Role of Salivary Biomarkers in Detection of Cardiovascular Diseases (CVD)
Abstract
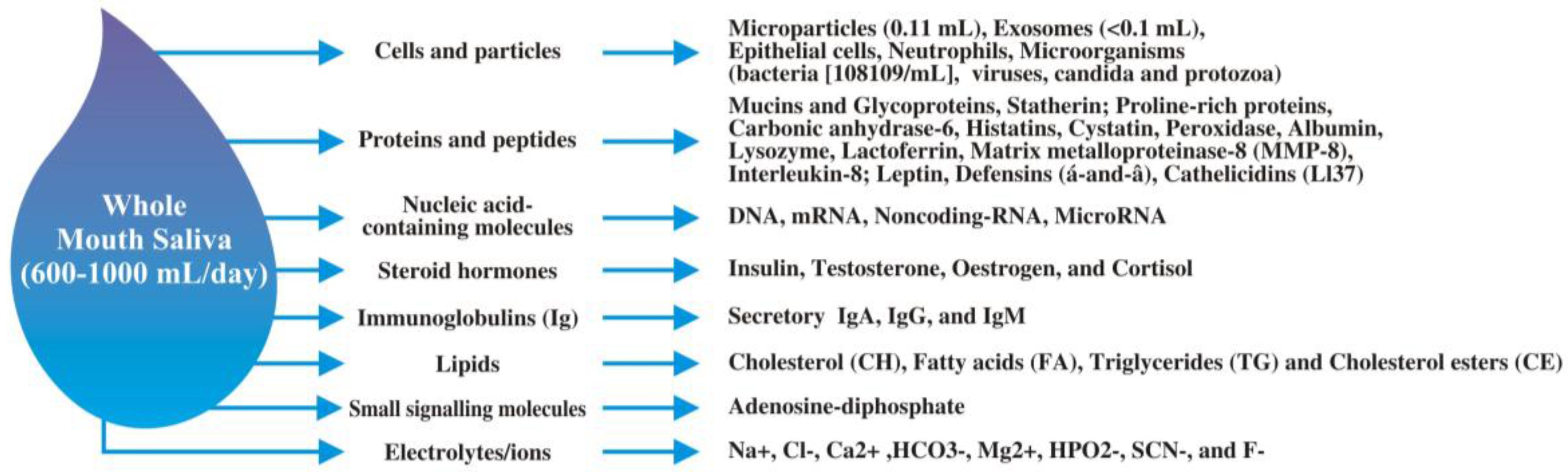
:1. Introduction
2. Salivary Biomarkers in Cardiovascular Disease (CVD) Detection
3. Future Prospects
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rathnayake, N.; Åkerman, S.; Klinge, B.; Lundegren, N.; Jansson, H.; Tryselius, Y.; Sorsa, T.; Gustafsson, A. Salivary Biomarkers for Detection of Systemic Diseases. PLoS ONE 2013, 8, e61356. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.S.; Foley, J.D.; Bailey, A.L.; Campell, C.L.; Humphries, R.L.; Christodoulides, N.; Floriano, P.N.; Simmons, G.; Bhagwandin, B.; Jacobson, J.W.; et al. Current developments in salivary diagnostics. Biomarkers Med. 2010, 4, 171–189. [Google Scholar] [CrossRef]
- Rahim, M.A.A.; Abdul Rahim, Z.H.; Wan Ahmad, W.A.; Hashim, O.H. Can saliva proteins be used to predict the onset of acute myocardial infarction among high-risk patients? Int. J. Med. Sci. 2015, 12, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.S.; Foley, J.D.; Floriano, P.N.; Christodoulides, N.; Ebersole, J.L.; Campbell, C.L.; Bailey, A.L.; Rose, B.G.; Kinane, D.F.; Novak, M.J.; et al. Utility of Salivary Biomarkers for Demonstrating Acute Myocardial Infarction. J. Dent. Res. 2014, 93, 72S–79S. [Google Scholar] [CrossRef] [PubMed]
- Javaid, M.A.; Ahmed, A.S.; Durand, R.; Tran, S.D. Saliva as a diagnostic tool for oral and systemic diseases. J. Oral Biol. Craniofac. Res. 2016, 6, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; White, H.D. Universal definition of myocardial infarction. Eur. Heart J. 2007, 28, 2525–2538. [Google Scholar] [CrossRef] [PubMed]
- Punyadeera, C. New frontiers in heart failure detection: Saliva testing. BMJ Innov. 2016, 2, 106–108. [Google Scholar] [CrossRef]
- Khurshid, Z.; Naseem, M.; Sheikh, Z.; Najeeb, S.; Shahab, S.; Zafar, M.S. Oral antimicrobial peptides: Types and role in the oral cavity. Saudi Pharm. J. 2015, 24, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Najeeb, S.; Mali, M.; Moin, S.F.; Raza, S.Q.; Zohaib, S.; Sefat, F.; Zafar, M.S. Histatin peptides: Pharmacological functions and its applications in dentistry. Saudi Pharm. J. 2016. [Google Scholar]
- Editorial, G. Salivaomics: An Emerging Approach in Dentistry. J. Pak. Dent. Assoc. 2016, 25, 1–3. [Google Scholar]
- Najeeb, S.; Zafar, M.; Khurshid, Z.; Zohaib, S.; Almas, K. The Role of Nutrition in Periodontal Health: An Update. Nutrients 2016, 8, 530. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Zafar, M.S.; Najeeb, S.; Shahab, S. Human Saliva: A Future Diagnostic Tool. Dent. Sci. 2015, 2, 260–265. [Google Scholar]
- Khurshid, Z.; Zohaib, S.; Najeeb, S.; Zafar, M.S.; Slowey, P.D.; Almas, K. Human saliva collection devices for proteomics: An update. Int. J. Mol. Sci. 2016, 17, 846. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Zohaib, S.; Najeeb, S.; Zafar, M.; Rehman, R.; Rehman, I. Advances of Proteomic Sciences in Dentistry. Int. J. Mol. Sci. 2016, 17, 728. [Google Scholar] [CrossRef] [PubMed]
- Floriano, P.N.; Christodoulides, N.; Miller, C.S.; Ebersole, J.L.; Spertus, J.; Rose, B.G.; Kinane, D.F.; Novak, M.J.; Steinhubl, S.; Acosta, S.; et al. Use of saliva-based nano-biochip tests for acute myocardial infarction at the point of care: A feasibility study. Clin. Chem. 2009, 55, 1530–1538. [Google Scholar] [CrossRef] [PubMed]
- Foo, J.Y.Y.; Wan, Y.; Kostner, K.; Arivalagan, A.; Atherton, J.; Cooper-White, J.; Dimeski, G.; Punyadeera, C. NT-ProBNP Levels in Saliva and Its Clinical Relevance to Heart Failure. PLoS ONE 2012, 7, e48452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khalyfa, A.; Gozal, D. Exosomal miRNAs as potential biomarkers of cardiovascular risk in children. J. Transl. Med. 2014, 12, 162. [Google Scholar] [CrossRef] [PubMed]
- Labat, C.; Temmar, M.; Nagy, E.; Bean, K.; Brink, C.; Benetos, A.; Bäck, M. Inflammatory mediators in saliva associated with arterial stiffness and subclinical atherosclerosis. J. Hypertens. 2013, 31, 2251–2258. [Google Scholar] [CrossRef] [PubMed]
- Thul, S.; Labat, C.; Temmar, M. Short report Low salivary resolvin D1 to leukotriene B 4 ratio predicts carotid intima media thickness: A novel biomarker of non-resolving vascular inflammation. Eur. J. Prev. Cardiol. 2017, 24, 903–906. [Google Scholar] [CrossRef] [PubMed]
- Hlawaty, H.; Labat, C.; Michel, J.; Brink, C. The Oral Cavity and Age: A Site of Chronic Inflammation? PLoS ONE 2007, 2, e1351. [Google Scholar]
- Foley, J.D.; Sneed, J.D.; Steinhubl, S.R.; Kolasa, J.; Ebersole, J.L.; Lin, Y.; Kryscio, R.J.; McDevitt, J.T.; Campbell, C.L.; Miller, C.S. Oral fluids that detect cardiovascular disease biomarkers. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Aizat, M.; Rahim, A.; Haji, Z.; Rahim, A.; Azman, W.; Ahmad, W.; Haji, O. Can Saliva Proteins Be Used to Predict the Onset of Acute Myocardial Infarction among High-Risk Patients? Int. J. Med. Sci. 2015, 12, 329–335. [Google Scholar]
- Foley, J.D.; Sneed, J.D.; Steinhubl, S.R.; Kolasa, J.R.; Ebersole, J.L.; Lin, Y.; Kryscio, R.J.; McDevitt, J.T.; Campbell, C.L.; Miller, C.S. Salivary biomarkers associated with myocardial necrosis: Results from an alcohol septal ablation model. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 616–623. [Google Scholar] [CrossRef] [PubMed]
- Mirzaii-Dizgah, I.; Riahi, E. Salivary troponin I as an indicator of myocardial infarction. Indian J. Med. Res. 2013, 138, 861–865. [Google Scholar] [PubMed]
- Khan, R.S.; Khurshid, Z. Advancing Point-of-Care (PoC) Testing Using Human Saliva as Liquid Biopsy. Diagnostics 2017, 7, 39. [Google Scholar] [CrossRef] [PubMed]

| Author Name and Year | Sample Type | Disease Conditions | Biomarkers | Result |
|---|---|---|---|---|
| Foley et al. (2012) [23] | Saliva and serum | Myocardial necrosis | TnI, CK-MB, and MYO | Elevated in both, but less dramatically in saliva. Significant correlation (p < 0.001) serum and saliva for matrix metalloproteinase-9, C-reactive protein, and myeloperoxidase observed. |
| Foley et al. (2012) [23] | Saliva and serum | Inflammation, tissue injury and remodeling | CRP, MMP-9, MPO | Elevated in both saliva and serum, but a greater MPO downward trend in saliva |
| Foley et al. (2012) [21] | Saliva and serum | Pre-existing CVD diseased patients who underwent an invasive cardiac procedure | CRP, TNF-α, sCD40L, IL-1β, IL-6, adiponectin, MMP-9, MPO, sICAM-1 | Adiponectin, BNP, CK-MB, CRP, IL-6, MMP-9, MPO, MYO, TNFα, sCD40-L and sICAM-1 more elevated in serum; IL-1β higher in UWS TnI same levels |
| Miller et al. (2014) [4] | Saliva and serum | AMI | CRP, IL-6, IL-1β, MPO, sCD40L, TNF-α, Adip, sICAM-1, MMP-9 | Tnl, BNP, CK-MB, MYO, CRP were all detected in statistically significant levels in the serum; Adip, sICAM, CRP were detected in statistically significant levels in saliva |
| Labat et al. (2013) [18] | Saliva and serum | Hypertension | Lysozyme | Saliva |
| Labat et al. (2013) [18] | Saliva and serum | IMT | CRP, MMP-9 | CRP more in serum; MMP-9 more in saliva |
| Labat et al. (2013) [18] | Saliva and serum | Arterial stiffness | LTB4 and PGE2 | Only saliva |
| Rathnayake et al. (2013) [1] | Saliva | Patients who had under gone heart surgery | MMP-8 | Elevated |
| Rathnayake et al. [1] | Saliva | Hypertension | MMP-8, lysozyme | Elevated |
| Floriano et al. (2009) [15] | Serum and saliva | AMI patients within 48 h of chest pain onset | CRP, MMP-9, IL-1beta, sICAM-1, adiponectin, MCP-1, Gro-alpha, E-selectin, IL-18, ENA-78, sVCAM-1 were upregulated in saliva more than serum; MPO, MYO, CK-MB, TnI, BNP, sCD40-L TNF-α were upregulated more in serum than saliva; Fractalkine, IL-6, Adiponectin, MCP-1, Gro-alpha, E-selectin were downregulated. | |
| Dizgah et al. (2013) [24] | Saliva and serum | 12 and 24 h of onset of MI | cTnI | Elevated |
| Foo et al. (2012) [16] | Saliva and serum | Heart Failure patients | NT-proBNP | No correlation found between saliva and serum, though elevated in both. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdul Rehman, S.; Khurshid, Z.; Hussain Niazi, F.; Naseem, M.; Al Waddani, H.; Sahibzada, H.A.; Sannam Khan, R. Role of Salivary Biomarkers in Detection of Cardiovascular Diseases (CVD). Proteomes 2017, 5, 21. https://doi.org/10.3390/proteomes5030021
Abdul Rehman S, Khurshid Z, Hussain Niazi F, Naseem M, Al Waddani H, Sahibzada HA, Sannam Khan R. Role of Salivary Biomarkers in Detection of Cardiovascular Diseases (CVD). Proteomes. 2017; 5(3):21. https://doi.org/10.3390/proteomes5030021
Chicago/Turabian StyleAbdul Rehman, Saad, Zohaib Khurshid, Fayez Hussain Niazi, Mustafa Naseem, Hamed Al Waddani, Haafsa Arshad Sahibzada, and Rabia Sannam Khan. 2017. "Role of Salivary Biomarkers in Detection of Cardiovascular Diseases (CVD)" Proteomes 5, no. 3: 21. https://doi.org/10.3390/proteomes5030021






