Human Factors and Human-Computer Considerations in Teleradiology and Telepathology
Abstract
:1. Introduction
2. Medical Image Displays and Perceptual Processes Underlying Image Interpretation
2.1. Perception Basics
2.2. Implications for Choosing Displays
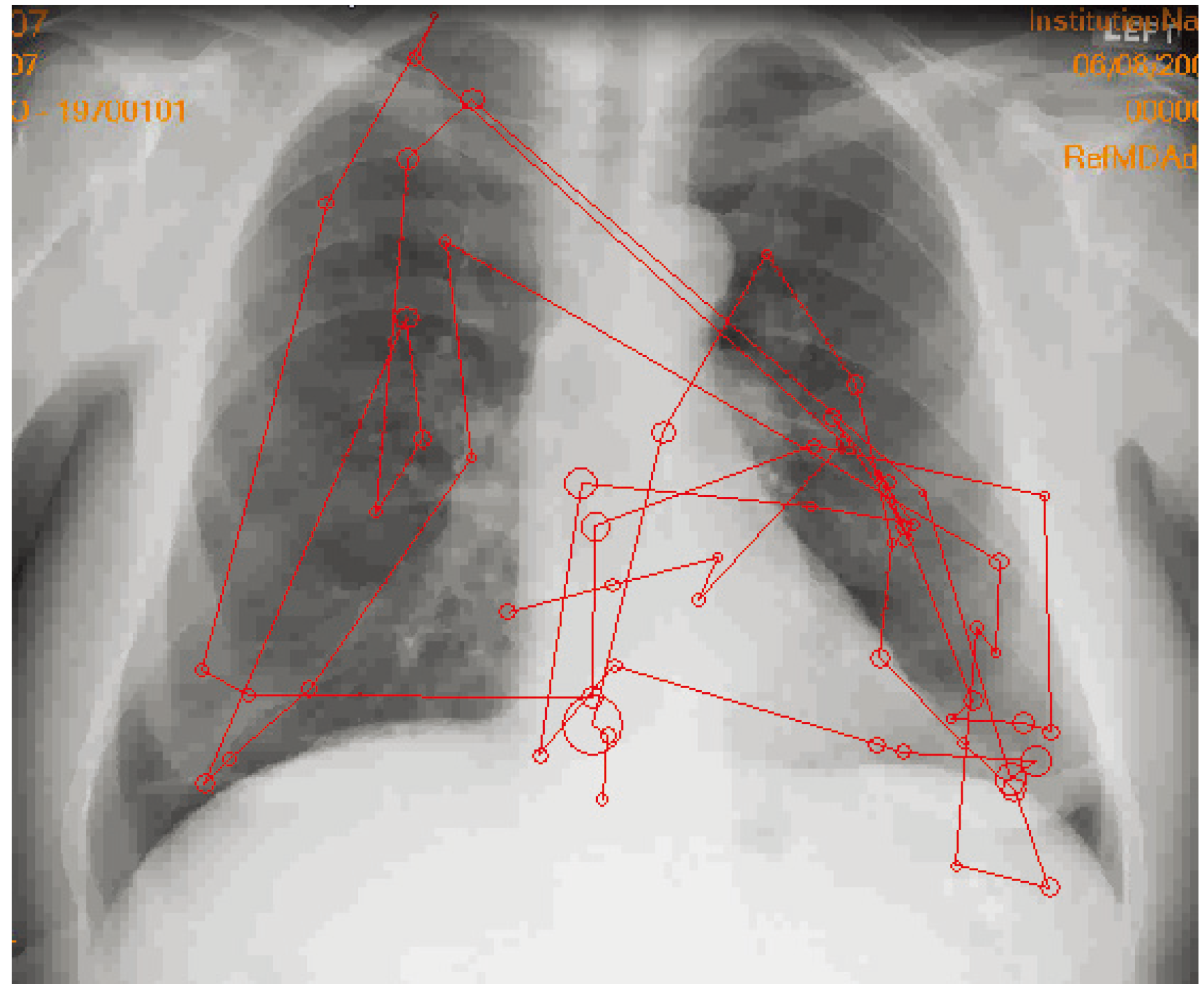
3. Display Calibration
3.1. Grayscale Calibration
3.2. Color Calibration
3.3. Calibration Tools
5. Reading Environment
5.1. Understanding Workflow
5.2. Input Devices
5.3. Other Environmental Considerations
5.4. Software Tools and Image Presentation
6. Fatigue in the Digital Reading Environment
6.1. Why Study Fatigue?
6.2. Steps to Avoid Fatigue
7. Mobile Devices and SmartPhones
7.1. Are Mobile Devices Adequate to Use Clinically?
7.2. Other Options on the Horizon
8. Conclusions
Conflicts of Interest
References
- Silva, E.; Breslau, J.; Barr, R.M.; Liebscher, L.A.; Bohl, M.; Hoffman, T.; Boland, G.W.L.; Sherry, C.; Kim, W.; Shah, S.S.; et al. ACR white paper on teleradiology practice: A report from the task force on teleradiology practice. J. Am. Coll. Radiol. 2013, 10, 575–585. [Google Scholar] [CrossRef]
- European Society of Radiology. The future role of radiology in healthcare. Insights Imaging 2010, 1, 2–11. [Google Scholar]
- Barisoni, L.; Nast, C.C.; Jennette, J.C.; Hodgin, J.B.; Herzenberg, A.M.; Lemley, K.V.; Conway, C.M.; Kopp, J.B.; Kretzler, M.; Lienczewski, C.; et al. Digital pathology evaluation ion the multicenter Nephrotic Syndrome Study Network (NEPTUNE). Clin. J. Am. Soc. Nephrol. 2013, 8, 1449–1459. [Google Scholar] [CrossRef]
- Ghaznavi, F.; Evans, A.; Madabhushi, A.; Feldman, M. Digital imaging in pathology: Whole-slide imaging and beyond. Annu. Rev. Pathol. 2013, 8, 331–359. [Google Scholar] [CrossRef]
- Lauro, G.R.; Cable, W.; Lesniak, A.; Tseytlin, E.; McHugh, J.; Parwani, A.; Pantanowitz, L. Digital pathology consultations—A new era in digital imaging, challenges and practical applications. J. Digit. Imaging 2013, 26, 668–677. [Google Scholar] [CrossRef]
- Al-Janabi, S.; Huisman, A.; van Diest, P.J. Digital pathology: Current status and future perspectives. Histopathology 2012, 61, 1–9. [Google Scholar] [CrossRef]
- Pantanowitz, L.; Wiley, C.A.; Demetris, A.; Lesniak, A.; Ahmed, I.; Cable, W.; Contis, L.; Parwani, A.V. Experience with multimodality telepathology at the University of Pittsburgh Medical Center. J. Pathol. Inform. 2012, 3, 45. [Google Scholar] [CrossRef]
- Stathonikos, N.; Veta, M.; Huisman, A.; van Diest, P.J. Going fully digital: Perspective of a Dutch academic pathology lab. J. Pathol. Inform. 2013, 4, 15. [Google Scholar] [CrossRef]
- Bautista, P.A.; Yagi, Y. Improving the visualization and detection of tissue folds in whole slide images through color enhancement. J. Pathol. Inform. 2010, 1, 25. [Google Scholar] [CrossRef]
- Cooper, L.A.D.; Carter, A.B.; Farris, A.B.; Wang, F.; Kong, J.; Gutman, D.A.; Widener, P.; Pan, T.C.; Cholleti, S.R.; Sharma, A.; et al. Digital pathology: Data-intensive frontier in medical imaging. IEEE. Proc. 2012, 100, 991–1003. [Google Scholar] [CrossRef]
- Gavrilovic, M.; Azar, J.C.; Lindblad, J.; Wahlby, C.; Bengtsson, E.; Busch, C.; Carlbom, I.B. Blind color decomposition of histological images. IEEE Trans. Med. Imaging 2013, 32, 983–994. [Google Scholar] [CrossRef]
- Ishikawa, M.; Ahi, S.T.; Murakami, Y.; Kimura, F.; Yamaguchi, M.; Abe, T.; Akinori, H.; Michiie, S. Automatic segmentation of hepatocellular structure from HE-stained liver tissue. In Proceedings of the SPIE Medical Imaging, Lake Buena Vista (Orlando Area), FL, USA, 9–14 February 2013; Volume 8676.
- Bahlmann, C.; Patel, A.H.; Johnson, J.P.; Ni, J.; Chekkoury, A.; Khurd, P.K.; Kamen, A.; Grady, L.; Krupinski, E.A.; Graham, A.R.; et al. Automated detection of diagnostically relevant regions in H&E stained digital pathology slides. In Proceedings of the SPIE Medical Imaging, San Diego, CA, USA, 4–9 February 2012; Volume 8315.
- Chekkoury, A.; Khurd, P.; Ni, J.; Bahlmann, C.; Kamen, A.; Patel, A.; Leo, G.; Maneesh, S.; Martin, G.; Nassir, N.; et al. Automated malignancy detection in breast histopathological images. In Proceedings of the SPIE Medical Imaging, San Diego, CA, USA, 4–9 February 2012; Volume 8315.
- Yagi, Y. Color standardization and optimization in Whole Slide Imaging. Diagn. Pathol. 2011, 6, S15. [Google Scholar]
- Cheng, W.C.; Keay, T.; O’Flaherty, N.; Wang, J.; Ivansky, A.; Gavrielides, M.A.; Gallas, B.D.; Badano, A. Assessing color reproducibility of whole-slide imaging scanners. In Proceedings of the SPIE Medical Imaging, Lake Buena Vista (Orlando Area), FL, USA, 9–14 February 2013; Volume 8676.
- Rojo, M.G.; Garcia, G.B.; Mateos, C.P.; Garcia, J.G.; Vicente, M.C. Critical comparison of 31 commercially available digital slide systems in pathology. Int. J. Surg. Pathol. 2006, 14, 285–305. [Google Scholar] [CrossRef]
- Rojo, M.G.; Daniel, C.; Schrader, T. Standardization efforts of digital pathology in Europe. Anal. Cell. Pathol. 2012, 35, 19–23. [Google Scholar]
- ICC Summit on Color in Medical Imaging at the FDA, 8–9 May 2013. Available online: http://www.fda.gov/MedicalDevices/NewsEvents/WorkshopsConferences/ucm342138.htm#webcast/ (accessed on 2 January 2014).
- Lowe, J. Telepathology: Guidance from The Royal College of Pathologists. Available online: http://www.rcpath.org/Resources/RCPath/Migrated%20Resources/Documents/G/G026_Telepathology_Oct13.pdf (accessed on 2 January 2014).
- Canadian Association of Pathologists Telepathology Guidelines. Available online: http://www.cap-acp.org/cmsUploads/CAP/File/Telepathology_Guidelines_Final_v_13.pdf (accessed on 2 January 2014).
- American Telemedicine Association Clinical Guidelines for Telepathology. Available online: http://www.americantelemed.org/practice/standards/ata-standards-guidelines/clinical-guidelines-for-telepathology (accessed on 2 January 2014).
- Pantanowitz, L.; Valenstein, P.N.; Evans, A.J.; Kaplan, K.J.; Pfeifer, J.D.; Wilbur, D.C.; Collins, L.C.; Colgan, T.J. Review of the current state of whole slide imaging in pathology. J. Pathol. Inform. 2011, 2, 36. [Google Scholar] [CrossRef]
- Collins, B.T. Telepathology in cytopathology: Challenges and opportunities. Acta Cytol. 2013, 57, 221–232. [Google Scholar] [CrossRef]
- Carter, A.B. Stepping across borders into the future of telepathology. J. Pathol. Inform. 2011, 2, 24. [Google Scholar] [CrossRef]
- Weinstein, R.S.; Graham, A.R.; Lian, F.; Braunhut, B.L.; Barker, G.R.; Krupinski, E.A.; Bhattacharyya, A.K. Reconciliation of diverse telepathology system designs. Historic issues and implications for emerging markets and new applications. APMIS 2012, 120, 256–275. [Google Scholar] [CrossRef]
- Bellis, M.; Metias, S.; Naugler, C.; Pollett, A.; Jothy, S.; Yousef, G.M. Digital pathology: Attitudes and practices in the Canadian pathology community. J. Pathol. Inform. 2013, 4, 3. [Google Scholar]
- Pospislova, E.; Cernochova, D.; Lichovska, R.; Erdosova, B.; Krajci, D. Application and evaluation of teaching practical histology with the use of virtual microscopy. Diagn. Pathol. 2013, 8, S7. [Google Scholar]
- Gabril, M.Y.; Yousef, G.M. Informatics for practicing anatomical pathologists: Marking a new era in pathology practice. Mod. Pathol. 2010, 23, 349–358. [Google Scholar] [CrossRef]
- Kundel, H.; Nodine, C. A short history of image perception in medical radiology. In The Handbook of Medical Image Perception and Techniques; Samei, E., Krupinski, E., Eds.; Cambridge University Press: London, UK, 2010; pp. 9–20. [Google Scholar]
- Krupinski, E.; Samei, E. Future of medical image perception. In The Handbook of Medical Image Perception and Techniques; Samei, E., Krupinski, E., Eds.; Cambridge University Press: London, UK, 2010; pp. 413–416. [Google Scholar]
- Krupinski, E. Perceptual factors in reading medical images. In The Handbook of Medical Image Perception and Techniques; Samei, E., Krupinski, E., Eds.; Cambridge University Press: London, UK, 2010; pp. 81–90. [Google Scholar]
- Saunders, R.; Samei, E. Image quality and its perceptual relevance. In The Handbook of Medical Image Perception and Techniques; Samei, E., Krupinski, E., Eds.; Cambridge University Press: London, UK, 2010; pp. 157–164. [Google Scholar]
- Krupinski, E.; Roehrig, H. Optimization of display systems. In The Handbook of Medical Image Perception and Techniques; Samei, E., Krupinski, E., Eds.; Cambridge University Press: London, UK, 2010; pp. 395–405. [Google Scholar]
- Forrester, J.; Dick, A.; McMenamin, P.; Lee, W. The Eye: Basic Sciences in Practice; WB Saunders: London, UK, 1996. [Google Scholar]
- Norweck, J.T.; Seibert, J.A.; Andriole, K.P.; Clunie, D.A.; Curran, B.H.; Flynn, M.J.; Krupinski, E.; Lieto, R.P.; Peck, D.J.; Mian, T.A.; et al. ACR-AAPM-SIIM technical standard for electronic practice of medical imaging. J. Digit. Imaging 2013, 26, 38–52. [Google Scholar] [CrossRef]
- Digital Imaging and Communications in Medicine (DICOM) Part 14: Grayscale Standard Display Function. Available online: http://medical.nema.org/dicom/2006/06_14pu.pdf (accessed on 3 January 2014).
- Krupinski, E.A.; Roehrig, H. The influence of a perceptually linearized display on observer performance and visual search. Acad. Radiol. 2000, 7, 8–13. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Silverstein, L.D.; Hashmi, S.F.; Graham, A.R.; Weinstein, R.S.; Roehrig, H. Observer performance using virtual pathology slides: Impact of LCD color reproduction accuracy. J. Digit. Imaging 2013, 25, 738–743. [Google Scholar]
- Weinstein, R.S.; Graham, A.R.; Richter, L.C.; Barker, G.P.; Krupinski, E.A.; Lopez, A.M.; Erps, K.A.; Bhattacharyya, A.H.; Yagi, Y.; Gilbertson, J.R. Overview of telepathology, virtual microscopy, and whole slide imaging: Prospects for the future. Hum. Pathol. 2009, 40, 1057–1069. [Google Scholar] [CrossRef]
- Silverstein, L.D.; Hashmi, S.F.; Lang, K.; Krupinski, E.A. Paradigm for achieving color-reproduction accuracy in LCDs for medical imaging. J. Soc. Inf. Disp. 2012, 20, 53–62. [Google Scholar] [CrossRef]
- Llewellyn-Thomas, E.; Lansdown, E.L. Visual search patterns of radiologists in training. Radiology 1963, 81, 288–291. [Google Scholar]
- Kundel, H.L.; Nodine, C.F.; Carmody, D.P. Visual scanning, pattern recognition and decision-making in pulmonary tumor detection. Invest. Radiol. 1978, 13, 175–181. [Google Scholar] [CrossRef]
- Kundel, H.L.; Nodine, C.F.; Krupinski, E.A. Searching for lung nodules: Visual dwell indicates locations of false-positive and false-negative decisions. Invest. Radiol. 1989, 24, 472–478. [Google Scholar] [CrossRef]
- Nodine, C.F.; Mello-Thoms, C.; Kundel, H.L.; Weinstein, S.P. Time course of perception and decision making during mammographic interpretation. Am. J. Roentgenol. 2002, 179, 917–923. [Google Scholar] [CrossRef]
- Krupinski, E.A. Visual scanning patterns of radiologists searching mammograms. Acad. Radiol. 1996, 3, 137–144. [Google Scholar] [CrossRef]
- Nodine, C.F.; Kundel, H.L.; Lauver, S.C.; Toto, L.C. Nature of expertise in searching mammograms for breast masses. Acad. Radiol. 1996, 3, 1000–1006. [Google Scholar] [CrossRef]
- Krupinski, E.A. Visual search of mammographic images: Influence of lesion subtlety. Acad. Radiol. 2005, 12, 965–969. [Google Scholar] [CrossRef]
- Lesgold, A.M.; Rubinson, H.; Feltovich, P.; Glaser, R.; Klopfer, D.; Wang, Y. Expertise in a complex skill: Diagnosing X-ray pictures. In The Nature of Expertise; Chi, M.T.H., Glaser, R., Farr, M.J., Eds.; Erlbaum Publishers: Hillsdale, NJ, USA, 1988; pp. 311–342. [Google Scholar]
- Krupinski, E.A.; Roehrig, H.; Furukawa, T. Influence of film and monitor display luminance on observer performance and visual search. Acad. Radiol. 1999, 6, 411–418. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Roehrig, H. Pulmonary nodule detection and visual search: P45 and P104 monochrome vs. color monitor displays. Acad. Radiol. 2002, 9, 638–645. [Google Scholar] [CrossRef]
- Tiersma, E.S.M.; Peters, A.A.W.; Mooij, H.A.; Fleuren, G.J. Visualizing scanning patterns of pathologists in the grading of cervical intraepithelial neoplasia. J. Clin. Pathol. 2003, 56, 677–680. [Google Scholar] [CrossRef]
- Roa-Pena, L.; Gomez, F.; Romero, F. An experimental study of pathologist’s navigation patterns in virtual microscopy. Diagn. Pathol. 2010, 5, 71. [Google Scholar] [CrossRef]
- Lund, P.J.; Krupinski, E.A.; Pereles, S.; Mockbee, B. Comparison of conventional and computer radiography: Assessment of image quality and reader performance in skeletal extremity trauma. Acad. Radiol. 1997, 4, 570–576. [Google Scholar] [CrossRef]
- Nodine, C.F.; Mello-Thoms, C. The role of expertise in radiologic image interpretation. In The Handbook of Medical Image Perception and Techniques; Samei, E., Krupinski, E., Eds.; Cambridge University Press: London, UK, 2010; pp. 139–156. [Google Scholar]
- Prak, E.T.L.; Young, D.S.; Kamoun, M.; Nachamkin, I.; Alexander, C.B.; Spitalnik, S.L.; Peerschke, E.I.; Smith, B.R. 2008 ACLPS panel discussion on resident education in clinical pathology. Am. J. Clin. Pathol. 2009, 131, 618–622. [Google Scholar] [CrossRef]
- Ford, J.C.; Pinder, K.E.; Ovalle, W.K.; Li, C.H. Pathology education in a multisite urban/rural distributed curriculum. Hum. Pathol. 2008, 39, 811–816. [Google Scholar] [CrossRef]
- Weiss, R.L.; McKenna, B.J.; Lord-Toof, M.; Thompson, N.N.; Work Group Members. A consensus curriculum for laboratory management training for pathology residents. Am. J. Clin. Pathol. 2011, 136, 671–678. [Google Scholar] [CrossRef]
- Alexander, C.B. Pathology graduate medical education (overview from 2006–2010). Hum. Pathol. 2011, 42, 763–769. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Tillack, A.A.; Richter, L.; Henderson, J.T.; Bhattacharyya, A.K.; Scott, K.M.; Graham, A.R.; Descour, M.R.; Davis, J.R.; Weinstein, R.S. Eye-movement study and human performance using telepathology virtual slides. Implications for medical education and differences with experience. Hum. Pathol. 2006, 37, 1543–1556. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Graham, A.R.; Weinstein, R.S. Characterizing the development of visual search expertise in pathology residents viewing whole slide images. Hum. Pathol. 2013, 44, 357–364. [Google Scholar] [CrossRef]
- Pantanowitz, L.; Parawni, A.; Tseytlin, E.; Mello-Thoms, C. Analysis of slide exploration of cytologists when reading digital slides. In Proceedings of the SPIE Medical Imaging, San Diego, CA, USA, 4–9 February 2012; Volume 8318.
- Haygood, T.M.; Arribas, E.; Brennan, P.C.; Atkinson, E.N.; Herndon, M.; Dieber, J.; Geiser, W.; Santiago, L.; Mills, C.M.; Davis, P.; et al. Conspicuity of microcalcifications on digital screening mammograms using varying degrees of monitor zooming. Acad. Radiol. 2009, 16, 1509–1517. [Google Scholar] [CrossRef]
- Krupinski, E.A. Optimizing the pathology workstation “cockpit”: Challenges and solutions. J. Pathol. Inform. 2010, 1, 19. [Google Scholar] [CrossRef]
- Randell, R.; Ruddle, R.A.; Thomas, R.; Treanor, D. Diagnosis at the microscope: A workplace study of histopathology. Cogn. Technol. Work 2012, 14, 319–335. [Google Scholar] [CrossRef]
- Randell, R.; Ruddle, R.A.; Quirke, P.; Thomas, R.G.; Treanor, D. Working at the microscope: Analysis of activities involved in diagnostic pathology. Histopathology 2012, 60, 504–510. [Google Scholar] [CrossRef]
- Ho, J.; Aridor, O.; Parwani, A.V. Use of contextual inquiry to understand anatomic pathology workflow: Implications for digital pathology adoption. J. Pathol. Inform. 2012, 3, 35. [Google Scholar] [CrossRef]
- Lindskold, L.; Wintell, M.; Aspelin, P.; Lundberg, N. Simulation of radiology workflow and throughput. Radiol. Manag. 2012, 34, 47–55. [Google Scholar]
- Benjamin, M.; Aradj, Y.; Shreiber, R. From shared data to sharing workflow: Merging PACS and teleradiology. Eur. J. Radiol. 2010, 73, 3–9. [Google Scholar] [CrossRef]
- Ruess, L.; O’Connor, S.C.; Cho, K.H.; Hussain, F.H.; Howard, W.J.; Slaughter, R.C.; Hedge, A. Carpal tunnel syndrome and cubital tunnel syndrome: Work-related musculoskeletal disorders in four symptomatic radiologists. Am. J. Roentgenol. 2003, 181, 37–42. [Google Scholar] [CrossRef]
- Yagi, Y.; Yoshioka, S.; Kyusojin, H.; Onozato, M.; Mizutani, Y.; Osato, K.; Yada, H.; Mark, E.J.; Frosch, M.P.; Louis, D.N. An ultra-high speed whole slide image viewing system. Anal. Cell. Pathol. 2012, 35, 65–73. [Google Scholar]
- Tan, J.H.; Chao, C.; Zawaideh, M.; Roberts, A.C.; Kinney, T.B. Informatics in radiology: Developing a touchless user interface for interoperative image control during interventional radiology procedures. Radiographics 2013, 33, 61–70. [Google Scholar] [CrossRef]
- Fares, R.; Fang, S.; Komogortsev, O. Can we beat the mouse with MAGIC? In Proceedings of the SIGCHI Conference on Human Factors in Computing Systems, New York, NY, USA, 2013; pp. 1387–1390.
- Brennan, P.C.; McEntee, M.; Evanoff, M.; Phillips, P.; O’Connor, W.T.; Manning, D.J. Ambient lighting: Effect of illumination on soft-copy viewing of radiographs of the wrist. Am. J. Roentgenol. 2007, 188, 177–180. [Google Scholar] [CrossRef]
- Moise, A.; Atkins, S. Designing better radiology workstations: Impact of two user interfaces on interpretation errors and user satisfaction. J. Digit. Imaging 2005, 18, 109–115. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Radvany, M.; Levy, A.; Balenger, D.; Tucker, J.; Chacko, A.; VanMetter, R. Enhanced visualization processing: Effect on workflow. Acad. Radiol. 2001, 8, 1127–1133. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Berbaum, K.S.; Caldwell, R.T.; Schartz, K.M.; Kim, J. Long radiology workdays reduce detection accommodation accuracy. J. Am. Coll. Radiol. 2010, 7, 698–704. [Google Scholar] [CrossRef]
- Krupinski, E.A.; Berbaum, K.S.; Caldwell, R.T.; Schartz, K.M.; Madsen, M.T.; Kramer, D.J. Do long radiology workdays affect nodule detection in dynamic CT interpretation? J. Am. Coll. Radiol. 2012, 9, 191–198. [Google Scholar] [CrossRef]
- Rosenfield, M. Computer vision syndrome: A review of ocular causes and potential treatments. Ophthalmic Physiol. Opt. 2011, 31, 502–515. [Google Scholar] [CrossRef]
- Krupinski, E.A. Potential method for relieving fatigue in radiologists. In Proceedings of the SPIE Medical Imaging, Lake Buena Vista (Orlando Area), FL, USA, 9–14 February 2013; Volume 8673.
- Food & Drug Administration Mobile Medical Applications Guidance. Issued September 25, 2013. Available online: http://www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/UCM263366.pdf (accessed on 3 January 2014).
- Modi, J.; Sharma, P.; Earl, A.; Simpson, M.; Mitchell, J.R.; Goyal, M. iPhone-based teleradiology for the diagnosis of acute cervico-dorsal spine trauma. Can. J. Neurol. Sci. 2010, 37, 849–854. [Google Scholar]
- Toomey, R.J.; Ryan, J.T.; McEntee, M.F.; Evanoff, M.G.; Chakraborty, D.P.; McNulty, J.P.; Manning, D.J.; Thomas, E.M.; Brennan, P.C. Diagnostic efficacy of handheld devices for emergency radiologic consultation. Am. J. Roentgenol. 2010, 194, 469–474. [Google Scholar] [CrossRef]
- Johnson, P.T.; Zimmerman, S.L.; Heath, D.; Eng, J.; Horton, K.M.; Scott, W.W.; Fishman, E.K. The iPad as a mobile device for CT display and interpretation: Diagnostic accuracy for identification of pulmonary embolism. Emerg. Radiol. 2012, 19, 323–327. [Google Scholar] [CrossRef]
- Bhatia, A.; Patel, S.; Pantol, G.; Wu, Y.Y.; Plitnikas, M.; Hancock, C. Intra and inter-observer reliability of mobile tablet PACS viewer system vs. standard PACS viewing station-diagnosis of acute nervous system events. Open J. Radiol. 2013, 3, 91–98. [Google Scholar] [CrossRef]
- Abboud, S.; Weiss, F.; Siegel, E.; Jeudy, J. TB or not TB: Interreader and intrareader variability in screening diagnosis on an iPad vs. a traditional display. J. Am. Coll. Radiol. 2013, 10, 42–44. [Google Scholar] [CrossRef]
- Rodrigues, M.A.; Visvanathan, A.; Murchison, J.T.; Brady, R.R. Radiology smartphone applications; current provision and caution. Insights Imaging 2013, 4, 555–562. [Google Scholar] [CrossRef]
- Ege, T.; Kose, O.; Koca, K.; Demiralp, B.; Basbozkurt, M. Use of the iPhone for radiographic evaluation of the hallux valgus. Skelet. Radiol. 2013, 42, 269–273. [Google Scholar] [CrossRef]
- John, S.; Poh, A.C.C.; Lim, T.C.C.; Chan, E.H.Y.; Chong, L.R. The iPad tablet computer for mobile on-call radiology diagnosis? Auditing discrepancy in CT and MRI reporting. J. Digit. Imaging 2012, 25, 628–634. [Google Scholar] [CrossRef]
- Panughpath, S.G.; Kalyanpur, A. Radiology and the mobile device: Radiology in motion. Indian J. Radiol. Imaging 2012, 22, 246–250. [Google Scholar] [CrossRef]
- Navruz, I.; Coskun, A.F.; Wong, J.; Mohammad, S.; Tseng, D.; Nagi, R.; Phillips, S.; Ozcan, A. Smart-phone based computational microscopy using multi-frame contact imaging on a fiber-optic array. Lab Chip 2013, 13, 4015–4023. [Google Scholar] [CrossRef]
- Speiser, J.J.; Hughes, I.; Mehta, V.; Wojcik, E.M.; Hutchens, K.A. Mobile teledermatopathology: Using a tablet PC as a novel and cost-efficient method to remotely diagnose dermatopathology cases. Am. J. Dermatopathol. 2014, 36, 54–57. [Google Scholar]
- Treanor, D.; Jordan-Owers, N.; Hodrien, J.; Wood, J.; Quirke, P.; Ruddle, R.A. Virtual reality Powerwall vs. conventional microscope for viewing pathology slides: An experimental comparison. Histopathology 2009, 55, 294–300. [Google Scholar] [CrossRef]
- Krupinski, E.; Roehrig, H.; Berger, W.; Dalal, S.; Stanton, D. Potential use of a large-screen display for interpreting radiographic images. In Proceedings of the SPIE Medical Imaging, San Diego, CA, USA, 11 February 2006; Volume 6146.
- Reiner, B. One size (doesn’t) fit all. J. Am. Coll. Radiol. 2008, 4, 567–570. [Google Scholar] [CrossRef]
- Reiner, B.I.; Krupinski, E. Demystifying occupational stress and fatigue through the creation of adaptive end-user profiling system. J. Digit. Imaging 2012, 25, 201–205. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Krupinski, E.A. Human Factors and Human-Computer Considerations in Teleradiology and Telepathology. Healthcare 2014, 2, 94-114. https://doi.org/10.3390/healthcare2010094
Krupinski EA. Human Factors and Human-Computer Considerations in Teleradiology and Telepathology. Healthcare. 2014; 2(1):94-114. https://doi.org/10.3390/healthcare2010094
Chicago/Turabian StyleKrupinski, Elizabeth A. 2014. "Human Factors and Human-Computer Considerations in Teleradiology and Telepathology" Healthcare 2, no. 1: 94-114. https://doi.org/10.3390/healthcare2010094
APA StyleKrupinski, E. A. (2014). Human Factors and Human-Computer Considerations in Teleradiology and Telepathology. Healthcare, 2(1), 94-114. https://doi.org/10.3390/healthcare2010094



