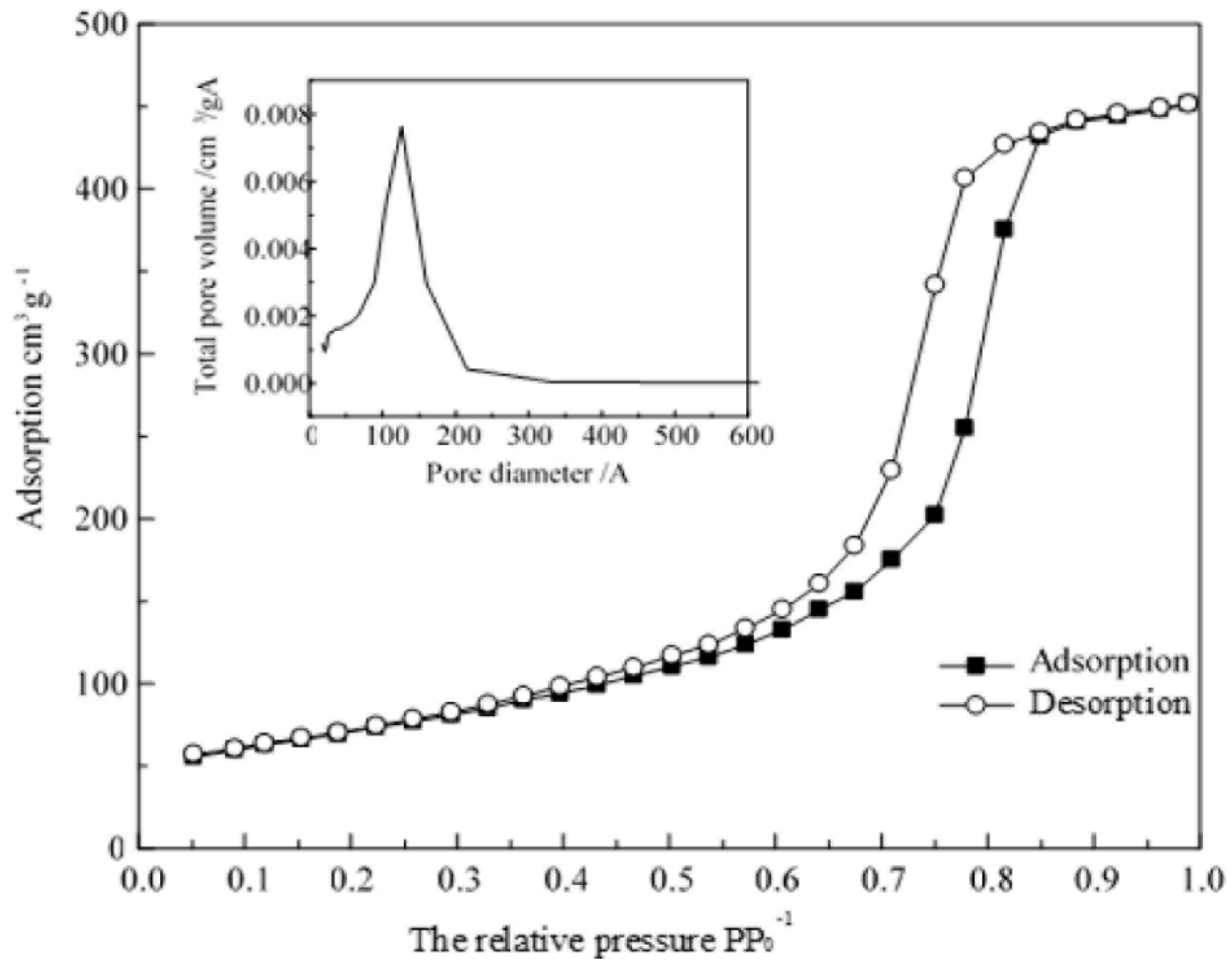
2.3. Catalyst Characterization Instruments
An X-ray diffractometer was used to analyze the crystal structure; a scanning electron microscope was used to observe particle morphological characteristics, particle size, and agglomeration; a specific surface area analyzer was used to determine the specific composite surface.
SEM test: An FEI QuantaTM 250 scanning electron microscope (USA) was used in high vacuum mode with magnification of 6× to 1,000,000× and acceleration voltage of 0.2 kV to 30 kV.
EDS test: A Bruker QUANTAX400-10 X-ray detector with electric cooling (Germany) was used. This test was performed under the following conditions: effective detection area of detection chip, 10 mm2; energy resolution, MnKa resolution superior to 129 eV (1000 cps to 100,000 cps testing condition); maximum input count, 1,000,000 cps; maximum output count, 400,000 cps; window type, ultrathin light element detection window; element detection range, Be (4)-Am (95), which can support a pixel resolution of 4096 × 3072; and mapping system stability, input count (peak-to-peak) offset within 1,000,000 cps not exceeding 1 eV.
XRD test: A Bruker D8 Advance XRD (Germany) was used. The specific parameters are as follows: X-ray tube with a voltage of 40 kV and current of 30 mA; Cu target as anode target material with Ka radiation and a goniometer radius of 250 mm; Slit systems, including a divergence slit (DS) of 0.6 mm and anti-scatter slit (SS) of 8 mm; Ni filter to filter Cu-Kβ ray with a detector opening of 2.82°; Soller slit of 2.5° between an incident side and a diffraction side with a sampling interval of 0.019450 (step) and a measuring range (2θ) of 3° to 90°; and Linx array detector.
XPS test: A Thermo Fisher ESCALAB 250Xi X-ray photoelectron spectrometer (USA) was used.
BET test: A Micromeritics ASAP 2460 surface area and porosimetry analyzer (USA) was used with N2 as an adsorbate at 77.4 K. Before the measurements were performed, the samples were subjected to vacuum pumping at 373 K for more than 10 h. The specific surface area and pore size distributions of the samples were obtained using BET and BJH methods.
XRF test: A Bruker S8 TIGER X-ray fluorescence spectrometer (Germany) was used. Samples were crushed to 200-mesh (74 μm) or less and dried at 105 °C for 2 h. Scanning was performed at a sequential mode with power of 4 kW, maximum voltage of 60 kV, and maximum current of 170 mA.
FT-IR test: A Bruker Vertex 80v FTIR instrument and a micro-infrared system (Germany) were used. A sample was dried, crushed to 200-mesh or less, and transformed into a KBr tablet. Scanning was performed at a spectral range of 4000 cm−1 to 400 cm−1, a resolution of 0.06 cm−1, and a beam diameter of 40 mm.
2.4. Test Methods
- (1)
Heterogeneous catalysis testing: The effluent (200 mL) after coagulation pretreatment was collected and placed in a 350 mL conical flask. A specific amount of catalyst and H2O2 was added to adjust the initial pH. At 25 °C, the sample was oscillated in a thermostat shaker at 150 rpm. After a predetermined reaction time was reached, 10 mL of solution was filtered with a 0.45 μm membrane. The pH of the filtered water was adjusted to >10. After the effect of H2O2 residue was removed, CODCr was determined. Using this method, we determined the effects of different catalyst dosage, H2O2 dosage, and initial pH values, as well as iron dissolution on the treatment of the effluent from coagulation pretreatment.
- (2)
Heterogeneous/homogeneous catalysis comparison testing: Heterogeneous/homogeneous catalysis comparison testing was conducted in two conical flasks (350 mL) with 200 mL effluent. The temperature was 25 °C, H2O2 concentration was 40 mmol/L, the initial pH was 5.1. In one flask, 0.8 g of Fe-γ-Al2O3 added in to the solution for heterogeneous Fenton reaction. In the other flask, FeSO4•7H2O (0.856 mmol) was added to the solution for homogenous Fenton reaction. The molar ratio of Fe2+ in two flasks were controlled to be 1:1. The samples were oscillated in a thermostat shaker at 150 rpm. After a predetermined reaction time was reached, 10 mL of solution was filtered with a 0.45 μm membrane. The pH of the filtered water was adjusted to >10. After the effect of H2O2 residue was removed, CODCr was determined.
5. Reaction Kinetics
Coalbed methane-produced water in the backflow stage contains a complex composition; therefore, the degradation mechanism via Fenton reaction is also complicated. Organic compounds in coalbed methane-produced water are rich in organic matter, and reaction mechanisms in the Fenton treatment of coalbed methane-produced water are also complex. Thus, the elementary reaction is not easily determined. It is not necessary to clarify the reaction process of each element from the point of water treatment and reuse; we only need to determine the reaction kinetics of CODCr degradation in coalbed methane-produced water during Fenton catalytic oxidation.
In a heterogeneous Fenton system, organic compounds can be directly oxidized by H
2O
2 and HO• generated through H
2O
2 decomposition. This reaction is shown in Equations (8) and (9).
The COD
Cr degradation rate can be expressed as follows:
where [COD
Cr] is the COD
Cr concentration at time
t, mg L
−1;
t is the reaction time, s; [H
2O
2] is the mass fraction of H
2O
2 at time
t, mg L
−1; [HO•] is the mass concentration of HO• at time
t, mg L
−1;
is the reaction rate constant of H
2O
2 degradation of COD
Cr, L (mol s)
−1; and [HO•] is the reaction rate constant of HO• degradation of COD
Cr, L (mol s)
−1.
Preliminary experiments indicated that H
2O
2 alone elicits a negligible degradation effect on COD
Cr in coalbed methane-produced water in backflow stage; therefore, Equation (10) can be simplified as follows:
If kHO•[HO•] is considered as k, then k is the apparent reaction rate constant of a heterogeneous Fenton reaction that degrades CODCr.
By integrating Equation (11),
Based on the apparent reaction kinetics, ln ([CODCr]/[CODCr]0) was plotted against t, followed by linear fitting. Thus, the apparent reaction rate equation of the heterogeneous Fenton reaction of CODCr degradation was obtained.
In our earlier experiment, CODCr degradation completely occurred in 75 min in coalbed methane-produced water via heterogeneous Fenton reaction. Thus, for the following discussion of kinetics, the data from the reaction at 75 min were used.
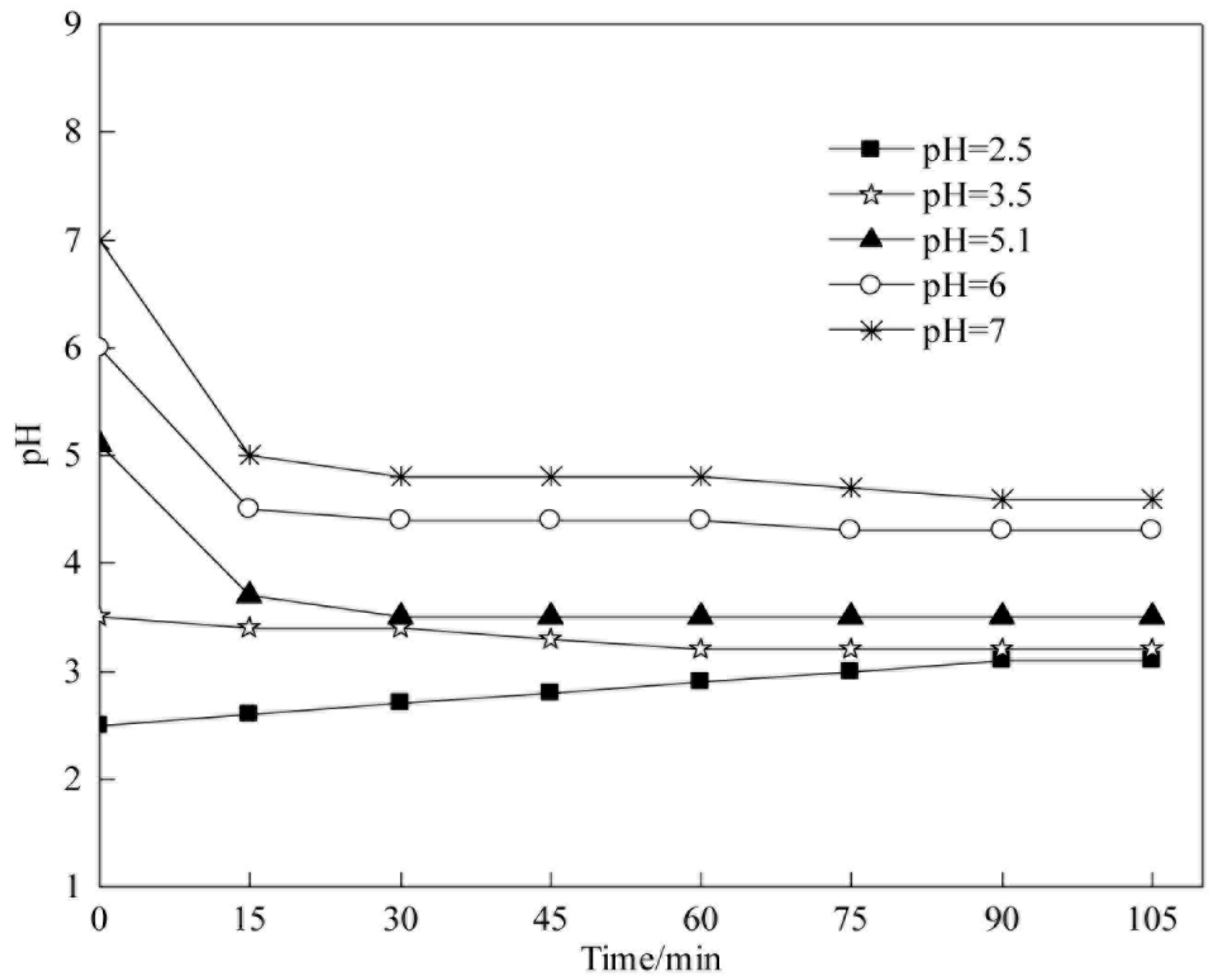
5.1. Degradation Kinetics of CODCr in Coalbed Methane-Produced Water with Different Initial pH Values
In this section, we discuss the influence of initial pH values on the degradation kinetics of COD
Cr via heterogeneous Fenton reaction. The experimental conditions were as follows: 25 °C, H
2O
2 concentration of 40 mmol L
−1, catalyst concentration of 4 g L
−1, initial pH of 2.5, 3.5, 5.1 (raw water), 6, and 7. The fitting results are shown in
Figure 13 and
Table 5.
Figure 13 and
Table 5 show that COD
Cr degradation in coalbed methane-produced water in the experimental range of initial pH (2.5 to 7) is in accordance with pseudo-first-order kinetics. The apparent reaction rate constants of COD
Cr degradation are −0.0133, −0.0172, −0.0113, −0.005, and −0.003 min
−1 when initial pH values are 2.5, 3.5, 5.1, 6, and 7, respectively.
The initial pH is necessary to stimulate Fenton reaction; an acidic condition is favorable for HO• generation. As the initial pH decreases, the apparent reaction rate constant increases. When the pH is very low, active components may on the catalyst surface exist as M-OH
2− (M is a metal ion), which adversely affects HO• production [
61]; therefore, the apparent reaction rate constant decreases. The reaction proceeds at the fastest speed when the initial pH is 3.5, and the apparent reaction rate constant is −0.0172 min
−1.
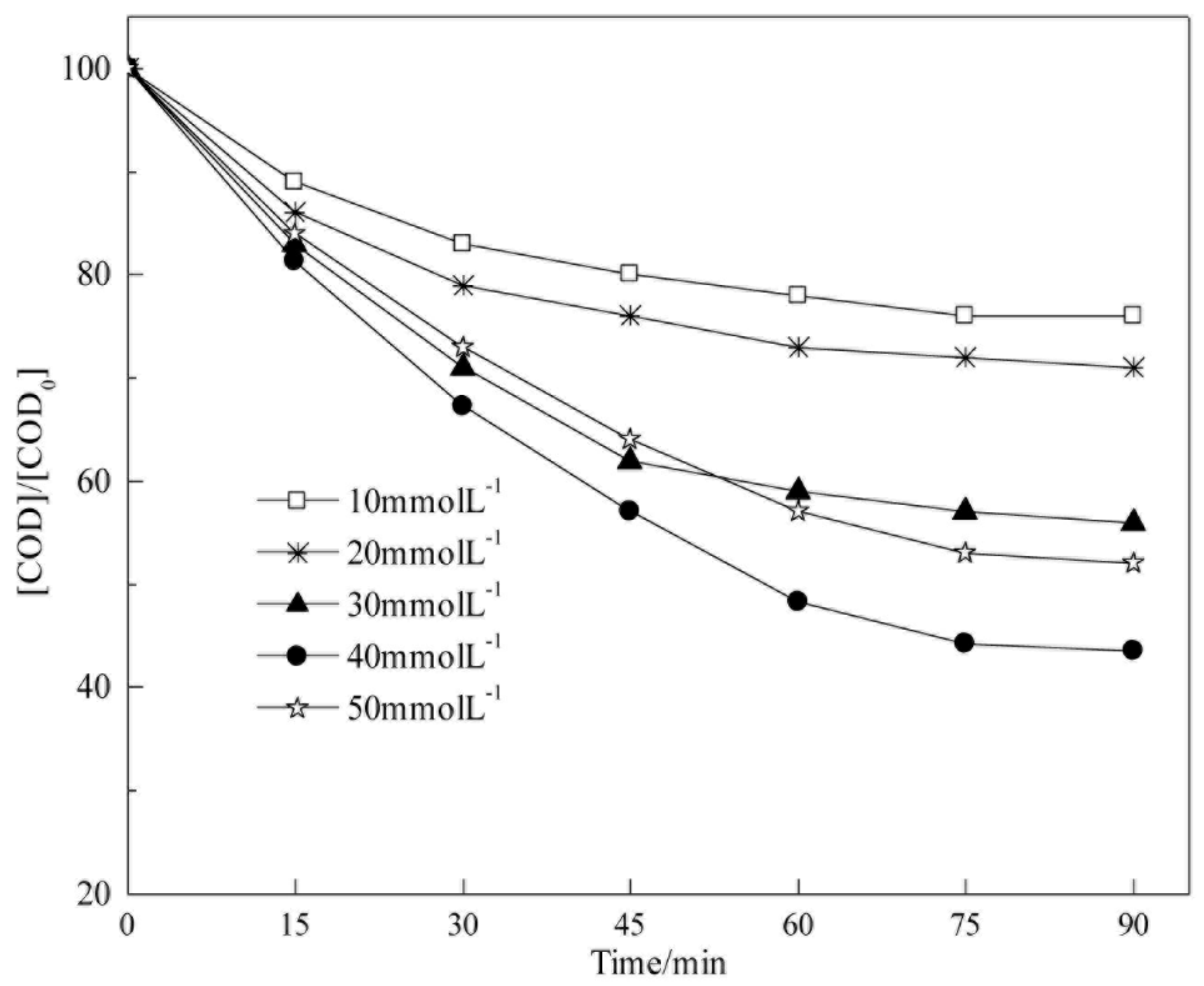
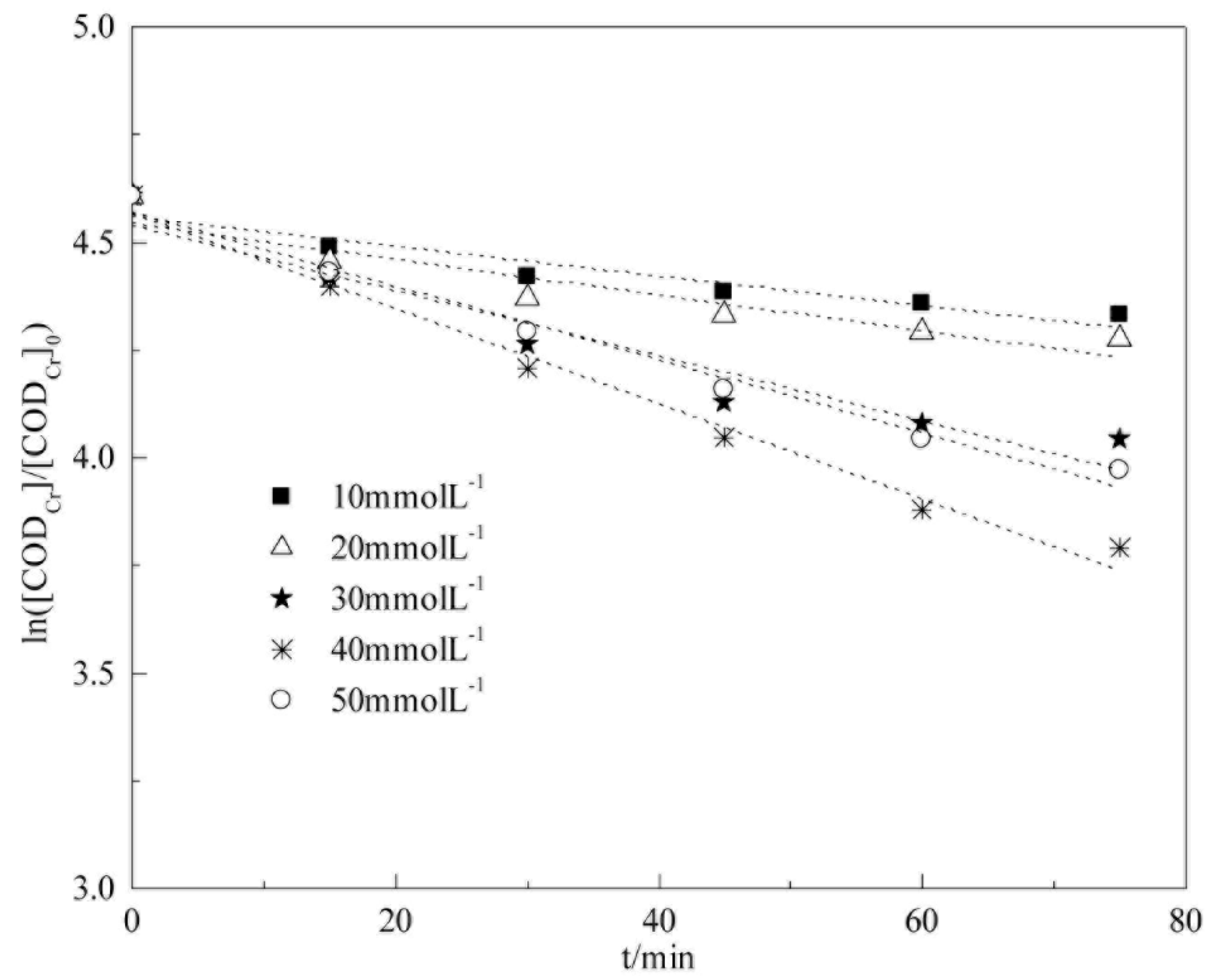
5.2. Analysis of Degradation Kinetics of CODCr in Coalbed Methane-Produced Water with Different H2O2 Concentrations
We examine the influence of H
2O
2 concentration on COD
Cr degradation kinetics via heterogeneous Fenton reaction in this section. The experimental conditions were as follows: 25 °C, initial pH of 5.1 (raw water), catalyst dosage of 4 g L
−1, H
2O
2 concentrations of 10, 20, 30, 40, and 50 mmol L
−1, respectively. The fitting results are shown in
Figure 14 and
Table 6.
Figure 14 and
Table 6 show that COD
Cr degradation in coalbed methane-produced water is in accordance with pseudo-first-order kinetics in the experimental range of H
2O
2 concentration (10 mmol L
−1 to 50 mmol L
−1). The apparent reaction rate constants of COD
Cr degradation are −0.0034, −0.0041, −0.0076, −0.0111, and −0.0085 min
−1 when H
2O
2 concentrations are 10, 20, 30, 40, and 50 mmol L
−1, respectively. The H
2O
2 concentration is directly related to the HO• yield. As the H
2O
2 concentration increases, the apparent reaction rate constant increases; however, H
2O
2 becomes a scavenging agent of HO• when the H
2O
2 concentration is very high; therefore, the apparent reaction rate constant decreases. The reaction reaches the largest value when the H
2O
2 concentration is 40 mmol L
−1, and the apparent reaction rate constant is −0.0111 min
−1.
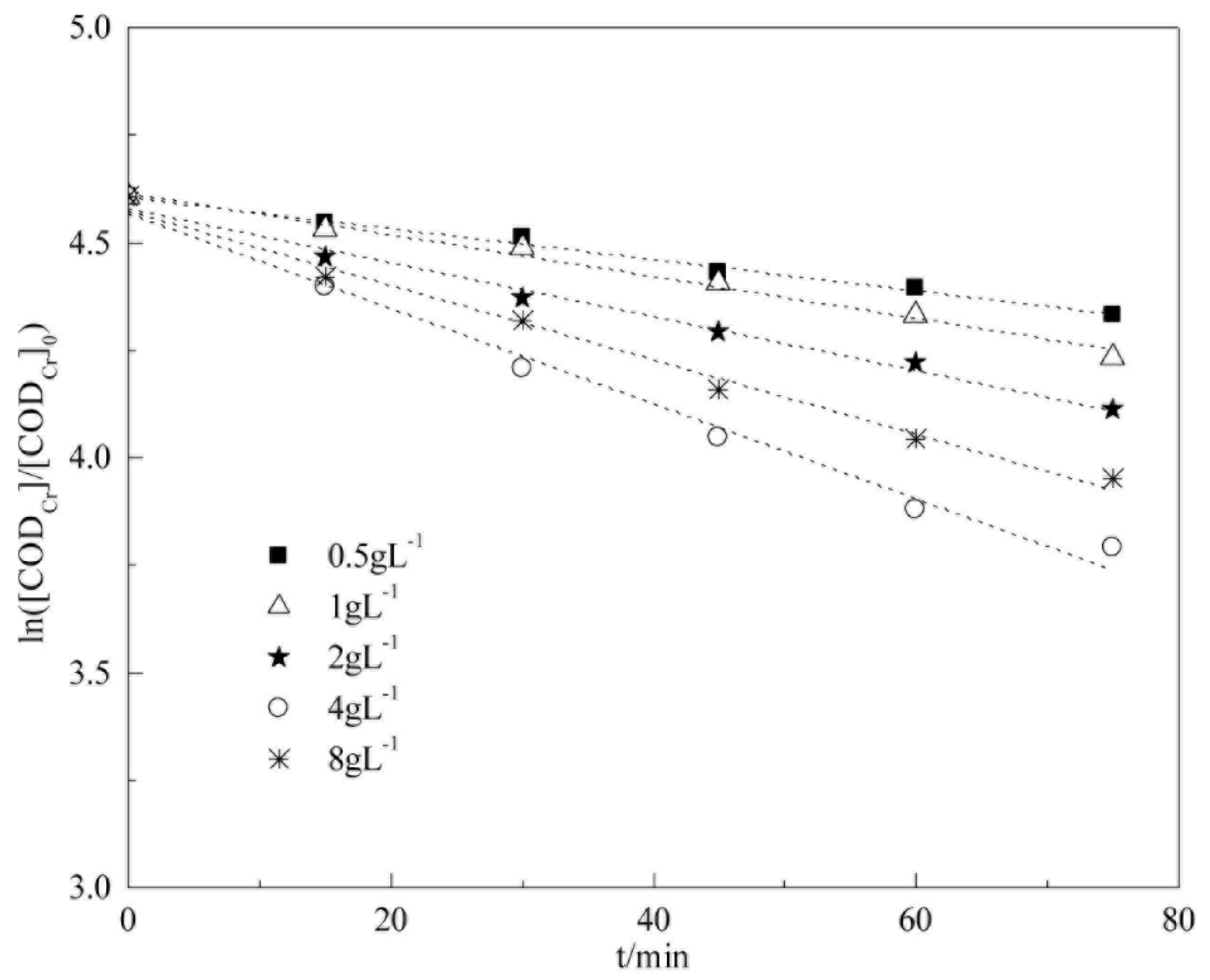
5.3. Analysis on Degradation Kinetics of CODCr in Coalbed Methane-Produced Water with Different Catalyst Concentrations
This section investigates the effect of catalyst concentration on COD
Cr degradation kinetics via heterogeneous Fenton reaction. The experimental conditions were as follows: 25 °C, H
2O
2 concentration of 40 mmol L
−1, initial pH of 5.1 (raw water), catalyst concentrations of 0.5, 1, 2, 4, and 8 g L
−1, respectively. The fitting results are shown in
Figure 15 and
Table 7.
Figure 15 and
Table 7 show that COD
Cr degradation in coalbed methane-produced water in the experimental catalyst dosage range (0.5 L
−1 to 8 L
−1) is in accordance with pseudo-first-order kinetics. The apparent reaction rate constants of COD
Cr degradation are −0.0036, −0.0048, −0.0063, −0.0111, and −0.0087 min
−1 when catalyst concentrations are 0.5, 1, 2, 4, and 8 g L
−1, respectively.
As the catalyst concentration increases, the apparent reaction rate constant increases. This increase occurs because more active sites become available for catalysis when high catalyst concentrations are present. However, the apparent reaction rate constant decreases when the catalyst concentration exceeds a specific limit because excess amounts of HO• interact. The reaction speed reaches the largest value when the catalyst concentration is 4 g L−1, and the apparent reaction rate constant is −0.0111 min−1.
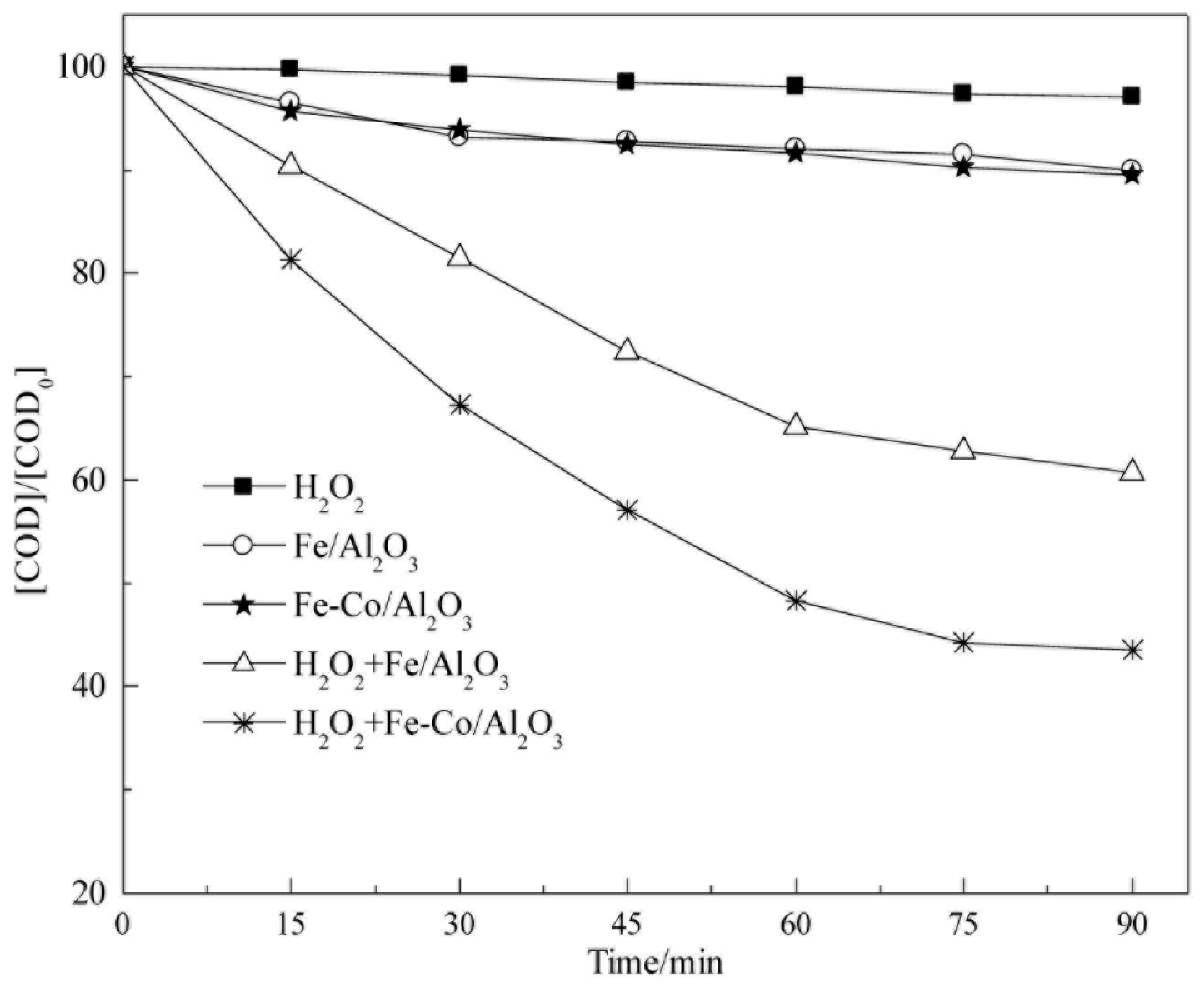
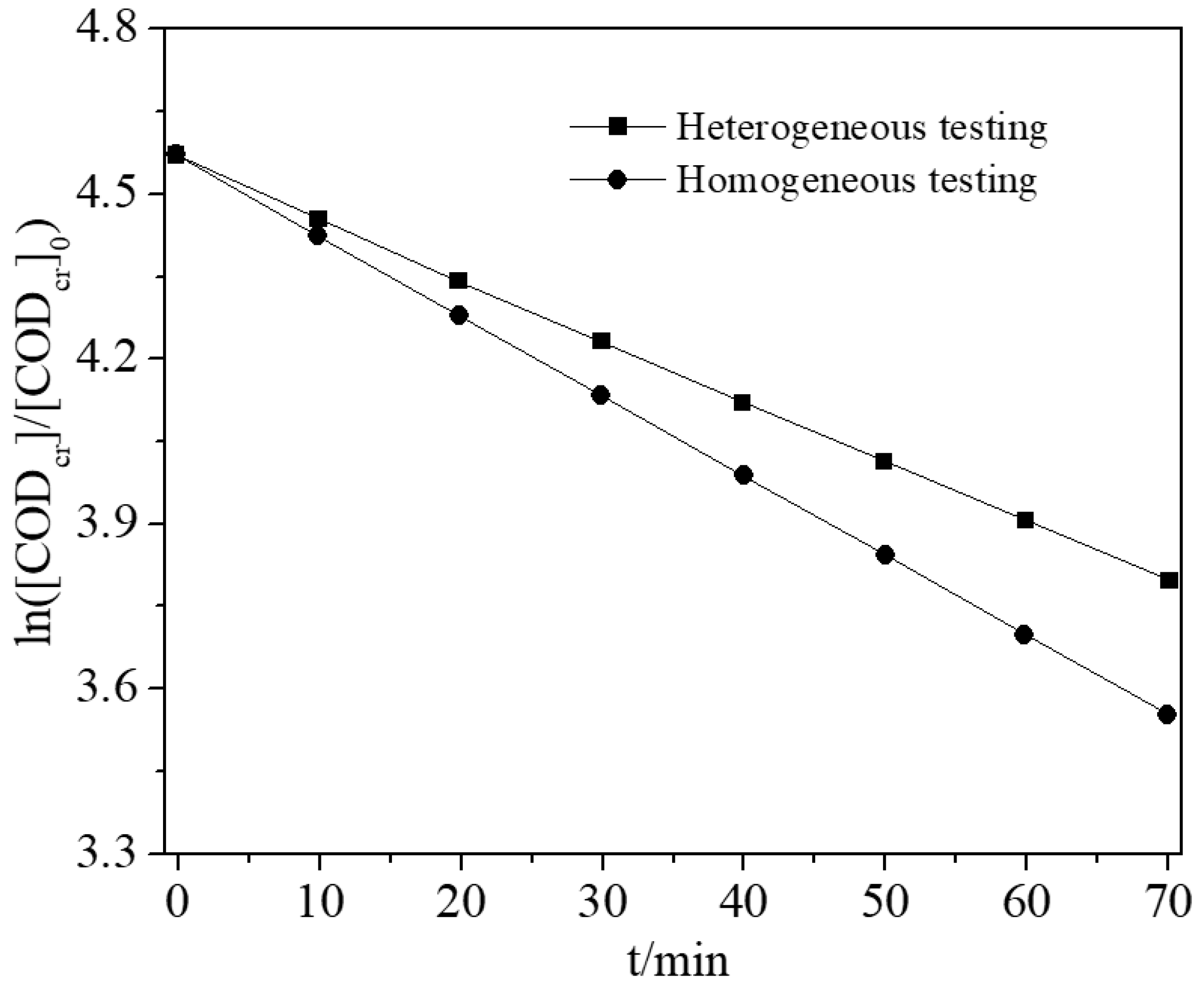
5.4. Heterogeneous/Homogeneous Catalysis Comparison Testing
The results for heterogeneous/homogeneous catalysis comparison testing is shown in
Figure 16. Both reactions are tested under the same reaction conditions, except for the type of the catalysts. It can be seen that homogeneous catalysis reaction resulted in a higher performance of in COD
cr degradation than that of heterogeneous catalysis. This could be attributed to the fact that, due to the pore and channel overlap in the heterogeneous catalyst of Fe-γ-Al
2O
3, not all Fe species can be exposed to the solution during the reaction. While all Fe
2+ can be involved to the reaction in the homogeneous catalysis. Similar results with a higher catalytic performance of homogeneous catalyst over a heterogeneous catalyst were reported previously [
62]. Even though the homogenous catalyst shows excellent performance in purifying wastewater, the heterogeneous catalyst is more superior in the practical reactions. This is because the heterogeneous catalyst can be easily separated from the water by filtration or other techniques after the Fenton reaction, and does not leave any residues. However, the homogenous catalyst (Fe
2+) is harder to separate from the water after reaction, and forms secondary pollutants [
26].