Role of the HPRG Component of Striated Muscle AMP Deaminase in the Stability and Cellular Behaviour of the Enzyme
Abstract
:1. Introduction
2. Distribution and Localization of AMPD Isoforms
3. Human Skeletal Muscle AMPD Deficiency. Correlation between the Level of Enzyme Activity and the Muscle Content of HPRG
4. A Highly Differentiated N-Terminal Region of AMPD Is Produced by Alternative Splicing Events
5. The Inherent N-Terminal Proteolysis of Striated Muscle AMPD Isoforms
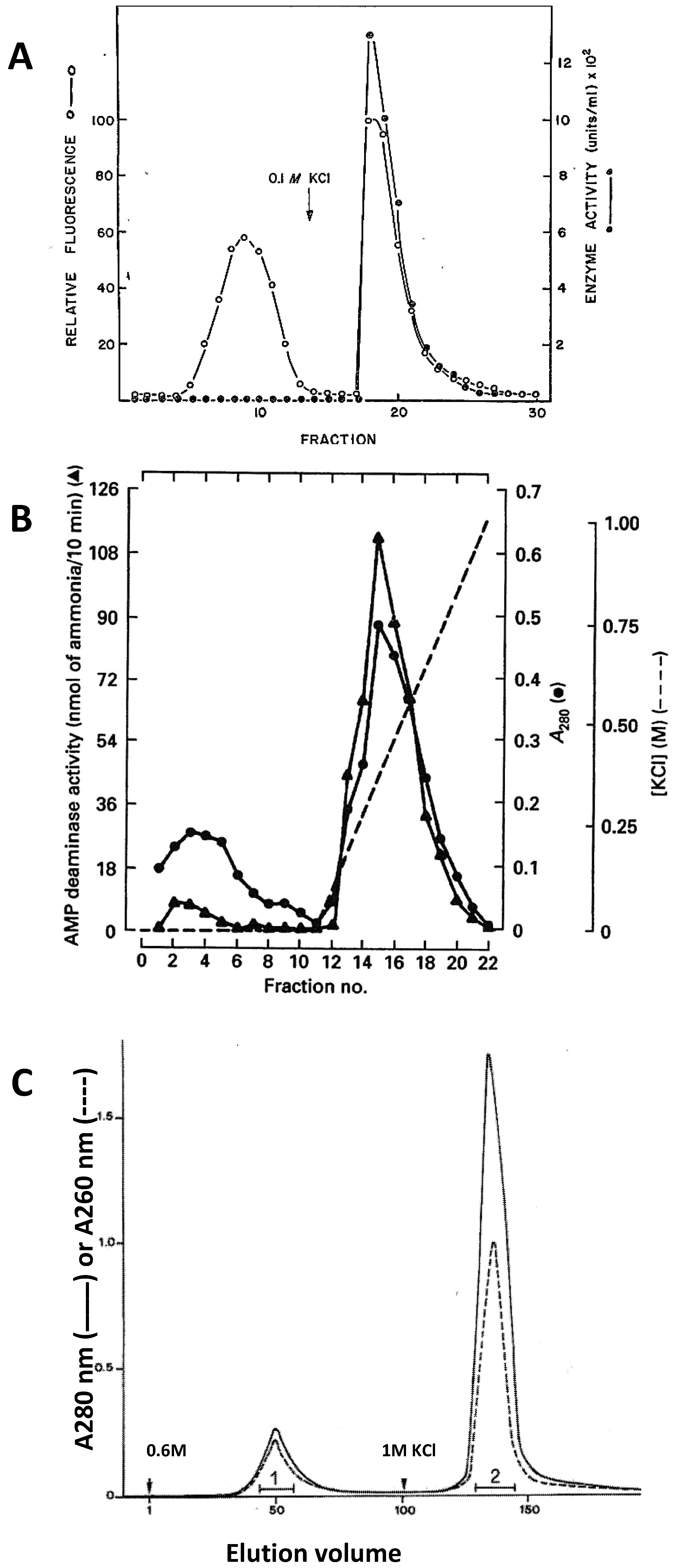
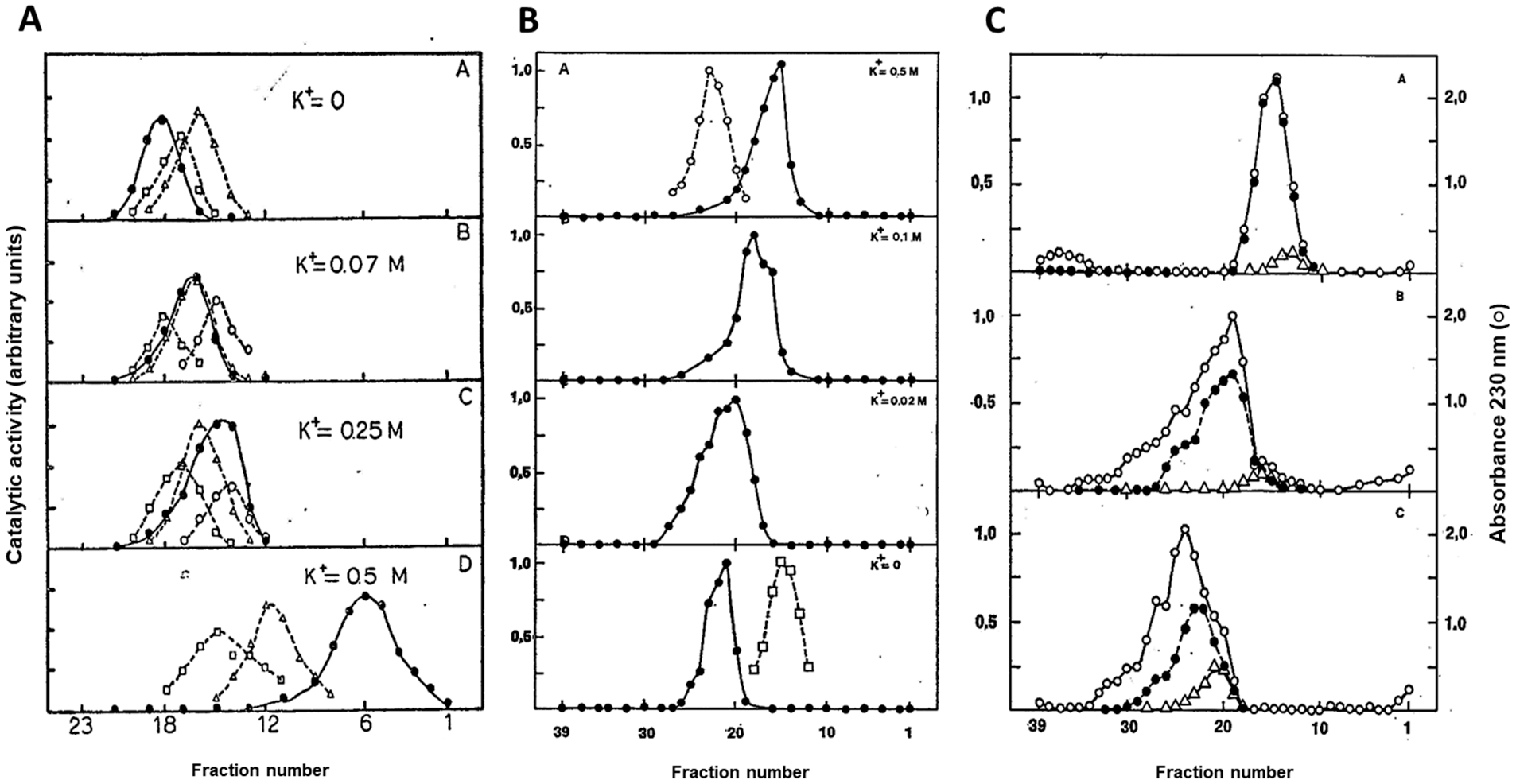
6. Importance of Inorganic Phosphate for the Stability of the AMPD-HPRG Complex
7. Intracellular Distribution Data of Striated Muscle AMPD. Evidence of the Interaction of the Enzyme with the Thin Filament
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Setlow, B.; Lowenstein, J.M. Adenylate deaminase II. Purification and some regulatory properties of the enzyme from calf brain. J. Biol. Chem. 1967, 242, 607–615. [Google Scholar] [PubMed]
- Chapman, A.G.; Atkinson, D.E. Stabilization of adenylate energy charge by the adenylate deaminase reaction. J. Biol. Chem. 1973, 248, 8309–8312. [Google Scholar] [PubMed]
- Lowenstein, J.M.; Goodman, M.N. The purine nucleotide cycle in skeletal muscle. Fed. Proc. 1978, 37, 2308–2312. [Google Scholar] [PubMed]
- Purzycka, J. AMP and adenosine aminohydrolases in rat tissues. Acta Biochim. Pol. 1962, 9, 83–93. [Google Scholar] [PubMed]
- Parnas, J.K. Ueber die Ammoniakbildung im muskel und ihren Zusammenhang mit funktion und Zustandsaenderung. Der Zusammenhang der Ammoniakbildung mit der umwaldung des Adeninnucleotids zu Inosinsaeure. Biochem. Z. 1929, 206, 16–30. (In German) [Google Scholar]
- Hancock, C.R.; Brault, J.J.; Terjung, R.L. Protecting the cellular energy state during contractions: Role of AMP deaminase. J. Physiol. Pharmacol. 2006, 57, 17–29. [Google Scholar] [PubMed]
- Ronca, F.; Raggi, A. Role of troponin T and AMP deaminase in the modulation of skeletal muscle contraction Rend. Rend. Lincei 2017, 28, 143–158. [Google Scholar] [CrossRef]
- Bendall, J.R.; Davey, C.L. Ammonia liberation during rigor mortis and its relation to changes in the adenine and inosine nucleotides of rabbit muscle. Biochim. Biophys. Acta 1957, 26, 93–103. [Google Scholar] [CrossRef]
- Ranieri-Raggi, M.; Moir, A.J.; Raggi, A. Interaction with troponin T from white skeletal muscle restores in white skeletal muscle AMP deaminase those allosteric properties removed by limited proteolysis. Biochim. Biophys. Acta 1985, 827, 93–100. [Google Scholar] [CrossRef]
- Mangani, S.; Benvenuti, M.; Moir, A.J.G.; Ranieri-Raggi, M.; Martini, D.; Sabbatini, A.R.M.; Raggi, A. Characterization of the metallocenter of rabbit skeletal muscle AMP deaminase. Evidence for a dinuclear zinc site. Biochim. Biophys. Acta 2007, 1774, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Shiraki, H.; Miyamoto, S.; Matsuda, Y.; Momose, E.; Nakagawa, H. Possible correlation between binding of muscle type AMP deaminase to myofibrils and ammoniagenesis in rat skeletal muscle on electrical stimulation. Biochem. Biophys. Res. Commun. 1981, 100, 1099–1103. [Google Scholar] [CrossRef]
- Rundell, K.W.; Tullson, P.C.; Terjung, R.L. AMP deaminase binding in rat skeletal muscle after high-intensity running. J. Appl. Physiol. 1993, 74, 2004–2006. [Google Scholar] [CrossRef] [PubMed]
- Ranieri-Raggi, M.; Moir, A.J.G.; Raggi, A. The role of histidine proline- rich glycoprotein as zinc chaperone for skeletal muscle AMP deaminase. Biomolecules 2014, 4, 474–497. [Google Scholar] [CrossRef] [PubMed]
- Marquetant, R.; Desai, N.M.; Sabina, R.L.; Holmes, E.W. Evidence for sequential expression of multiple AMP deaminase isoforms during skeletal muscle development. Proc. Natl. Acad. Sci. USA 1987, 184, 2345–2349. [Google Scholar] [CrossRef]
- Ogasawara, N.; Goto, H.; Yamada, Y. AMP deaminase isozymes in rabbit red and white muscles and heart. Comp. Biochem. Physiol. B 1983, 76, 471–473. [Google Scholar] [CrossRef]
- Ogasawara, N.; Goto, H.; Yamada, Y.; Watanabe, T. Distribution of AMP deaminase isozymes in various human blood cells. Int. J. Biochem. 1984, 16, 269–273. [Google Scholar] [CrossRef]
- Ogasawara, N.; Goto, H.; Watanabe, T. Multiple forms of AMP deaminase in various rat tissues. FEBS Lett. 1974, 44, 63–66. [Google Scholar] [CrossRef] [Green Version]
- Ogasawara, N.; Goto, H.; Yamada, Y.; Watanabe, T. Distribution of AMP-deaminase isozymes in rat tissues. Eur. J. Biochem. 1978, 87, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, N.; Goto, H.; Yamada, Y.; Watanabe, T.; Asano, T. AMP deaminase isozymes in human tissues. Biochim. Biophys. Acta 1982, 714, 298–306. [Google Scholar] [CrossRef]
- Bausch-Jurken, M.T.; Mahnke-Zizelman, D.K.; Morisaki, T.; Sabina, R.L. Molecular cloning of AMP deaminase isofom L: Sequence and bacterial expression of human AMPD2 cDNA. J. Biol. Chem. 1992, 267, 22407–22413. [Google Scholar] [PubMed]
- Mahnke-Zizelman, D.K.; Eddy, R.; Shows, T.B.; Sabina, R.L. Characterization of the human AMPD3 gene reveals that 59 exon usage is subject to transcriptional control by three tandem promoters and alternative splicing. Biochim. Biophys. Acta 1996, 1306, 75–92. [Google Scholar] [CrossRef]
- Morisaki, T.; Sabina, R.L.; Holmes, E.W. Adenylate deaminase. A multigene family in humans and rats. J. Biol. Chem. 1990, 265, 11482–11486. [Google Scholar] [PubMed]
- Sabina, R.L.; Morisaki, T.; Clarke, P.R.H.; Eddy, R.; Shows, T.B.; Morton, C.C.; Holmes, E.W. Characterization of the human and rat myoadenylate deaminase genes. J. Biol. Chem. 1990, 265, 9423–9433. [Google Scholar] [PubMed]
- Fishbein, W.N.; Sabina, R.L.; Ogasawara, N.; Holmes, E.W. Immunologic evidence for three isoforms of AMP deaminase (AMPD) in mature skeletal muscle. Biochim. Biophys. Acta 1993, 1163, 97–104. [Google Scholar] [CrossRef]
- Wang, X.; Morisaki, H.; Sermsuvitayawong, K.; Mineo, I.; Toyama, K.; Ogasawara, N.; Mukai, T.; Morisaki, T. Cloning and expression of cDNA encoding heart-type isoform of AMP deaminase. Gene 1997, 188, 285–290. [Google Scholar] [CrossRef]
- Lazarides, E. From genes to structural morphogenesis: The genesis and epigenesis of a red blood cell. Cell 1987, 51, 345–356. [Google Scholar] [CrossRef]
- Rao, S.N.; Hara, L.; Askari, A. Alkali cation-activated AMP deaminase of erythrocytes: Some properties of the membrane-bound enzyme. Biochim. Biophys. Acta 1968, 151, 651–654. [Google Scholar] [CrossRef]
- Pipoly, G.M.; Nathans, G.R.; Chang, D.; Deuel, T.F. Regulation of the interaction of purified human erythrocyte AMP deaminase and the human erythrocyte membrane. J. Clin. Investig. 1979, 63, 1066–1076. [Google Scholar] [CrossRef] [PubMed]
- Sabina, R.L.; Marquetant, R.; Desai, N.M.; Kaletha, K.; Holmes, E.W. Cloning and sequence of rat myoadenylate deaminase cDNA: Evidence for tissue-specific and developmental regulation. J. Biol. Chem. 1987, 262, 12397–12400. [Google Scholar] [PubMed]
- Ogasawara, N.; Goto, H.; Watanabe, T. Isozymes of rat brain AMP deaminase: Developmental changes and characterizations of five forms. FEBS Lett. 1975, 58, 245–248. [Google Scholar] [CrossRef] [Green Version]
- Sammons, D.W.; Chilson, O.P. AMP deaminase: Stage-specific isozymes in differentiating chick muscle. Arch. Biochem. Biophys. 1978, 191, 561–570. [Google Scholar] [CrossRef]
- Spychała, J.; Kaletha, K.; Makarewicz, W. Developmental changes of chicken liver AMP deaminase. Biochem. J. 1985, 231, 329–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruckeberg, W.C.; Chilson, O.P. Regulation of avian erythrocyte AMP-deaminase. Comp. Biochem. Physiol. B 1979, 62, 251–258. [Google Scholar] [CrossRef]
- Kaletha, K.; Skladanowski, A. Regulatory properties of 14 day embryo and adult hen heart AMP-deaminase. Int. J. Biochem. 1984, 16, 75–81. [Google Scholar] [CrossRef]
- Mahnke-Zizelman, D.K.; D’cunha, J.; Wojnar, J.M.; Brogley, M.A.; Sabina, R.L. Regulation of rat AMP deaminase 3 (isoform C) by development and skeletal muscle fibre type. Biochem. J. 1997, 326, 521–529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahnke-Zizelman, D.K.; Sabina, R.L. Cloning of human AMP deaminase isoform E cDNAs. Evidence for a third AMPD gene exhibiting alternatively spliced 5′-exons. J. Biol. Chem. 1992, 267, 20866–20877. [Google Scholar] [PubMed]
- Raggi, A.; Ronca-Testoni, S.; Ronca, G. Muscle AMP aminohydrolase II. Distribution of AMP aminohydrolase, myokinase and creatine kinase activities in skeletal muscle. Biochim. Biophys. Acta 1969, 178, 619–622. [Google Scholar] [CrossRef]
- Raggi, A.; Bergamini, C.; Ronca, G. Isozymes of AMP deaminase in red and white skeletal muscles. FEBS Lett. 1975, 58, 19–23. [Google Scholar] [CrossRef] [Green Version]
- Raggi, A.; Ranieri-Raggi, M. Regulatory properties of AMP deaminase isoenzymes from rabbit red muscle. Biochem. J. 1987, 242, 875–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabbatini, A.R.M.; Ranieri-Raggi, M.; Pollina, L.; Viacava, P.; Ashby, J.R.; Moir, A.J.G.; Raggi, A. Presence in human skeletal muscle of an AMP deaminase-associated protein that reacts with an antibody to human plasma histidine-proline-rich glycoprotein. J. Histochem. Cytochem. 1999, 47, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Van Kuppevelt, T.H.; Veerkamp, J.H.; Fishbein, W.N.; Ogasawara, N.; Sabina, R.L. Immunolocalization of AMP-deaminase isozymes in human skeletal muscle and cultured muscle cells: Concentration of isoform M at the neuromuscular junction. J. Histochem. Cytochem. 1994, 42, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Ranieri-Raggi, M.; Montali, U.; Ronca, F.; Sabbatini, A.; Brown, P.E.; Moir, A.J.G.; Raggi, A. Association of purified skeletal-muscle AMP deaminase with a histidine-proline-rich-glycoprotein-like molecule. Biochem. J. 1997, 326, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Ranieri-Raggi, M.; Martini, D.; Sabbatini, A.R.M.; Moir, A.J.G.; Raggi, A. Isolation by zinc-affinity chromatography of the histidine-proline-rich-glycoprotein molecule associated with rabbit skeletal muscle AMP deaminase. Evidence that the formation of a protein-protein complex between the catalytic subunit and the novel component is critical for the stability of the enzyme. Biochim. Biophys. Acta 2003, 1645, 81–88. [Google Scholar] [PubMed]
- Mangani, S.; Meyer-Klaucke, W.; Moir, A.J.G.; Ranieri-Raggi, M.; Martini, D.; Raggi, A. Characterization of the Zn binding site of the HPRG protein associated with rabbit skeletal muscle AMP deaminase. J. Biol. Chem. 2003, 278, 3176–3184. [Google Scholar] [CrossRef] [PubMed]
- Ronca, F.; Raggi, A. Structure-function relationships in mammalian histidine-proline-rich glycoprotein. Biochimie 2015, 118, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Sabbatini, A.R.M.; Mattii, L.; Battolla, B.; Polizzi, E.; Martini, D.; Ranieri-Raggi, M.; Moir, A.J.G.; Raggi, A. Evidence that muscle cells do not express the histidine-rich glycoprotein associated with AMP deaminase but can internalise the plasma protein. Eur. J. Histochem. 2011, 55, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, S. New insights into the functions of histidine-rich glycoprotein. Int. Rev. Cell Mol. Biol. 2013, 304, 467–493. [Google Scholar] [PubMed]
- Tugues, S.; Roche, F.; Noguer, O.; Orlova, A.; Bhoi, S.; Padhan, N.; Akerud, P.; Honjo, S.; Selvaraju, R.K.; Mazzone, M.; et al. Histidine-rich glycoprotein uptake and turnover is mediated by mononuclear phagocytes. PLoS ONE 2014, 9, e107483. [Google Scholar] [CrossRef] [PubMed]
- Husic, H.D.; Suelter, C.H. Circulatory clearance, internalization, and degradation of muscle AMP aminohydrolase. J. Biol. Chem. 1984, 259, 4359–4364. [Google Scholar] [PubMed]
- Husic, H.D.; Suelter, C.H. Binding, internalization, and degradation of AMP aminohydrolase by avian hepatocyte monolayers. J. Biol. Chem. 1984, 259, 4365–4371. [Google Scholar] [PubMed]
- Burch, M.K.; Blackburn, M.N.; Morgan, W.T. Further characterization of the interaction of histidine-rich glycoprotein with heparin: Evidence for the binding of two molecules of histidine-rich glycoprotein by high molecular weight heparin and for the involvement of histidine residues in heparin binding. Biochemistry 1987, 26, 7477–7482. [Google Scholar] [PubMed]
- Mattii, L.; Rossi, L.; Ippolito, C.; Alì, G.; Martini, D.; Raggi, A.; Sabbatini, A.R.M. Immunohistochemical localization of histidine-rich glycoprotein in human skeletal muscle: Preferential distribution of the protein at the sarcomeric I-band. Histochem. Cell Biol. 2017, 148, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Morisaki, T.; Gross, M.; Morisaki, H.; Pongratz, D.; Zӧllner, N.; Holmes, E.W. Molecular basis of AMP deaminase deficiency in skeletal muscle. Proc. Natl. Acad. Sci. USA 1992, 89, 6457–6461. [Google Scholar] [CrossRef] [PubMed]
- Verzijl, H.T.; van Engelen, B.G.; Luyten, J.A.; Steenbergen, G.C.; van den Heuvel, L.P.; ter Laak, H.J.; Padberg, G.W.; Wevers, R.A. Genetic characteristics of myoadenylate deaminase deficiency. Ann. Neurol. 1998, 44, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Tarnopolsky, M.A.; Parise, G.; Gibala, M.J.; Graham, T.E.; Rush, J.W. Myoadenylate deaminase deficiency does not affect muscle anaplerosis during exhaustive exercise in humans. J. Physiol. 2001, 533, 881–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayes, D.J.; Summers, B.A.; Morgan-Hughes, J.A. Myoadenylate deaminase deficiency or not? Observations on two brothers with exercise-induced muscle pain. J. Neurol. Sci. 1982, 53, 125–136. [Google Scholar] [CrossRef]
- Sinkeler, S.P.; Joosten, E.M.; Wevers, R.A.; Oei, T.L.; Jacobs, A.E.; Veerkamp, J.H.; Hamel, B.C. Myoadenylate deaminase deficiency: A clinical, genetic and biochemical study in nine families. Muscle Nerve 1988, 11, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Fishbein, W.N. Myoadenylate deaminase deficiency: Inherited and acquired forms. Biochem. Med. 1985, 33, 158–169. [Google Scholar] [CrossRef]
- Morisaki, H.; Morisaki, T.; Newby, L.K.; Holmes, E.W. Alternative splicing: A mechanism for phenotypic rescue of a common inherited defect. J. Clin. Investig. 1993, 91, 2275–2280. [Google Scholar] [CrossRef] [PubMed]
- Mahnke-Zizelman, D.K.; Tullson, P.C.; Sabina, R.L. Novel aspects of tetramer assembly and N-terminal domain structure and function are revealed by recombinant expression of human AMP deaminase isoforms. J. Biol. Chem. 1998, 273, 35118–35125. [Google Scholar] [CrossRef] [PubMed]
- Fortuin, F.D.; Morisaki, T.; Holmes, E.W. Subunit composition of AMPD varies in response to changes in AMPD1 and AMPD3 gene expression in skeletal muscle. Proc. Assoc. Am. Phys. 1996, 108, 329–333. [Google Scholar] [PubMed]
- Sabbatini, A.R.; Toscano, A.; Aguennouz, M.; Martini, D.; Polizzi, E.; Ranieri-Raggi, M.; Moir, A.J.G.; Migliorato, A.; Musumeci, O.; Vita, G.; et al. Immunohistochemical analysis of human skeletal muscle AMP deaminase deficiency. Evidence of a correlation between the muscle HPRG content and the level of the residual AMP deaminase activity. J. Muscle Res. Cell Motil. 2006, 27, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Goto, H.; Ogasawara, N. Cloning and nucleotide sequence of the cDNA encoding human erythrocyte-specific AMP deaminase. Biochim. Biophys. Acta 1992, 1171, 125–128. [Google Scholar] [CrossRef]
- Mineo, I.; Clarke, P.R.H.; Sabina, R.L.; Holmes, E.W. A novel pathway for alternative splicing: Identification of an RNA intermediate that generates an alternative splice donor in the primary transcript of AMPD1. Mol. Cell. Biol. 1990, 10, 5271–5278. [Google Scholar] [CrossRef] [PubMed]
- Van den Bergh, F.; Sabina, R.L. Characterization of human AMP deaminase 2 (AMPD2) gene expression reveals alternative transcripts encoding variable N-terminal extensions of isoform L. Biochem. J. 1995, 312, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Chang, Z.Y.; Nygaard, P.; Chinault, A.C.; Kellems, R.E. Deduced amino acid sequence of Escherichia coli adenosine deaminase reveals evolutionarily conserved amino acid residues: Implications for catalytic function. Biochemistry 1991, 30, 2273–2280. [Google Scholar] [CrossRef] [PubMed]
- Mahnke-Zizelman, D.K.; Van den Bergh, F.; Bausch-Jurken, M.T.; Eddy, R.; Sait, S.; Shows, T.B.; Sabina, R.L. Cloning, sequence, and characterization of the human AMPD2 gene: Evidence for transcriptional regulation by two closely spaced promoters. Biochim. Biophys. Acta 1996, 1308, 122–132. [Google Scholar] [CrossRef]
- Gross, M.; Morisaki, H.; Morisaki, T.; Holmes, E.W. Identification of functional domains in AMPD1 by mutational analysis. Biochem. Biophys. Res. Commun. 1994, 205, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- Mahnke-Zizelman, D.K.; Sabina, R.L. Localization of N-terminal sequences in human AMP deaminase isoforms that influence contractile protein binding. Biochem. Biophys. Res. Commun. 2001, 285, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Rundell, K.W.; Tullson, P.C.; Terjung, R.L. AMP deaminase binding in contracting rat skeletal muscle. Am. J. Physiol. 1992, 263, C287–C293. [Google Scholar] [CrossRef] [PubMed]
- Rundell, K.W.; Tullson, P.C.; Terjung, R.L. Altered kinetics of AMP deaminase by myosin binding. Am. J. Physiol. 1992, 263, C294–C299. [Google Scholar] [CrossRef] [PubMed]
- Hohl, A.M.; Hohl, C.M. Isolation and regulation of piglet cardiac AMP deaminase. Mol. Cell. Biochem. 1999, 201, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Woźniak, M.; Kossowska, E.; Purzycka-Preis, J.; Zydowo, M.M. The influence of phosphatidate bilayers on pig heart AMP deaminase. Crucial role of pH-dependent lipid-phase transition. Biochem. J. 1988, 255, 977–981. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanfani, F.; Kossowska, E.; Purzycka-Preis, J.; Zydowo, M.M.; Wozniak, M.; Tartaglini, E.; Bertoli, E. The interaction of phospholipid bilayers with pig heart AMP deaminase:Fourier-transform infrared spectroscopic and kinetic studies. Biochem. J. 1993, 291, 921–926. [Google Scholar] [CrossRef] [PubMed]
- Tanfani, F.; Kulawiak, D.; Kossowska, E.; Preis, J.P.; Zydowo, M.M.; Sarkissova, E.; Bertoli, E.; Wozniak, M. Structural-functional relationships in pig heart AMP-deaminase in the presence of ATP, orthophosphate, and phosphatidate bilayers. Mol. Genet. Metab. 1998, 65, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Sabina, R.L.; Mahnke-Zizelman, D.K. Towards an understanding of the functional significance of N-terminal domain divergence in human AMP deaminase isoforms. Pharmacol. Ther. 2000, 87, 279–283. [Google Scholar] [CrossRef]
- Martini, D.; Ranieri-Raggi, M.; Sabbatini, A.R.M.; Moir, A.J.G.; Polizzi, E.; Mangani, S.; Raggi, A. Characterization of the metallocenter of rabbit skeletal muscle AMP deaminase. A new model for substrate interactions at a dinuclear cocatalytic Zn site. Biochim. Biophys. Acta 2007, 1774, 1508–1518. [Google Scholar] [CrossRef] [PubMed]
- Raggi, A.; Ranieri, M.; Taponeco, G.; Ronca-Testoni, S.; Ronca, G.; Rossi, C.A. Interaction of the rat muscle AMP aminohydrolase with chelating agents and metal ions. FEBS Lett. 1970, 10, 101–104. [Google Scholar] [CrossRef]
- Zielke, C.L.; Suelter, C.H. Rabbit muscle adenosine 5′-monophosphate aminohydrolase. Characterization as a zinc metalloenzyme. J. Biol. Chem. 1971, 246, 2179–2186. [Google Scholar] [PubMed]
- Merkler, D.J.; Schramm, V.L. Catalytic mechanism of yeast adenosine 5′-monophosphate deaminase. Zinc content, substrate specificity, pH studies and solvent isotope effects. Biochemistry 1993, 32, 5792–5799. [Google Scholar] [CrossRef] [PubMed]
- Bazin, R.J.; Mc Donald, G.A.; Phillips, C. Adenosine Monophosphate Deaminase Crystal Structure. UK Patent GB2373504, 23 September 2003. [Google Scholar]
- Han, B.W.; Bingman, C.A.; Mahnke, D.K.; Bannen, R.M.; Bednarek, S.Y.; Sabina, R.L.; Phillips, G.N., Jr. Membrane association, Mechanism of action, and structure of Arabidopsis embryonic factor 1 (FAC1). J. Biol. Chem. 2006, 281, 14939–14947. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.K.; Rudolph, F.B.; Quiocho, F.A. Atomic structure of adenosine deaminase complexed with a transition-state analog: Understanding catalysis and immunodeficiency mutations. Science 1991, 252, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Ranieri-Raggi, M.; Raggi, A. Effect of pH and KCl on aggregation state and sulphydryl groups reactivity of rat skeletal muscle AMP deaminase. Ital. J. Biochem. 1984, 33, 155–176. [Google Scholar] [PubMed]
- Smiley, K.L., Jr.; Berry, A.J.; Suelter, C.H. An improved purification, crystallization, and some properties of rabbit muscle 5′-adenylic acid deaminase. J. Biol. Chem. 1967, 242, 2502–2506. [Google Scholar] [PubMed]
- Ampd cDNAs. Available online: https://www.ncbi.nlm.nih.gov/ (accessed on 1 June 2018).
- Boosman, A.; Chilson, O.P. Subunit structure of AMP-deaminase from chicken and rabbit skeletal muscle. J. Biol. Chem. 1976, 251, 1847–1852. [Google Scholar] [PubMed]
- Stankiewicz, A.; Spychała, J.; Składanowski, A.; Zydowo, M. Comparative studies on muscle AMP-deaminase—I. Purification, molecular weight, subunit structure and metal content of the enzymes from rat, rabbit, hen, frog and pikeperch. Comp. Biochem. Physiol. B 1979, 62, 363–369. [Google Scholar] [CrossRef]
- Ogasawara, N.; Goto, H.; Yamada, Y.; Yoshino, M. Subunit structures of AMP deaminase isozymes in rat. Biochem. Biophys. Res. Commun. 1977, 79, 671–676. [Google Scholar] [CrossRef]
- Coffee, C.J.; Kofke, W.A. Rat muscle 5′-adenylic acid aminohydrolase. I. Purification and subunit structure. J. Biol. Chem. 1975, 250, 6653–6658. [Google Scholar] [PubMed]
- Coffee, C.J. AMP deaminase from rat skeletal muscle. Methods Enzymol. 1978, 51, 490–497. [Google Scholar] [PubMed]
- Thakkar, J.K.; Janero, D.R.; Yarwood, C.; Sharif, H.; Hreniuk, D. Isolation and characterization of AMP deaminase from mammalian (rabbit) myocardium. Biochem. J. 1993, 290, 335–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janero, D.R.; Yarwood, C. Oxidative modulation and inactivation of rabbit cardiac adenylate deaminase. Biochem. J. 1995, 306, 421–427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kruckeberg, W.C.; Lemley, S.; Chilson, O.P. Purification, subunit structure, and amino acid composition of avian erythrocyte adenosine monophosphate deaminase. Biochemistry 1978, 17, 4376–4383. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, M.; Kawamura, Y.; Ogasawara, N. Regulation of AMP deaminase from chicken erythrocytes. A kinetic study of the allosteric interactions. J. Biochem. 1976, 80, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Ranieri-Raggi, M.; Raggi, A. Effects of storage on activity and subunit structure of rabbit skeletal-muscle AMP deaminase. Biochem. J. 1980, 189, 367–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chilson, O.P.; Kelly-Chilson, A.E.; Siegel, N.R. AMP-deaminases from chicken and rabbit muscle: Partial primary sequences of homologous 17-kDa CNBr fragments: Autorecognition by rabbit anti-[chicken AMPD]. Comp. Biochem. Physiol. B 1997, 116, 371–377. [Google Scholar] [CrossRef]
- Haas, A.L.; Sabina, R.L. Expression, purification, and inhibition of in-vitro proteolysis of human AMPD2 (isoform L) recombinant enzymes. Protein Expr. Purif. 2003, 27, 293–303. [Google Scholar] [CrossRef]
- Tompa, P.; Buzder-Lantos, P.; Tantos, A.; Farkas, A.; Szilagyi, A.; Banoczi, Z.; Hudecz, F.; Friedrich, P. On the sequential determinants of calpain cleavage. J. Biol. Chem. 2004, 279, 20775–20785. [Google Scholar] [CrossRef] [PubMed]
- Martini, D.; Montali, U.; Ranieri-Raggi, M.; Sabbatini, A.R.M.; Thorpe, S.J.; Moir, A.J.G.; Raggi, A. A calpain-like proteolytic activity produces the limited cleavage at the N-terminal regulatory domain of rabbit skeletal muscle AMP deaminase: Evidence of a protective molecular mechanism. Biochim. Biophys. Acta 2004, 1702, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Ranieri-Raggi, M.; Raggi, A. Regulation of skeletal muscle AMP deaminase. Effects of limited proteolysis on the activity of the rabbit enzyme. FEBS Lett. 1979, 102, 59–63. [Google Scholar] [CrossRef]
- Currie, R.D.; Webster, H.L. Preparation of 5′-adenylic acid deaminase based on phosphate-induced dissociation of rat actomyosin-deaminase complexes. Biochim. Biophys. Acta 1962, 64, 30–40. [Google Scholar] [CrossRef]
- Ronca, F.; Ranieri-Raggi, M.; Brown, P.E.; Moir, A.J.G.; Raggi, A. Evidence of a species-differentiated regulatory domain within the N-terminal region of skeletal muscle AMP deaminase. Biochim. Biophys. Acta 1994, 1209, 123–129. [Google Scholar] [CrossRef]
- Ranieri-Raggi, M.; Raggi, A. Regulation of skeletal muscle AMP deaminase. Evidence for a highly pH-dependent inhibition by ATP of the homogeneous derivative of the rabbit enzyme yielded by limited proteolysis. Biochem. J. 1990, 272, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, J.K.; Janero, D.R.; Sharif, H.M.; Hreniuk, D.; Yarwood, C. Cardiac adenylate deaminase: Molecular, kinetic and regulatory properties under phosphate-free conditions. Biochem. J. 1994, 300, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Ashman, L.K.; Atwell, J.L. AMP deaminase from rabbit skeletal muscle: The effect of monovalent cations on catalytic activity and molecular weight. Biochim. Biophys. Acta 1972, 258, 618–625. [Google Scholar] [CrossRef]
- Ashby, B.; Frieden, C. Interaction of AMP-aminohydrolase with myosin and its subfragments. J. Biol. Chem. 1977, 252, 1869–1872. [Google Scholar] [PubMed]
- Koretz, J.F.; Frieden, C. Adenylate deaminase binding to synthetic thick filaments of myosin. Proc. Natl. Acad. Sci. USA 1980, 77, 7186–7188. [Google Scholar] [CrossRef] [PubMed]
- Ashby, B.; Frieden, C.; Bischoff, R. Immunofluorescent and histochemical localization of AMP deaminase in skeletal muscle. J. Cell. Biol. 1979, 81, 361–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, L.Y.; Ackermann, M.A.; Kontrogianni-Konstantopoulos, A. The sarcomeric M-region: A molecular command center for diverse cellular processes. Biomed. Res. Int. 2015, 2015, 714197. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.; Trinick, J. Binding and location of AMP deaminase in rabbit psoas muscle myofibrils. J. Mol. Biol. 1984, 177, 137–152. [Google Scholar] [CrossRef]
- Koretz, J.F.; Irving, T.C.; Wang, K. Filamentous aggregates of native titin and binding of C-protein and AMP-deaminase. Arch. Biochem. Biophys. 1993, 304, 305–309. [Google Scholar] [CrossRef] [PubMed]
- Raggi, A.; Laboratory of Biochemistry, Department of Pathology, University of Pisa, Pisa, Italy. Actin filament co-purification with HPRG-AMPD1 complex. Personal communication, 2018. [Google Scholar]
- Guan, X.; Juarez, J.C.; Qi, X.; Shipulina, N.V.; Shaw, D.E.; Morgan, W.T.; McCrae, K.R.; Mazar, A.P.; Donate, F. Histidine-proline-rich glycoprotein (HPRG) binds and transduces anti-angiogenic signals through cell surface tropomyosin on endothelial cells. Thromb. Haemost. 2004, 92, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.P. Evolution, regulation, and function of N-terminal variable region of troponin T: Modulation of muscle contractility and beyond. Int. Rev. Cell. Mol. Biol. 2016, 321, 1–28. [Google Scholar] [PubMed]
- Zhang, Z.; Biesiadecki, B.J.; Jin, J.P. Selective deletion of the NH2-terminal variable region of cardiac troponin T in ischemia reperfusion by myofibril-associated l-calpain cleavage. Biochemistry 2006, 45, 11681–11694. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.Z.; Biesiadecki, B.J.; Yu, Z.B.; Hossain, M.M.; Jin, J.P. Restricted N-terminal truncation of cardiac troponin T: A novel mechanism for functional adaptation to energetic crisis. J. Physiol. 2008, 586, 3537–3550. [Google Scholar] [CrossRef] [PubMed]
- Fredericks, S.; Degens, H.; McKoy, G.; Bainbridge, K.; Collinson, P.O.; Coulton, G.; Elmahdi, H.; Holt, D.W. Effect of denervation on the content of cardiac troponin-T and cardiac troponin-I in rat skeletal muscle. Clin. Biochem. 2007, 40, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Lecker, S.H.; Jagoe, R.T.; Gilbert, A.; Gomes, M.; Baracos, V.; Bailey, J.; Price, S.R.; Mitch, W.E.; Goldberg, A.L. Multiple types of skeletal muscle atrophy involve a common program of changes in gene expression. FASEB J. 2004, 18, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Pereyra, A.S.; Wang, Z.M.; Birbrair, A.; Reisz, J.A.; Files, D.C.; Purcell, L.; Feng, X.; Messi, M.L.; Feng, H.; et al. Calpain inhibition rescues troponin T3 fragmentation, increases Cav1.1, and enhances skeletal muscle force in aging sedentary mice. Aging Cell 2016, 15, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida-Straeten, N.; Ensslen, S.; Schäfer, C.; Wöltje, M.; Denecke, B.; Moser, M.; Gräber, S.; Wakabayashi, S.; Koide, T.; Jahnen-Dechent, W. Enhanced blood coagulation and fibrinolysis in mice lacking histidine-rich glycoprotein (HRG). J. Thromb. Haemost. 2005, 3, 865–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Species | RABBIT | RAT | CHICKEN | |||
|---|---|---|---|---|---|---|
| Isoform | Reported (kDa) | Predicted (kDa) | Reported (kDa) | Predicted (kDa) | Reported (kDa) | Predicted (kDa) |
| AMPD1 (M) | 80–85 a 68 b–70 c | 86 | 60 d 68 e–70 f | 86.3–85.9 | 69 b–70 c | 85.1–85.6 |
| AMPD3 (E) | 80–81 g | 93.9 | n.d. | 88.4–92 | 69 h–70 i | 88.8–93.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ronca, F.; Raggi, A. Role of the HPRG Component of Striated Muscle AMP Deaminase in the Stability and Cellular Behaviour of the Enzyme. Biomolecules 2018, 8, 79. https://doi.org/10.3390/biom8030079
Ronca F, Raggi A. Role of the HPRG Component of Striated Muscle AMP Deaminase in the Stability and Cellular Behaviour of the Enzyme. Biomolecules. 2018; 8(3):79. https://doi.org/10.3390/biom8030079
Chicago/Turabian StyleRonca, Francesca, and Antonio Raggi. 2018. "Role of the HPRG Component of Striated Muscle AMP Deaminase in the Stability and Cellular Behaviour of the Enzyme" Biomolecules 8, no. 3: 79. https://doi.org/10.3390/biom8030079




