Role of Pyroptosis in Intervertebral Disc Degeneration and Its Therapeutic Implications
Abstract
:1. Introduction
2. Pyroptosis
2.1. Canonical Inflammasome-Induced Pyroptosis
2.2. Noncanonical Inflammasome-Induced Pyroptosis
2.3. Other Relevant Approaches
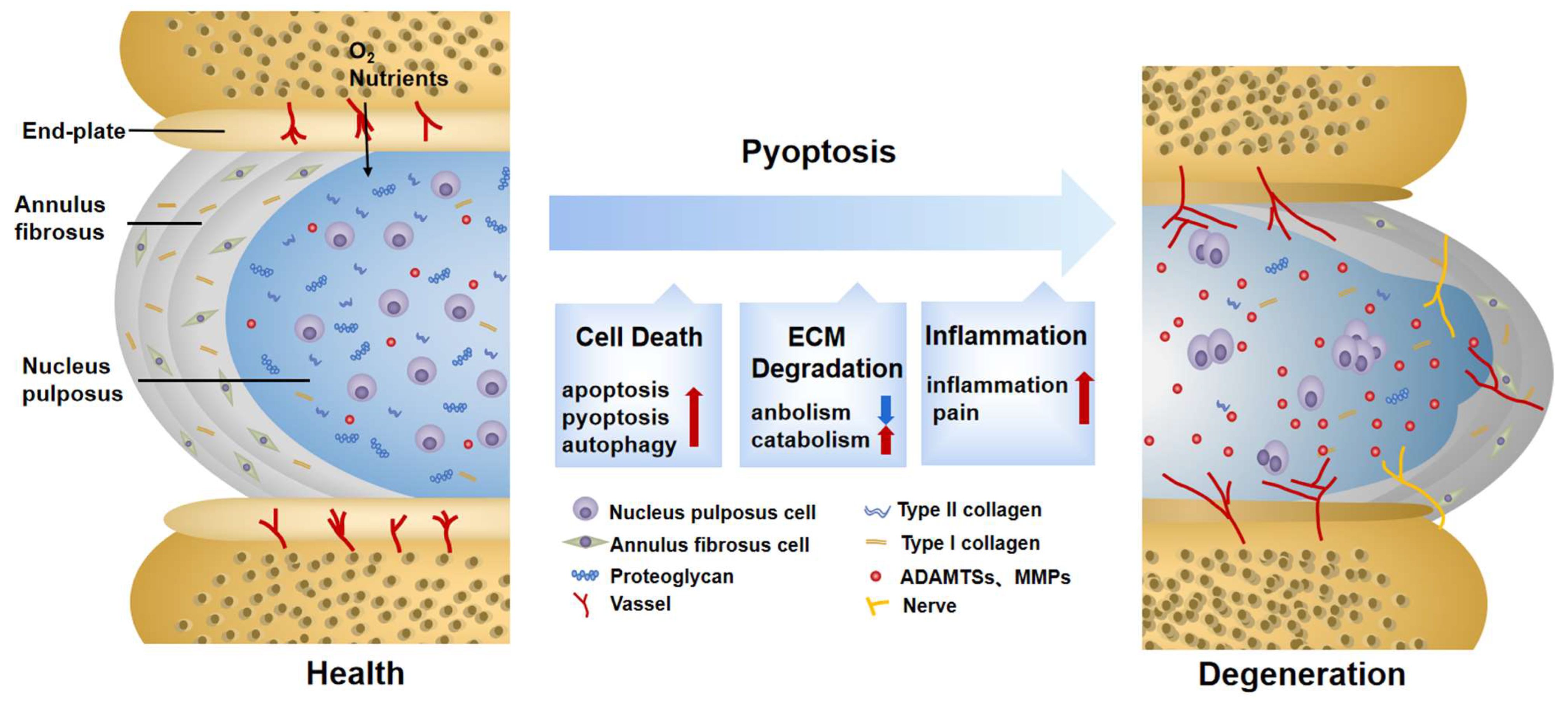
3. The Pathogenesis of IDD
4. Roles of Pyroptosis in IDD
4.1. Pyroptosis Triggers Cell Death in IDD
4.2. Pyroptosis Provokes ECM Disorder in IDD
4.3. Pyroptosis Induces Secondary Inflammation in IDD
5. Therapeutic Strategies Targeting Pyroptosis and NLRP3 Inflammasome in IDD
5.1. Exogenous Drugs
| Type | Compound | Dose | Model Type | Mechanism | References | ||||
|---|---|---|---|---|---|---|---|---|---|
| Pathway | Cell Death | ECM | Inflammation | Others | |||||
| Plant-derived natural compounds | Ganoderic Acid A | 08 μM | Rat NP tissue (in vivo/in vitro) | Inactivated TLR4/NLRP3 | Suppressed apoptosis | Suppressed MMP3, MMP13, ADAMTS4, ADAMTS5. Upregulated Col II, aggrecan | Alleviated IL-6, IL-1β, TNF-α | Reduced oxidative stress by restoring GSH, SOD, GPX | [96] |
| Plant-derived natural compounds | Magnoflorine | 100 μg/mL | Human NP cells (in vitro) | Inactivated HMGB1/ MyD88/NF-κB/NLRP3 | Suppressed apoptosis | Suppressed MMP3, MMP13, ADAMTS4, ADAMTS5. Upregulated Col II, aggrecan | Alleviated HMGB1, IL-1β, IL-6, TNF-α, IL-18 | Alleviated “M1” polarized macrophage | [98] |
| Plant-derived natural compounds | Paeoniflorin | 20 mg/kg | Rat NP tissue (in vivo/in vitro) | Inactivated IL-1β/NLRP3 | Suppressed pyroptosis | Suppressed MMP3, MMP13. Upregulated Col II, aggrecan | Alleviated IL-1β, IL-18 | Reduced calcium concentration | [103] |
| Plant-derived natural compounds | Gamma oryzanol | 40 μM | Rat NP tissue (in vivo/in vitro) | Inactivated IL-1β/NF-κB/NLRP3 | Suppressed pyroptosis | Suppressed MMP13, ADAMTS5. Upregulated Col II, aggrecan | Alleviated IL-1β, IL-18 | —— | [104] |
| Plant-derived natural compounds | Morin | 50 mg/kg | Rat NP tissue (in vivo/in vitro) | Inactivated TXNIP/ NLRP3 | Suppressed pyroptosis | —— | Alleviated IL-1β, IL-18, TNF-α | —— | [106] |
| Plant-derived natural compounds | Honokiol | 40 μM | Rat NP tissue (in vivo/in vitro) | Inactivated NF-κB, JNK, TXNIP/NLRP3 | Suppressed apoptosis | Suppressed MMP3, MMP13, ADAMTS4, ADAMTS5. Upregulated Col II, SOX9 | Alleviated IL-6, COX-2, iNOS | Reduced oxidative stress by restoring MDA, SOD, GPX | [107] |
| Chemical compound | Bay11-7082 | 5 mg/kg | Rat NP tissue (in vivo/in vitro) | Inactivated NF-κB/NLRP3 | —— | —— | Alleviated IL-1β, IL-18. Downregulated CGRP in DRG neurons | —— | [90] |
| Chemical compound | Atorvastatin | 20 μM | Rat NP tissue (in vivo/in vitro) | Inactivated NF-κB/NLRP3 | Promoted autophagy. Suppressed pyroptosis | Suppressed MMP3, MMP13, ADAMTS4, ADAMTS5. Upregulated Col II, aggrecan | Alleviated IL-1β, IL-18, TNF-α | Alleviated SREBP1 mediated cholesterol-induced pyroptosis and ER stress | [72] |
5.2. Endogenous Small Molecules
5.3. Stem Cells and Bioengineering
6. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Maher, C.; Underwood, M.; Buchbinder, R. Non-specific low back pain. Lancet 2017, 389, 736–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Disease, G.B.D.; Injury, I.; Prevalence, C. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef] [Green Version]
- Dieleman, J.L.; Cao, J.; Chapin, A.; Chen, C.; Li, Z.; Liu, A.; Horst, C.; Kaldjian, A.; Matyasz, T.; Scott, K.W.; et al. US Health Care Spending by Payer and Health Condition, 1996–2016. JAMA 2020, 323, 863–884. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.M.; Karppinen, J.; Chan, D.; Ho, D.W.; Song, Y.Q.; Sham, P.; Cheah, K.S.; Leong, J.C.; Luk, K.D. Prevalence and pattern of lumbar magnetic resonance imaging changes in a population study of one thousand forty-three individuals. Spine 2009, 34, 934–940. [Google Scholar] [CrossRef] [PubMed]
- de Schepper, E.I.; Damen, J.; van Meurs, J.B.; Ginai, A.Z.; Popham, M.; Hofman, A.; Koes, B.W.; Bierma-Zeinstra, S.M. The association between lumbar disc degeneration and low back pain: The influence of age, gender, and individual radiographic features. Spine 2010, 35, 531–536. [Google Scholar] [CrossRef]
- Jo, M.; Chae, S.W. Stress analysis of intervertebral disc during occupational activities. Comput. Methods Programs Biomed. 2021, 208, 106298. [Google Scholar] [CrossRef]
- Ding, F.; Shao, Z.W.; Xiong, L.M. Cell death in intervertebral disc degeneration. Apoptosis 2013, 18, 777–785. [Google Scholar] [CrossRef]
- Liang, H.; Luo, R.; Li, G.; Zhang, W.; Song, Y.; Yang, C. The Proteolysis of ECM in Intervertebral Disc Degeneration. Int. J. Mol. Sci. 2022, 23, 1715. [Google Scholar] [CrossRef]
- Navone, S.E.; Marfia, G.; Giannoni, A.; Beretta, M.; Guarnaccia, L.; Gualtierotti, R.; Nicoli, D.; Rampini, P.; Campanella, R. Inflammatory mediators and signalling pathways controlling intervertebral disc degeneration. Histol. Histopathol. 2017, 32, 523–542. [Google Scholar] [CrossRef]
- Raj, P.P. Intervertebral disc: Anatomy-physiology-pathophysiology-treatment. Pain Pract. 2008, 8, 18–44. [Google Scholar] [CrossRef]
- Zhao, L.; Manchikanti, L.; Kaye, A.D.; Abd-Elsayed, A. Treatment of Discogenic Low Back Pain: Current Treatment Strategies and Future Options—A Literature Review. Curr. Pain Headache Rep. 2019, 23, 86. [Google Scholar] [CrossRef] [PubMed]
- Eisenstein, S.M.; Balain, B.; Roberts, S. Current Treatment Options for Intervertebral Disc Pathologies. Cartilage 2020, 11, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.H.; Kim, H.S.; Jang, I.T. Intervertebral Disc Diseases PART 2: A Review of the Current Diagnostic and Treatment Strategies for Intervertebral Disc Disease. Int. J. Mol. Sci. 2020, 21, 2135. [Google Scholar] [CrossRef] [Green Version]
- Zychlinsky, A.; Prevost, M.C.; Sansonetti, P.J. Shigella flexneri induces apoptosis in infected macrophages. Nature 1992, 358, 167–169. [Google Scholar] [CrossRef]
- Cookson, B.T.; Brennan, M.A. Pro-inflammatory programmed cell death. Trends Microbiol. 2001, 9, 113–114. [Google Scholar] [CrossRef]
- Bergsbaken, T.; Fink, S.L.; Cookson, B.T. Pyroptosis: Host cell death and inflammation. Nat. Rev. Microbiol. 2009, 7, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Wang, Z.; Yu, H.; Zhuang, W.; Chen, J.; Jiang, Y.; Guo, Z.; Huang, X.; Liu, Q. Cell pyroptosis in picornavirus and its potential for treating viral infection. J. Med. Virol. 2022, 94, 3570–3580. [Google Scholar] [CrossRef]
- Yu, P.; Zhang, X.; Liu, N.; Tang, L.; Peng, C.; Chen, X. Pyroptosis: Mechanisms and diseases. Signal Transduct. Target Ther. 2021, 6, 128. [Google Scholar] [CrossRef]
- Voet, S.; Srinivasan, S.; Lamkanfi, M.; van Loo, G. Inflammasomes in neuroinflammatory and neurodegenerative diseases. EMBO Mol. Med. 2019, 11, e10248. [Google Scholar] [CrossRef]
- Tang, P.; Zhu, R.; Ji, W.P.; Wang, J.Y.; Chen, S.; Fan, S.W.; Hu, Z.J. The NLRP3/Caspase-1/Interleukin-1beta Axis is Active in Human Lumbar Cartilaginous Endplate Degeneration. Clin. Orthop. Relat. Res. 2016, 474, 1818–1826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, G.; Han, X.; Lin, Z.; Qian, H.; Chen, B.; Zhou, C.; Chen, Y.; Jiang, W. Propionibacterium acnes Accelerates Intervertebral Disc Degeneration by Inducing Pyroptosis of Nucleus Pulposus Cells via the ROS-NLRP3 Pathway. Oxid. Med. Cell. Longev. 2021, 2021, 4657014. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; An, R.; Xiang, Q.; Li, G.; Wang, K.; Song, Y.; Liao, Z.; Li, S.; Hua, W.; Feng, X.; et al. Acid-sensing ion channels regulate nucleus pulposus cell inflammation and pyroptosis via the NLRP3 inflammasome in intervertebral disc degeneration. Cell Prolif. 2021, 54, e12941. [Google Scholar] [CrossRef] [PubMed]
- Martinon, F.; Burns, K.; Tschopp, J. The inflammasome: A molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol. Cell 2002, 10, 417–426. [Google Scholar] [CrossRef]
- Broz, P.; Dixit, V.M. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 2016, 16, 407–420. [Google Scholar] [CrossRef]
- Burdette, B.E.; Esparza, A.N.; Zhu, H.; Wang, S. Gasdermin D in pyroptosis. Acta Pharm. Sin. B 2021, 11, 2768–2782. [Google Scholar] [CrossRef]
- Hornung, V.; Ablasser, A.; Charrel-Dennis, M.; Bauernfeind, F.; Horvath, G.; Caffrey, D.R.; Latz, E.; Fitzgerald, K.A. AIM2 recognizes cytosolic dsDNA and forms a caspase-1-activating inflammasome with ASC. Nature 2009, 458, 514–518. [Google Scholar] [CrossRef] [Green Version]
- Boucher, D.; Monteleone, M.; Coll, R.C.; Chen, K.W.; Ross, C.M.; Teo, J.L.; Gomez, G.A.; Holley, C.L.; Bierschenk, D.; Stacey, K.J.; et al. Caspase-1 self-cleavage is an intrinsic mechanism to terminate inflammasome activity. J. Exp. Med. 2018, 215, 827–840. [Google Scholar] [CrossRef] [Green Version]
- Kuang, S.; Zheng, J.; Yang, H.; Li, S.; Duan, S.; Shen, Y.; Ji, C.; Gan, J.; Xu, X.W.; Li, J. Structure insight of GSDMD reveals the basis of GSDMD autoinhibition in cell pyroptosis. Proc. Natl. Acad. Sci. USA 2017, 114, 10642–10647. [Google Scholar] [CrossRef] [Green Version]
- Ding, J.; Wang, K.; Liu, W.; She, Y.; Sun, Q.; Shi, J.; Sun, H.; Wang, D.C.; Shao, F. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 2016, 535, 111–116. [Google Scholar] [CrossRef]
- Ruhl, S.; Shkarina, K.; Demarco, B.; Heilig, R.; Santos, J.C.; Broz, P. ESCRT-dependent membrane repair negatively regulates pyroptosis downstream of GSDMD activation. Science 2018, 362, 956–960. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Newton, K.; Dixit, V.M.; Kayagaki, N. Dying cells fan the flames of inflammation. Science 2021, 374, 1076–1080. [Google Scholar] [CrossRef] [PubMed]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [Green Version]
- Bauernfeind, F.G.; Horvath, G.; Stutz, A.; Alnemri, E.S.; MacDonald, K.; Speert, D.; Fernandes-Alnemri, T.; Wu, J.; Monks, B.G.; Fitzgerald, K.A.; et al. Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J. Immunol. 2009, 183, 787–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Py, B.F.; Kim, M.S.; Vakifahmetoglu-Norberg, H.; Yuan, J. Deubiquitination of NLRP3 by BRCC3 critically regulates inflammasome activity. Mol. Cell 2013, 49, 331–338. [Google Scholar] [CrossRef] [Green Version]
- Song, N.; Liu, Z.S.; Xue, W.; Bai, Z.F.; Wang, Q.Y.; Dai, J.; Liu, X.; Huang, Y.J.; Cai, H.; Zhan, X.Y.; et al. NLRP3 Phosphorylation is an Essential Priming Event for Inflammasome Activation. Mol. Cell 2017, 68, 185–197.e186. [Google Scholar] [CrossRef] [Green Version]
- Downs, K.P.; Nguyen, H.; Dorfleutner, A.; Stehlik, C. An overview of the non-canonical inflammasome. Mol. Asp. Med. 2020, 76, 100924. [Google Scholar] [CrossRef]
- Aglietti, R.A.; Estevez, A.; Gupta, A.; Ramirez, M.G.; Liu, P.S.; Kayagaki, N.; Ciferri, C.; Dixit, V.M.; Dueber, E.C. GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proc. Natl. Acad. Sci. USA 2016, 113, 7858–7863. [Google Scholar] [CrossRef] [Green Version]
- Man, S.M.; Karki, R.; Kanneganti, T.D. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol. Rev. 2017, 277, 61–75. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Chen, Z.M.; Wu, X.; Zhang, L.; Cao, Y.; Zhou, P. Distinct Molecular Mechanisms Underlying Potassium Efflux for NLRP3 Inflammasome Activation. Front. Immunol. 2020, 11, 609441. [Google Scholar] [CrossRef]
- Yang, D.; He, Y.; Munoz-Planillo, R.; Liu, Q.; Nunez, G. Caspase-11 Requires the Pannexin-1 Channel and the Purinergic P2X7 Pore to Mediate Pyroptosis and Endotoxic Shock. Immunity 2015, 43, 923–932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Gao, W.; Shi, X.; Ding, J.; Liu, W.; He, H.; Wang, K.; Shao, F. Chemotherapy drugs induce pyroptosis through caspase-3 cleavage of a gasdermin. Nature 2017, 547, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Rogers, C.; Fernandes-Alnemri, T.; Mayes, L.; Alnemri, D.; Cingolani, G.; Alnemri, E.S. Cleavage of DFNA5 by caspase-3 during apoptosis mediates progression to secondary necrotic/pyroptotic cell death. Nat. Commun. 2017, 8, 14128. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Zhang, Y.; Xia, S.; Kong, Q.; Li, S.; Liu, X.; Junqueira, C.; Meza-Sosa, K.F.; Mok, T.M.Y.; Ansara, J.; et al. Gasdermin E suppresses tumour growth by activating anti-tumour immunity. Nature 2020, 579, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; He, H.; Wang, K.; Shi, X.; Wang, Y.; Su, Y.; Wang, Y.; Li, D.; Liu, W.; Zhang, Y.; et al. Granzyme A from cytotoxic lymphocytes cleaves GSDMB to trigger pyroptosis in target cells. Science 2020, 368, eaaz7548. [Google Scholar] [CrossRef] [PubMed]
- Kambara, H.; Liu, F.; Zhang, X.; Liu, P.; Bajrami, B.; Teng, Y.; Zhao, L.; Zhou, S.; Yu, H.; Zhou, W.; et al. Gasdermin D Exerts Anti-inflammatory Effects by Promoting Neutrophil Death. Cell Rep. 2018, 22, 2924–2936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, M.A.; Roughley, P.J. What is intervertebral disc degeneration, and what causes it? Spine 2006, 31, 2151–2161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, W.C.; Sze, K.L.; Samartzis, D.; Leung, V.Y.; Chan, D. Structure and biology of the intervertebral disk in health and disease. Orthop. Clin. N. Am. 2011, 42, 447–464. [Google Scholar] [CrossRef]
- Guerrero, J.; Hackel, S.; Croft, A.S.; Hoppe, S.; Albers, C.E.; Gantenbein, B. The nucleus pulposus microenvironment in the intervertebral disc: The fountain of youth? Eur. Cell Mater. 2021, 41, 707–738. [Google Scholar] [CrossRef]
- Antoniou, J.; Steffen, T.; Nelson, F.; Winterbottom, N.; Hollander, A.P.; Poole, R.A.; Aebi, M.; Alini, M. The human lumbar intervertebral disc: Evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J. Clin. Investig. 1996, 98, 996–1003. [Google Scholar] [CrossRef] [Green Version]
- Visse, R.; Nagase, H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003, 92, 827–839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apte, S.S. A disintegrin-like and metalloprotease (reprolysin-type) with thrombospondin type 1 motif (ADAMTS) superfamily: Functions and mechanisms. J. Biol. Chem. 2009, 284, 31493–31497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sivan, S.S.; Wachtel, E.; Roughley, P. Structure, function, aging and turnover of aggrecan in the intervertebral disc. Biochim. Biophys. Acta 2014, 1840, 3181–3189. [Google Scholar] [CrossRef] [PubMed]
- Nerurkar, N.L.; Elliott, D.M.; Mauck, R.L. Mechanical design criteria for intervertebral disc tissue engineering. J. Biomech. 2010, 43, 1017–1030. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, G.; Shi, C.; Lin, J.; Wang, S.; Wang, H.; Liu, T.; Yang, H.; Li, B. Biomechanics in Annulus Fibrosus Degeneration and Regeneration. Adv. Exp. Med. Biol. 2018, 1078, 409–420. [Google Scholar] [CrossRef]
- Anitua, E.; Padilla, S. Biologic therapies to enhance intervertebral disc repair. Regen. Med. 2018, 13, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Ashinsky, B.; Smith, H.E.; Mauck, R.L.; Gullbrand, S.E. Intervertebral disc degeneration and regeneration: A motion segment perspective. Eur. Cell Mater. 2021, 41, 370–380. [Google Scholar] [CrossRef]
- Roberts, S.; Urban, J.P.; Evans, H.; Eisenstein, S.M. Transport properties of the human cartilage endplate in relation to its composition and calcification. Spine 1996, 21, 415–420. [Google Scholar] [CrossRef]
- Wong, J.; Sampson, S.L.; Bell-Briones, H.; Ouyang, A.; Lazar, A.A.; Lotz, J.C.; Fields, A.J. Nutrient supply and nucleus pulposus cell function: Effects of the transport properties of the cartilage endplate and potential implications for intradiscal biologic therapy. Osteoarthr. Cartil. 2019, 27, 956–964. [Google Scholar] [CrossRef]
- Gullbrand, S.E.; Ashinsky, B.G.; Martin, J.T.; Pickup, S.; Smith, L.J.; Mauck, R.L.; Smith, H.E. Correlations between quantitative T2 and T1rho MRI, mechanical properties and biochemical composition in a rabbit lumbar intervertebral disc degeneration model. J. Orthop. Res. 2016, 34, 1382–1388. [Google Scholar] [CrossRef]
- Chen, Z.H.; Jin, S.H.; Wang, M.Y.; Jin, X.L.; Lv, C.; Deng, Y.F.; Wang, J.L. Enhanced NLRP3, caspase-1, and IL- 1beta levels in degenerate human intervertebral disc and their association with the grades of disc degeneration. Anat. Rec. 2015, 298, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Fu, F.; Bao, R.; Yao, S.; Zhou, C.; Luo, H.; Zhang, Z.; Zhang, H.; Li, Y.; Yan, S.; Yu, H.; et al. Aberrant spinal mechanical loading stress triggers intervertebral disc degeneration by inducing pyroptosis and nerve ingrowth. Sci. Rep. 2021, 11, 772. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Zhang, C.; Zhou, Z.M.; Wang, K.; Gao, J.W.; Qian, Z.Y.; Bao, J.P.; Ji, H.Y.; Cabral, V.L.F.; Wu, X.T. A20 attenuates pyroptosis and apoptosis in nucleus pulposus cells via promoting mitophagy and stabilizing mitochondrial dynamics. Inflamm. Res. 2022, 71, 695–710. [Google Scholar] [CrossRef]
- Wang, Y.; Kanneganti, T.D. From pyroptosis, apoptosis and necroptosis to PANoptosis: A mechanistic compendium of programmed cell death pathways. Comput. Struct. Biotechnol. J. 2021, 19, 4641–4657. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, X.; Li, J.; Du, L.; Wang, X.; Cao, J.; Li, H.; Huo, Z.; Li, G.; Pan, D.; et al. Circ_0004354 might compete with circ_0040039 to induce NPCs death and inflammatory response by targeting miR-345-3p-FAF1/TP73 axis in intervertebral disc degeneration. Oxid. Med. Cell Longev. 2022, 2022, 2776440. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yuan, Y.; Huang, Z.X.; Chen, H.; Lan, R.; Wang, Z.; Lai, K.; Chen, H.; Chen, Z.; Zou, Z.; et al. GSDME-mediated pyroptosis promotes inflammation and fibrosis in obstructive nephropathy. Cell Death Differ. 2021, 28, 2333–2350. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Li, S.; Markova, D.Z.; Liang, A.; Kepler, C.K.; Huang, Y.; Zhou, J.; Yan, J.; Chen, W.; Huang, D.; et al. Bromodomain-containing protein 4 inhibition alleviates matrix degradation by enhancing autophagy and suppressing NLRP3 inflammasome activity in NP cells. J. Cell Physiol. 2020, 235, 5736–5749. [Google Scholar] [CrossRef]
- Chen, J.; Yan, J.; Li, S.; Zhu, J.; Zhou, J.; Li, J.; Zhang, Y.; Huang, Z.; Yuan, L.; Xu, K.; et al. Atorvastatin inhibited TNF-alpha induced matrix degradation in rat nucleus pulposus cells by suppressing NLRP3 inflammasome activity and inducing autophagy through NF-kappaB signaling. Cell Cycle 2021, 20, 2160–2173. [Google Scholar] [CrossRef]
- Liao, Z.; Li, S.; Liu, R.; Feng, X.; Shi, Y.; Wang, K.; Li, S.; Zhang, Y.; Wu, X.; Yang, C. Autophagic Degradation of Gasdermin D Protects against Nucleus Pulposus Cell Pyroptosis and Retards Intervertebral Disc Degeneration In Vivo. Oxid. Med. Cell Longev. 2021, 2021, 5584447. [Google Scholar] [CrossRef]
- Le Maitre, C.L.; Pockert, A.; Buttle, D.J.; Freemont, A.J.; Hoyland, J.A. Matrix synthesis and degradation in human intervertebral disc degeneration. Biochem. Soc. Trans. 2007, 35, 652–655. [Google Scholar] [CrossRef]
- Huang, Y.; Peng, Y.; Sun, J.; Li, S.; Hong, J.; Zhou, J.; Chen, J.; Yan, J.; Huang, Z.; Wang, X.; et al. Nicotinamide Phosphoribosyl Transferase Controls NLRP3 Inflammasome Activity through MAPK and NF-kappaB Signaling in Nucleus Pulposus Cells, as Suppressed by Melatonin. Inflammation 2020, 43, 796–809. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Li, S.; Zhang, Y.; Deng, Z.; Wu, J.; Huang, Z.; Qin, T.; Xiao, Y.; Zhou, J.; Xu, K.; et al. Cholesterol Induces Pyroptosis and Matrix Degradation via mSREBP1-Driven Endoplasmic Reticulum Stress in Intervertebral Disc Degeneration. Front. Cell Dev. Biol. 2021, 9, 803132. [Google Scholar] [CrossRef] [PubMed]
- Lawson, L.Y.; Harfe, B.D. Developmental mechanisms of intervertebral disc and vertebral column formation. Wiley Interdiscip. Rev. Dev. Biol. 2017, 6, e283. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.J.; Yu, X.H.; Wang, C.; Yang, W.; He, W.S.; Zhang, S.J.; Yan, Y.G.; Zhang, J. MMPs and ADAMTSs in intervertebral disc degeneration. Clin. Chim. Acta 2015, 448, 238–246. [Google Scholar] [CrossRef]
- Wang, Y.; Che, M.; Xin, J.; Zheng, Z.; Li, J.; Zhang, S. The role of IL-1beta and TNF-alpha in intervertebral disc degeneration. Biomed. Pharmacother. 2020, 131, 110660. [Google Scholar] [CrossRef]
- Brand, F.J., 3rd; Forouzandeh, M.; Kaur, H.; Travascio, F.; de Rivero Vaccari, J.P. Acidification changes affect the inflammasome in human nucleus pulposus cells. J. Inflamm. 2016, 13, 29. [Google Scholar] [CrossRef] [Green Version]
- Song, Y.; Wang, Y.; Zhang, Y.; Geng, W.; Liu, W.; Gao, Y.; Li, S.; Wang, K.; Wu, X.; Kang, L.; et al. Advanced glycation end products regulate anabolic and catabolic activities via NLRP3-inflammasome activation in human nucleus pulposus cells. J. Cell Mol. Med. 2017, 21, 1373–1387. [Google Scholar] [CrossRef]
- Johnson, Z.I.; Schoepflin, Z.R.; Choi, H.; Shapiro, I.M.; Risbud, M.V. Disc in flames: Roles of TNF-alpha and IL-1beta in intervertebral disc degeneration. Eur. Cell Mater. 2015, 30, 104–116; discussion 107–116. [Google Scholar] [CrossRef]
- Capossela, S.; Schlafli, P.; Bertolo, A.; Janner, T.; Stadler, B.M.; Potzel, T.; Baur, M.; Stoyanov, J.V. Degenerated human intervertebral discs contain autoantibodies against extracellular matrix proteins. Eur. Cell Mater. 2014, 27, 251–263; discussion 263. [Google Scholar] [CrossRef]
- Sun, Z.; Liu, B.; Luo, Z.J. The Immune Privilege of the Intervertebral Disc: Implications for Intervertebral Disc Degeneration Treatment. Int. J. Med. Sci. 2020, 17, 685–692. [Google Scholar] [CrossRef]
- Risbud, M.V.; Shapiro, I.M. Role of cytokines in intervertebral disc degeneration: Pain and disc content. Nat. Rev. Rheumatol. 2014, 10, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Le Maitre, C.L.; Freemont, A.J.; Hoyland, J.A. The role of interleukin-1 in the pathogenesis of human intervertebral disc degeneration. Arthritis Res. Ther. 2005, 7, R732–R745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jimbo, K.; Park, J.S.; Yokosuka, K.; Sato, K.; Nagata, K. Positive feedback loop of interleukin-1beta upregulating production of inflammatory mediators in human intervertebral disc cells in vitro. J. Neurosurg. Spine 2005, 2, 589–595. [Google Scholar] [CrossRef]
- Penolazzi, L.; Bergamin, L.S.; Lambertini, E.; Poma, V.V.; Sarti, A.C.; De Bonis, P.; Di Virgilio, F.; Piva, R. The P2X7 purinergic receptor in intervertebral disc degeneration. J. Cell Physiol. 2022, 237, 1418–1428. [Google Scholar] [CrossRef]
- Chen, Z.; Cao, P.; Zhou, Z.; Yuan, Y.; Jiao, Y.; Zheng, Y. Overview: The role of Propionibacterium acnes in nonpyogenic intervertebral discs. Int. Orthop. 2016, 40, 1291–1298. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Zhou, M.; Bai, Z.; Wen, Y.; Shen, J.; Hu, Z. Propionibacterium acnes induces intervertebral disc degeneration by promoting nucleus pulposus cell pyroptosis via NLRP3-dependent pathway. Biochem. Biophys. Res. Commun. 2020, 526, 772–779. [Google Scholar] [CrossRef]
- Stefanakis, M.; Al-Abbasi, M.; Harding, I.; Pollintine, P.; Dolan, P.; Tarlton, J.; Adams, M.A. Annulus fissures are mechanically and chemically conducive to the ingrowth of nerves and blood vessels. Spine 2012, 37, 1883–1891. [Google Scholar] [CrossRef]
- Johnson, W.E.; Sivan, S.; Wright, K.T.; Eisenstein, S.M.; Maroudas, A.; Roberts, S. Human intervertebral disc cells promote nerve growth over substrata of human intervertebral disc aggrecan. Spine 2006, 31, 1187–1193. [Google Scholar] [CrossRef]
- Arkless, K.; Argunhan, F.; Brain, S.D. CGRP Discovery and Timeline. Handb. Exp. Pharmacol. 2019, 255, 1–12. [Google Scholar] [CrossRef]
- Zhang, A.; Wang, K.; Ding, L.; Bao, X.; Wang, X.; Qiu, X.; Liu, J. Bay11-7082 attenuates neuropathic pain via inhibition of nuclear factor-kappa B and nucleotide-binding domain-like receptor protein 3 inflammasome activation in dorsal root ganglions in a rat model of lumbar disc herniation. J. Pain Res. 2017, 10, 375–382. [Google Scholar] [CrossRef]
- Sun, Y.; Leng, P.; Song, M.; Li, D.; Guo, P.; Xu, X.; Gao, H.; Li, Z.; Li, C.; Zhang, H. Piezo1 activates the NLRP3 inflammasome in nucleus pulposus cell-mediated by Ca2+/NF-kappaB pathway. Int. Immunopharmacol. 2020, 85, 106681. [Google Scholar] [CrossRef] [PubMed]
- Mamet, J.; Lazdunski, M.; Voilley, N. How nerve growth factor drives physiological and inflammatory expressions of acid-sensing ion channel 3 in sensory neurons. J. Biol. Chem. 2003, 278, 48907–48913. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sen, T.; Samanta, S.K. Medicinal plants, human health and biodiversity: A broad review. Adv. Biochem. Eng. Biotechnol. 2015, 147, 59–110. [Google Scholar] [CrossRef] [PubMed]
- Pantziarka, P.; Pirmohamed, M.; Mirza, N. New uses for old drugs. BMJ 2018, 361, k2701. [Google Scholar] [CrossRef]
- Lim, K.H.; Staudt, L.M. Toll-like receptor signaling. Cold Spring Harb. Perspect. Biol. 2013, 5, a011247. [Google Scholar] [CrossRef] [Green Version]
- Wang, D.; Cai, X.; Xu, F.; Kang, H.; Li, Y.; Feng, R. Ganoderic Acid A alleviates the degeneration of intervertebral disc via suppressing the activation of TLR4/NLRP3 signaling pathway. Bioengineered 2022, 13, 11684–11693. [Google Scholar] [CrossRef]
- Yang, H.; Wang, H.; Andersson, U. Targeting Inflammation Driven by HMGB1. Front. Immunol. 2020, 11, 484. [Google Scholar] [CrossRef] [Green Version]
- Zhao, F.; Guo, Z.; Hou, F.; Fan, W.; Wu, B.; Qian, Z. Magnoflorine Alleviates “M1” Polarized Macrophage-Induced Intervertebral Disc Degeneration through Repressing the HMGB1/Myd88/NF-kappaB Pathway and NLRP3 Inflammasome. Front. Pharmacol. 2021, 12, 701087. [Google Scholar] [CrossRef]
- Li, W.; Deng, M.; Loughran, P.A.; Yang, M.; Lin, M.; Yang, C.; Gao, W.; Jin, S.; Li, S.; Cai, J.; et al. LPS Induces Active HMGB1 Release from Hepatocytes into Exosomes through the Coordinated Activities of TLR4 and Caspase-11/GSDMD Signaling. Front. Immunol. 2020, 11, 229. [Google Scholar] [CrossRef]
- Xu, J.; Jiang, Y.; Wang, J.; Shi, X.; Liu, Q.; Liu, Z.; Li, Y.; Scott, M.J.; Xiao, G.; Li, S.; et al. Macrophage endocytosis of high-mobility group box 1 triggers pyroptosis. Cell Death Differ. 2014, 21, 1229–1239. [Google Scholar] [CrossRef]
- Yilmaz, I.; Akalan, H.; Oznam, K.; Karaarslan, N.; Yasar Sirin, D.; Ozbek, H. Does oseltamivir protect human chondrocyte and nucleus pulposus cells from degeneration by inhibiting senescence and proinflammation mediated by the NLRP3 inflammasome and NF-kappaB? Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 4816–4827. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tong, Z.; Jiang, S.; Zheng, W.; Zhao, J.; Zhou, X. The Roles of Endoplasmic Reticulum in NLRP3 Inflammasome Activation. Cells 2020, 9, 1219. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.Y.; Luo, Z.P. Paeoniflorin inhibits pyroptosis of nucleus pulposus cells in an acidic environment and alleviates the degeneration of the intervertebral disc in rats. Cell. Signal. 2022, 91, 110243. [Google Scholar] [CrossRef]
- Xu, H.; Dai, Z.H.; He, G.L.; Cai, H.C.; Chen, X.Y.; Chen, Y.L.; Xu, C.; Sheng, S.R. Gamma-oryzanol alleviates intervertebral disc degeneration development by intercepting the IL-1beta/NLRP3 inflammasome positive cycle. Phytomedicine 2022, 102, 154176. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Tardivel, A.; Thorens, B.; Choi, I.; Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 2010, 11, 136–140. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, Z.; Yang, X.; Cao, X.; Liang, Z.; Ma, H.; Zhao, J. Morin attenuates pyroptosis of nucleus pulposus cells and ameliorates intervertebral disc degeneration via inhibition of the TXNIP/NLRP3/Caspase-1/IL-1beta signaling pathway. Biochem. Biophys. Res. Commun. 2021, 559, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.; Gu, J.M.; Xie, Z.A.; Gu, Y.; Jie, Z.W.; Huang, K.M.; Wang, J.Y.; Fan, S.W.; Jiang, X.S.; Hu, Z.J. Honokiol alleviates the degeneration of intervertebral disc via suppressing the activation of TXNIP-NLRP3 inflammasome signal pathway. Free Radic. Biol. Med. 2018, 120, 368–379. [Google Scholar] [CrossRef]
- Bofill, A.; Jalencas, X.; Oprea, T.I.; Mestres, J. The human endogenous metabolome as a pharmacology baseline for drug discovery. Drug Discov. Today 2019, 24, 1806–1820. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Gong, Y.; Zheng, X.; Qiu, J.; Jiang, T.; Chen, L.; Lu, F.; Wu, X.; Cheng, F.; Hong, Z. Platelet-Derived Growth Factor-BB Inhibits Intervertebral Disc Degeneration via Suppressing Pyroptosis and Activating the MAPK Signaling Pathway. Front. Pharmacol. 2021, 12, 799130. [Google Scholar] [CrossRef]
- Wang, Y.H.; Li, Y.; Wang, J.N.; Zhao, Q.X.; Jin, J.; Wen, S.; Wang, S.C.; Sun, T. Maresin 1 Attenuates Radicular Pain through the Inhibition of NLRP3 Inflammasome-Induced Pyroptosis via NF-kappaB Signaling. Front. Neurosci. 2020, 14, 831. [Google Scholar] [CrossRef]
- Chen, F.; Jiang, G.; Liu, H.; Li, Z.; Pei, Y.; Wang, H.; Pan, H.; Cui, H.; Long, J.; Wang, J.; et al. Melatonin alleviates intervertebral disc degeneration by disrupting the IL-1beta/NF-kappaB-NLRP3 inflammasome positive feedback loop. Bone Res. 2020, 8, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Qiu, C.; Wang, W.; Peng, J.; Cheng, X.; Shangguan, Y.; Xu, M.; Li, J.; Qu, R.; Chen, X.; et al. Cortistatin protects against intervertebral disc degeneration through targeting mitochondrial ROS-dependent NLRP3 inflammasome activation. Theranostics 2020, 10, 7015–7033. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xie, G.; Yang, W.; Wang, W.; Zuo, Z.; Wang, W. Platelet-rich plasma attenuates intervertebral disc degeneration via delivering miR-141-3p-containing exosomes. Cell Cycle 2021, 20, 1487–1499. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Xie, C.; Chen, Z.; He, G.; Dai, Z.; Cai, H.; Zhang, H.; Lu, H.; Wu, H.; Hu, X.; et al. MFG-E8 alleviates intervertebral disc degeneration by suppressing pyroptosis and extracellular matrix degradation in nucleus pulposus cells via Nrf2/TXNIP/NLRP3 axis. Cell Death Discov. 2022, 8, 209. [Google Scholar] [CrossRef]
- Jin, J.; Xie, Y.; Shi, C.; Ma, J.; Wang, Y.; Qiao, L.; Li, K.; Sun, T. Lipoxin A4 Inhibits NLRP3 Inflammasome Activation in Rats with Non-compressive Disc Herniation through the JNK1/Beclin-1/PI3KC3 Pathway. Front. Neurosci. 2020, 14, 799. [Google Scholar] [CrossRef]
- Ma, Z.; Tang, P.; Dong, W.; Lu, Y.; Tan, B.; Zhou, N.; Hao, J.; Shen, J.; Hu, Z. SIRT1 alleviates IL-1beta induced nucleus pulposus cells pyroptosis via mitophagy in intervertebral disc degeneration. Int. Immunopharmacol. 2022, 107, 108671. [Google Scholar] [CrossRef]
- Hu, Z.L.; Li, H.Y.; Chang, X.; Li, Y.Y.; Liu, C.H.; Gao, X.X.; Zhai, Y.; Chen, Y.X.; Li, C.Q. Exosomes derived from stem cells as an emerging therapeutic strategy for intervertebral disc degeneration. World J. Stem Cells 2020, 12, 803–813. [Google Scholar] [CrossRef]
- Bowles, R.D.; Setton, L.A. Biomaterials for intervertebral disc regeneration and repair. Biomaterials 2017, 129, 54–67. [Google Scholar] [CrossRef]
- Yuan, X.; Li, T.; Shi, L.; Miao, J.; Guo, Y.; Chen, Y. Human umbilical cord mesenchymal stem cells deliver exogenous miR-26a-5p via exosomes to inhibit nucleus pulposus cell pyroptosis through METTL14/NLRP3. Mol. Med. 2021, 27, 91. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, J.; Zhang, Y.; Liu, W.; Ni, W.; Huang, X.; Yuan, J.; Zhao, B.; Xiao, H.; Xue, F. Mesenchymal stem cells-derived exosomes ameliorate intervertebral disc degeneration through inhibiting pyroptosis. J. Cell Mol. Med. 2020, 24, 11742–11754. [Google Scholar] [CrossRef]
- Xing, H.; Zhang, Z.; Mao, Q.; Wang, C.; Zhou, Y.; Zhou, X.; Ying, L.; Xu, H.; Hu, S.; Zhang, N. Injectable exosome-functionalized extracellular matrix hydrogel for metabolism balance and pyroptosis regulation in intervertebral disc degeneration. J. Nanobiotechnology 2021, 19, 264. [Google Scholar] [CrossRef] [PubMed]
- Liao, Z.; Liu, H.; Ma, L.; Lei, J.; Tong, B.; Li, G.; Ke, W.; Wang, K.; Feng, X.; Hua, W.; et al. Engineering Extracellular Vesicles Restore the Impaired Cellular Uptake and Attenuate Intervertebral Disc Degeneration. ACS Nano 2021, 15, 14709–14724. [Google Scholar] [CrossRef] [PubMed]
- Xia, K.S.; Li, D.D.; Wang, C.G.; Ying, L.W.; Wang, J.K.; Yang, B.; Shu, J.W.; Huang, X.P.; Zhang, Y.A.; Yu, C.; et al. An esterase-responsive ibuprofen nano-micelle pre-modified embryo derived nucleus pulposus progenitor cells promote the regeneration of intervertebral disc degeneration. Bioact. Mater. 2023, 21, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.S.; Yang, J.M.; Ding, R.J.; Liu, X.Z.; Jiang, X.B.; Yang, Z.J.; Ling, Z.M.; Hu, T.X.; Wei, F.X. Fabrication of a Polylactide-Glycolide/Poly-epsilon-Caprolactone/Dextran/Plastrum Testudinis Extract Composite Anti-Inflammation Nanofiber Membrane via Electrospinning for Annulus Fibrosus Regeneration. J. Biomed. Nanotechnol. 2021, 17, 873–888. [Google Scholar] [CrossRef]
- Vrentas, C.E.; Schaut, R.G.; Boggiatto, P.M.; Olsen, S.C.; Sutterwala, F.S.; Moayeri, M. Inflammasomes in livestock and wildlife: Insights into the intersection of pathogens and natural host species. Vet. Immunol. Immunopathol. 2018, 201, 49–56. [Google Scholar] [CrossRef]

| Compound | Dose | Model Type | Mechanism | References | ||||
|---|---|---|---|---|---|---|---|---|
| Pathway | Cell Death | ECM | Inflammation | Others | ||||
| Melatonin | 1000 μM | Human (in vitro)/Rat NP tissue (in vivo/in vitro) | Inactivated NAMPT/MAPK/NF-κB/NLRP3 | Suppressed pyroptosis | Upregulated Col II, aggrecan | Suppressed IL-1β, IL-18, TNF-α | —— | [71,111] |
| Platelet-Derived Growth Factor-BB | 50 ng/mL | Rat NP tissue (in vivo/in vitro) | Inactivated MAPK/PI3K/AKT/NLRP3 | Suppressed pyroptosis and apoptosis | Suppressed MMP3, MMP9, ADAMTS4, ADAMTS5. Upregulated Col II, aggrecan | Alleviated IL-1β, IL-18 | —— | [109] |
| Cortistatin | 50 μg/mL | Human (in vitro)/Mice NP tissue (in vitro) | Inactivated NF-κB/NLRP3 | Suppressed apoptosis | Suppressed MMP13, ADAMTS5. Upregulated Col II, aggrecan | Alleviated IL-1β, TNF-α | Alleviated respiratory chain. Suppressed mitochondrial ROS generation | [112] |
| Maresin 1 | 100 ng | Rat NP tissue (in vivo/in vitro) | Inactivated NF-κB/NLRP3 | Suppressed pyroptosis | —— | Alleviated IL-1β, IL-18, TNF-α | Alleviated mechanical allodynia. Improved radicular pain | [110] |
| Platelet-rich plasma | —— | Mice NP tissue (in vivo/in vitro) | Activated the Keap1-Nrf2 | Suppressed pyroptosis | —— | Alleviated IL-1β, IL-18, TGF-β, IL-6 | Alleviated ROS production | [113] |
| A20 | —— | Rat NP tissue (in vivo/in vitro) | Inactivated NF-κB/NLRP3 | Promoted mitophagy. Suppressed pyroptosis and apoptosis | —— | Alleviated iNOS, COX2, TNF-α, IL-1β, IL-6, IL-18 | Stabilized mitochondrial dynamics. Inhibited collapse of mitochondrial membrane potential and ROS | [63] |
| MFG-E8 | 100 ng/ml | Rat NP tissue (in vivo/in vitro) | Inactivated Nrf2/TXNIP/NLRP3 | Suppressed pyroptosis | Suppressed MMP3, ADAMTS5. Upregulated Col II, aggrecan | Alleviated IL-1β, IL-18 | Suppressed ROS and mitochondrial dysfunction | [114] |
| Lipoxin A4 | 10 µL, 100 ng | Rat NP tissue (in vivo/in vitro) | Activated JNK1/Beclin-1/PI3KC3/NLRP3 | Promoted autophagy | —— | Decreased TNF-α, IL-1β, IL-18. Increased IL-4, IL-10, TGF-β | Ameliorated the pain threshold | [115] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luo, J.; Yang, Y.; Wang, X.; Chang, X.; Fu, S. Role of Pyroptosis in Intervertebral Disc Degeneration and Its Therapeutic Implications. Biomolecules 2022, 12, 1804. https://doi.org/10.3390/biom12121804
Luo J, Yang Y, Wang X, Chang X, Fu S. Role of Pyroptosis in Intervertebral Disc Degeneration and Its Therapeutic Implications. Biomolecules. 2022; 12(12):1804. https://doi.org/10.3390/biom12121804
Chicago/Turabian StyleLuo, Jieya, Yuxuan Yang, Xuan Wang, Xingyu Chang, and Songbo Fu. 2022. "Role of Pyroptosis in Intervertebral Disc Degeneration and Its Therapeutic Implications" Biomolecules 12, no. 12: 1804. https://doi.org/10.3390/biom12121804






