A Rationally Designed, Spiropyran-Based Chemosensor for Magnesium
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Syntheses
2.1.1. Ethyl 4-(3′,3′-Dimethyl-6-fluorospiro[chromene-2,2′-indolin]-1′-yl)butanoate (4)
2.1.2. Ethyl 4-(3′,3′-Dimethyl-6-nitrospiro[chromene-2,2′-indolin]-1′-yl)butanoate (3)
2.1.3. 4-(6-Fluoro-3′,3′-dimethylspiro[chromene-2,2′-indolin]-1′-yl)butanoic acid (2)
2.1.4. 4-(3′,3′-Dimethyl-6-nitrospiro[chromene-2,2′-indolin]-1′-yl)butanoic acid (1)
2.2. Spectroscopic Analyses of Chemosensors 1–4
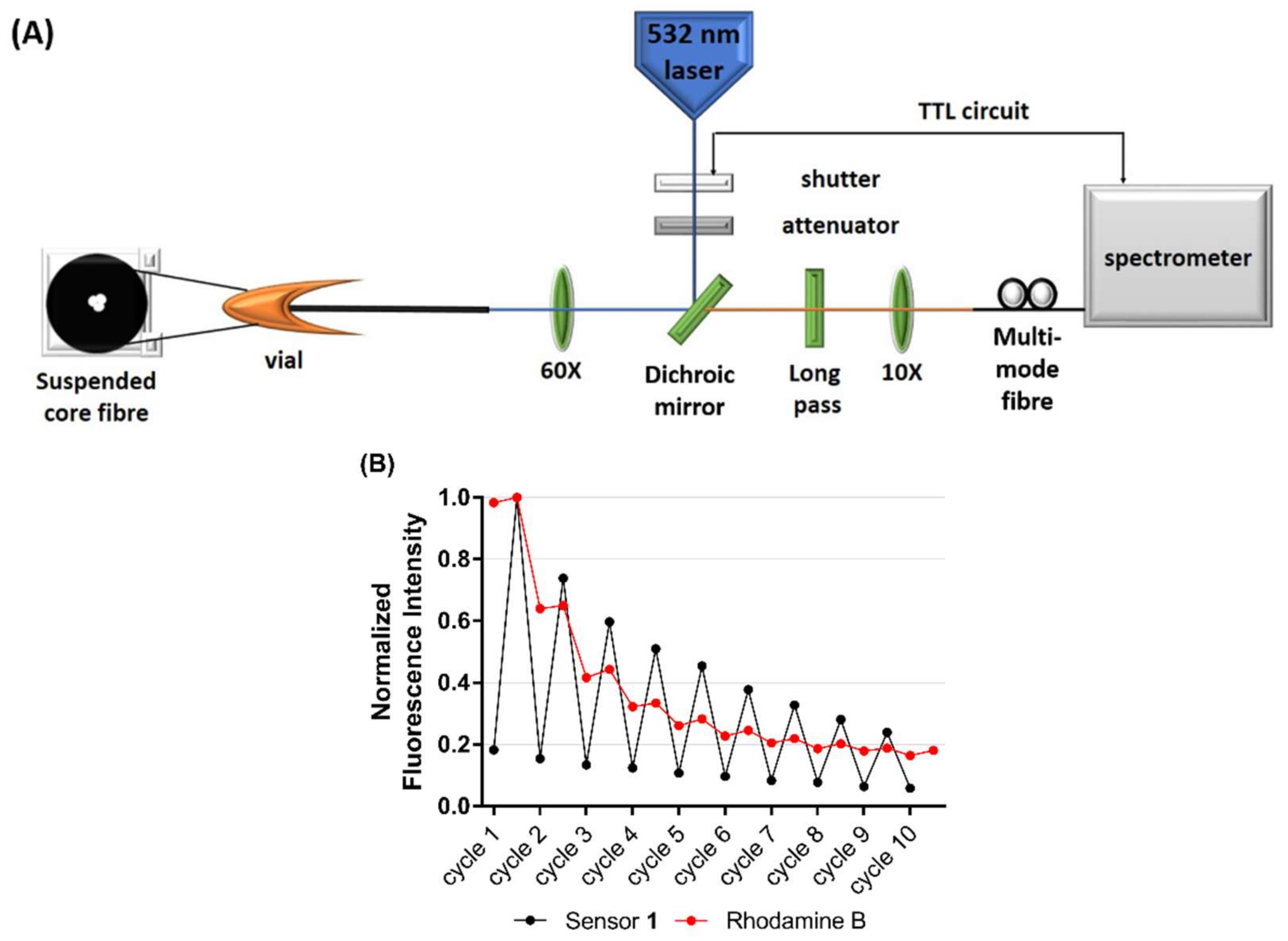
2.3. Photoswitching in Optical Fibre
3. Results and Discussion
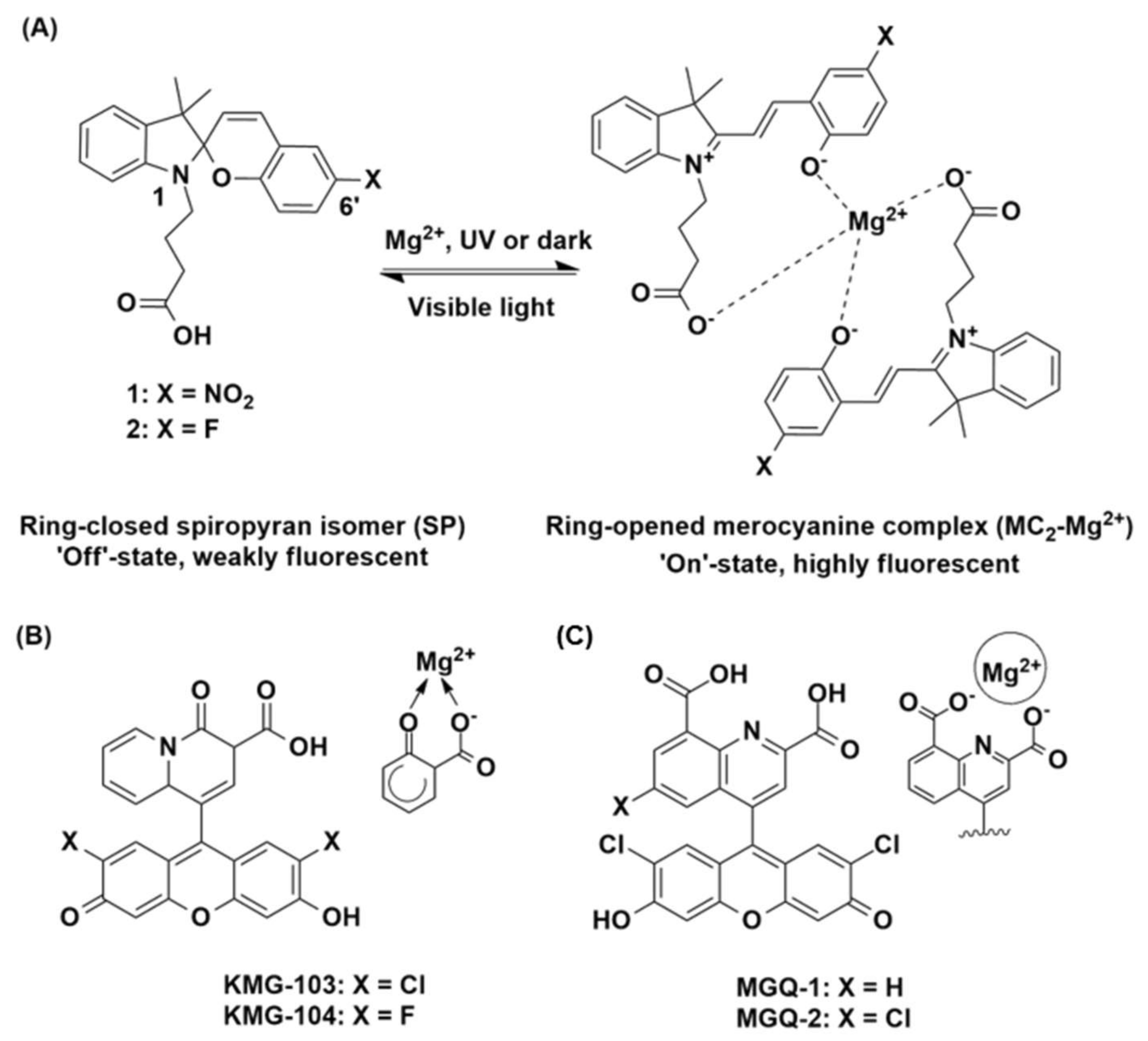
3.1. DFT Modelling
3.2. Synthesis
3.3. Sensor Characterisation
3.4. Photoswitching in an Optical Fibre
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Glasdam, S.M.; Glasdam, S.; Peters, G.H. The importance of magnesium in the human body: A systematic literature review. Adv. Clin. Chem. 2016, 73, 169–193. [Google Scholar] [PubMed]
- Wolf, F.I.; Trapani, V. Cell (patho)physiology of magnesium. Clin. Sci. 2008, 114, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Gunther, T. Concentration, compartmentation and metabolic function of intracellular free Mg2+. Magnes. Res. 2006, 19, 225–236. [Google Scholar] [PubMed]
- Romani, A.M. Cellular magnesium homeostasis. Arch. Biochem. Biophys. 2011, 512, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Ryan, M.F. The role of magnesium in clinical biochemistry: An overview. Ann. Clin. Biochem. 1991, 28 Pt 1, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Krishnamoorthy, G.; Yang, Y.; Hu, L.; Chaturvedi, N.; Harilal, D.; Qin, J.; Cui, J. Mechanism of magnesium activation of calcium-activated potassium channels. Nature 2002, 418, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Mubagwa, K.; Gwanyanya, A.; Zakharov, S.; Macianskiene, R. Regulation of cation channels in cardiac and smooth muscle cells by intracellular magnesium. Arch. Biochem. Biophys. 2007, 458, 73–89. [Google Scholar] [CrossRef] [PubMed]
- Nadler, M.J.; Hermosura, M.C.; Inabe, K.; Perraud, A.L.; Zhu, Q.; Stokes, A.J.; Kurosaki, T.; Kinet, J.P.; Penner, R.; Scharenberg, A.M.; et al. Ltrpc7 is a mg.Atp-regulated divalent cation channel required for cell viability. Nature 2001, 411, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Ko, Y.H.; Hong, S.; Pedersen, P.L. Chemical mechanism of atp synthase. Magnesium plays a pivotal role in formation of the transition state where atp is synthesized from adp and inorganic phosphate. J. Biol. Chem. 1999, 274, 28853–28856. [Google Scholar] [CrossRef] [PubMed]
- Sarrafzadegan, N.; Khosravi-Boroujeni, H.; Lotfizadeh, M.; Pourmogaddas, A.; Salehi-Abargouei, A. Magnesium status and the metabolic syndrome: A systematic review and meta-analysis. Nutrition 2016, 32, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Mooren, F.C. Magnesium and disturbances in carbohydrate metabolism. Diabetes Obes. Metab. 2015, 17, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Volpe, S.L. Magnesium, the metabolic syndrome, insulin resistance, and type 2 diabetes mellitus. Crit. Rev. Food Sci. Nutr. 2008, 48, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Zurlo, A.; Solmi, M.; Luchini, C.; Trevisan, C.; Bano, G.; Manzato, E.; Sergi, G.; Rylander, R. Magnesium status in alzheimer’s disease: A systematic review. Am. J. Alzheimer’s Dis. Other Dement. 2016, 31, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Belvedere, M.; Di Bella, G.; Dominguez, L.J. Altered ionized magnesium levels in mild-to-moderate alzheimer’s disease. Magnes. Res. 2011, 24, S115–S121. [Google Scholar] [PubMed]
- Kolte, D.; Vijayaraghavan, K.; Khera, S.; Sica, D.A.; Frishman, W.H. Role of magnesium in cardiovascular diseases. Cardiol. Rev. 2014, 22, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Kupetsky-Rincon, E.A.; Uitto, J. Magnesium: Novel applications in cardiovascular disease--a review of the literature. Ann. Nutr. Metab. 2012, 61, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Geiger, H.; Wanner, C. Magnesium in disease. Clin. Kidney J. 2012, 5, i25–i38. [Google Scholar] [CrossRef] [PubMed]
- Long, S.; Romani, A.M.P. Role of cellular magnesium in human diseases. Austin J. Nutr. Food Sci. 2014, 2, 1051. [Google Scholar] [PubMed]
- Carter, K.P.; Young, A.M.; Palmer, A.E. Fluorescent sensors for measuring metal ions in living systems. Chem. Rev. 2014, 114, 4564–4601. [Google Scholar] [CrossRef] [PubMed]
- Raju, B.; Murphy, E.; Levy, L.A.; Hall, R.D.; London, R.E. A fluorescent indicator for measuring cytosolic free magnesium. Am. J. Physiol.-Cell Physiol. 1989, 256, C540–C548. [Google Scholar] [CrossRef] [PubMed]
- Fluorescent Mg2+ indicators. The Molecular Probes Handbook—A Guide to Fluorescent Probes and Labeling Technologies, 11st ed.; ThermoFisher Scientific: Waltham, MA, USA, 2010; pp. 862–864. [Google Scholar]
- Levy, L.A.; Murphy, E.; Raju, B.; London, R.E. Measurement of cytosolic free magnesium ion concentration by fluorine-19 nmr. Biochemistry 1988, 27, 4041–4048. [Google Scholar] [CrossRef] [PubMed]
- Schachter, M.; Gallagher, K.L.; Sever, P.S. Measurement of intracellular magnesium in a vascular smooth muscle cell line using a fluorescent probe. Biochim. Biophys. Acta (BBA) Gen. Subj. 1990, 1035, 378–380. [Google Scholar] [CrossRef]
- Murphy, E.; Freudenrich, C.C.; Levy, L.A.; London, R.E.; Lieberman, M. Monitoring cytosolic free magnesium in cultured chicken heart cells by use of the fluorescent indicator furaptra. Proc. Natl. Acad. Sci. USA 1989, 86, 2981–2984. [Google Scholar] [CrossRef] [PubMed]
- Grubbs, R.; Walter, A. Determination of cytosolic Mg2+ activity and buffering in BC3H-1 cells with MAG-fura-2. Mol. Cell. Biochem 1994, 136, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Zaguilán, R.; Parnami, J.; Martinez, G.M. Mag-fura-2 (furaptra) exhibits both low (µm) and high (nm) affinity for Ca2+. Cell. Physiol. Biochem. 1998, 8, 158–174. [Google Scholar] [CrossRef] [PubMed]
- Hurley, T.W.; Ryan, M.P.; Brinck, R.W. Changes of cytosolic Ca2+ interfere with measurements of cytosolic Mg2+ using mag-fura-2. Am. J. Physiol.-Cell Physiol. 1992, 263, C300–C307. [Google Scholar] [CrossRef] [PubMed]
- Shindo, Y.; Fujii, T.; Komatsu, H.; Citterio, D.; Hotta, K.; Suzuki, K.; Oka, K. Newly developed Mg2+-selective fluorescent probe enables visualization of mg2+ dynamics in mitochondria. PLoS ONE 2011, 6, e23684. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, H.; Iwasawa, N.; Citterio, D.; Suzuki, Y.; Kubota, T.; Tokuno, K.; Kitamura, Y.; Oka, K.; Suzuki, K. Design and synthesis of highly sensitive and selective fluorescein-derived magnesium fluorescent probes and application to intracellular 3d Mg2+ imaging. J. Am. Chem. Soc. 2004, 126, 16353–16360. [Google Scholar] [CrossRef] [PubMed]
- Matsui, Y.; Sadhu, K.K.; Mizukami, S.; Kikuchi, K. Highly selective tridentate fluorescent probes for visualizing intracellular Mg2+ dynamics without interference from Ca2+ fluctuation. Chem. Commun. 2017, 53, 10644–10647. [Google Scholar] [CrossRef] [PubMed]
- Trapani, V.; Farruggia, G.; Marraccini, C.; Iotti, S.; Cittadini, A.; Wolf, F.I. Intracellular magnesium detection: Imaging a brighter future. Analyst 2010, 135, 1855–1866. [Google Scholar] [CrossRef] [PubMed]
- Trapani, V.; Schweigel-Rontgen, M.; Cittadini, A.; Wolf, F.I. Intracellular magnesium detection by fluorescent indicators. Methods Enzymol. 2012, 505, 421–444. [Google Scholar] [PubMed]
- Natali, M.; Giordani, S. Interaction studies between photochromic spiropyrans and transition metal cations: The curious case of copper. Org. Biomol. Chem. 2012, 10, 1162–1171. [Google Scholar] [CrossRef] [PubMed]
- Natali, M.; Giordani, S. Molecular switches as photocontrollable “smart” receptors. Chem. Soc. Rev. 2012, 41, 4010–4029. [Google Scholar] [CrossRef] [PubMed]
- Lukyanov, B.S.; Lukyanova, M.B. Spiropyrans: Synthesis, properties, and application. Chem. Heterocycl. Compd. 2005, 41, 281–311. [Google Scholar] [CrossRef]
- Klajn, R. Spiropyran-based dynamic materials. Chem. Soc. Rev. 2014, 43, 148–184. [Google Scholar] [CrossRef] [PubMed]
- Zakharova, M.I.; Pimienta, V.; Metelitsa, A.V.; Minkin, V.I.; Micheaua, J.C. Thermodynamic and kinetic analysis of metal ion complexation by photochromic spiropyrans. Russ. Chem. Bull. 2009, 58, 1329–1337. [Google Scholar] [CrossRef]
- Heng, S.; Mak, A.M.; Kostecki, R.; Zhang, X.Z.; Pei, J.X.; Stubing, D.B.; Ebendorff-Heidepriem, H.; Abell, A.D. Photoswitchable calcium sensor: ‘On’-’off’ sensing in cells or with microstructured optical fibers. Sens. Actuators B Chem. 2017, 252, 965–972. [Google Scholar] [CrossRef]
- Heng, S.; McDevitt, C.A.; Kostecki, R.; Morey, J.R.; Eijkelkamp, B.A.; Ebendorff-Heidepriem, H.; Monro, T.M.; Abell, A.D. Microstructured optical fiber-based biosensors: Reversible and nanoliter-scale measurement of zinc ions. ACS Appl. Mater. Interfaces 2016, 8, 12727–12732. [Google Scholar] [CrossRef] [PubMed]
- Heng, S.; Nguyen, M.-C.; Kostecki, R.; Monro, T.M.; Abell, A.D. Nanoliter-scale, regenerable ion sensor: Sensing with a surface functionalized microstructured optical fibre. RSC Adv. 2013, 3, 8308–8317. [Google Scholar] [CrossRef]
- Stubing, D.B.; Heng, S.; Monro, T.M.; Abell, A.D. A comparative study of the fluorescence and photostability of common photoswitches in microstructured optical fibre. Sens. Actuators B Chem. 2017, 239, 474–480. [Google Scholar] [CrossRef]
- Monro, T.M.; Warren-Smith, S.; Schartner, E.P.; François, A.; Heng, S.; Ebendorff-Heidepriem, H.; Afshar, S. Sensing with suspended-core optical fibers. Opt. Fiber Technol. 2010, 16, 343–356. [Google Scholar] [CrossRef]
- Schartner, E.P.; Henderson, M.R.; Purdey, M.; Dhatrak, D.; Monro, T.M.; Gill, P.G.; Callen, D.F. Cancer detection in human tissue samples using a fiber-tip pH probe. Cancer Res. 2016, 76, 6795–6801. [Google Scholar] [CrossRef] [PubMed]
- Purdey, M.S.; Schartner, E.P.; Sutton-McDowall, M.L.; Ritter, L.J.; Thompson, J.; Monro, T.M.; Abell, A.D. Localised hydrogen peroxide sensing for reproductive health. In Proceedings of the SPIE Optics + Optoelectronics, Prague, Czech Republic, 13–16 April 2015. [Google Scholar]
- Collins, G.E.; Choi, L.-S.; Ewing, K.J.; Michelet, V.; Bowen, C.M.; Winkler, J.D. Photoinduced switching of metal complexation by quinolinospiropyranindolines in polar solvents. Chem. Commun. 1999, 321–322. [Google Scholar] [CrossRef]
- Garcia, A.A.; Cherian, S.; Park, J.; Gust, D.; Jahnke, F.; Rosario, R. Photon-controlled phase partitioning of spiropyrans. J. Phys. Chem. A 2000, 104, 6103–6107. [Google Scholar] [CrossRef]
- Baldrighi, M.; Locatelli, G.; Desper, J.; Aakeroy, C.B.; Giordani, S. Probing metal ion complexation of ligands with multiple metal binding sites: The case of spiropyrans. Chem.-Eur. J. 2016, 22, 13976–13984. [Google Scholar] [CrossRef] [PubMed]
- Heng, S.; Mak, A.M.; Stubing, D.B.; Monro, T.M.; Abell, A.D. Dual sensor for cd(ii) and ca(ii): Selective nanoliter-scale sensing of metal ions. Anal. Chem. 2014, 86, 3268–3272. [Google Scholar] [CrossRef] [PubMed]
- Filley, J.; Ibrahim, M.A.; Nimlos, M.R.; Watt, A.S.; Blake, D.M. Magnesium and calcium chelation by a bis-spiropyran. J. Photochem. Photobiol. A Chem. 1998, 117, 193–198. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Frisch, M.J.E.A. Gaussian 09; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Magde, D.; Rojas, G.E.; Seybold, P.G. Solvent dependence of the fluorescence lifetimes of xanthene dyes. Photochem. Photobiol. 1999, 70, 737–744. [Google Scholar] [CrossRef]
- Zhao, J.B.; Jin, D.Y.; Schartner, E.P.; Lu, Y.Q.; Liu, Y.J.; Zvyagin, A.V.; Zhang, L.X.; Dawes, J.M.; Xi, P.; Piper, J.A.; et al. Single-nanocrystal sensitivity achieved by enhanced upconversion luminescence. Nat. Nanotechnol. 2013, 8, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Schartner, E.P.; Tsiminis, G.; Francois, A.; Kostecki, R.; Warren-Smith, S.C.; Nguyen, L.V.; Heng, S.; Reynolds, T.; Klantsataya, E.; Rowland, K.J.; et al. Taming the light in microstructured optical fibers for sensing. Int. J. Appl. Glass Sci. 2015, 6, 229–239. [Google Scholar] [CrossRef]
- Wiedemann, U.; Alt, W.; Meschede, D. Switching photochromic molecules adsorbed on optical microfibres. Opt. Express 2012, 20, 12710–12720. [Google Scholar] [CrossRef] [PubMed]


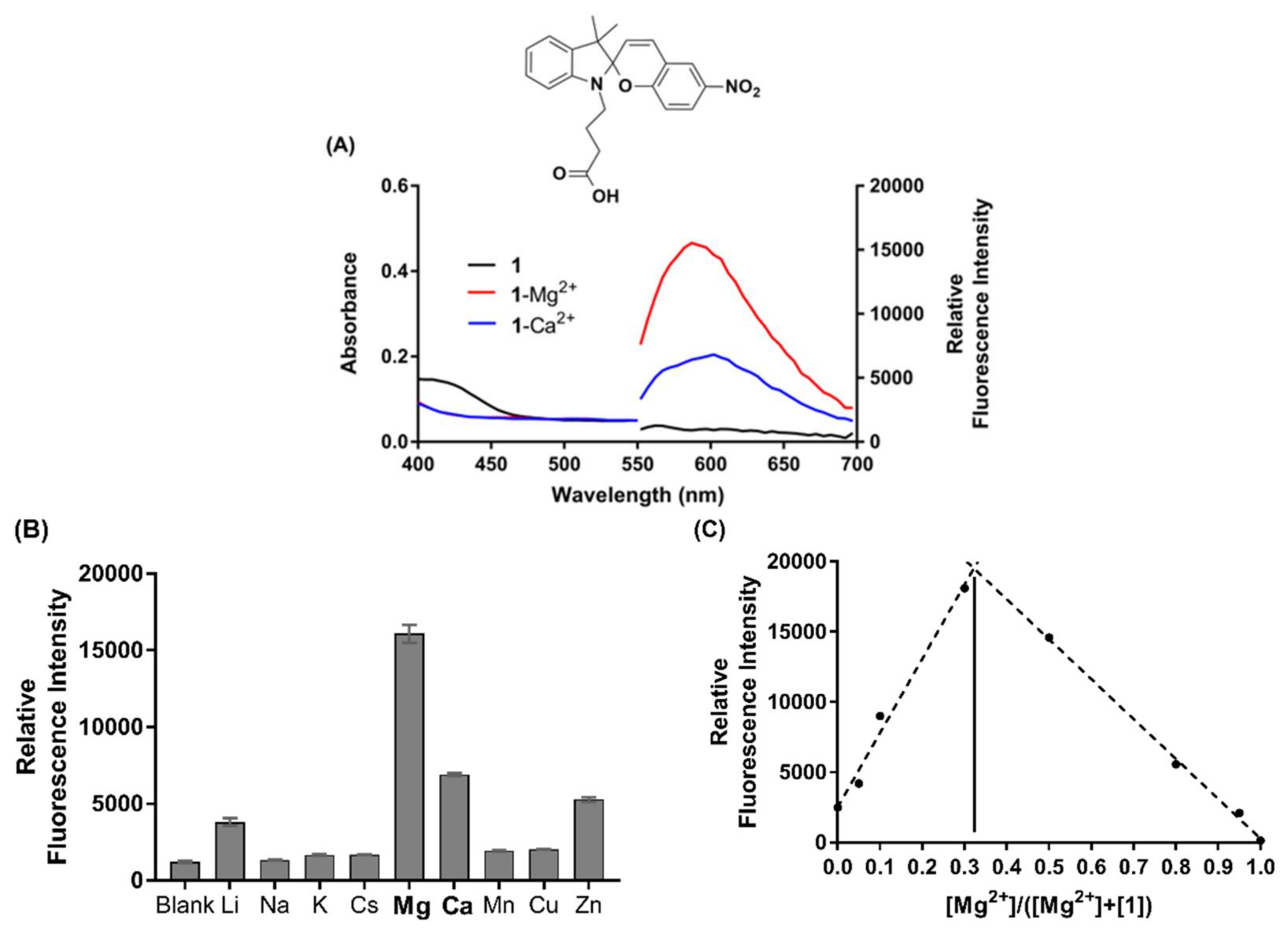
| Chemosensor 1 | Em λmax (nm) | Stoichiometry 2 | Φ 3 | Kd (µM) |
|---|---|---|---|---|
| MC(1) | 605 | * | * | * |
| MC(1)-Mg2+ | 590 | 2:1 (0.33) | 0.20 | 6.0 |
| MC(1)-Ca2+ | 605 | 2:1 (0.33) | 0.06 | 18.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sylvia, G.M.; Mak, A.M.; Heng, S.; Bachhuka, A.; Ebendorff-Heidepriem, H.; Abell, A.D. A Rationally Designed, Spiropyran-Based Chemosensor for Magnesium. Chemosensors 2018, 6, 17. https://doi.org/10.3390/chemosensors6020017
Sylvia GM, Mak AM, Heng S, Bachhuka A, Ebendorff-Heidepriem H, Abell AD. A Rationally Designed, Spiropyran-Based Chemosensor for Magnesium. Chemosensors. 2018; 6(2):17. https://doi.org/10.3390/chemosensors6020017
Chicago/Turabian StyleSylvia, Georgina M., Adrian M. Mak, Sabrina Heng, Akash Bachhuka, Heike Ebendorff-Heidepriem, and Andrew D. Abell. 2018. "A Rationally Designed, Spiropyran-Based Chemosensor for Magnesium" Chemosensors 6, no. 2: 17. https://doi.org/10.3390/chemosensors6020017





