Effect of Hydrophobic Pollution on Response of Thermo-Sensitive Hydrogel
Abstract
:1. Introduction
2. Experimental Section
2.1. Temperature-Response of a Single Spherical DEAA Gel Particle in Dilute Phenol Solution
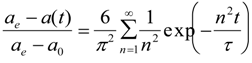
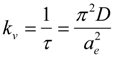
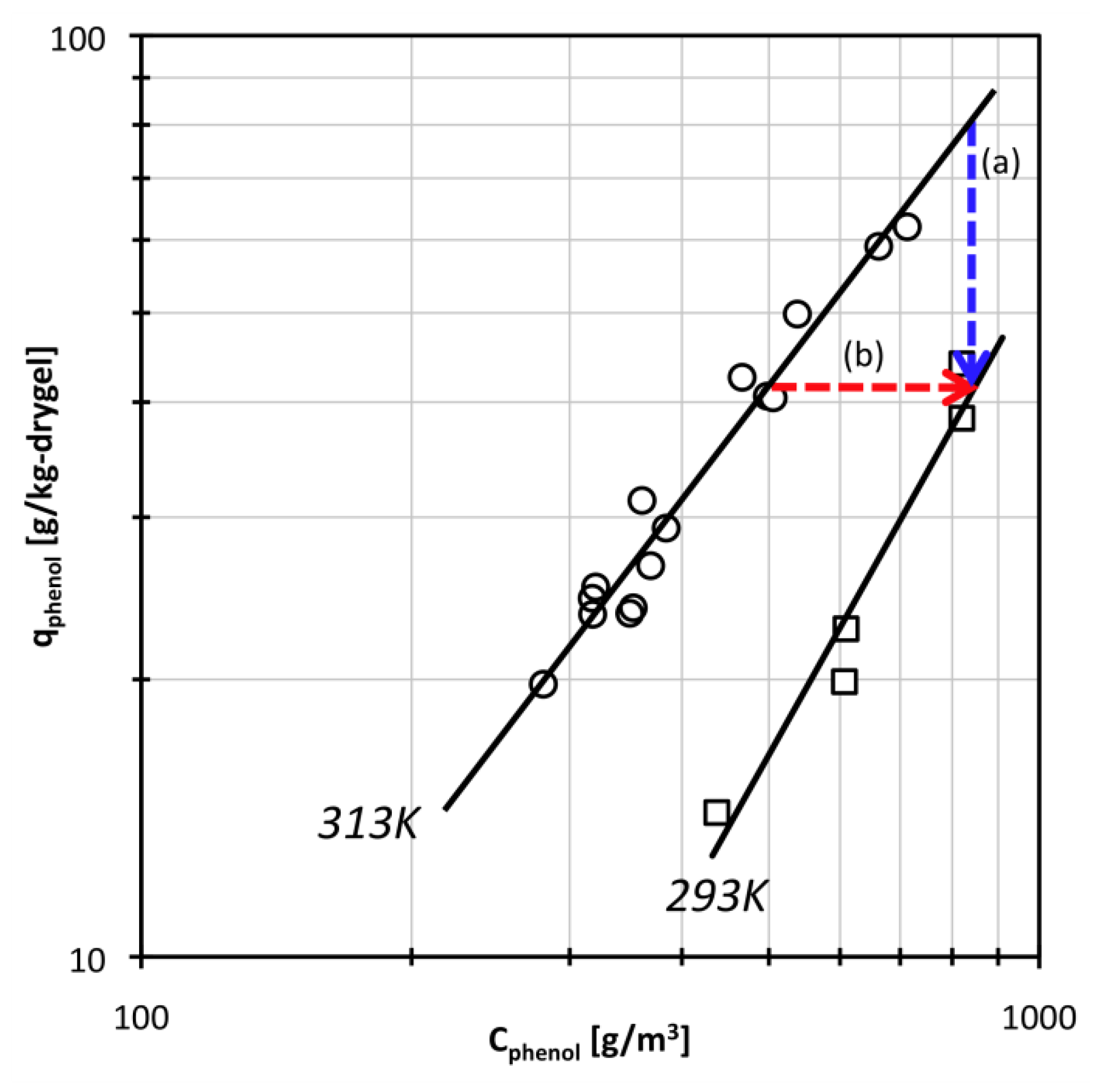
2.2. Phenol Adsorption Isotherm
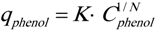
2.3. Swelling Behavior of DEAA Gel at Fixed amount of Adsorption Phenol
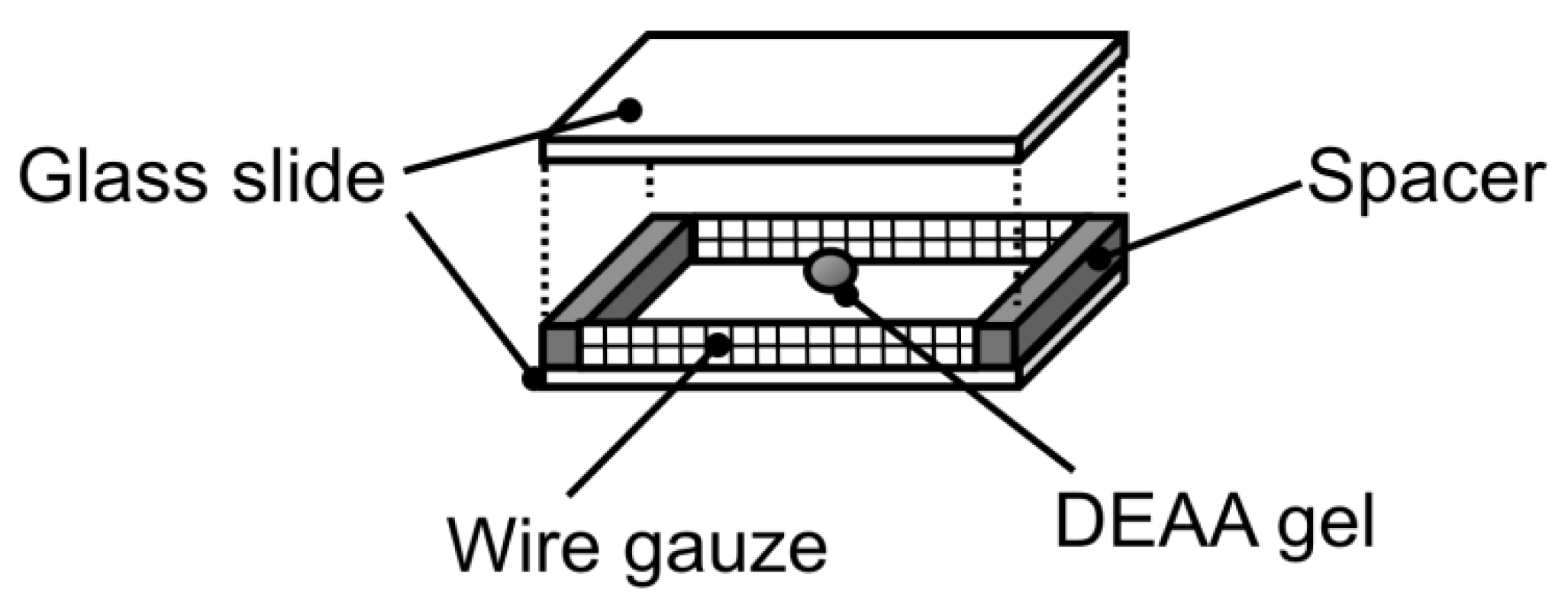
3. Results and Discussion
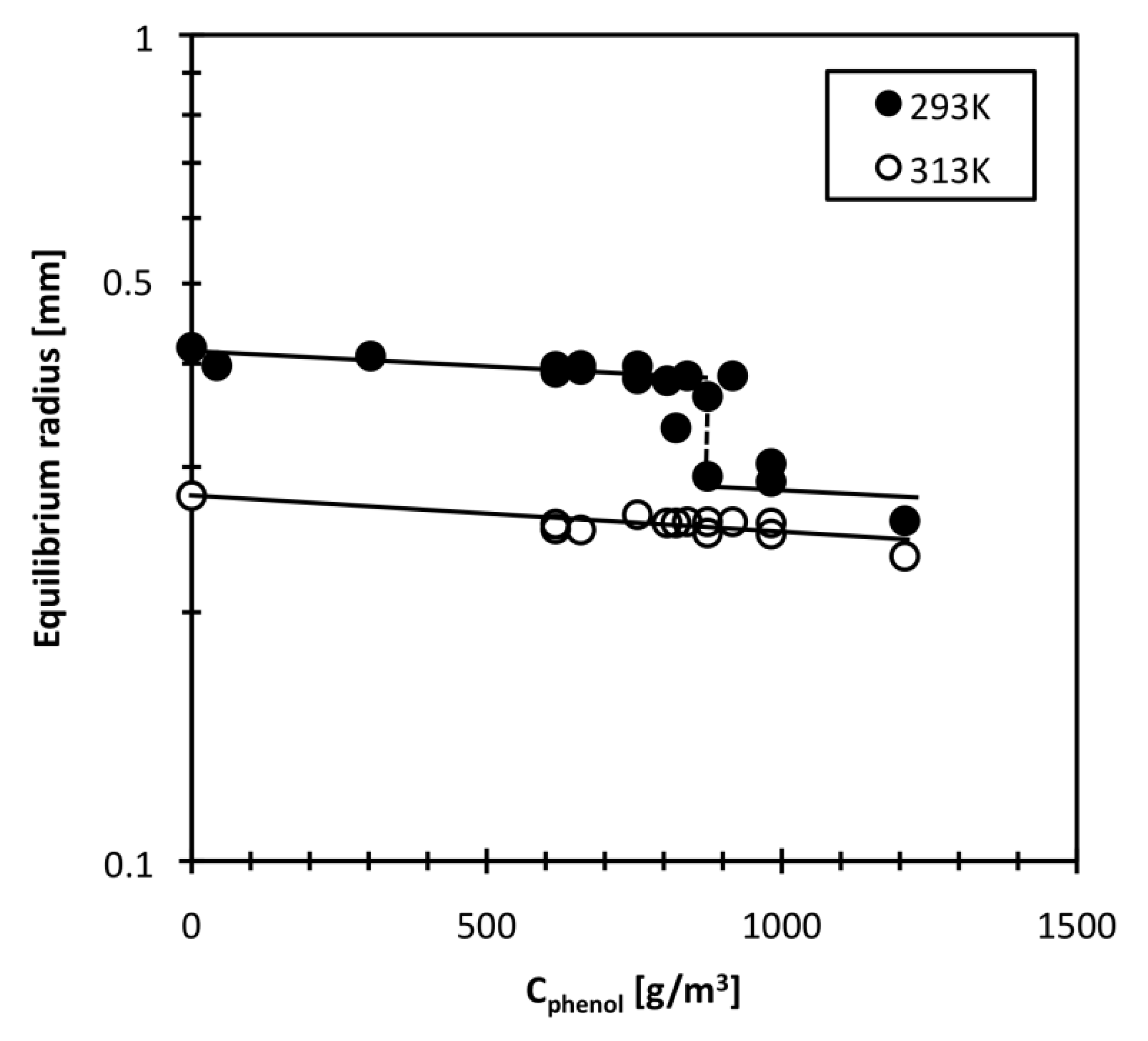
3.1. Equilibrium Radius and Swelling Behavior of DEAA Gel in Phenol Solution

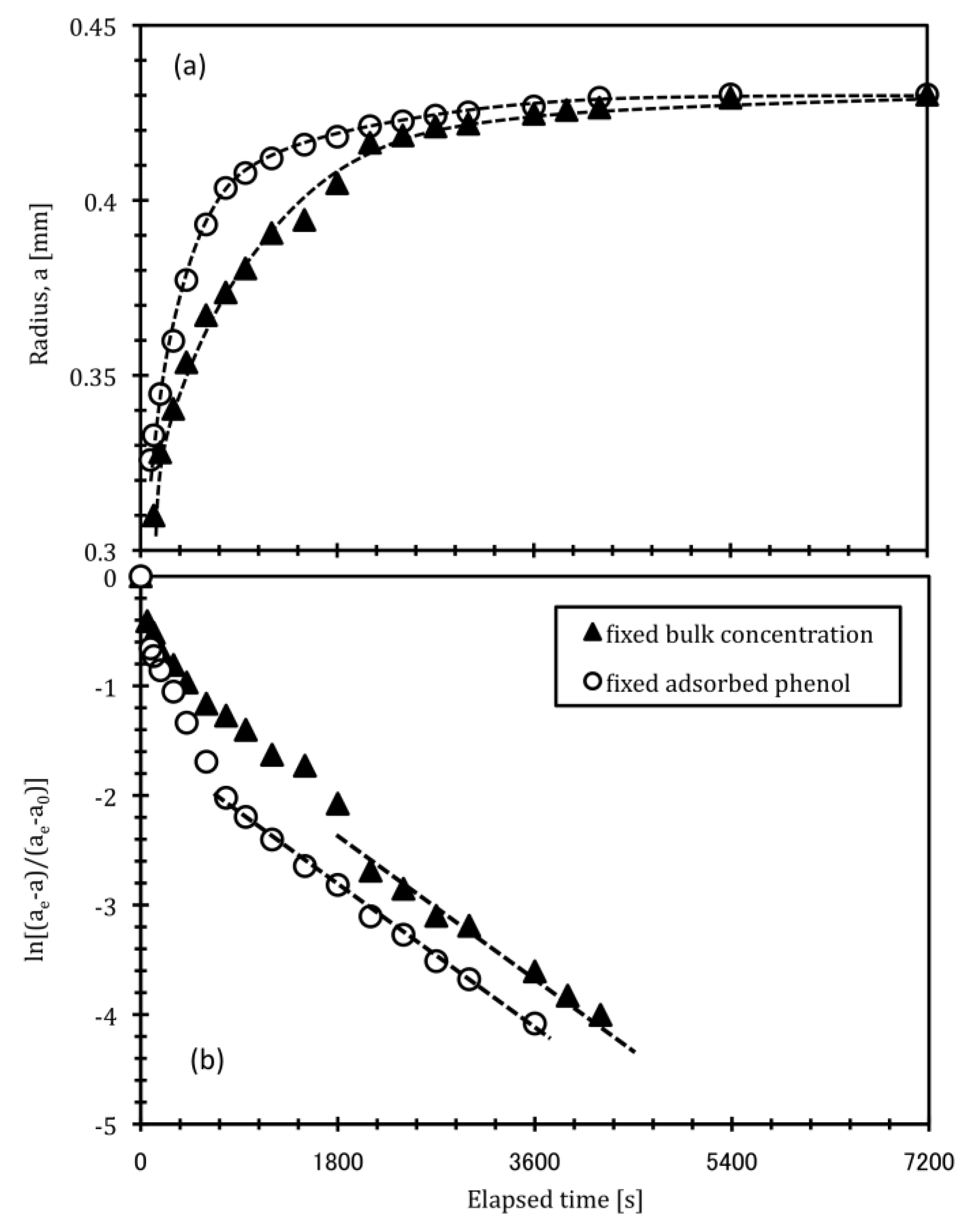
3.2. Effect of Phenol Adsorption-Desorption Process on Swelling Behavior
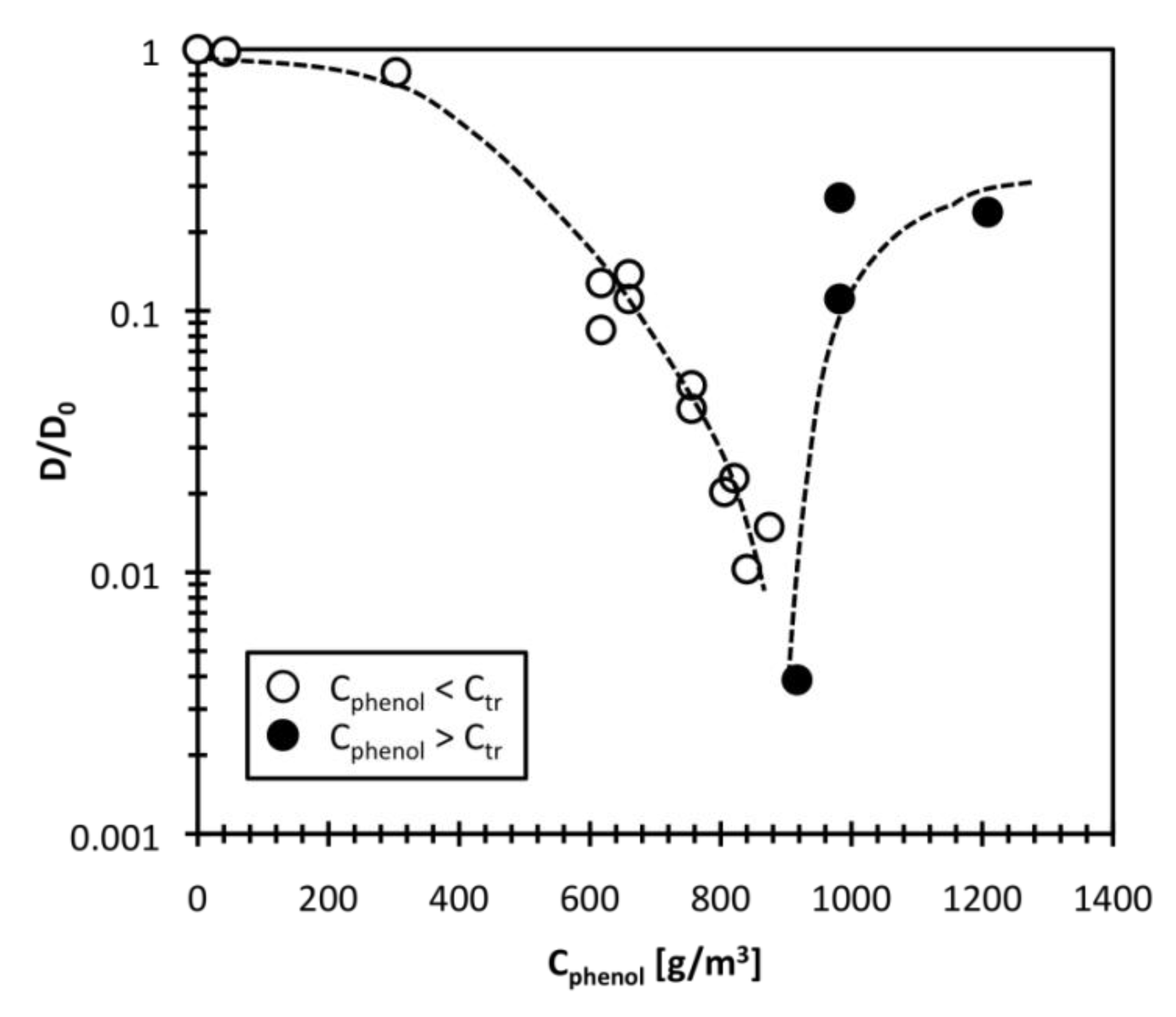
3.3. Effect of Phenol Adsorption on Collective Polymer Diffusion Constant
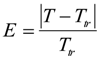
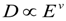
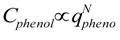
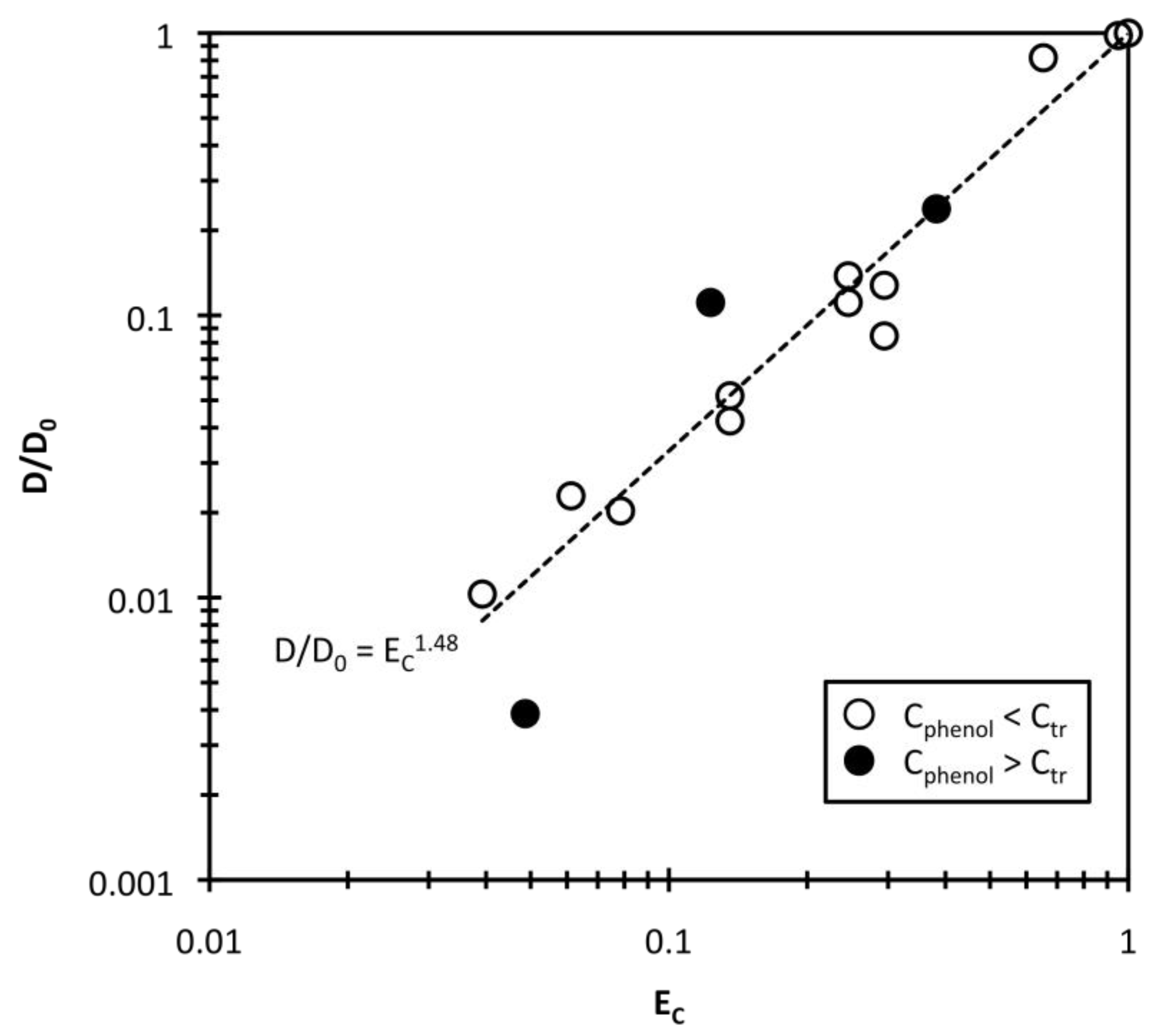
4. Conclusions
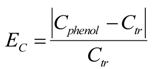
 , and that the exponent v = 1.48. The correlation will be useful as estimation of hydrogel response reduction associated with hydrophobic pollution.
, and that the exponent v = 1.48. The correlation will be useful as estimation of hydrogel response reduction associated with hydrophobic pollution.Acknowledgments
Conflicts of Interest
References
- Shin, J.; Braun, P.V.; Lee, W. Fast response photonic crystal pH sensor based on templated photo-polymerized hydrogel inverse opal. Sens. Actuators B 2010, 150, 183–190. [Google Scholar] [CrossRef]
- Lee, S.; Ibey, B.L.; Coté, G.L.; Pishko, M.V. Measurement of pH and dissolved oxygen within cell culture media using a hydrogel microarray sensor. Sens. Actuators B 2008, 128, 388–398. [Google Scholar] [CrossRef]
- Pi, S.; Ju, X.; Wu, H.; Xie, R.; Chu, L. Smart responsive microcapsules capable of recognizing heavy metal ions. J. Colloid Interface Sci. 2010, 349, 512–518. [Google Scholar] [CrossRef]
- Herber, S.; Olthuis, W.; Bergveld, P. A swelling hydrogel-based PCO2 sensor. Sens. Actuators B 2003, 91, 378–382. [Google Scholar] [CrossRef]
- Meskath, S.; Urban, G.; Heinzea, J. A new optochemical chlorine gas sensor based on the application of amphiphilic co-networks as matrices. Sens. Actuators B 2011, 151, 327–332. [Google Scholar] [CrossRef]
- Zguris, J.; Pishko, M.V. Nitric oxide sensitive fluorescent poly(ethylene glycol) hydrogel microstructures. Sens. Actuators B 2006, 115, 503–509. [Google Scholar] [CrossRef]
- Endo, T.; Yanagida, Y.; Hatsuzawa, T. Colorimetric detection of volatile organic compounds using a colloidal crystal-based chemical sensor for environmental applications. Sens. Actuators B 2007, 125, 589–595. [Google Scholar] [CrossRef]
- Yamagiwa, K.; Komi, T.; Kumakura, A.; Yokoyama, S.; Yoshida, M.; Ohkawa, A. Photo-crosslinked copolymer gel as an adsorbent for temperature-swing adsorption process. J. Chem. Eng. Jpn. 2004, 37, 1274–1278. [Google Scholar] [CrossRef]
- Yamagiwa, K.; Komi, T.; Yoshida, M.; Ohkawa, A.; Iida, T. Temperature-swing adsorption of nonionic surfactant with photo-crosslinked polymeric gel. J. Chem. Eng. Jpn. 2001, 34, 1171–1176. [Google Scholar] [CrossRef]
- Tokuyama, H.; Yanagawa, K.; Sakohara, S. Temperature swing adsorption of heavy metals on novel phosphate-type adsorbents using thermosensitive gels and/or polymers. Sep. Purif. Technol. 2006, 50, 8–14. [Google Scholar] [CrossRef]
- Tokuyama, H.; Kanehara, A. Temperature swing adsorption of gold(III) ions on poly(N-isopropylacrylamide) gel. React. Funct. Polym. 2007, 67, 136–143. [Google Scholar] [CrossRef]
- Takeshita, K.; Matsumura, T.; Nakao, Y. Separation of americium(III) and europium(III) by thermal-swing extraction using thermosensitive polymer gel. Prog. Nucl. Energy 2008, 50, 466–469. [Google Scholar] [CrossRef]
- Huber, D.L.; Manginell, R.P.; Samara, M.A.; Kim, B.; Bunker, B.C. Programmed adsorption and release of proteins in a microfluidic device. Science 2003, 301, 352–354. [Google Scholar] [CrossRef]
- Klouda, L.; Mikos, A.G. Thermoresponsive hydrogels in biomedical applications. Eur. J. Pharm. Biopharm. 2008, 68, 34–45. [Google Scholar] [CrossRef]
- Sershen, S.; West, J. Implantable, polymeric systems for modulated drug delivery. Adv. Drug Deliv. Rev. 2002, 54, 1225–1235. [Google Scholar] [CrossRef]
- Tokuyama, H.; Kanazawa, R.; Sakohara, S. Equilibrium and kinetics for temperature swing adsorption of a target metal on molecular imprinted thermosensitive gel adsorbents. Sep. Purif. Technol. 2005, 44, 152–159. [Google Scholar] [CrossRef]
- Tanaka, T.; Fillmore, J. Kinetics of swelling of gels. J. Chem. Phys. 1979, 70, 1214–1218. [Google Scholar] [CrossRef]
- Li, Y.; Tanaka, T. Kinetics of swelling and shrinking of gels. J. Chem. Phys. 1990, 92, 1365–1371. [Google Scholar] [CrossRef]
- Sato Matsuo, E.; Tanaka, T. Kinetics of discontinuous volume-phase transition of gels. J. Chem. Phys. 1988, 89, 1695–1703. [Google Scholar] [CrossRef]
- Kosik, K.; Wilk, E.; Geissler, E.; Laszlo, K. Interaction of phenols with thermo-responsive hydrogels. Colloids Surf. A Physicochem. Eng. Aspects 2008, 319, 159–164. [Google Scholar] [CrossRef]
- Tajima, H.; Yoshida, Y.; Yamagiwa, K. Experimental study of swelling and shrinking kinetics of spherical poly (N,N-diethylacrylamide) gel with continuous phase transition. Polymer 2011, 52, 732–738. [Google Scholar] [CrossRef]
- Tajima, H.; Yoshida, Y.; Abiko, S.; Yamagiwa, K. Size adjustment of spherical temperature-sensitive hydrogel beads by liquid–liquid dispersion using a Kenics static mixer. Chem. Eng. J. 2010, 156, 479–486. [Google Scholar] [CrossRef]
- Idziak, I.; Avoce, D.; Lessaed, D.; Grabel, D.; Zhu, X.X. Thermosensitivity of aqueous solutions of poly(N,N-diethylacrylamide). Macromolecules 1999, 32, 1260–1263. [Google Scholar] [CrossRef]
- Kawashima, T.; Koga, S.; Annaka, M.; Sasaki, S. Roles of hydrophobic interaction in a volume transition of alkylacrylamide gel induced by the hydrogen-bond-driving alkylphenol binding. J. Phys. Chem. B 2005, 109, 1055–1062. [Google Scholar] [CrossRef]
- Koga, S.; Kawashima, T.; Sasaki, S. Elastic relaxation of collapsed poly(alkylacrylamide) gels and their complexes with phenol. J. Phys. Chem. B 2004, 108, 10838–10844. [Google Scholar] [CrossRef]
- Suzuki, Y.; Suzuki, N.; Takasu, Y.; Nishio, I. A study on the structure of water in an aqueous solution by the solvent effect on a volume phase transition of N-isopropylacrylamide gel and low-frequency Raman spectropy. J. Chem. Phys. 1997, 107, 5890–5897. [Google Scholar] [CrossRef]
- Laszio, K.; Kosik, K.; Rochas, C.; Geissler, E. Phase transition in Poly (N-isopropylacrylamide) hydrogels induced by phenols. Macromolecules 2003, 36, 7771–7776. [Google Scholar] [CrossRef]
- Kosik, K.; Geissler, E.; Zrinyi, M.; Laszlo, K. Interaction of non-ionic hydrogels with small aromatic molecules. Polym. Adv. Technol. 2003, 14, 771–775. [Google Scholar] [CrossRef]
- Takahashi, K.; Takigawa, T.; Masuda, T. Swelling and deswelling kinetics of poly(N-isopropylacrylamide) gels. J. Chem. Phys. 2004, 120, 2972–2979. [Google Scholar] [CrossRef] [Green Version]
- Tokita, M.; Tanaka, T. Reversible decrease of gel-solvemt friction. Science 1991, 253, 1121–1123. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tajima, H.; Sato, F.; Yamagiwa, K. Effect of Hydrophobic Pollution on Response of Thermo-Sensitive Hydrogel. Chemosensors 2013, 1, 21-32. https://doi.org/10.3390/chemosensors1030021
Tajima H, Sato F, Yamagiwa K. Effect of Hydrophobic Pollution on Response of Thermo-Sensitive Hydrogel. Chemosensors. 2013; 1(3):21-32. https://doi.org/10.3390/chemosensors1030021
Chicago/Turabian StyleTajima, Hideo, Fumiaki Sato, and Kazuaki Yamagiwa. 2013. "Effect of Hydrophobic Pollution on Response of Thermo-Sensitive Hydrogel" Chemosensors 1, no. 3: 21-32. https://doi.org/10.3390/chemosensors1030021
APA StyleTajima, H., Sato, F., & Yamagiwa, K. (2013). Effect of Hydrophobic Pollution on Response of Thermo-Sensitive Hydrogel. Chemosensors, 1(3), 21-32. https://doi.org/10.3390/chemosensors1030021



