Detection and Discrimination of Non-Melanoma Skin Cancer by Multimodal Imaging
Abstract
:1. Introduction
2. Experimental
2.1. Sample Preparation
2.2. Experimental Setup
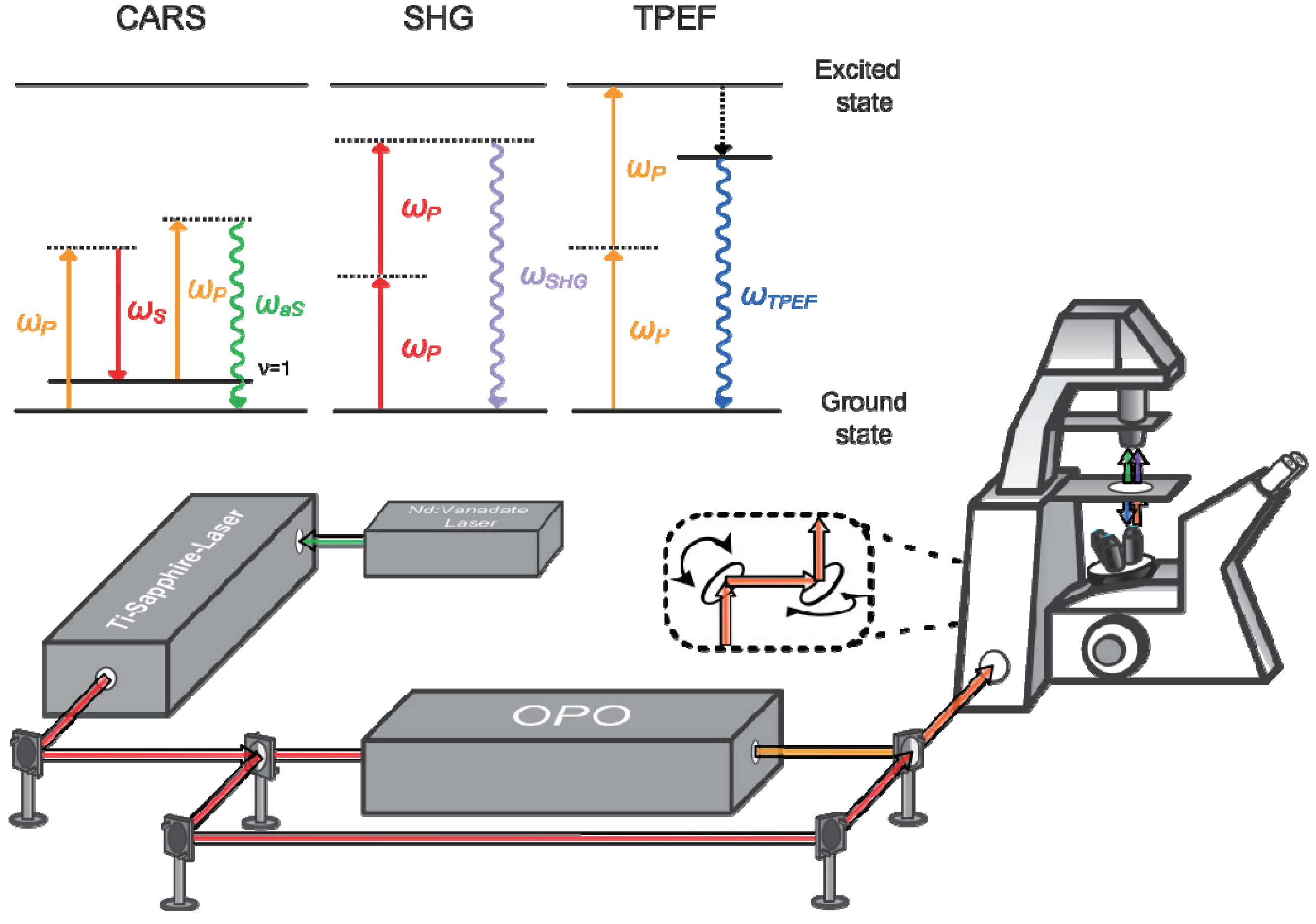
| Configuration | Excitation source | Central wavelength | FWHM/nm | Average power at the sample/mW | Peak irradiance at the sample/(W/cm2) |
|---|---|---|---|---|
| 1st | ||||
| CARS @ 2,850 cm−1 | OPO (Pump) | ~671 | 0.6 | ~20 | ≈9 × 109 |
| TPEF @ 435–485 nm | Ti: sapphire | ~830 | 0.5 | ~20 | ≈6 × 109 |
| (Stokes) | ||||
| 2nd | ||||
| SHG @ 415 nm | Ti: sapphire | ~830 | 0.5 | ~80 | ≈2 × 1010 |
2.3. Statistical Analysis
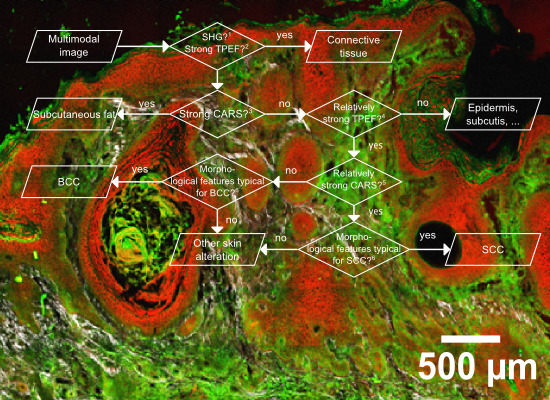
3. Results
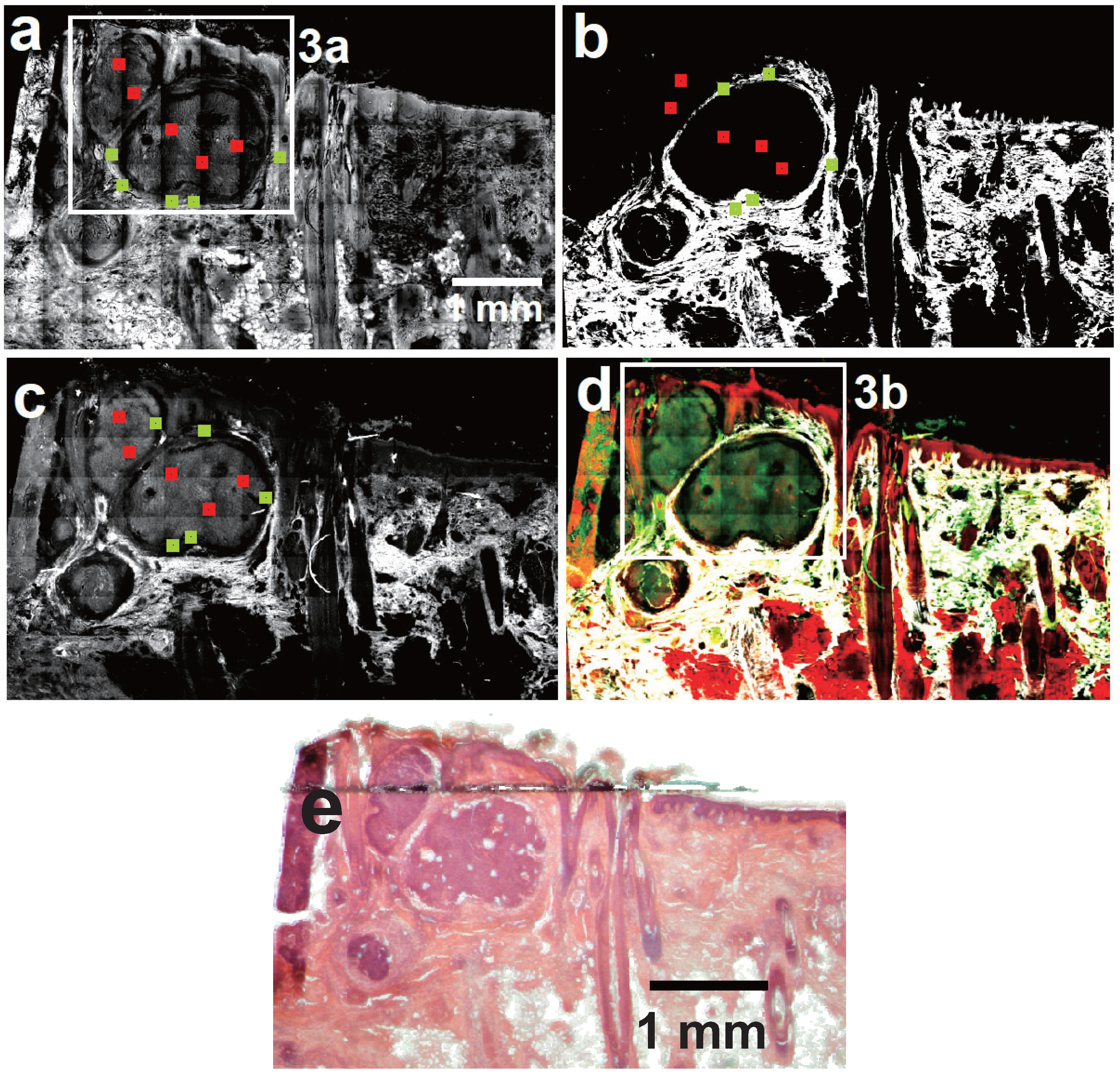
3.1. Basal Cell Carcinoma

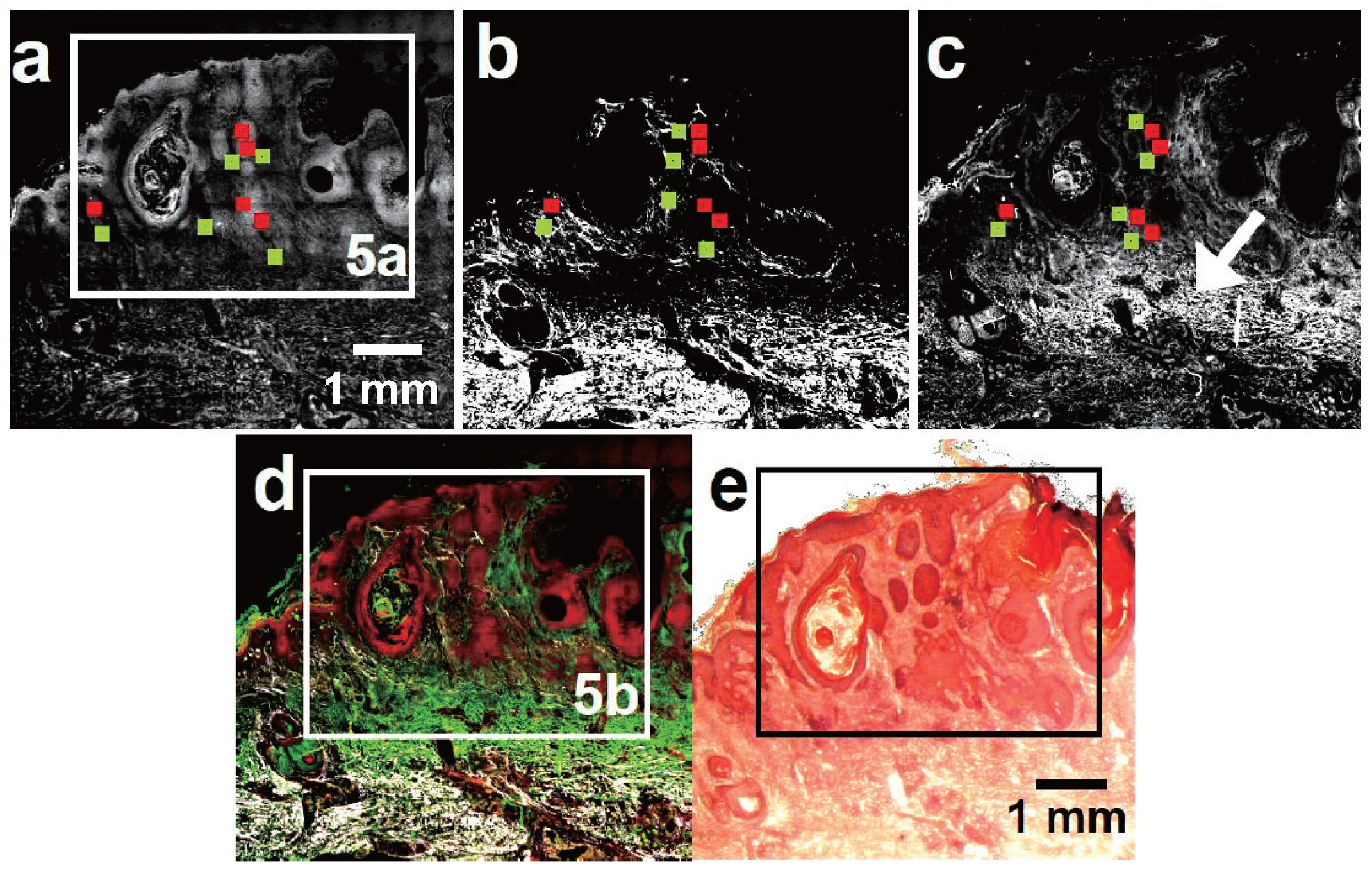
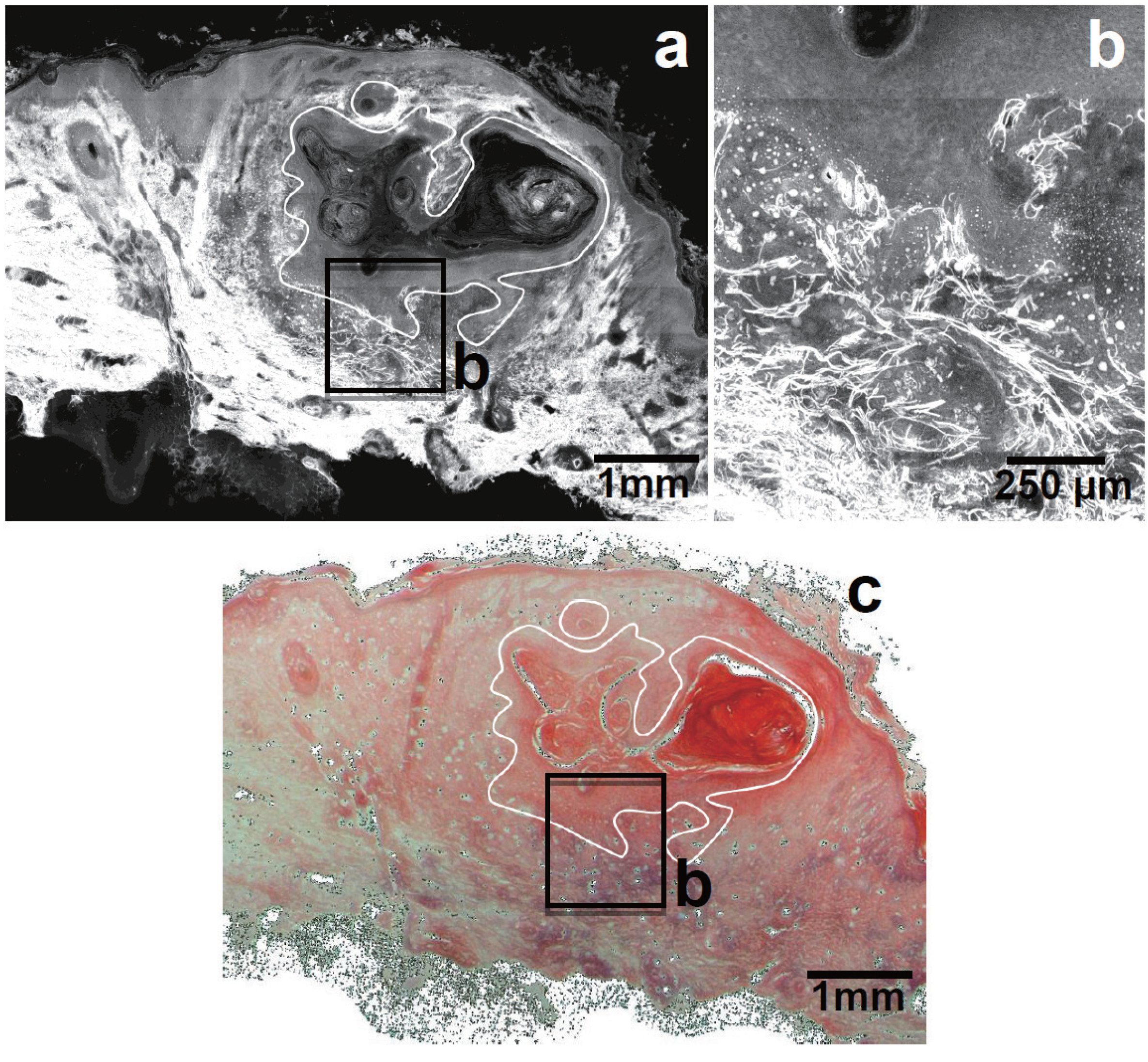
3.2. Squamous Cell Carcinoma
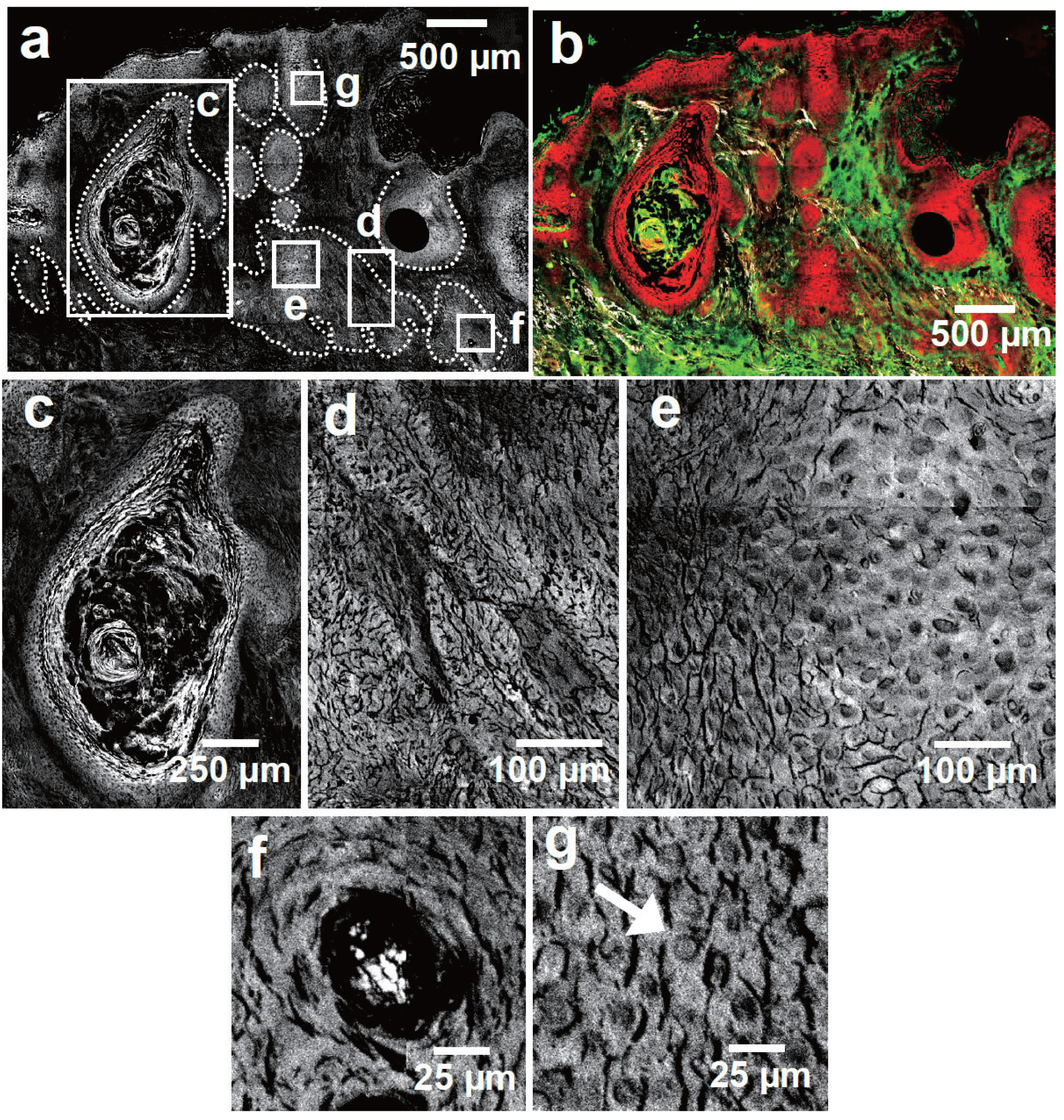
4. Discussion
| p-values (H0) | CARS | SHG | TPEF |
|---|---|---|---|
| BCC | 2.086 × 10−7 (H0: µ < 0) | 2.98 × 10−8 (H0: µ <0) | 2.98 × 10−8 (H0: µ > 0) |
| SCC | 9.711 × 10−6 (H0: µ > 0) | 2.98 × 10−8 (H0: µ <0) | 2.98 × 10−8 (H0: µ > 0) |
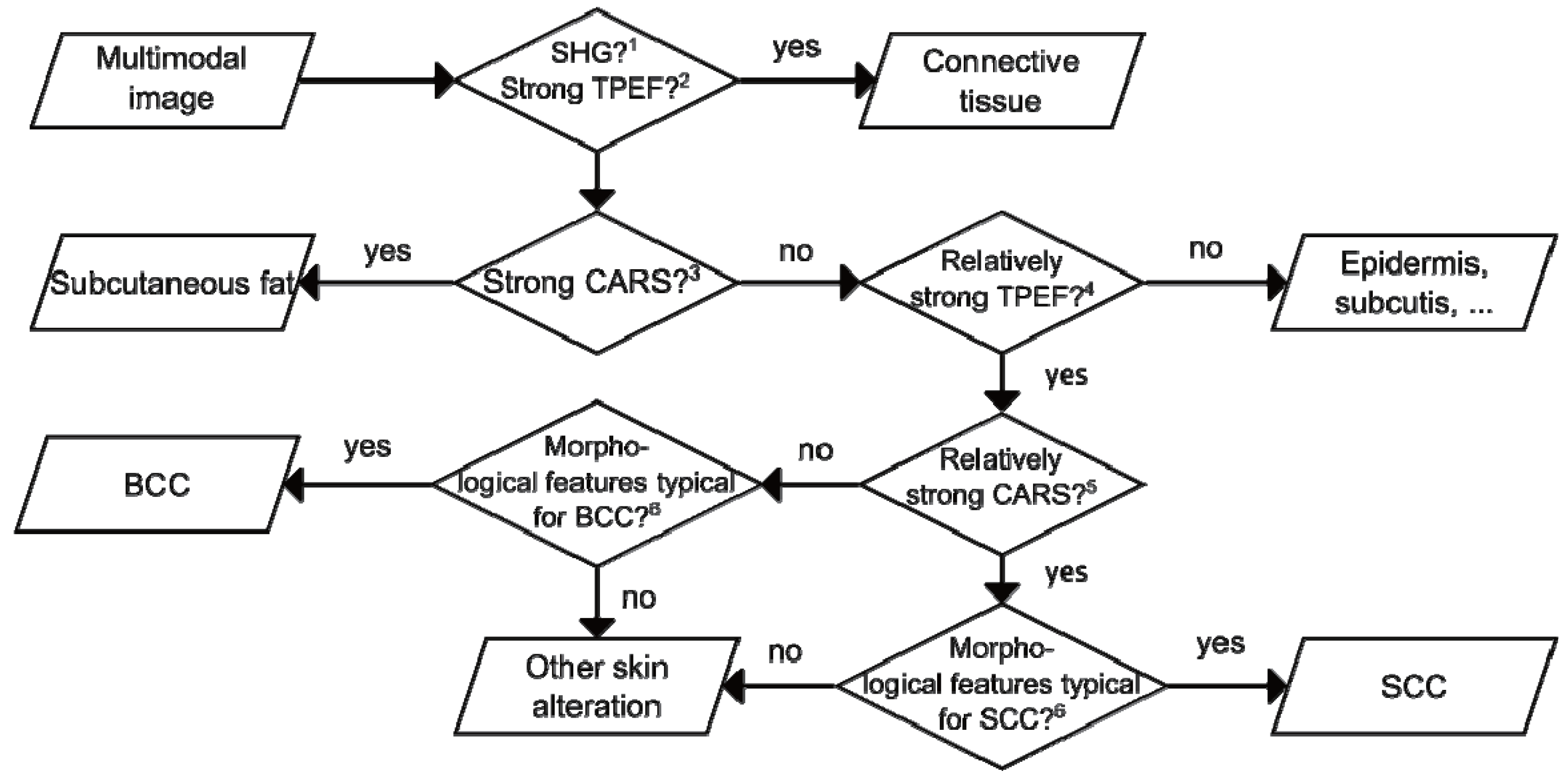
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- National Cancer Intelligence Network (NCIN). Non-Melanoma Skin Cancer in England, Scotland, Northern Ireland, and Ireland; NCIN: London, UK, 2013. [Google Scholar]
- Stern, R.S. The mysteries of geographic variability in nonmelanoma skin cancer incidence. Arch. Dermatol. 1999, 135, 843–844. [Google Scholar] [CrossRef]
- Rogers, H.W.; Weinstock, M.A.; Harris, A.R.; Hinckley, M.R.; Feldman, S.R.; Fleischer, A.B.; Coldiron, B.M. Incidence estimate of nonmelanoma skin cancer in the United States, 2006. Arch. Dermatol. 2010, 146, 283–287. [Google Scholar] [CrossRef]
- Østerlind, A.; Hjalgrim, H.; Kulinsky, B.; Frentz, G. Skin cancer as a cause of death in Denmark. Br. J. Dermatol. 1991, 125, 580–582. [Google Scholar] [CrossRef]
- Mogensen, M.; Jemec, G.B.E. Diagnosis of nonmelanoma skin cancer/keratinocyte, carcinoma: A review of diagnostic accuracy of nonmelanoma, skin cancer diagnostic tests and technologies. Dermatol. Surg. 2007, 33, 1158–1174. [Google Scholar] [CrossRef]
- Mogensen, M.; Thrane, L.; Jørgensen, T.M.; Andersen, P.E.; Jemec, G.B.E. OCT imaging of skin cancer and other dermatological diseases. J. Biophotonics 2009, 2, 442–451. [Google Scholar]
- Hinz, T.; Ehler, L.K.; Voth, H.; Fortmeier, I.; Hoeller, T.; Hornung, T.; Schmid-Wendtner, M.-H. Assessment of tumor thickness in melanocytic skin lesions: Comparison of optical coherence tomography, 20-MHz ultrasound and histopathology. Dermatology 2011, 223, 161–168. [Google Scholar] [CrossRef]
- Tang, S.; Zhou, Y.; Ju, M.J. Multimodal optical imaging with multiphoton microscopy and optical coherence tomography. J. Biophotonics 2012, 5, 396–403. [Google Scholar] [CrossRef]
- Sansa, N.; Farucha, M.; Chiavassa-Gandoisa, H.; de Ribes, C.L.C.; Paul, C.; Railhac, J.J. High-resolution magnetic resonance imaging in study of the skin: Normal patterns. Eur. J. Radiol. 2011, 80, e176–e181. [Google Scholar] [CrossRef]
- König, K.; Martin, M.S.; Köhler, J.; Scharenberg, R.; Kaatz, M. Clinical application of multiphoton tomography in combination with high-frequency ultrasound for evaluation of skin diseases. J. Biophotonics 2010, 3, 759–773. [Google Scholar] [CrossRef]
- Smith, L.; MacNeil, S. State of the art in non-invasive imaging of cutaneous melanoma. Skin Res. Technol. 2011, 17, 257–269. [Google Scholar] [CrossRef]
- Lin, S.-J.; Jee, S.-H.; Dong, C.-Y. Multiphoton microscopy: A new paradigm in dermatological imaging. Eur. J. Dermatol. 2007, 17, 361–366. [Google Scholar]
- Perry, S.W.; Burke, R.M.; Brown, E.B. Two-photon and second harmonic microscopy in clinical and translational cancer research. Annu. Biomed. Eng. 2012, 40, 277–291. [Google Scholar] [CrossRef]
- Tsai, T.H.; Jee, S.H.; Dong, C.Y.; Lin, S.J. Multiphoton microscopy in dermatological imaging. J. Dermatol. Sci. 2009, 56, 1–8. [Google Scholar] [CrossRef]
- König, K.; Ehlers, A.; Stracke, F.; Riemann, I. In vivo drug screening in human skin using femtosecond laser multiphoton tomography. Skin Pharmacol. Physiol. 2006, 19, 78–88. [Google Scholar] [CrossRef]
- Lyubovitsky, J.G.; Krasieva, T.B.; Xu, X.; Andersen, B.; Tromberg, B.J. In situ multiphoton optical tomography of hair follicles in mice. J. Biomed. Opt. 2007, 12, e044003. [Google Scholar] [CrossRef]
- Mulholland, W.; Arbuthnott, E.A.H.; Bellhouse, B.; Cornhill, J.F.; Austyn, J.M.; Kendall, M. Multiphoton high-resolution 3D imaging of Langerhans cells and keratinocytes in the mouse skin model adopted for epidermal powdered immunization. J. Invest. Dermatol. 2006, 126, 1541–1548. [Google Scholar] [CrossRef]
- Roediger, B.; Ng, L.G.; Smith, A.L.; de St Groth, B.F.; Weninger, W. Visualizing dendritic cell migration within the skin. Histochem. Cell Biol. 2008, 130, 1131–1146. [Google Scholar] [CrossRef]
- Giorgi, V.D.; Massi, D.; Sestini, S.; Cicchi, R.; Pavone, F.; Lotti, T. Combined non-linear laser imaging (two-photon excitation fluorescence microscopy, fluorescence lifetime imaging microscopy, multispectral multiphoton microscopy) in cutaneous tumours: First experiences. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 314–316. [Google Scholar] [CrossRef]
- Lin, S.J.; Wu, R.J.; Tan, H.Y.; Lo, W.; Lin, W.C.; Young, T.H.; Hsu, C.J.; Chen, J.S.; Jee, S.H.; Dong, C.Y. Evaluating cutaneous photoaging by use of multiphoton fluorescence and second-harmonic generation microscopy. Opt. Lett. 2005, 30, 2275–2277. [Google Scholar] [CrossRef]
- König, K. Clinical multiphoton tomography. J. Biophotonics 2008, 1, 13–23. [Google Scholar] [CrossRef]
- Lin, S.J.; Jee, S.H.; Kuo, C.J.; Wu, R.J.; Lin, W.C.; Chen, J.S.; Liao, Y.H.; Hsu, C.J.; Tsai, T.F.; Chen, Y.F.; et al. Discrimination of basal cell carcinoma from normal dermal stroma by quantitative multiphoton imaging. Opt. Lett. 2006, 31, 2756–2758. [Google Scholar] [CrossRef]
- Paoli, J.; Smedh, M.; Ericson, M.B. Multiphoton laser scanning microscopy—A novel diagnostic method for superficial skin cancers. Semin. Cutan. Med. Surg. 2009, 28, 190–195. [Google Scholar] [CrossRef]
- Seidenari, S.; Arginelli, F.; Bassoli, S.; Cautela, J.; Cesinaro, A.M.; Guanti, M.; Guardoli, D.; Magnoni, C.; Manfredini, M.; Ponti, G.; et al. Diagnosis of BCC by multiphoton laser tomography. Skin Res. Technol. 2012, 19, e297–e304. [Google Scholar]
- Tsai, M.R.; Shieh, D.B.; Lou, P.J.; Lin, C.F.; Sun, C.K. Characterization of oral squamous cell carcinoma based on higher-harmonic generation microscopy. J. Biophotonics 2012, 5, 415–424. [Google Scholar] [CrossRef]
- Gerger, A.; Horn, M.; Koller, S.; Weger, W.; Massone, C.; Leinweber, B.; Kerl, H.; Smolle, J. Confocal examination of untreated fresh specimens from basal cell carcinoma. Arch. Dermatol. 2005, 141, 1269–1274. [Google Scholar] [CrossRef]
- Pellacani, G.; Guitera, P.; Longo, C.; Avramidis, M.; Seidenari, S.; Menzies, S. The impact of in vivo reflectance confocal microscopy for the diagnostic accuracy of melanoma and equivocal melanocytic lesions. J. Invest. Dermatol. 2007, 127, 2759–2765. [Google Scholar]
- Peng, T.; Xie, H.; Ding, Y.; Wang, W.; Li, Z.; Jin, D.; Tang, Y.; Ren, Q.; Xi, P. CRAFT: Multimodality confocal skin imaging for early cancer diagnosis. J. Biophotonics 2012, 5, 469–476. [Google Scholar] [CrossRef]
- Borst, J.W.; Visser, A.J.W.G. Fluorescence lifetime imaging microscopy in life sciences. Meas. Sci. Technol. 2010, 21, e102002. [Google Scholar] [CrossRef]
- Cicchi, R.; Massi, D.; Sestini, S.; Carli, P.; Giorgi, V.D.; Lotti, T.; Pavone, F.S. Multidimensional non-linear laser imaging of basal cell carcinoma. Opt. Express 2007, 15, 10135–10148. [Google Scholar] [CrossRef]
- Meyer, T.; Bergner, N.; Bielecki, C.; Krafft, C.; Akimov, D.; Romeike, B.F.M.; Reichart, R.; Kalff, R.; Dietzek, B.; Popp, J. Nonlinear microscopy, infrared, and Raman microspectroscopy for brain tumor analysis. J. Biomed. Opt. 2011, 16, e021113. [Google Scholar] [CrossRef]
- Meyer, T.; Guntinas-Lichius, O.; von Eggeling, F.; Ernst, G.; Akimov, D.; Schmitt, M.; Dietzek, B.; Popp, J. Multimodal nonlinear microscopic investigations on head and neck squamous cell carcinoma: Toward intraoperative imaging. Head Neck 2013, 35, E280–E287. [Google Scholar] [CrossRef]
- Yang, Y.; Li, F.; Gao, L.; Wang, Z.; Thrall, M.J.; Shen, S.S.; Wong, K.K.; Wong, S.T.C. Differential diagnosis of breast cancer using quantitative, label-free and molecular vibrational imaging. Biomed. Opt. Express 2011, 2, 2160–2174. [Google Scholar] [CrossRef]
- König, K.; Breunig, H.; Bückle, R.; Kellner-Höfer, M.; Weinigel, M.; Büttner, E.; Sterry, W.; Lademann, J. Optical skin biopsies by clinical CARS and multiphoton fluorescence/SHG tomography. Laser Phys. Lett. 2011, 8, 465–468. [Google Scholar] [CrossRef]
- Vogler, N.; Meyer, T.; Akimov, D.; Latka, I.; Krafft, C.; Bendsoe, N.; Svanberg, K.; Dietzek, B.; Popp, J. Multimodal imaging to study the morphochemistry of basal cell carcinoma. J. Biophotonics 2010, 3, 728–736. [Google Scholar] [CrossRef]
- Levenson, M.; Bloembergen, N. Dispersion of the nonlinear optical susceptibility tensor on centrosymmetric media. Phys. Rev. B 1974, 10, 4447–4464. [Google Scholar] [CrossRef]
- Meyer, T.; Akimov, D.; Tarcea, T.; Chatzipapadopoulos, S.; Muschiolok, G.; Kobow, J.; Schmitt, M.; Popp, J. Three-dimensional molecular mapping of a multiple emulsion by means of CARS microscopy. J. Phys. Chem. B 2008, 112, 1420–1426. [Google Scholar] [CrossRef]
- Heuke, S.; Vogler, N.; Meyer, T.; Akimova, D.; Kluschke, F.; Röwert-Huber, H.J.; Lademann, J.; Dietzek, B.; Popp, J. Multimodal mapping of human skin. Br. J. Dermatol. 2013. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. Available online: http://www.R-project.org/ (accessed on 3 April 2013).
- Urbanek, S. PNG: Read and Write PNG Images. R Package Version 0.1-4. 2011. Available online: http://CRAN.R-project.org/package=png/ (accessed on 3 April 2013).
- Evans, C.L.; Xie, X.S. Coherent anti-Stokes Raman scattering microscopy: Chemical imaging for biology and medicine. Annu. Rev. Anal. Chem. 2008, 1, 883–909. [Google Scholar] [CrossRef]
- Lim, R.S.; Suhalim, J.L.; Miyazaki-Anzai, S.; Miyazaki, M.; Levi, M.; Potma, E.O.; Tromberg, B.J. Identification of cholesterol crystals in plaques of atherosclerotic mice using hyperspectral CARS imaging. J. Lipid Res. 2011, 52, 2177–2186. [Google Scholar] [CrossRef]
- Meyer, T.; Bergner, N.; Medyukhina, A.; Dietzek, B.; Krafft, C.; Romeike, B.F.M.; Reichart, R.; Kalff, R.; Popp, J. Interpreting CARS images of tissue within the C-H-stretching region. J. Biophotonics 2012, 5, 729–733. [Google Scholar] [CrossRef]
- Zipfel, W.R.; Williams, R.M.; Christie, R.; Nikitin, A.Y.; Hyman, B.T.; Webb, W.W. Live tissue intrinsic emission microscopy using multiphoton-excited native fluorescence and second harmonic generation. Proc. Natl. Acad. Sci. USA 2003, 100, 7075–7080. [Google Scholar] [CrossRef]
- Campagnola, P.J.; Millard, A.C.; Terasaki, M.; Hoppe, P.E.; Malone, C.J.; Mohler, W.A. Three-dimensional high-resolution second-harmonic generation imaging of endogenous structural proteins in biological tissues. Biophys. J. 2002, 81, 493–508. [Google Scholar]
- Guo, Y.; Ho, P.P.; Tirksliunas, A.; Liu, F.; Alfano, R.R. Optical harmonic generation from animal tissues by the use of picosecond and femtosecond laser pulses. Appl. Opt. 1996, 35, 6810–6813. [Google Scholar] [CrossRef]
- Mohler, W.; Millard, A.C.; Campagnolab, P.J. Second harmonic generation imaging of endogenous structural proteins. Methods 2003, 29, 97–109. [Google Scholar] [CrossRef]
- Freund, I.; Deutsch, M.; Sprecher, A. Connective tissue polarity: Optical second-harmonic microscopy, crossed-beam summation, and small-angle scattering in rat-tail tendon. Biophys. J. 1986, 50, 693–712. [Google Scholar] [CrossRef]
- Theodossiou, T.A.; Thrasivoulou, C.; Ekwobi, C.; Becker, D.L. Second harmonic generation confocal microscopy of collagen Type I from rat tendon cryosections. Biophys. J. 2006, 91, 4665–4677. [Google Scholar] [CrossRef]
- Huang, S.; Heikal, A.A.; Webb, W.W. Two-photon fluorescence spectroscopy and microscopy of NAD(P)H and flavoprotein. Biophys. J. 2002, 82, 2811–2825. [Google Scholar] [CrossRef]
- Paoli, J.; Smedh, M.; Wennberg, A.M.; Ericson, M.B. Multiphoton laser scanning microscopy on non-melanoma skin cancer: Morphologic features for future non-invasive diagnostics. J. Invest. Dermatol. 2008, 128, 1248–1255. [Google Scholar] [CrossRef]
- Morgan, M.B.; Hamill, J.R.; Spencer, J.M. Atlas of Mohs and Frozen Section Cutaneous Pathology; Springer: New York, NY, USA, 2009. [Google Scholar]
- Rapini, R.P. Practical Dermatopathology; Elsevier Health Sciences: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Fritsch, P. Dermatologie und Venerologie für das Studium; Springer Medizin Verlag: Heidelberg, Germany, 2009. [Google Scholar]
- Van Kempen, L.C.; Rijntjes, J.; Claes, A.; Blokx, W.A.; Gerritsen, M.J.P.; Ruiter, D.J.; van Muijen, G.N. Type I collagen synthesis parallels the conversion of keratinocytic intraepidermal neoplasia to cutaneous squamous cell carcinoma. J. Pathol. 2004, 204, 333–339. [Google Scholar]
- Thrasivoulou, C.; Virich, G.; Krenacs, T.; Korom, I.; Becker, D.L. Optical delineation of human malignant melanoma using second harmonic imaging of collagen. Biomed. Opt. Express 2011, 2, 1285–1295. [Google Scholar]
- Szeimies, R.M.; Hauschild, A.; Garbe, C.; Kaufmann, R.; Landthaler, M. Tumoren der Haut; Georg Thieme Verlag KG: Stuttgart, Germany, 2010. [Google Scholar]
- Marks, D.B.; Smith, C.; Marks, A.D.; Liebermann, M. Marks Basic Medical Biochemistry: A Clinical Approach; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2004. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 4, 646–674. [Google Scholar] [CrossRef]
- Talmadge, J.E.; Fidler, I.J. AACR centennial series: The biology of cancer metastasis: Historical perspective. Cancer Res. 2010, 70, 5649–5669. [Google Scholar] [CrossRef]
- Joyce, J.A.; Pollard, J.W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 2009, 9, 239–252. [Google Scholar] [CrossRef]
- Provenzano, P.P.; Eliceiri, K.W.; Campbell, J.M.; Inman, D.R.; White, J.G.; Keely, P.J. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 2006, 4, 38–52. [Google Scholar] [CrossRef]
- Heikal, A.A. Intracellular coenzymes as natural biomarkers for metabolic activities and mitochondrial anomalies. Biomark Med. 2010, 4, 241–263. [Google Scholar] [CrossRef]
- Breunig, H.G.; Studier, H.; König, K. Multiphoton excitation characteristics of cellular fluorophores of human skin in vivo. Opt. Express 2010, 18, 7857–7871. [Google Scholar] [CrossRef]
- Uppal, A.; Gupta, P.K. Measurement of NADH concentration in normal and malignant human tissues from breast and oral cavity. Biotechnol. Appl. Biochem. 2003, 37, 45–50. [Google Scholar] [CrossRef]
- Yu, Q.; Heikal, A.A. Two-photon autofluorescence dynamics imaging reveals sensitivity of intracellular NADH concentration and conformation to cell physiology at the single-cell level. J. Photochem. Photobiol. B 2009, 95, 46–57. [Google Scholar] [CrossRef]
- Gupta, P.K.; Majumder, S.K.; Uppal, A. Breast cancer diagnosis using N2 laser excited autofluorescence spectroscopy. Lasers Surg. Med. 1997, 21, 417–422. [Google Scholar] [CrossRef]
- Schmuth, M.; Jiang, Y.J.; Dubrac, S.; Elias, P.M.; Feingold, K.R. Peroxisome proliferator-activated receptors and liver X receptors in epidermal biology. J. Lipid Res. 2008, 49, 499–509. [Google Scholar] [CrossRef]
- Sandilands, A.; Sutherland, C.; Irvine, A.D.; McLean, W.H.I. Filaggrin in the frontline: Role in skin barrier function and disease. J. Cell Sci. 2009, 122, 1285–1294. [Google Scholar]
- Plathow, C.; Weber, W.A. Tumor cell metabolism imaging. J. Nucl. Med. 2008, 49, 43S–63S. [Google Scholar] [CrossRef]
- Santos, C.R.; Schulze, A. Lipid metabolism in cancer. FEBS J. 2012, 279, 2610–2623. [Google Scholar] [CrossRef]
- Zhang, F.; Du, G. Dysregulated lipid metabolism in cancer. World J. Biol. Chem. 2012, 3, 167–174. [Google Scholar] [CrossRef]
- Yecies, J.L.; Manning, B.D. Chewing the fat on tumor cell metabolism. Cell 2010, 8, 28–30. [Google Scholar] [CrossRef]
- Kuhajda, F.P. Fatty acid synthase and cancer: New application of an old pathway. Cancer Res. 2006, 66, 5977–5980. [Google Scholar] [CrossRef]
- Menendez, J.A.; Lupu, R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat. Rev. Cancer 2007, 7, 763–777. [Google Scholar] [CrossRef]
- Le, T.T.; Huff, T.B.; Cheng, J.X. Coherent anti-Stokes Raman scattering imaging of lipids in cancer metastasis. BMC Cancer 2009, 9, 42–55. [Google Scholar] [CrossRef]
- Weinstock, M.; Bogaars, H.; Ashley, M.; Litle, V.; Bilodeau, E.; Kimmel, S. Nonmelanoma skin cancer mortality: A population-based study. Arch. Dermatol. 1991, 127, 1194–1197. [Google Scholar] [CrossRef]
- Baumgartl, M.; Gottschall, T.; Abreu-Afonso, J.; Díez, A.; Meyer, T.; Dietzek, B.; Rothhardt, M.; Popp, J.; Limpert, J.; Tünnermann, A. Alignment-free, all-spliced fiber laser source for CARS microscopy based on four-wave-mixing. Opt. Express 2012, 20, 21010–21018. [Google Scholar]
Appendix
Tumor-Associated Collagen Signatures

Elastin Fiber Distribution in NMSC
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Heuke, S.; Vogler, N.; Meyer, T.; Akimov, D.; Kluschke, F.; Röwert-Huber, H.-J.; Lademann, J.; Dietzek, B.; Popp, J. Detection and Discrimination of Non-Melanoma Skin Cancer by Multimodal Imaging. Healthcare 2013, 1, 64-83. https://doi.org/10.3390/healthcare1010064
Heuke S, Vogler N, Meyer T, Akimov D, Kluschke F, Röwert-Huber H-J, Lademann J, Dietzek B, Popp J. Detection and Discrimination of Non-Melanoma Skin Cancer by Multimodal Imaging. Healthcare. 2013; 1(1):64-83. https://doi.org/10.3390/healthcare1010064
Chicago/Turabian StyleHeuke, Sandro, Nadine Vogler, Tobias Meyer, Denis Akimov, Franziska Kluschke, Hans-Joachim Röwert-Huber, Jürgen Lademann, Benjamin Dietzek, and Jürgen Popp. 2013. "Detection and Discrimination of Non-Melanoma Skin Cancer by Multimodal Imaging" Healthcare 1, no. 1: 64-83. https://doi.org/10.3390/healthcare1010064