An In Vitro Procedure for Phenotypic Screening of Growth Parameters and Symbiotic Performances in Lotus corniculatus Cultivars Maintained in Different Nutritional Conditions
Abstract
:1. Introduction
2. Results
2.1. Experimental Design
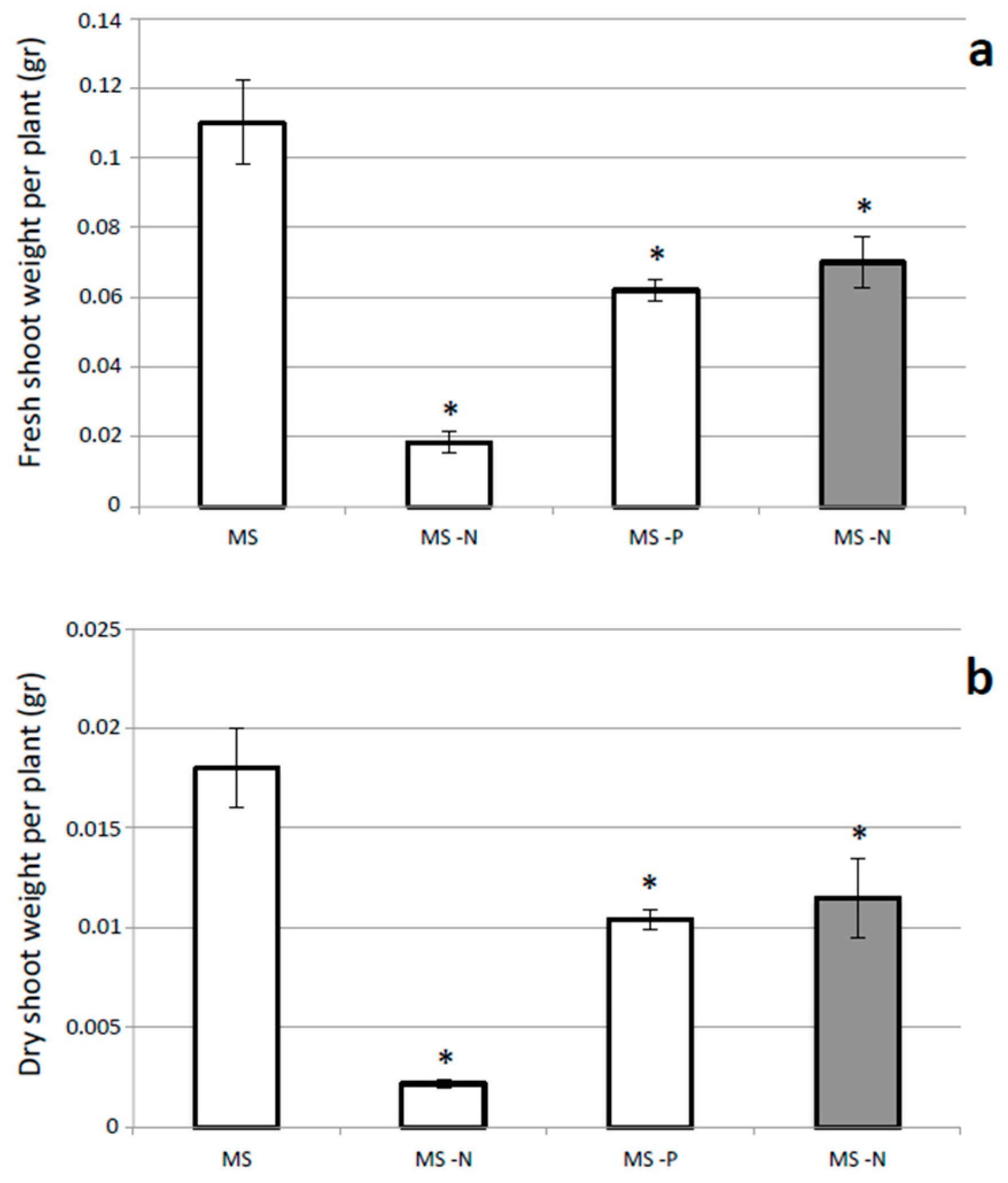
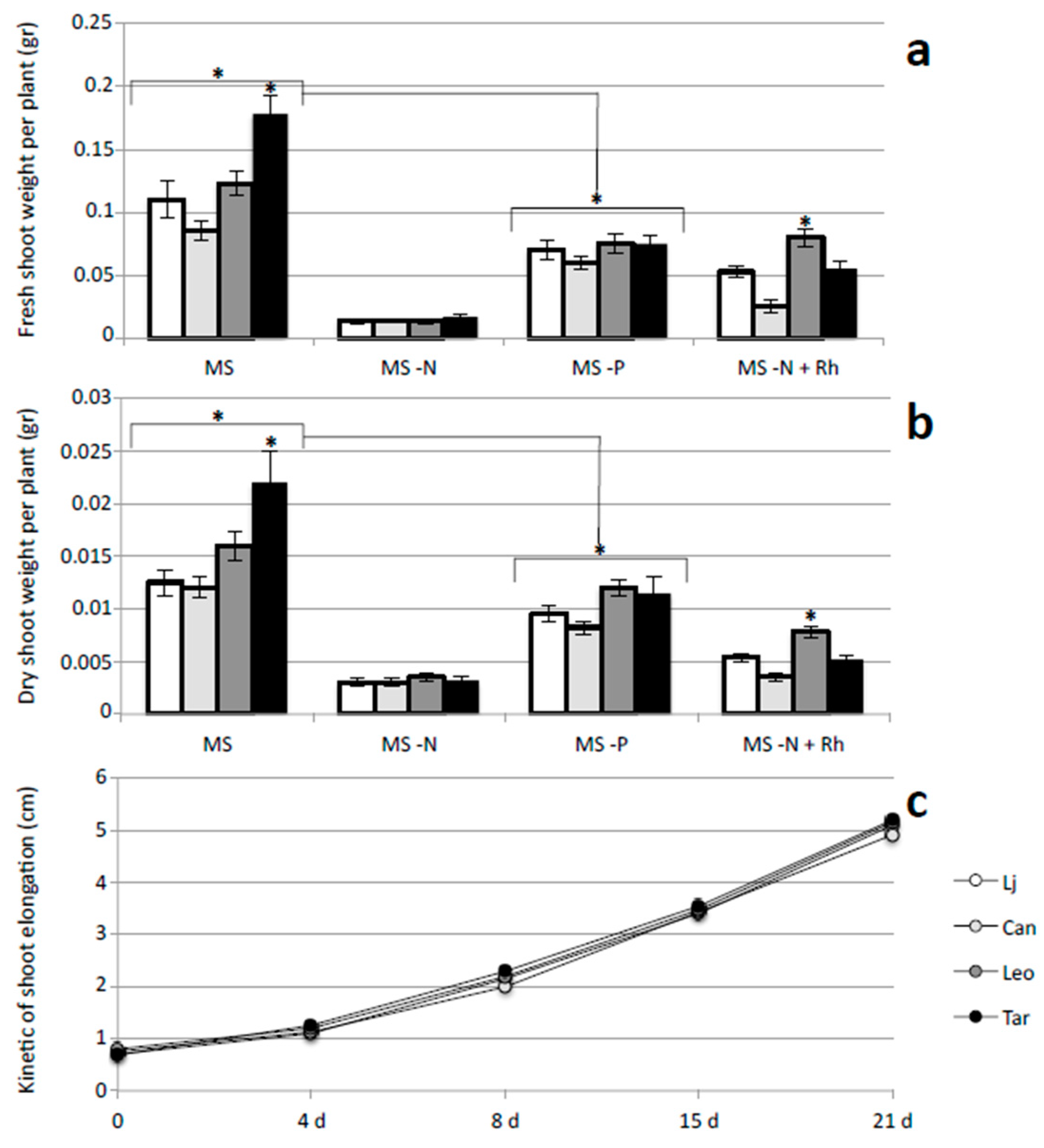
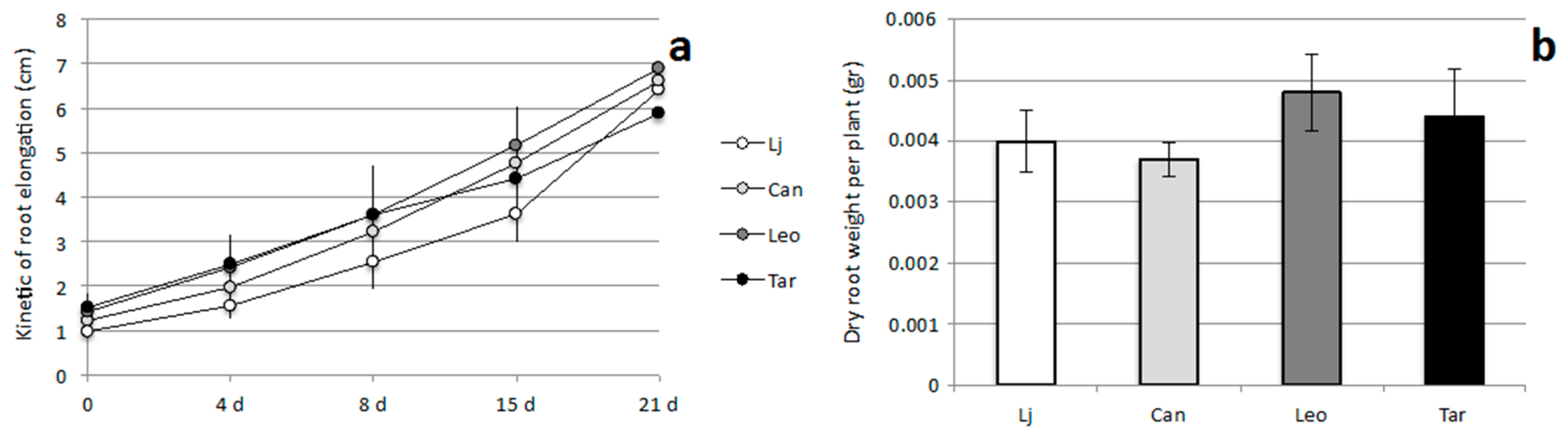
2.2. Growth Responses of L. corniculatus cvs. to N and P Starvation
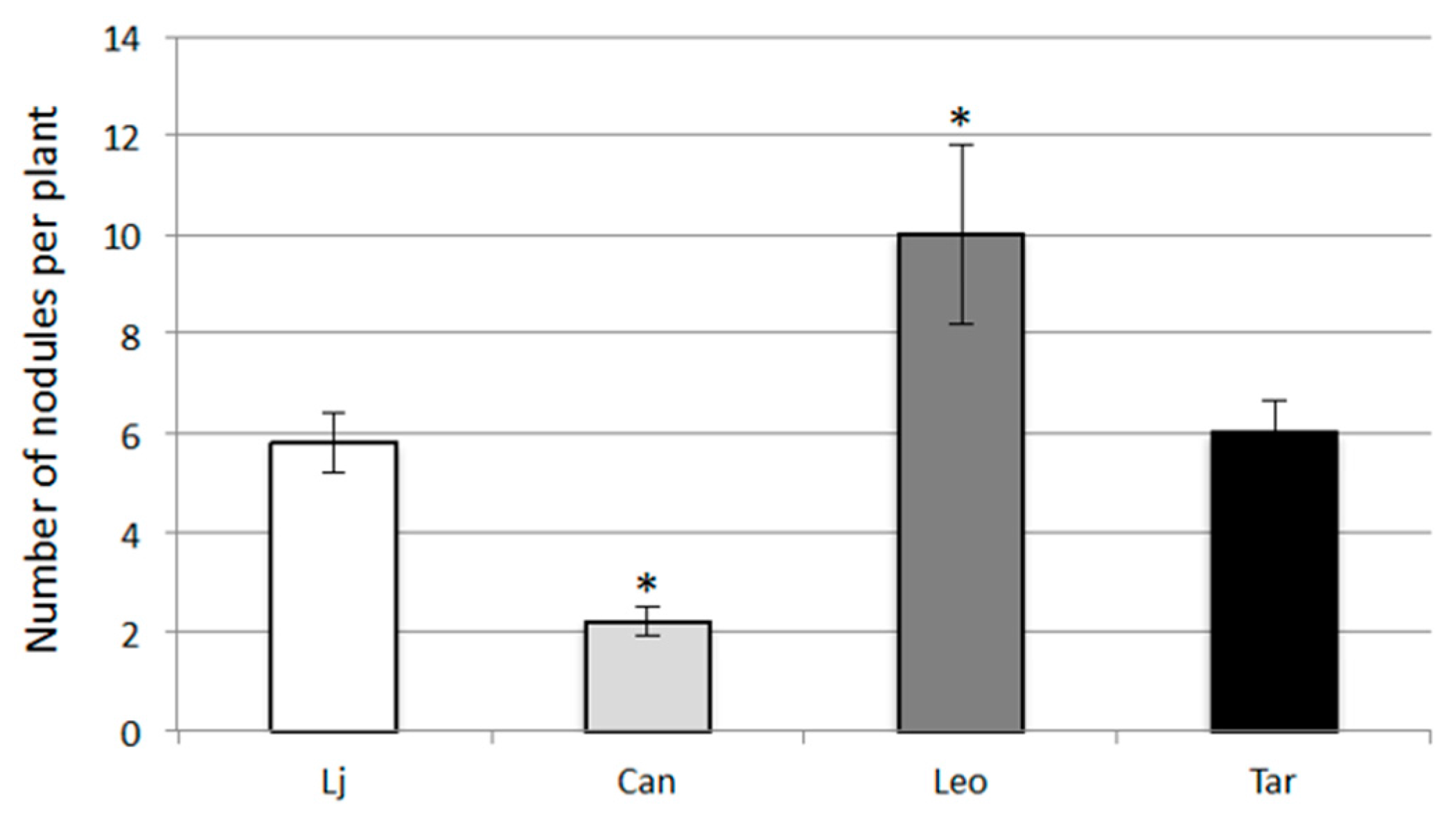
2.3. Symbiotic Performances of L. corniculatus cvs.
3. Discussion
4. Experimental Section
4.1. Plant Material and Growth Conditions
4.2. Inoculation Procedure and Nodulation Test
4.3. Root Analyses
4.4. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Graham, P.H.; Vance, C.P. Legumes: Importance and constraints to greater use. Plant Physiol. 2003, 131, 872–877. [Google Scholar] [CrossRef] [PubMed]
- Kinkema, M.; Scott, P.T.; Gresshoff, P.M. Legume nodulation: Successful symbiosis through short- and long-distance signalling. Funct. Plant Biol. 2006, 33, 707–721. [Google Scholar] [CrossRef]
- Rochon, J.J.; Doyle, C.J.; Greef, J.M.; Hopkins, A.; Molle, G.; Sitzia, M.; Scholefield, D.; Smith, C.J. Grazing legumes in Europe: A review of their status, management, benefits, research needs and future prospects. Grass Forage Sci. 2004, 59, 197–214. [Google Scholar] [CrossRef]
- Luscher, A.; Mueller-Harvey, J.F.; Soussana, R.M.; Peyraud, J.L. Potential of legume-based grassland-livestock systems in Europe: A review. Grass Forage Sci. 2014, 69, 206–228. [Google Scholar] [CrossRef] [PubMed]
- Kusvuran, A.; Ralice, Y.; Saglamtimur, T. Determining the biomass production capacities of certain forage grasses and legumes and their mixtures under Mediterranean regional conditions. Acta Adv. Agric. Sci. 2014, 2, 13–24. [Google Scholar]
- Pypers, P.S.; Verstraete, C.P.; Merckx, R. Changes in mineral nitrogen, phosphorus availability and salt-extractable aluminium following the application of green manure residues in two weathered soils of south Vietnam. Soil Biol. Biochem. 2005, 37, 163–172. [Google Scholar] [CrossRef]
- Vanlawe, B.J.; Wendt, K.E.; Giller, M.; Corbeels, B.; Nolte, C. A fourth principle is required to define conservation agriculture in sub-Saharan Africa: The appropriate use of fertilizer to enhance crop productivity. Field Crops Res. 2014, 155, 10–13. [Google Scholar] [CrossRef]
- Caporali, F.; Campiglia, E.; Mancinelli, R.; Paolini, R. Maize performances as influenced by winter cover crop green manuring. Ital. J. Agron. 2004, 8, 37–45. [Google Scholar]
- Carroll, B.; Gresshoff, P.M. Nitrate inhibition of nodulation and nitrogen fixation in white clover. Z. Pflanzenphysiol. 1983, 110, 69–76. [Google Scholar] [CrossRef]
- Day, D.A.; Carroll, B.J.; Delves, A.C.; Gresshoff, P.M. Relationship between autoregulation and nitrate inhibition of nodulation in soybeans. Physiol. Plant. 1989, 75, 37–42. [Google Scholar] [CrossRef]
- Fujikake, H.; Yamazaki, A.; Ohtake, N.; Sueyoshi, K.; Matsuhashi, S.; Ito, T.; Mizuniwa, C.; Kume, T.; Hashimoto, S.; Ishioka, N.S.; et al. Quick and reversible inhibition of soybean root nodule growth by nitrate involves a decrease in sucrose supply to nodules. J. Exp. Bot. 2003, 54, 1379–1388. [Google Scholar] [CrossRef] [PubMed]
- Barbulova, A.; Rogato, A.; D’Apuzzo, E.; Omrane, S.; Chiurazzi, M. Differential effects of combined N sources on early steps of the Nod factor-dependent transduction pathway in Lotus japonicus. Mol. Plant-Microbes Interact. 2007, 20, 994–1003. [Google Scholar] [CrossRef] [PubMed]
- Omrane, S.; Ferrarini, A.; D’Apuzzo, E.; Rogato, A.; Delledonne, M.; Chiurazzi, M. Symbiotic competence in Lotus japonicus is affected by plant nitrogen status: Transcriptomic identification of genes affected by a new signalling pathway. New Phytol. 2009, 183, 380–394. [Google Scholar] [CrossRef] [PubMed]
- Omrane, S.; Chiurazzi, M. A variety of regulatory mechanisms are involved in the nitrogen-dependent modulation of the nodule organogenesis program in legume roots. Plant Signal Behav. 2009, 4, 1066–1068. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, S.; Ohnishi, E.; Sato, S.; Takahashi, H.; Nakazono, M.; Tabata, S.; Kawaguchi, M. Nod factor/nitrate-induced CLE genes that drive HAR1-mediated systemic regulation of nodulation. Plant Cell Physiol. 2009, 50, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Reid, D.E.; Ferguson, B.J.; Gresshoff, P.M. Inoculation- and nitrate-induced CLE peptides of soybean control NARK-dependent nodule formation. Mol. Plant Microbe Interact. 2011, 24, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Jeudy, C.; Ruffell, S.; Freixes, S.; Tillard, P.; Santoni, A.L.; Morel, S.; Journet, E.P.; Duc, G.; Gojon, A.; Lepetit, M. Adaptation of Medicago truncatula to nitrogen limitation is modulated via local and systemic nodule developmental responses. New Phytol. 2010, 185, 817–828. [Google Scholar] [CrossRef] [PubMed]
- Ledgard, S.F.; Steele, K.W. Biological nitrogen fixation in mixed legume/grass pastures. Plant Soil. 1992, 141, 137–153. [Google Scholar] [CrossRef]
- Vasileva, V.; Ilieva, A. Sustainable yield index in some mixtures. J. Glob. Innov. Agric. Soc. Sci. 2016, 4, 55–61. [Google Scholar] [CrossRef]
- Waghorn, G.; Ulyatt, M.; John, A.; Fisher, M. The effect of condensed tannins on the site of digestion of amino acids and other nutrients in sheep fed on Lotus corniculatus L. Br. J. Nutr. 1987, 57, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Jean-Blain, C. Nutritional and toxicological aspects of tannins. Rev. Med. Vet. 1998, 149, 911–920. [Google Scholar]
- Banuelos, G.; Cardon, G.; Mackey, B.; Ben-Asher, J.; Wu, L.; Beuselink, P. Boron and selenium removal in boron-laden soil by birdsfoot trefoil. Lotus Newsl. 1992, 23, 32–35. [Google Scholar]
- Blumenthal, M.; McGraw, R. Lotus adaptation, use and management. In Trefoil: The Science and Technology of Lotus; Beuselink, P., Ed.; American Society of Agronomy: Fitchburg, WI, USA, 1999; pp. 97–120. [Google Scholar]
- Weeb, K.; Jones, S.; Robbins, M.; Minchin, F. Characterization of transgenic root culture of Trifolium repens, T. pratense and Lotus corniculatus and transgenic plants of L. corniculatus. Plant Sci. 1990, 70, 243–254. [Google Scholar] [CrossRef]
- Handberg, K.; Stougaard, J. Lotus japonicus, an autogamous, diploid legume species for classical and molecular genetics. Plant J. 1992, 2, 487–496. [Google Scholar] [CrossRef]
- Jiang, Q.; Gresshoff, P.M. Classical and molecular genetics of the model legume Lotus japonicus. Mol. Plant-Microbes Interact. 1997, 10, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, S.; Andria, V.; Reichenauer, T.G.; Smalla, K.; Sessitsch, A. Phylogenetic and functional diversity of alkane degrading bacteria associated with Italian ryegrass (Lolium multiflorum) and Birdsfoot trefoil (Lotus corniculatus) in a petroleum oil-contaminated environment. J. Hazard. Mater. 2010, 184, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, S.; Ripka, K.; Reichenauer, T.G.; Andria, V.; Azfal, M.; Sessitsch, A. Hydrocarbon degradation and plant colonization by selected bacterial strains isolated from Italian ryegrass and birdsfoot trefoil. J. Appl. Microbiol. 2010, 109, 1389–1401. [Google Scholar] [CrossRef] [PubMed]
- Keith, C.N.; McKersie, B.D. The effect of abscisic acid on the freezing tolerance of callus cultures of Lotus corniculatus. Plant Physiol. 1986, 80, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Pofelis, S.; Le, H.; Grant, W.F. The development of sulfonylurea herbicide-resistant birdsfoot trefoil (Lotus corniculatus) plants from in vitro selection. Theor. Appl. Genet. 1992, 83, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Carron, T.R.; Robbins, M.P.; Morris, P. Genetic modification of condensed tannins biosynthesis in Lotus corniculatus. 1. heterologous antisense dihydroflavonol reductase down-regulates tannin accumulation in “hairy root” cultures. Theor. Appl. Genet. 1994, 87, 1006–1015. [Google Scholar] [CrossRef] [PubMed]
- Robbins, M.P.; Paolocci, F.; Hughes, J.W.; Turchetti, V.; Allison, G.; Arcioni, S.; Morris, P.; Damiani, F. Sn, a maize bHLH gene, modulates anthocyanin and condensed tannin pathways in Lotus corniculatus. J. Exp. Bot. 2003, 54, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Paolocci, F.; Bovone, T.; Tosti, N.; Arcioni, S.; Damiani, F. Light and an exogenous transcription factor qualitatively and quantitatively affect the biosynthetic pathway of condensed tannins in Lotus corniculatus leaves. J. Exp. Bot. 2005, 56, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Paolocci, F.; Robbins, M.P.; Madeo, L.; Arcioni, S.; Martens, S.; Damiani, F. Ectopic expression of a basic helix-loop-helix gene transactivates parallel pathways of proanthocyanidin biosynthesis. structure, expression analysis, and genetic control of leucoanthocyanidin 4-reductase and anthocyanin reductase genes in Lotus corniculatus. Plant Physiol. 2007, 143, 504–516. [Google Scholar] [PubMed]
- Sun, Z.M.; Zhou, M.L.; Xiao, X.G.; Tang, Y.X.; Wu, Y.M. Genome-wide analysis of AP2/ERF family genes from Lotus corniculatus shows LcERF054 enhances salt tolerance. Funct. Integr. Genom. 2014, 14, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.M.; Zhou, M.L.; Wang, D.; Tang, Y.X.; Lin, M.; Wu, Y.M. Overexpression of the Lotus corniculatus soloist gene LcAP2/ERF107 enhances tolerance to salt stress. Protein Pept. Lett. 2016, 23, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Ilieva, A.; Vasileva, V.; Katova, A. The effect of mixed planting of birdfoot trefoil, sainfoin, subterranean clover and tall fescue on nodulation and nitrate reductase activity in shoots. J. Glob. Agric. Ecol. 2015, 4, 222–228. [Google Scholar]
- Ilieva, A.; Vasileva, V. Biochemical composition and phosphorus use efficiency in some mixtures. Intern. J. Bioassays 2016, 5, 4694–4698. [Google Scholar]
- Nelissen, H.; Moloney, M.; Inzè, D. Translational research: From pot to plot. Plant Biotechnol. J. 2014, 12, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Ogunkule, A.O.; Beckett, P.H.T. The efficiency of pot trials, or trials on undisturbed soil cores, as predictors of crop behaviour in the field. Plant Soil 1988, 107, 85–93. [Google Scholar] [CrossRef]
- Sherrell, C.G.; Saunders, W.M.H. Factors affecting growth and response of white clover in pots to appled phosphorus: I level of watering at which pots are maintained. N. Z. J. Agric. Res. 1974, 17, 19–29. [Google Scholar] [CrossRef]
- Stevenson, D.S. Interaction between soil volume and added fertilizers under continuous watering. Can. J. Sci. 1978, 58, 271–274. [Google Scholar] [CrossRef]
- Dhondt, S.; Wuyts, N.; Inzè, D. Cell to whole-plant phenotyping: The best is yet to come. Trends Plant Sci. 2013, 18, 428–439. [Google Scholar] [CrossRef] [PubMed]
- Fiorani, F.; Schurr, U. Future scenarios for plant phenotyping. Annu. Rev. Plant Biol. 2013, 64, 267–291. [Google Scholar] [CrossRef] [PubMed]
- Israel, D.W. Investigation of the role of phosphorous in symbiotic dinitrogen fixation. Plant Physiol. 1987, 84, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorous acquisition and use: Critical adaptations by plants for securing a non-renewable resource. New Phytol. 2003, 157, 427–447. [Google Scholar] [CrossRef]
- Kleinert, A.; Venter, M.; Kossmann, J.; Valentine, A. The reallocation of carbon in P deficient lupins affects biological nitrogen fixation. J. Plant Physiol. 2014, 171, 1619–1624. [Google Scholar] [CrossRef] [PubMed]
- Schwendiman, J.L.; Foster, R.B.; Haglund, O.K. The influence of climate, soils and management on the root development of grass species in western states. In Proceedings of the Annual Meeting American Forage and Grassland Council, New Orleans, LA, USA, 1–4 February 1966; pp. 40–57.
- Torales, A.T.A.; Deregibus, V.A.; Moauro, P.R. Phosphorous absorption capacity of Lotus corniculatus and Festuca arundinacea during sward establishment. N. Z. J. Agric. Res. 1998, 41, 307–312. [Google Scholar] [CrossRef]
- Complainville, A.; Brocard, L.; Roberts, I.; Dax, E.; Sever, N.; Sauer, N.; Kondorosi, A.; Wolf, S.; Oparka, K.; Crespi, M. Nodule initiation involves the creation of a new symplasmic field in specific root cells of medicago species. Plant Cell 2003, 120, 2778–2791. [Google Scholar] [CrossRef] [PubMed]
- Vance, C.P. Carbon and Nitrogen metabolism in legume nodules. In Nitrogen-Fixing Leguminous Symbioses; Dilworth, M.J., James, E.K., Sprent, J.I., Eds.; Springer: Berlin, Germany, 2008; pp. 293–320. [Google Scholar]
- D’Apuzzo, E.; Valkov, T.V.; Parlati, A.; Omrane, S.; Barbulova, A.; Sainz, M.M.; Lentini, M.; Esposito, S.; Rogato, A.; Chiurazzi, M. PII overexpression in Lotus japonicus affects nodule activity in permissive low nitrogen conditions and increases nodule numbers in high nitrogen treated plants. Mol. Plant-Microbes Interact. 2014, 28, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Barbulova, A.; D’Apuzzo, E.; Rogato, A.; Chiurazzi, M. Improved procedures for in vitro regeneration and for phenotypical analysis in the model legume Lotus japonicus. Func. Plant Biol. 2005, 32, 529–536. [Google Scholar]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valkov, V.T.; Chiurazzi, M. An In Vitro Procedure for Phenotypic Screening of Growth Parameters and Symbiotic Performances in Lotus corniculatus Cultivars Maintained in Different Nutritional Conditions. Plants 2016, 5, 40. https://doi.org/10.3390/plants5040040
Valkov VT, Chiurazzi M. An In Vitro Procedure for Phenotypic Screening of Growth Parameters and Symbiotic Performances in Lotus corniculatus Cultivars Maintained in Different Nutritional Conditions. Plants. 2016; 5(4):40. https://doi.org/10.3390/plants5040040
Chicago/Turabian StyleValkov, Vladimir Totev, and Maurizio Chiurazzi. 2016. "An In Vitro Procedure for Phenotypic Screening of Growth Parameters and Symbiotic Performances in Lotus corniculatus Cultivars Maintained in Different Nutritional Conditions" Plants 5, no. 4: 40. https://doi.org/10.3390/plants5040040





