Endocytic Pathways and Recycling in Growing Pollen Tubes
Abstract
:1. Introduction
2. Endocytic Pathways
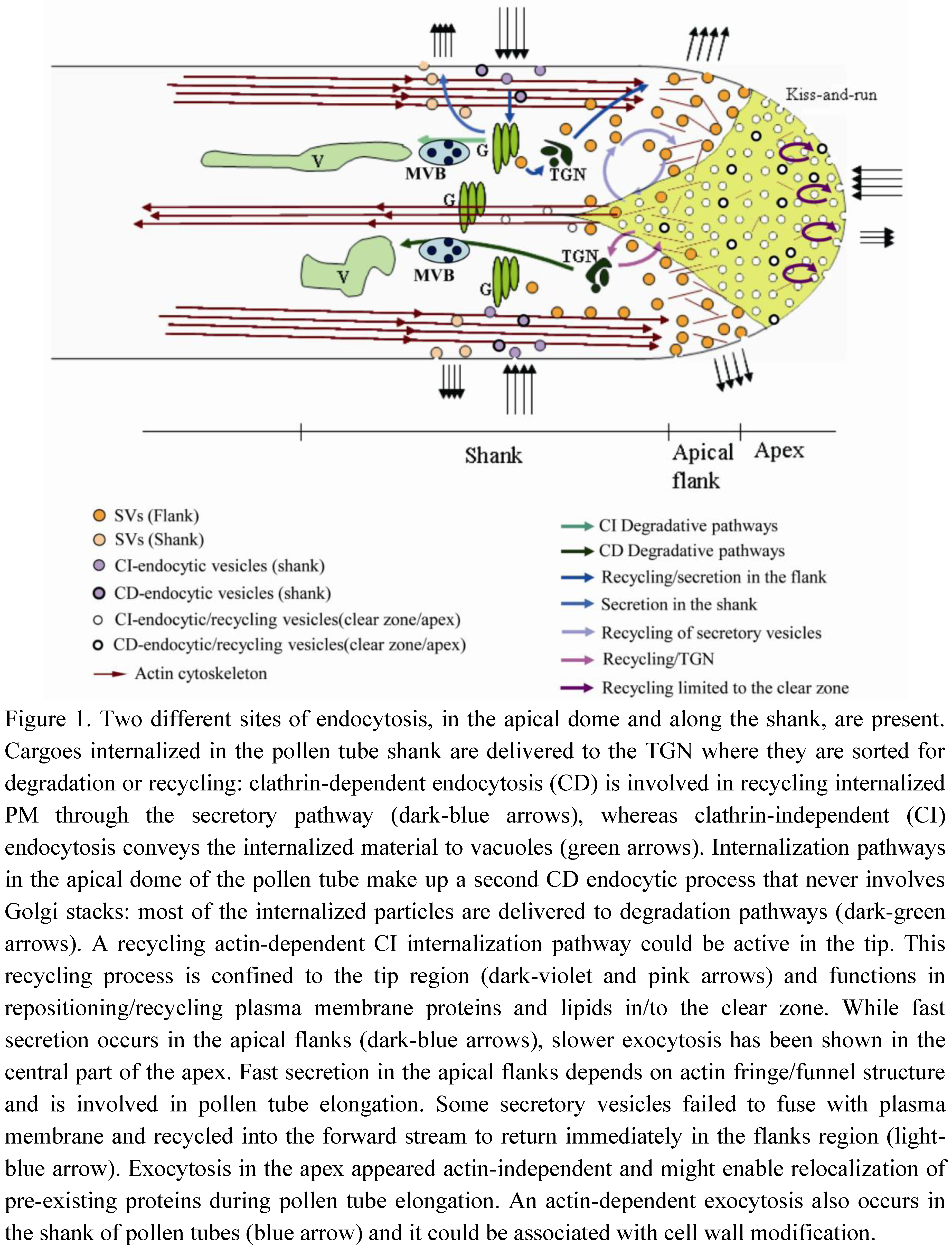

3. Exocytosis/Recycling
4. Clathrin-Dependent and Clathrin-Independent Endocytosis
5. Conclusions
References
- Cheung, A.Y.; Wu, H. Structural and functional compartmentalization in pollen tubes. J. Exp. Bot. 2007, 58, 75–82. [Google Scholar] [CrossRef]
- Li, Y.Q.; Moscatelli, A.; Cai, G.; Cresti, M. Functional interactions among cytoskeleton, membranes, and cell wall in the pollen tube of flowering plants. Int. Rev. Cytol. 1997, 176, 133–199. [Google Scholar] [CrossRef]
- Taylor, L.P.; Hepler, P.K. Pollen germination and tube growth. Annu. Rev. Physiol. Plant Mol. Biol. 1997, 48, 461–491. [Google Scholar] [CrossRef]
- Hepler, P.K.; Vidali, L.; Cheung, A.Y. Polarized cell growth in higher plants. Annu. Rev. Cell Dev. Biol. 2001, 17, 159–187. [Google Scholar] [CrossRef]
- Heslop-Harrison, J.; Heslop-Harrison, Y. Dynamic aspects of the apical zonation in the angiosperm pollen tube. Sex. Plant Reprod. 1990, 3, 187–194. [Google Scholar]
- Cárdenas, L.; Lovy-Wheeler, A.; Wilsen, K.L.; Hepler, P.K. Actin polymerization promotes the reversal of streaming in the apex of pollen tubes. Cell Motil. Cytoskel. 2005, 61, 112–127. [Google Scholar] [CrossRef]
- Vidali, L.; McKenna, S.T.; Hepler, P.K. Actin polymerization is essential for pollen tube growth. Mol. Biol. Cell. 2001, 12, 2534–2545. [Google Scholar]
- Bove, J.; Vaillancourt, B.; Kroeger, J.; Hepler, P.K.; Wiseman, P.W.; Geitmann, A. Magnitude and direction of vesicle dynamics in growing pollen tubes using spatiotemporal image correlation spectroscopy and fluorescence recovery after photobleaching. Plant Physiol. 2008, 147, 1646–1658. [Google Scholar] [CrossRef]
- Kroeger, J.H.; Bou Daher, F.; Grant, M.; Geitmann, A. Microfilament orientation constrains vesicle flow and spatial distribution in growing pollen tubes. Biophys. J. 2009, 97, 1822–1831. [Google Scholar] [CrossRef]
- Heslop-Harrison, J.; Heslop-Harrison, Y. The actin cytoskeleton in unfixed pollen tubes following microwave-accelerated DMSO permeabilization and TRITC-phalloidin staining. Sex. Plant Reprod. 1991, 4, 6–11. [Google Scholar]
- Derksen, J.; Rutten, T.L.M.; van Amstel, T.; de Win, A.; Doris, F.; Steer, M. Regulation of pollen tube growth. Acta Bot. Neerl. 1995, 44, 93–119. [Google Scholar]
- Lovy-Wheeler, A.; Wilsen, K.L.; Baskin, T.I.; Hepler, P.K. Enhanced fixation reveals the apical cortical fringe of actin filaments as a consistent feature of the pollen tube. Planta 2005, 221, 95–104. [Google Scholar] [CrossRef]
- Lovy-Wheeler, A.; Kunkel, J.G.; Allwood, E.G.; Hussey, P.J.; Hepler, P. Oscillatory increases in alkalinity anticipate growth and may regulate actin dynamics in pollen tubes of lily. Plant Cell 2006, 18, 2182–2193. [Google Scholar] [CrossRef]
- Cárdenas, L.; Lovy-Wheeler, A.; Kunkel, J.G.; Hepler, P.K. Pollen tube growth oscillations and intracellular calcium levels are reversibly modulated by actin polymerization. Plant Physiol. 2008, 146, 1611–1621. [Google Scholar] [CrossRef]
- Vidali, L.; Round, C.M.; Hepler, P.K.; Bezanilla, M. Lifeact-mEGFP reveals a dynamic apical Factin network in tip growing plant cells. PLoS One 2009, 4, 1–15. [Google Scholar]
- Moscatelli, A.; Ciampolini, F.; Rodighiero, S.; Onelli, E.; Cresti, M.; Santo, N.; Idilli, A.I. Distinct endocytic pathways identified in tobacco pollen tubes using charged nanogold. J. Cell Sci. 2007, 120, 3804–3819. [Google Scholar] [CrossRef]
- Zonia, L.; Munnik, T. Vesicle trafficking dynamics and visualization of zones of exocytosis and endocytosis in tobacco pollen tubes. J. Exp. Bot. 2008, 59, 861–873. [Google Scholar] [CrossRef]
- Parton, R.M.; Fischer-Parton, S.; Watahiki, M.K.; Trewavas, A.J. Dynamics of the apical vesicle accumulation and the rate of growth are related in individual pollen tubes. J. Cell Sci. 2001, 114, 2685–2695. [Google Scholar]
- Wang, Q.; Kong, L.; Hao, H.; Wang, X.; Lin, J.; Samaj, J.; Baluska, F. Effects of Brefeldin A on pollen germination and tube growth. Antagonistic effects on endocytosis and secretion. Plant Physiol. 2005, 139, 1692–1703. [Google Scholar] [CrossRef]
- Steer, M.W.; Steer, J.M. Pollen tube tip growth. New Phytol. 1989, 111, 323–358. [Google Scholar] [CrossRef]
- Murphy, A.S.; Bandyopadhyay, A.; Holstein, S.E.; Peer, W.A. Endocytotic cycling of membrane proteins. Annu. Rev. Plant Biol. 2005, 56, 221–251. [Google Scholar] [CrossRef]
- Fowke, L.C.; Tanchak, M.A.; Galway, M.E. Ultrastructural cytology of the endocytic pathways in plants. In Endocytosis, Exocytosis and Vesicle Traffic in Plants; Hawes, C.R., Coleman, J.O.D., Evans, D.E., Eds.; UK Cambridge University Press: Cambridge, UK, 1991; pp. 15–40. [Google Scholar]
- Dettmer, J.; Hong-Hermesdorf, A.; Stierhof, Y.D.; Schumacher, K. Vacuolar H+ATPase activity is required for endocytic and secretory trafficking in Arabidopsis. Plant Cell 2006, 18, 715–730. [Google Scholar] [CrossRef]
- Lam, S.K.; Siu, C.L.; Hillmer, S.; Jang, S.; An, G.; Robinson, D.G.; Jiang, L. Rice SCAMP1 defines clathrin coated, trans Golgi-located tubular-vesicular structures as an early endosome in tobacco BY-2 cells. Plant Cell 2007, 19, 296–319. [Google Scholar] [CrossRef]
- Viotti, C.; Bubeck, J.; Stierhof, Y.D.; Krebs, M.; Langhans, M.; van der Berg, W.; van Dongen, W.; Richter, S.; Geldner, N.; Takano, J.; et al. Endocytic and secretory traffic in arabidopsis merge in the Trans Golgi Network/Early endosome, an independent and highly dynamic organelle. Plant Cell 2010, 22, 1344–1357. [Google Scholar] [CrossRef]
- Gendre, D.; Oh, J.; Boutté, Y.; Best, J.G.; Samuels, L.; Nilsson, R.; Uemura, T.; Marchant, A.; Bennett, M.J.; Grebe, M.; et al. Conserved Arabidopsis ECHIDNA protein mediates trans-Golgi-network trafficking and cell elongation. Proc. Natl. Acad. Sci.USA 2011, 108, 8048–8053. [Google Scholar] [CrossRef]
- Herberth, S.; Shahriari, M.; Bruderek, M.; Hessner, F.; Müller, B.; Hülskamp, M.; Schellmann, S. Artificial ubiquitylation is sufficient for sorting of a plasma membrane ATPase to the vacuolar lumen of Arabidopsis cells. Planta 2012, 236, 63–77. [Google Scholar] [CrossRef]
- Raiborg, C.; Stenmark, H. The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature 2009, 458, 445–452. [Google Scholar]
- Scheuring, D.; Künzl, F.; Viotti, C.; San Wan Yan, M.; Jiang, L.; Schellmann, S.; Robinson, D.G.; Pimpl, P. Ubiquitin initiates sorting of Golgi and plasma membrane proteins into the vacuolar degradation pathway. BMC Plant Biol. 2012. [Google Scholar] [CrossRef]
- Onelli, E.; Prescianotto-Baschong, C.; Caccianiga, M.; Moscatelli, A. Clathrin-dependent and independent endocytic pathways in tobacco protoplasts revealed by labelling with charged nanogold. J. Exp. Bot. 2008, 59, 3051–3068. [Google Scholar] [CrossRef]
- Foresti, O.; Gershlick, D.C.; Bottanelli, F.; Hummel, E.; Hawes, C.; Denecke, J. A recycling-defective vacuolar sorting receptor reveals an intermediate compartment situated between prevacuoles and vacuoles in tobacco. Plant Cell 2010, 22, 3992–4008. [Google Scholar] [CrossRef]
- Niemes, S.; Langhans, M.; Viotti, C.; Scheuring, D.; San Wan Yan, M.; Jiang, L.; Hillmer, S.; Robinson, D.G.; Pimpl, P. Retromer recycles vacuolar sorting receptors from the trans-Golgi network. Plant J. 2010, 61, 107–121. [Google Scholar] [CrossRef]
- Chavarría-Krauser, A.; Yejie, D. A model of plasma membrane flow and cytosis regulation in growing pollen tubes. J. Theor. Biol. 2011, 285, 10–24. [Google Scholar] [CrossRef]
- Wang, H.; Tse, Y.C.; Law, A.H.; Sun, S.S.; Sun, Y.B.; Xu, Z.F.; Hillmer, S.; Robinson, D.G.; Jiang, L. Vacuolar sorting receptors (VSRs) and secretory carrier membrane proteins (SCAMPs) are essential for pollen tube growth. Plant J. 2010, 61, 826–838. [Google Scholar] [CrossRef]
- Moscatelli, A.; Idilli, A.I. Pollen tube growth: A delicate equilibrium between secretory and endocytic pathways. J. Integr. Plant Biol. 2009, 51, 727–739. [Google Scholar] [CrossRef]
- Cheung, A.Y.; Wu, H. Structural and signaling networks for the polar cell growth machinery in pollen tubes. Annu. Rev. Plant Biol. 2008, 59, 547–572. [Google Scholar] [CrossRef]
- Zou, Y.; Aggarwal, M.; Zheng, W.G.; Wu, H.M.; Cheung, A.Y. Receptor-like kinases as surface regulators for RAC/ROP-mediated pollen tube growth and interaction with the pistil. AoB Plants 2011. [Google Scholar] [CrossRef]
- Lovy-Wheeler, A.; Cárdenas, L.; Kunkel, J.G.; Hepler, P.K. Differential organelle movement on the actin cytoskeleton in lily pollen tubes. Cell Motil. Cytoskel. 2007, 64, 217–232. [Google Scholar] [CrossRef]
- Moscatelli, A.; Idilli, A.I.; Rodighiero, S.; Caccianiga, M. Inhibition of actin polymerisation by low concentration Latrunculin B affects endocytosis and alters exocytosis in shank and tip of tobacco pollen tubes. Plant Biol. 2012. [Google Scholar] [CrossRef]
- Kost, B.; Spielhofer, P.; Chua, N.H. A GFP-mouse talin fusion protein labels plant actin filaments in vivo and visualizes the actin cytoskeleton in growing pollen tubes. Plant J. 1998, 16, 393–401. [Google Scholar] [CrossRef]
- Gibbon, B.C.; Kovar, D.R.; Staiger, C.J. Latrunculin B has different effects on pollen germination and tube growth. Plant Cell 1999, 11, 2349–2363. [Google Scholar]
- Chen, C.Y.; Wong, E.I.; Vidali, L.; Estavillo, A.; Hepler, P.K.; Wu, H.M.; Cheung, A.Y. The regulation of actin organization by actin-depolymerizing factor in elongating pollen tubes. Plant Cell 2002, 14, 2175–2190. [Google Scholar] [CrossRef]
- Kost, B.; Lemichez, E.; Spielhofer, P.; Hong, Y.; Tolias, K.; Carpenter, C.; Chua, N.H. Rac homologues and compartmentalized phosphatidylinositol 4,5-bisphosphate act in a common pathway to regulate polar pollen tube growth. J. Cell Biol. 1999, 145, 317–330. [Google Scholar] [CrossRef]
- Fu, Y.; Wu, G.; Yang, Z. Rop GTPase-dependent dynamics of tip-localized F-actin controls tip growth in pollen tubes. J. Cell Biol. 2001, 152, 1019–1032. [Google Scholar] [CrossRef]
- Kroeger, J.H.; Geitmann, A. Pollen tube growth: Getting a grip on cell biology through modelling. Mech. Res. Commun. 2012, 42, 32–39. [Google Scholar] [CrossRef]
- Cole, R.A.; Synek, L.; Zarsky, V.; Fowler, J.E. SEC8, a subunit of the putative Arabidopsis exocyst complex, facilitates pollen germination and competitive pollen tube growth. Plant Physiol. 2005, 138, 2005–2018. [Google Scholar] [CrossRef]
- Wen, T.J.; Hochholdinger, F.; Sauer, M.; Bruce, W.; Schnable, P.S. The roothairless1 gene of maize encodes a homolog of sec3, which is involved in polar exocytosis. Plant Physiol. 2005, 138, 1637–1643. [Google Scholar] [CrossRef]
- Richter, S.; Müller, L.M.; Stierhof, Y.D.; Mayer, U.; Takada, N.; Kost, B.; Vieten, A.; Geldner, N.; Koncz, C.; Jürgens, G. Polarized cell growth in Arabidopsis requires endosomal recycling mediated by GBF1-related ARF exchange factors. Nat. Cell Biol. 2012, 14, 80–86. [Google Scholar]
- Zheng, Z.L.; Yang, Z. The Rop GTPase: An emerging signaling switch in plants. Plant Mol. Biol. 2000, 44, 1–9. [Google Scholar] [CrossRef]
- Fu, Y.; Yang, Z. Rop GTPase: A master switch of cell polarity development in plants. Trends Plant Sci. 2001, 6, 545–547. [Google Scholar] [CrossRef]
- Gu, Y.; Vernoud, V.; Fu, Y.; Yang, Z. ROP GTPase regulation of pollen tube growth through the dynamics of tip-localized F-actin. J. Exp. Bot. 2003, 54, 93–101. [Google Scholar] [CrossRef]
- Gu, Y.; Wang, Z.; Yang, Z. ROP/RAC GTPase: An old new master regulator for plant signaling. Curr. Opin. Plant Biol. 2004, 7, 527–536. [Google Scholar] [CrossRef]
- Pollard, T.D. Regulation of actin filament assembly by Arp2/3 complex and formins. Annu. Rev. Biophys. Biomol. Struct. 2007, 36, 451–477. [Google Scholar] [CrossRef]
- Goode, B.L.; Eck, M.J. Mechanism and function of formins in the control of actin assembly. Annu. Rev. Biochem. 2007, 76, 593–627. [Google Scholar] [CrossRef]
- Ishizaki, T.; Morishima, Y.; Okamoto, M.; Furuyashiki, T.; Kato, T.; Narumiya, S. Coordination of microtubules and actin cytoskeleton by the Rho effector mDia1. Nat. Cell Biol. 2001, 3, 8–14. [Google Scholar] [CrossRef]
- Lancelle, S.A.; Cresti, M.; Hepler, P.K. Ultrastructure of the cytoskeleton in freeze-substituted pollen tubes of Nicotiana alata. Protoplasma 1987, 140, 141–150. [Google Scholar] [CrossRef]
- Lancelle, S.A.; Hepler, P.K. Ultrastructure of freeze-substituted pollen tubes of Lilium longiflorum. Protoplasma 1992, 167, 215–230. [Google Scholar] [CrossRef]
- Idilli, A.I.; Onelli, E.; Moscatelli, A. Low concentration of LatB dramatically changes the microtubule organization and the timing of vegetative nucleus/generative cell entrance in tobacco pollen tubes. Plant Signal. Behav. 2012, 7, 947–950. [Google Scholar] [CrossRef]
- Cheung, A.Y.; Wu, H.M. Overexpression of an Arabidopsis formin stimulates supernumerary actin cable formation from pollen tube cell membrane. Plant Cell 2004, 16, 257–269. [Google Scholar] [CrossRef]
- Ye, J.; Zheng, Y.; Yan, A.; Chen, N.; Wang, Z.; Huang, S.; Yang, Z. Arabidopsis formin3 directs the formation of actin cables and polarized growth in pollen tubes. Plant Cell 2009, 21, 3868–3884. [Google Scholar] [CrossRef]
- Cheung, A.Y.; Niroomand, S.; Zou, Y.; Wu, H.M. A transmembrane formin nucleates subapical actin assembly and controls tip-focused growth in pollen tubes. Proc. Natl. Acad. Sci. USA 2010, 107, 16390–16395. [Google Scholar]
- Stevenson, J.M.; Perera, I.Y.; Heilmann, I.I.; Persson, S.; Boss, W.F. Inositol signaling and plant growth. Trends Plant Sci. 2000, 5, 252–258. [Google Scholar] [CrossRef]
- Martin, T.F.J. Phosphoinositides as spatial regulators of membrane traffic. Curr. Opin. Neurobiol. 1997, 7, 331–338. [Google Scholar] [CrossRef]
- Kock, M.; Holt, M. Coupling exo- and endocytosis: An essential role for PIP2 at the synapse. Biochim. Biophys. Acta 2012, 1821, 1114–1132. [Google Scholar]
- Helling, D.; Possart, A.; Cottier, S.; Klahre, U.; Kost, B. Pollen tube tip growth depends on plasma membrane polarization mediated by tobacco PLC3 activity and endocytic membrane recycling. Plant Cell 2006, 18, 3519–3534. [Google Scholar] [CrossRef]
- Sousa, E.; Kost, B.; Malhó, R. Arabidopsis phosphatidylinositol-4-monophosphate 5-kinase 4 regulates pollen tube growth and polarity by modulating membrane recycling. Plant Cell 2008, 20, 3050–3064. [Google Scholar]
- Dowd, P.E.; Coursol, S.; Skirpan, A.L.; Kao, T.H.; Gilroy, S. Petunia phospholipase C1 is involved in pollen tube growth. Plant Cell 2006, 18, 1438–1453. [Google Scholar] [CrossRef]
- Ischebeck, T.; Stenzel, I.; Heilmann, I. Type B phosphatidylinositol-4-phosphate 5-kinases mediate Arabidopsis and Nicotiana tabacum pollen tube growth by regulating apical pectin secretion. Plant Cell 2008, 20, 3312–3330. [Google Scholar] [CrossRef]
- Franklin-Tong, V.E.; Drobak, B.K.; Allan, A.C.; Watkins, P.; Trewavas, A.J. Growth of Pollen Tubes of Papaver rhoeas Is Regulated by a Slow-Moving Calcium Wave Propagated by Inositol 1,4,5-Trisphosphate. Plant Cell 1996, 8, 1305–1321. [Google Scholar]
- Monteiro, D.; Liu, Q.; Lisboa, S.; Scherer, G.E.; Quader, H.; Malhó, R. Phosphoinositides and phosphatidic acid regulate pollen tube growth and reorientation through modulation of [Ca2+]c and membrane secretion. J. Exp. Bot. 2005, 56, 1665–1674. [Google Scholar] [CrossRef]
- Ischebeck, T.; Stenzel, I.; Hempel, F.; Jin, X.; Mosblech, A.; Heilmann, I. Phosphatidylinositol-4,5-bisphosphate influences Nt-Rac5-mediated cell expansion in pollen tubes of Nicotiana tabacum. Plant J. 2011, 65, 453–468. [Google Scholar] [CrossRef]
- Kost, B. Spatial control of Rho (Rac-Rop) signaling in tip-growing plant cells. Trends Cell Biol. 2008, 18, 119–127. [Google Scholar] [CrossRef]
- Klahre, U.; Becker, C.; Schmitt, A.C.; Kost, B. Nt-RhoGDI2 regulates Rac/Rop signaling and polar cell growth in tobacco pollen tubes. Plant J. 2006, 46, 1018–1031. [Google Scholar] [CrossRef]
- De Win, A.H.N.; Pierson, E.S.; Derksen, J. Rational analysis of organelle trajectories in tobacco pollen tubes reveal characteristics of actomyosin cytoskeleton. Biophys. J. 1999, 76, 1648–1658. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, E.S.; Choi, Y.; Hwang, I.; Staiger, C.J.; Chung, Y.Y.; Lee, Y. The Arabidopsis phosphatidylinositol 3-kinase is important for pollen development. Plant Physiol. 2008, 147, 1886–1897. [Google Scholar] [CrossRef]
- Silva, P.A.; Ul-Rehman, R.; Rato, C.; di Sansebastiano, G.P.; Malhó, R. Asymmetric localization of Arabidopsis SYP124 syntaxin at the pollen tube apical and sub-apical zones is involved in tip growth. BMC Plant Biol. 2010, 10, 179–191. [Google Scholar] [CrossRef]
- Geitmann, A. The rheological properties of the pollen tube cell wall. In Fertilization in Higher Plants; Springer-Verlag: Heidelberg/Berlin, Germany, 1999; pp. 283–297. [Google Scholar]
- Li, Y.Q.; Mareck, A.; Faleri, C.; Moscatelli, A.; Liu, Q.; Cresti, M. Detection and localization of pectin methylesterase isoforms in pollen tubes of Nicotiana tabacum L. Planta 2002, 214, 734–740. [Google Scholar] [CrossRef]
- Chebli, Y.; Kaneda, M.; Zerzour, R.; Geitmann, A. The cell wall of the Arabidopsis thaliana pollen tube-spatial distribution, recycling and network formation of polysaccharides. Plant Physiol. 2012. [Google Scholar] [CrossRef]
- Fayant, P.; Girlanda, O.; Chebli, Y.; Aubin, C. E.; Villemure, I.; Geitmann, A. Finite element model of polar growth in pollen tubes. Plant Cell 2010, 22, 2579–2593. [Google Scholar] [CrossRef]
- Willats, W.G.; Orfila, C.; Limberg, G. Buchholt, H.C.; van Alebeek, G.J.; Voragen, A.G.; Marcus, S.E.; Christensen, T.M.; Mikkelsen, J.D.; Murray, B.S.; Knox, J.P. Modulation of the degree and pattern of methyl-esterification of pectic homogalacturonan in plant cell walls. Implications for pectin methyl esterase action, matrix properties, and cell adhesion. J. Biol. Chem. 2001, 276, 19404–19413. [Google Scholar] [CrossRef]
- Röckel, N.; Wolf, S.; Kost, B.; Rausch, T.; Greiner, S. Elaborate spatial patterning of cell-wall PME and PMEI at the pollen tube tip involves PMEI endocytosis, and reflects the distribution of esterified and de-esterified pectins. Plant J. 2008, 53, 133–143. [Google Scholar] [CrossRef]
- Heslop-Harrison, J. Pollen germination and pollen-tube growth. Int. Rev. Cytol. 1987, 107, 1–78. [Google Scholar] [CrossRef]
- Cai, G.; Faleri, C.; del Casino, C.; Emons, A.M.; Cresti, M. Distribution of callose synthase, cellulose synthase, and sucrose synthase in tobacco pollen tube is controlled in dissimilar ways by actin filaments and microtubules. Plant Physiol. 2011, 155, 1169–1190. [Google Scholar] [CrossRef]
- Brownfield, L.; Wilson, S.; Newbigin, E.; Bacic, A.; Read, S. Molecular control of the glucan synthaselike protein NaGSL1 and callose synthesis during growth of Nicotiana alata pollen tubes. Biochem. J. 2008, 414, 43–52. [Google Scholar] [CrossRef]
- Ito, E.; Fujimoto, M.; Ebine, K.; Uemura, T.; Ueda, T.; Nakano, A. Dynamic behavior of clathrin in Arabidopsis thaliana unveiled by live imaging. Plant J. 2012, 69, 204–216. [Google Scholar] [CrossRef]
- Baluska, F.; Samaj, J.; Hlavacka, A.; Kendrik-Jones, J.; Volkmann, D. Actin-dependent fluid phase endocytosis in inner cortex cells of maize root apices. J. Exp. Bot. 2004, 55, 463–473. [Google Scholar] [CrossRef]
- Bandmann, V.; Homann, U. Clathrin-independent endocytosis contributes to uptake of glucose into BY-2 protoplasts. Plant J. 2012, 70, 578–584. [Google Scholar] [CrossRef]
- Simons, K.; Gerl, M.-J. Revitalizing membrane rafts: New tools and insights. Nat. Rev. Mol. Cell Biol. 2010, 11, 688–699. [Google Scholar] [CrossRef]
- Ewers, H.; Helenius, A. Lipid mediated endocytosis. Cold Spring Harb. Perspect. Biol. 2011. [Google Scholar] [CrossRef]
- Cacas, J.L.; Furt, F.; Le Guédard, M.; Schmitter, J.M.; Buré, C.; Gerbeau-Pissot, P.; Moreau, P.; Bessoule, J.J.; Simon-Plas, F.; Mongrand, S. Lipids of plant membrane rafts. Prog. Lipid Res. 2012, 51, 272–299. [Google Scholar] [CrossRef]
- Morel, J.S.; Claverol, S.; Mongrand, F.; Furt, J.; Fromentin, J.J.; Bessoule, J.P.; Blein, F.; Simon-Plas, F. Proteomics of plant detergent-resistant membranes. Mol. Cell Proteomics 2006, 5, 1396–1411. [Google Scholar] [CrossRef]
- Lefebvre, B.F.; Furt, M.A.; Hartmann, L.V.; Michaelson, J.P.; Carde, F.; Sargueil-Boiron, M.; Rossignol, J.A.; Napier, J.; Cullimore, J.J.; Bessoule, J.J.; et al. Characterization of lipid rafts from Medicago truncatula root plasma membranes: A proteomic study reveals the presence of a raft-associated redox system. Plant Physiol. 2007, 144, 402–418. [Google Scholar] [CrossRef]
- Men, S.; Boutté, Y.; Ikeda, Y.; Li, X.; Palme, K.; Stierhof, Y.D.; Hartmann, M.A.; Moritz, T.; Grebe, M. Sterol-dependent endocytosis mediates post-cytokinetic acquisition of PIN2 auxin efflux carrier polarity. Nat. Cell Biol. 2008, 10, 237–244. [Google Scholar] [CrossRef]
- Li, R.; Liu, P.; Wan, Y.; Chen, T.; Wang, Q.; Mettbach, U.; Baluska, F.; Samaj, J.; Fang, X.; Lucas, W.J.; et al. A membrane microdomain-associated protein, Arabidopsis Flot1, is involved in a clathrin-independent endocytic pathway and is required for seedling development. Plant Cell 2012, 24, 2105–2122. [Google Scholar] [CrossRef]
- Frick, M.; Bright, N.A.; Riento, K.; Bray, A.; Merrified, C.; Nichols, B.J. Coassembly of flotillins induces formation of membrane microdomains, membrane curvature, and vesicle budding. Curr. Biol. 2007, 17, 1151–1156. [Google Scholar] [CrossRef]
- Chae, K.; Zhang, K.; Zhang, L.; Morikis, D.; Kim, S.T.; Mollet, J.C.; de la Rosa, N.; Tan, K.; Lord, E.M. Two SCA (stigma/style cysteine-rich adhesin) isoforms show structural differences that correlate with their levels of in vitro pollen tube adhesion activity. J. Biol. Chem. 2007, 282, 33845–33858. [Google Scholar] [CrossRef]
- Kim, S.T.; Zhang, K.; Dong, J.; Lord, E.M. Exogenous free ubiquitin enhances lily pollen tube adhesion to an in vitro stylar matrix and may facilitate endocytosis of SCA. Plant Physiol. 2006, 142, 1397–1411. [Google Scholar] [CrossRef]
- König, S.; Ischebeck, T.; Lerche, J.; Stenzel, I.; Heilmann, I. Salt-stress-induced association of phosphatidylinositol 4,5-bisphosphate with clathrin-coated vesicles in plants. Biochem. J. 2008, 415, 387–399. [Google Scholar] [CrossRef]
- Zhao, Y.; Yan, A.; Feijó, J.A.; Furutani, M.; Takenawa, T.; Hwang, I.; Fu, Y.; Yang, Z. Phosphoinositides regulate clathrin-dependent endocytosis at the tip of pollen tubes in Arabidopsis and tobacco. Plant Cell 2010, 22, 4031–4044. [Google Scholar] [CrossRef]
- Guo, F.; McCubbin, A.G. The pollen-specific R-SNARE/longin PiVAMP726 mediates fusion of endo- and exocytic compartments in pollen tube tip growth. J. Exp. Bot. 2012, 63, 3083–3095. [Google Scholar]
- Saheki, Y.; de Camilli, P. Synaptic vesicle endocytosis. Cold Spring Harb. Perspect. Biol. 2012. [Google Scholar] [CrossRef]
- Tiezzi, A.; Moscatelli, A.; Cai, G.; Bartalesi, A.; Cresti, M. An immunoreactive homolog of mammalian kinesin in Nicotiana tabacum pollen tubes. Cell Motil. Cytoskel. 1992, 21, 132–137. [Google Scholar] [CrossRef]
- Terasaka, O.; Niitsu, T. Kinesin localized in the pollen tube tips of Pinus densiflora. Jpn. J. Palynol. 1994, 40, 1–6. [Google Scholar]
- Cai, G.; Bartalesi, A.; del Casino, C.; Moscatelli, A.; Tiezzi, A.; Cresti, M. The kinesin-immunoreactive homologue from Nicotiana tabacum pollen tubes: Biochemical properties and subcellular localization. Planta 1993, 191, 496–506. [Google Scholar]
- Romagnoli, S.; Cai, G.; Cresti, M. In vitro assays demonstrate that pollen tube organelles use kinesin-related motor proteins to move along microtubules. Plant Cell 2003, 15, 251–269. [Google Scholar] [CrossRef]
- Cai, G.; Cresti, M. Are kinesins required for organelle trafficking in plant cells? Front. Plant Sci. 2012, 3, 170–179. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Onelli, E.; Moscatelli, A. Endocytic Pathways and Recycling in Growing Pollen Tubes. Plants 2013, 2, 211-229. https://doi.org/10.3390/plants2020211
Onelli E, Moscatelli A. Endocytic Pathways and Recycling in Growing Pollen Tubes. Plants. 2013; 2(2):211-229. https://doi.org/10.3390/plants2020211
Chicago/Turabian StyleOnelli, Elisabetta, and Alessandra Moscatelli. 2013. "Endocytic Pathways and Recycling in Growing Pollen Tubes" Plants 2, no. 2: 211-229. https://doi.org/10.3390/plants2020211



