Cutaneous Alternariosis Caused by Alternaria infectoria: Three Cases in Kidney Transplant Patients
Abstract
:1. Introduction
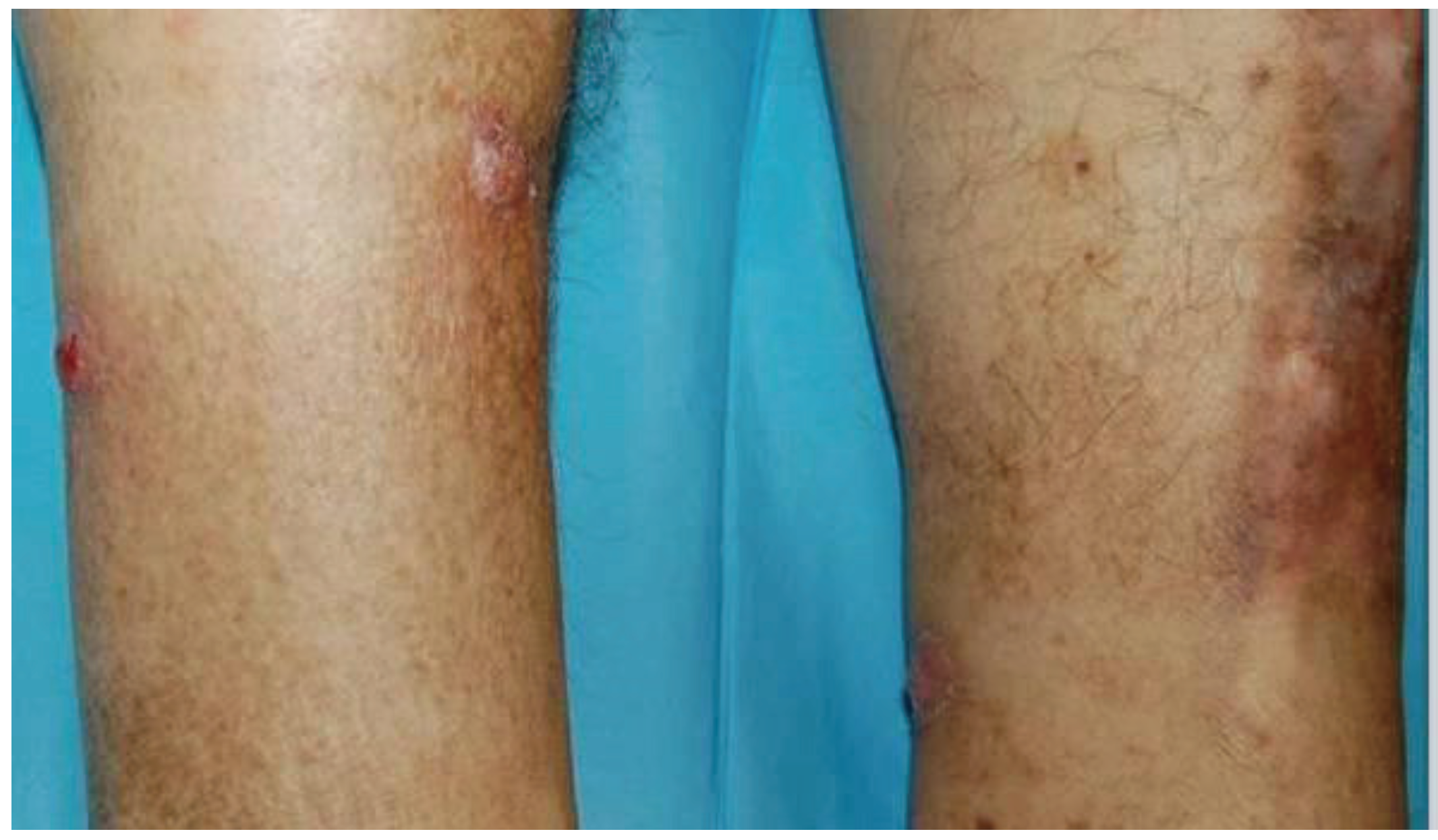
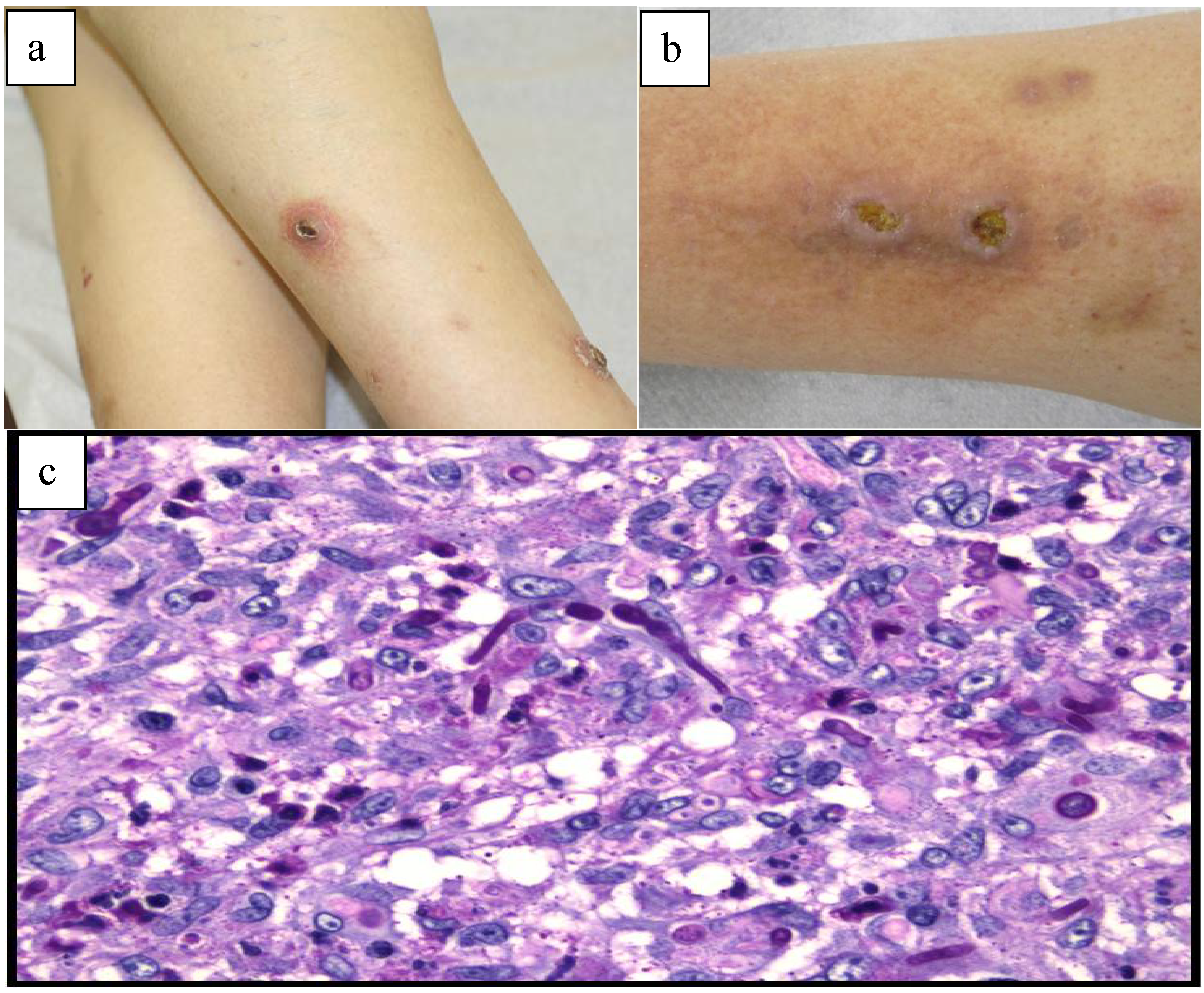
2. Results and Discussion

| Sex/Age | Immunosuppression | Medication | Clinical findings | Evolution | Diagnosis | Intervention | Follow-up | |
|---|---|---|---|---|---|---|---|---|
| 1 | M/61 years | Kidney transplant (6-months prior) | Tacrolimus Prednisolone | Five tender, violaceous nodules on both legs | 2 weeks | Histological examination Culture PCR | Itraconazole 100 mg/day, 3 months | 36-months No relapse |
| 2 | M/63 years | Kidney transplant (14-months prior) | Cyclosporine Prednisolone | Papulo-nodules and 2 ulcerations on the legs | 1 year | Histological examination Culture PCR | Cryotherapy Posaconazole 200 mg/day, 1 month | 12-months No relapse |
| 3 | M/56 years | Kidney transplant (11-years prior) | Tacrolimus Prednisolone Mycophenolate mofetil | 1 nodule on the third finger of the right hand | 5 months | Histological examination PCR | Surgical excision Itraconazole 100 mg/day, 3 months | 17-months No relapse |
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gallelli, B.; Viviani, M.; Nebuloni, M.; Marzano, A.V.; Pozzi, C.; Messa, P.; Fogazzi, G.B. Skin infection due to Alternaria species in kidney allograft recipients: Report of a new case and review of the literature. J. Nephrol. 2006, 19, 668–672. [Google Scholar]
- Shashikala, R.P.; Saikumar, A.; Ramesh, K.; Aparana, V. Alternaria alternata in a case of mass in the lung. Indian J. Med. Microbiol. 2013, 31, 308–310. [Google Scholar] [CrossRef]
- Gilaberte, M.; Bartralot, R.; Torres, J.M.; Reus, F.S.; Rodríguez, V.; Alomar, A.; Pujol, R.M. Cutaneous alternariosis in transplant recipients: Clinicopathologic review of 9 cases. J. Am. Acad. Dermatol. 2005, 52, 653–659. [Google Scholar] [CrossRef]
- Pastor, F.J.; Guarro, J. Alternaria infections: Laboratory diagnosis and relevant clinical features. Clin. Microbiol. Infect. 2008, 14, 734–746. [Google Scholar] [CrossRef]
- Leahy, T.R.; Punnett, A.S.; Richardson, S.E.; Gharabaghi, F.; Wadhwa, A. Molecular identification of phaeohyphomycosis due to Alternaria infectoria in a patient with acute myeloid leukemia—A case report. Diagn. Microbiol. Infect. Dis. 2010, 66, 318–321. [Google Scholar]
- Lyke, K.E.; Miller, N.S.; Towne, L.; Merz, W.G. A case of cutaneous ulcerative alternariosis: Rare association with diabetes mellitus and unusual failure of itraconazole treatment. Clin. Infect. Dis. 2001, 32, 1178–1187. [Google Scholar] [CrossRef]
- Santiago, F.; Serra, D.; Vieira, R.; Brites, M.M.; Figueiredo, A. Successful cryotherapy for a cutaneous alternariosis in a renal transplant recipient. Eur. J. Dermatol. 2010, 20, e841. [Google Scholar]
- Ara, M.; Aspiroz, C.; Zaballos, P.; Alcalde, V.; Alvarez, R.; Rezusta, A.; Giménez, J.A. Relapse of cutaneous Alternaria infectoria in a renal transplant recipient after 2 years. Acta Derm. Venereol. 2006, 86, 154–155. [Google Scholar]
- Robert, T.; Talarmin, J.; Leterrier, M.; Cassagnau, E.; Pape, P.L.; Danner-Boucher, I.; Malard, O.; Brocard, A.; Gay-Andrieu, F.; Miegeville, M.; et al. Phaeohyphomycosis due to Alternaria infectoria: A single-center experience with utility of PCR for diagnosis and species identification. Med. Mycol. 2012, 50, 594–600. [Google Scholar] [CrossRef]
- Cunha, D.; Amaro, C.; Vieira, M.R.; Martins, L.; Maduro, A.P.; Inácio, J.; Afonso, A.; Pinto, G.M.; Cardoso, J. Phaeohyphomycosis caused by Alternaria infectoria presenting as multiple vegetating lesions in a renal transplant patient. Rev. Iberoam. Micol. 2012, 29, 44–46. [Google Scholar] [CrossRef]
- Rammaert, B.; Aguilar, C.; Bougnoux, M.E.; Noël, N.; Charlier, C.; Denis, B.; Lecuit, M.; Lortholary, O. Success of posaconazole therapy in a heart transplanted patient with Alternaria infectoria cutaneous infection. Med. Mycol. 2012, 50, 518–521. [Google Scholar] [CrossRef]
- Halaby, T.; Boots, H.; Vermeulen, A.; van der Ven, A.; Beguin, H.; van Hooff, H.; Jacobs, J. Phaeohyphomycosis caused by Alternaria infectoria in a renal transplant recipient. J. Clin. Microbiol. 2001, 39, 1952–1955. [Google Scholar] [CrossRef]
- Brasch, J.; Busch, J.O.; de Hoog, G.S. Cutaneous phaeohyphomycosis caused by Alternaria infectoria. Acta Derm. Venereol. 2008, 88, 160–161. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lopes, L.; Borges-Costa, J.; Soares-Almeida, L.; Filipe, P.; Neves, F.; Santana, A.; Guerra, J.; Kutzner, H. Cutaneous Alternariosis Caused by Alternaria infectoria: Three Cases in Kidney Transplant Patients. Healthcare 2013, 1, 100-106. https://doi.org/10.3390/healthcare1010100
Lopes L, Borges-Costa J, Soares-Almeida L, Filipe P, Neves F, Santana A, Guerra J, Kutzner H. Cutaneous Alternariosis Caused by Alternaria infectoria: Three Cases in Kidney Transplant Patients. Healthcare. 2013; 1(1):100-106. https://doi.org/10.3390/healthcare1010100
Chicago/Turabian StyleLopes, Leonor, João Borges-Costa, Luís Soares-Almeida, Paulo Filipe, Fernanda Neves, Alice Santana, José Guerra, and Heinz Kutzner. 2013. "Cutaneous Alternariosis Caused by Alternaria infectoria: Three Cases in Kidney Transplant Patients" Healthcare 1, no. 1: 100-106. https://doi.org/10.3390/healthcare1010100




