1. Introduction
As the discharge of environmental pollutants in receiving waters continues to be a serious concern across the globe, it is important to develop effective approaches for their removal. Synthetic dyes found in wastewater effluents are of particular concern due to their high toxicity and low biodegradability in water [
1]. They are used widely in the textile, pharmaceutical, cosmetics, and food industries because of their coloring capabilities. In the textile industry, an estimated 10,000 different dyes and pigments are used, and over 7 × 10
5 tons of synthetic dyes are annually produced worldwide; up to 200,000 tons of these dyes are discharged to receiving environments every year [
2]. With the large quantities of dyes released and their known toxicity to aquatic environments, as well as to human health, the removal of textile dyes has received increasing research attention in recent years. Dyes have been reported to exhibit carcinogenicity, mutagenicity, and resistance to natural degradation [
3]. Some of these dyes also have the potential to cause kidney failure and dysfunction of the brain and reproductive and central nervous systems.
Another important aspect of textile wastewater treatment is the de-colouration of dyes. Even in small amounts, dyes can be toxic to aquatic life and lead to changes in salinity and the visible coloration of receiving waters. Additionally, they can also reduce sunlight penetration, which hinders natural photosynthesis and disinfection processes [
4]. Unfortunately, these dyes exhibit high stabilities to light, temperature, water, and detergents, allowing them to escape conventional physio-chemical and biological wastewater treatment processes [
2,
5]. Furthermore, textile industries consume a substantial amount of water in the dyeing process and have been classified as among the most polluting of all industrial sectors. Hence, as the demand for textile products and synthetic dyes increases with the growing global population, textile dye wastewater is becoming a more significant water pollution contributor.
Adsorption is one of the most commonly used approach for the treatment of wastewaters from the textile industry. The applications of most traditional wastewater treatment approaches are often limited by process cost, efficiency, and sludge handling requirements after treatment. Since adsorption processes are highly versatile in terms their applicability for a range of contaminants, their use has been growing in popularity. Additionally, their potential simplicity, economic feasibility, and high level of efficiency make them suitable solutions for the treatment of a number of wastewater contaminants. Adequate, cost-effective adsorbents that could provide sufficient adsorption capacity, adsorption rate, and mechanical strength are in increasing demand. Activated carbon has been reported to be very successful and efficient in adsorbing dyes, but its popularity has decreased due to its high market value and the operational costs associated with its use. Other adsorbents such as silicon polymers and kaolin have also been explored. Natural materials and polymers are often suggested alternatives to activated carbons because of their renewability and availability [
3]. These natural materials can be used with or without pretreatment and can often be modified to selectively target particular pollutants under specific conditions. Polymeric adsorbents are demonstrating increasing advantages over activated carbon because of their simple processing and possibility for modifications. Moreover, polymeric adsorbents can be tailored to have reversible adsorption capabilities, meaning the adsorbent could be recovered through separation processes to separate the adsorbent from the dye from for future reuse.
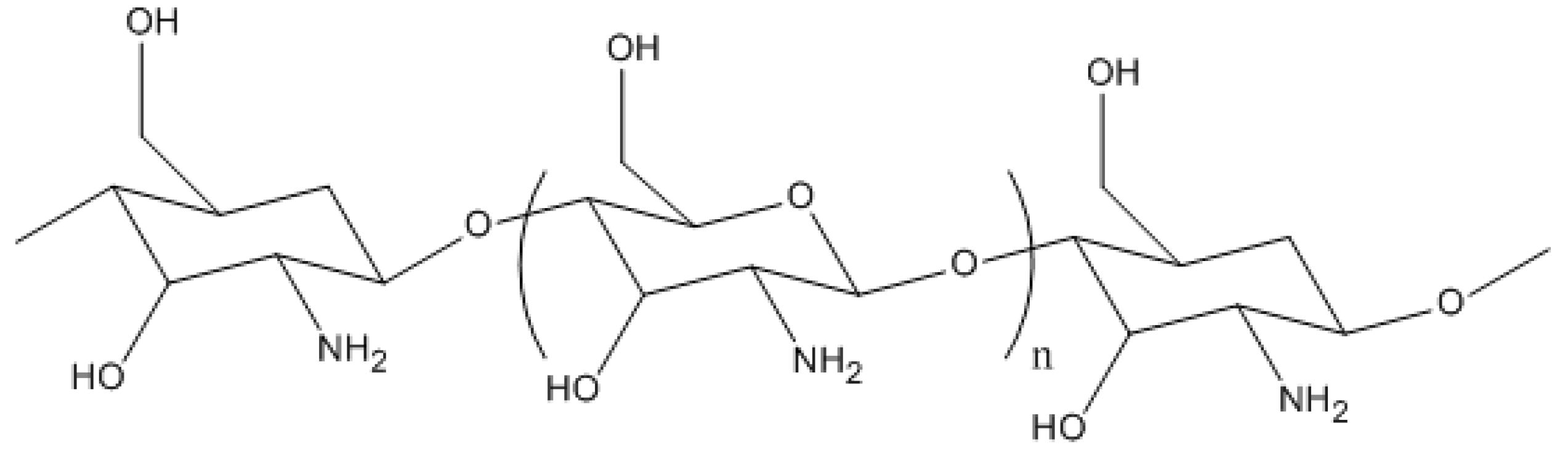
One example of a natural polymer is chitosan (CTS) (
Figure 1). Chitosan is a naturally abundant polysaccharide derived from chitin, which is found and harvested from crustacean shells. Chitin is one of the most abundant natural polymers in the world, second to cellulose, therefore making chitosan inexpensive and readily accessible. Among a wide range of applications, chitosan is a natural adsorbent and has been shown to adsorb substantial amounts of heavy metals and organic pollutants due to its high hydrophilicity and presence of amino and hydroxyl functional groups that serve as active sites for adsorption.
The ability for chitosan to adsorb textile dyes has been well documented over the past decade, showing promising results and expanding its potential applications. Azo dyes are one of many classes of dyes and are characterized by having one or more azo groups (–N=N–). Azo dyes represent 70% of dyes produced annually and consist of a wide range of classifications that describes their reactivity with the dyeing materials. Reactive azo dyes, specifically, are the most used due to their bright colors, excellent color fastness, and ease of application [
5]. Reactive Orange 16 (RO16) (
Figure 2) was selected as a representative reactive azo dye for this work as it is a commonly used textile dye.
Polymer graft modification can introduce desired properties and broaden the field of potential applications for chitosan through the selection of different types of polymer side chains with different functionalities. By grafting and functionalizing additional side chains onto chitosan, the adsorption capacity of the grafted copolymer could potentially be increased. For reactive azo dyes, Singh et al. reported a poly(methyl methacrylate) (PMMA) grafted chitosan with an adsorption capacity for azo dyes of three times that of pure chitosan. In addition, a separate study demonstrated that triphenylphosphine (TPP) crosslinked chitosan improved the adsorption capacity of chitosan by 40% in the removal of reactive azo dyes [
5]. These studies would suggest that with tailored polymeric side chains, the adsorption efficiency of chitosan can be improved extensively through functionalization. In this work, the adsorption capacity of glycidyl methacrylate (GMA) functionalized chitosan with grafted poly-(poly(ethylene glycol) methyl ether methacrylate) (PPEGMA) side chains (CTS-
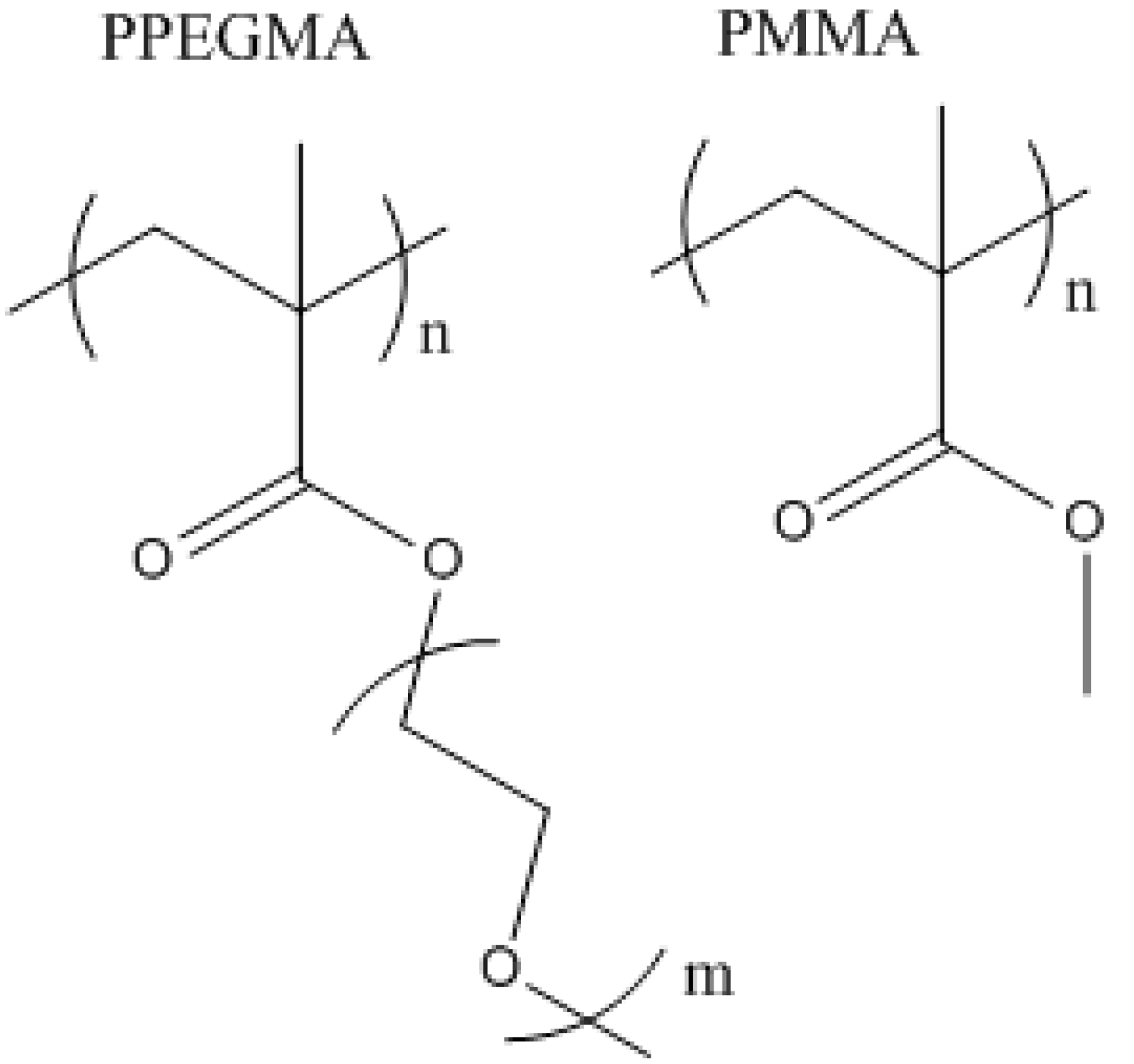
g-GMA-PPEGMA) was investigated and compared with pure chitosan for the removal of RO16 dye. PPEGMA has a similar structure to PMMA (
Figure 3), and would therefore be expected to exhibit similar adsorption properties. PPEGMA also has repeating ester groups, while PMMA only has one, which would suggest that PPEGMA could yield higher adsorption capacities. More importantly, PPEGMA is water soluble, whereas PMMA is water insoluble. Since chitosan is soluble in water under acidic conditions, the synthesis of PPEGMA grafted chitosan could be performed in an aqueous system, in comparison to the synthesis of PMMA grafted chitosan which would require organic solvents, leading to a much greener and cost efficient process resulting from the reduction in organic waste solvents and fewer synthesis steps.
The modification of chitosan to yield application-specific properties can generally take place at the amino (NH
2) or primary hydroxyl (OH) functionalities, although the amino functionality is mostly used. However, the natural adsorption capacity of chitosan is largely attributed to the NH
2 group. As such, the hydroxyl group was targeted for the modification of chitosan in this work, hence preserving the NH
2 functionality and its natural adsorption capacity. The modification of chitosan through polymer grafting was achieved via nitroxide-mediated polymerization and a grafting to approach [
6]. The objective was to examine the adsorption capacity of CTS-
g-GMA-PPEGMA for RO16 relative to pure chitosan in water for textile wastewater treatment applications. The present work describes the synthesis and application of PPEGMA-grafted chitosan for the removal of Reactive Orange 16.
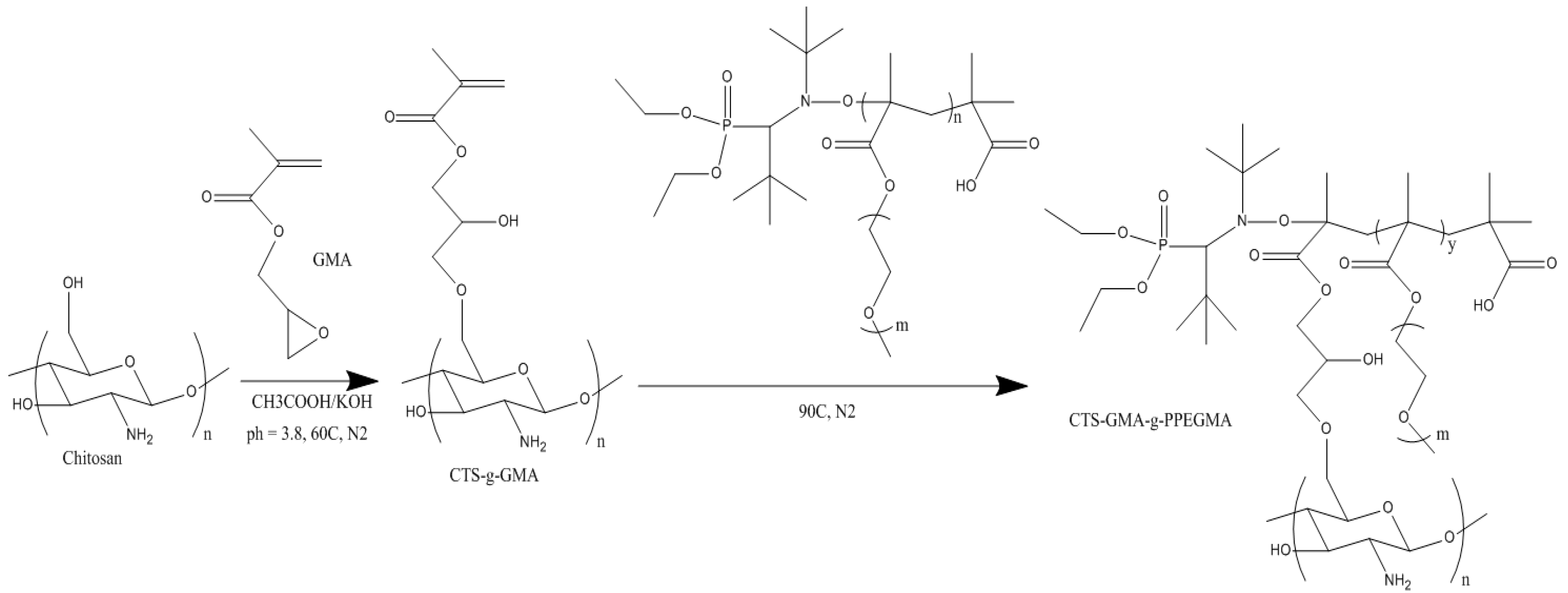
Figure 4 illustrates the steps we used in the synthesis of the CTS-
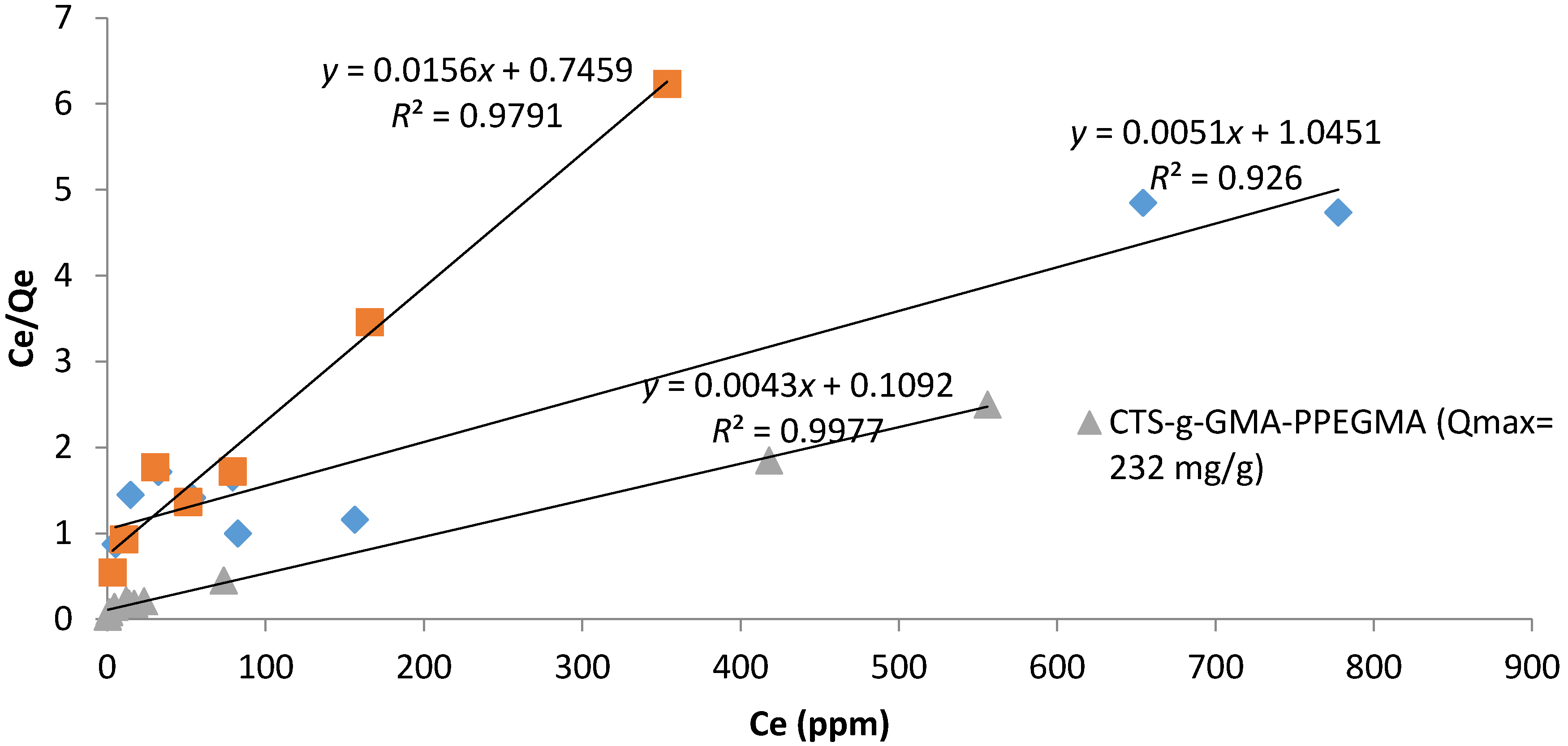
g-GMA-PPEGMA. Batch adsorption experiments were carried out to develop adsorption isotherms for both chitosan and PPEGMA-grafted chitosan and experimental data were fitted to the Langmuir and Freundlich isotherms for the computation of respective equation parameter constants.
2. Experimental
2.1. Materials
Chitosan (CTS, Aldrich, Oakville, ON, Canada, low molecular weight, 85% degree of deacetylation), glycidyl methacrylate (GMA, Aldrich, 97%), acetic acid (Fisher, Waltham, MA, USA, 99.7%), and acetone (ACP, 99.5%) were used as received. Poly(ethylene glycol) methyl ether methacrylate (PEGMA, Aldrich, Mn of 300 g/mol) and styrene (St, Aldrich, 99+%) were passed through aluminum oxide columns (Aldrich, ~150 mesh, 58 A) prior to polymerization. BlocBuilder (2-methyl-2-(N-tert-butyl-N-(1-diethoxyphosphoryl-2,2-dimethylpropyl)aminoxy)-propionic acid alkoxyamine) (BB, 99%) and SG1 (4-(diethoxyphosphinyl)-2,2,5,5-tetramethyl-3-azahexane-N-oxyl) (85%) were supplied by Arkema. Reactive Orange 16 was kindly supplied by the Ramsay research group in the Department of Chemical Engineering at Queen’s University (Kingston, ON, Canada).
2.2. Instrumentation
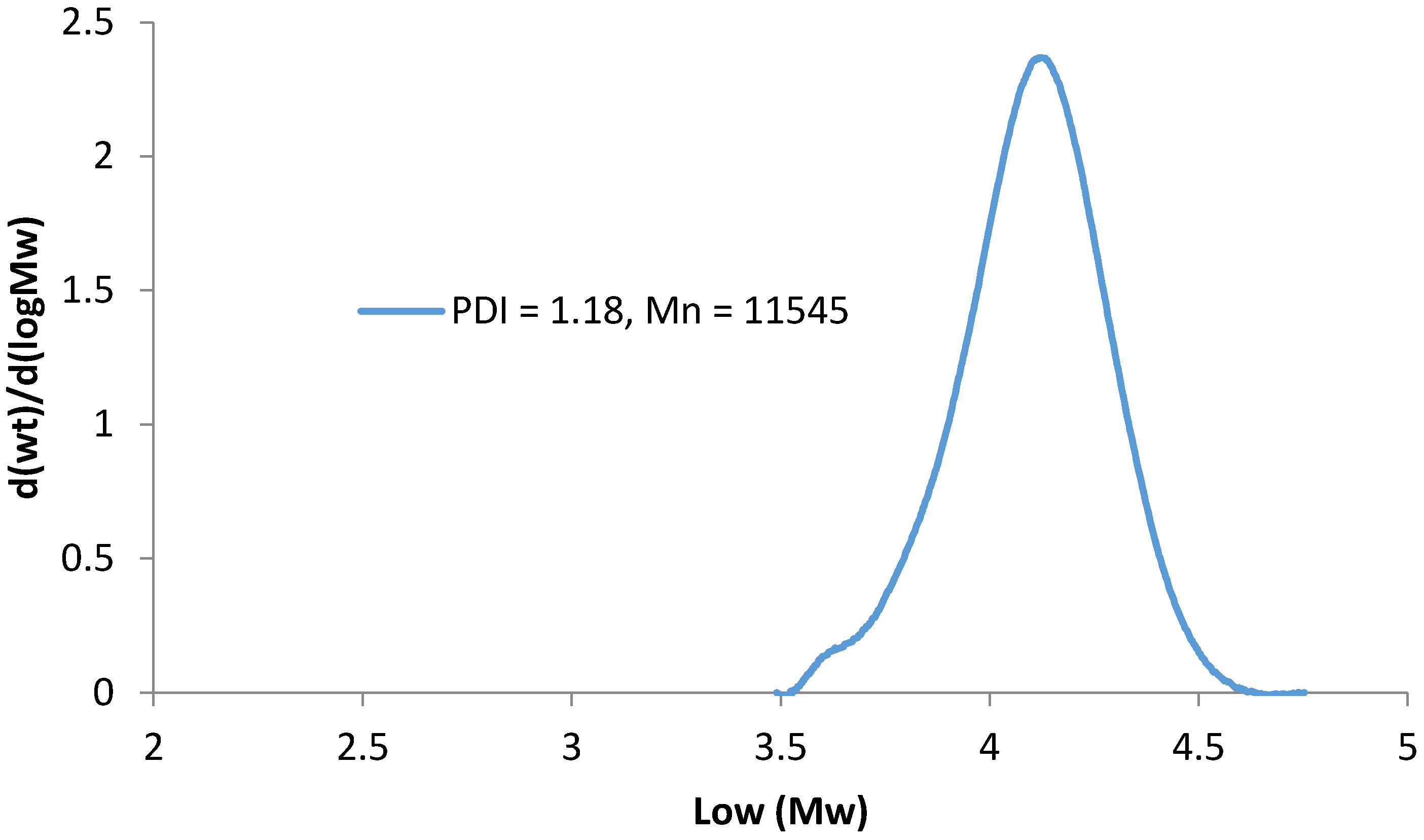
1H NMR spectroscopy was performed at room temperature on a FT-NMR Bruker Advance 400 MHz spectrometer (Billerica, MA, USA) with a total of 256 scans, using D2O as the solvent at 5 mg/mL. Thermogravimetric analysis (TGA) curves were recorded on TA Instruments Q500 TGA Analyzer (New Castle, DE, USA) by heating the sample from 75 °C to 600 °C at a rate of 10 °C per minute. Gel Permeation Chromatography (GPC, Waters, Milford, MA, USA) analyses were performed with a Waters 2690 Separation Module and Waters 410 Differential Refractometer with THF as the eluent. Adsorption experiments were conducted using a WSR Shaker Model 3500 (VWR, Radnor, PA, USA) and Lambda XLS Spectrometer (Perkin Elmer, Waltham, MA, USA).
2.3. Synthesis of CTS-g-GMA
The functionalization of chitosan with GMA followed a previously published synthesis approach by García-Valdez et al. [
6]. Chitosan (1 g) was first dissolved in 100 mL of acetic acid (0.4 M) in a three neck round bottom flask with 5 mL of 0.05 M potassium hydroxide (KOH) and 10 mL of 9.08 M hydroquinone. The flask was then purged with nitrogen for 30 min and heated to 60 °C in an oil bath. GMA was added drop wise at 60 °C and magnetically stirred for 2 h. CTS-
g-GMA was precipitated in acetonitrile, washed with THF and dried for
1H NMR and TGA analysis.
2.4. Synthesis of Poly(PEGMA) via Nitroxide-Mediated Polymerization
PEGMA monomer (50 g, 53 mL), styrene (1.92 g, 2.14 mL), BlocBuilder (0.70 g), and 0.06 mL of SG1 were mixed in a three neck round bottom flask with magnetic stirring and oxygen purging for 30 min. (A small amount of styrene comonomer provides better control over the PEGMA polymerization). The flask was then heated to 90 °C for one hour. Unreacted monomer was removed through precipitation in diethyl ether. The polymer was dried under vacuum and analyzed by 1H NMR and TGA.
2.5. Synthesis of CTS-GMA-g-PPEGMA via Grafting to Approach
The modification of CTS-
g-GMA was carried out following a previously reported approach developed in our research group [
5]. Briefly, CTS-
g-GMA (1 g) was dissolved in 100 mL of 0.1 M acetic acid in a three neck round bottom flask. KOH was added to the CTS-
g-GMA solution to increase the pH to 5 and mechanically stirred under nitrogen for 30 min before increasing the temperature to 90 °C. PPEGMA (0.5 g) was dissolved in 60 mL of de-ionized (DI) water and was purged of oxygen in an inert nitrogen atmosphere. 20 mL PPEGMA was added every 30 min once the CTS-
g-GMA solution reached 90 °C and the reaction was left to react for an additional 1.5 h. CTS-
g-GMA-PPEGMA was washed in THF and extracted using a rotary evaporator.
2.6. Dye Adsorption Experiments
A 100 ppm dye stock solution (100 mg in 1 L of DI water) was diluted to lower concentrations for the construction of a calibration curve. Concentrations ranged from 0.5 ppm to 80 ppm with a
R2 of 0.99. The λ
max of RO16 dye was measured to be 494 nm. Adsorption experiments were carried out using CTS-
g-GMA-PPEGMA, chitosan, chitosan-PPEGMA (50/50) physical mixture, and PPEGMA as adsorbents on an orbital shaker table set at 240 rpm. The experiments were conducted at room temperature for a predetermined equilibrium time of 36 h and neutral pH. Each adsorbent was thoroughly mixed with 20 mL dye solution, and once the samples were shaken for the desired experimental time, the suspensions were filtered through Whatman 0.90 mm filter paper. The aqueous dye samples were diluted, if necessary, and analyzed for dye concentration. Initial dye concentrations ranged from 20 to 1200 ppm with 50 mg of adsorbent and each adsorption batch experiment was conducted in duplicate. The adsorption capacity of each adsorbent from the equilibrium solution was calculated using Equation (1).
where
Qe is the adsorption capacity, describing the mass of dye adsorbed (mg) for each gram of adsorbent (mg/g),
Co is the initial concentration of dye in mg/L,
Ce is the equilibrium concentration of dye in solution (mg/L),
V (L) is the volume of dye solution and
W (g) is the weight of the adsorbent used.