Gut Microbiota, Microbial Metabolites and Human Physical Performance
Abstract
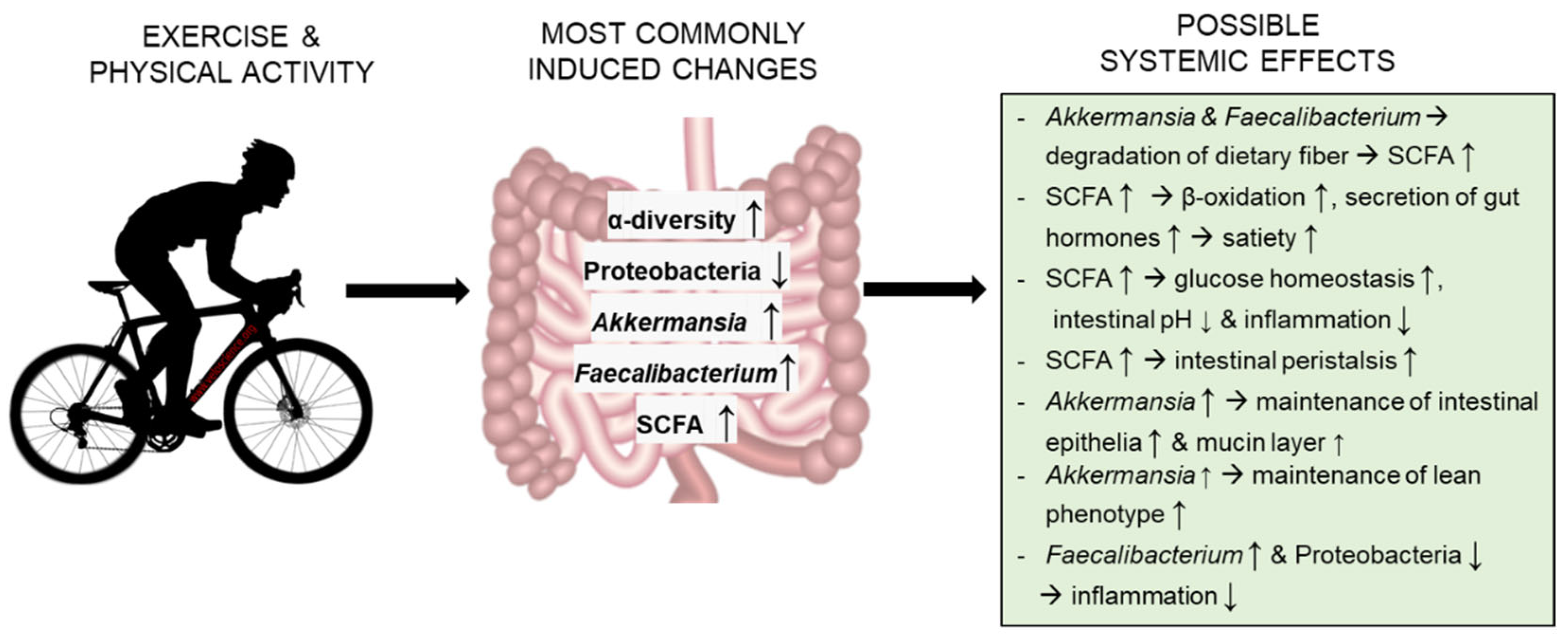
:1. Introduction
2. Cross-Sectional Studies Have Reported Associations between Exercise, Physical Performance, and the Gut Microbiota in Humans—Some Examples
3. Longitudinal Studies Showing Effects of Longer-Term Exercise on the Human Gut Microbiota and Microbial Metabolism
3.1. Endurance Exercise and Gut Microbiota
3.2. The Effects of Combined Resistance and Aerobic Exercise on the Gut Microbiota
3.3. High Intensity Exercise or Strenuous Training and Gut Microbiota
4. The Effects of Shorter-Term Exercise Challenges on the Gut Microbiome
5. Certain Gut Microbes Can Increase Physical Performance
6. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- WHO. Available online: https://www.who.int/health-topics/obesity#tab=tab_1 (accessed on 15 October 2021).
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Margolis, K.G.; Cryan, J.F.; Mayer, E.A. The Microbiota-Gut-Brain Axis: From Motility to Mood. Gastroenterology 2021, 160, 1486–1501. [Google Scholar] [CrossRef] [PubMed]
- van de Wouw, M.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. Microbiota-gut-brain axis: Modulator of host metabolism and appetite. J. Nutr. 2017, 147, 727–745. [Google Scholar] [CrossRef] [Green Version]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef] [Green Version]
- Koh, A.; Bäckhed, F. From Association to Causality: The Role of the Gut Microbiota and Its Functional Products on Host Metabolism. Mol. Cell 2020, 78, 584–596. [Google Scholar] [CrossRef]
- Macfarlane, S.; Macfarlane, G.T. Regulation of short-chain fatty acid production. Proc. Nutr. Soc. 2007, 62, 67–72. [Google Scholar] [CrossRef]
- Song, B.K.; Cho, K.O.; Jo, Y.; Oh, J.W.; Kim, Y.S. Colon transit time according to physical activity level in adults. J. Neurogastroenterol. Motil. 2012, 18, 64–69. [Google Scholar] [CrossRef]
- Li, M.; van Esch, B.; Wagenaar, G.T.M.; Garssen, J.; Folkerts, G.; Henricks, P.A.J. Pro- and anti-inflammatory effects of short chain fatty acids on immune and endothelial cells. Eur. J. Pharm. 2018, 831, 52–59. [Google Scholar] [CrossRef]
- Thomas, A.M.; Manghi, P.; Asnicar, F.; Pasolli, E.; Armanini, F.; Zolfo, M.; Beghini, F.; Manara, S.; Karcher, N.; Pozzi, C.; et al. Metagenomic analysis of colorectal cancer datasets identifies cross-cohort microbial diagnostic signatures and a link with choline degradation. Nat. Med. 2019, 25, 667–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leshem, A.; Segal, E.; Elinav, E. The Gut Microbiome and Individual-Specific Responses to Diet. mSystems 2020, 5, e00665-20. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [Green Version]
- Lim, M.Y.; You, H.J.; Yoon, H.S.; Kwon, B.; Lee, J.Y.; Lee, S.; Song, Y.M.; Lee, K.; Sung, J.; Ko, G. The effect of heritability and host genetics on the gut microbiota and metabolic syndrome. Gut 2017, 66, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Goodrich, J.K.; Waters, J.L.; Poole, A.C.; Sutter, J.L.; Koren, O.; Blekhman, R.; Beaumont, M.; Van Treuren, W.; Knight, R.; Bell, J.T.; et al. Human genetics shape the gut microbiome. Cell 2014, 159, 789–799. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.S.; Unno, T.; Kim, B.Y.; Park, M.S. Sex Differences in Gut Microbiota. World J. Mens Health 2020, 38, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Dwiyanto, J.; Hussain, M.H.; Reidpath, D.; Ong, K.S.; Qasim, A.; Lee, S.W.H.; Lee, S.M.; Foo, S.C.; Chong, C.W.; Rahman, S. Ethnicity influences the gut microbiota of individuals sharing a geographical location: A cross-sectional study from a middle-income country. Sci. Rep. 2021, 11, 2618. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhu, H.; Qiu, P. Aging progression of human gut microbiota. BMC Microbiol. 2019, 19, 236. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez, N.R.; Di Marco, N.M.; Langley, S. American College of Sports Medicine position stand. Nutrition and athletic performance. Med. Sci. Sports Exerc. 2009, 41, 709–731. [Google Scholar]
- Murtaza, N.; Burke, L.M.; Vlahovich, N.; Charlesson, B.; O’Neill, H.; Ross, M.L.; Campbell, K.L.; Krause, L.; Morrison, M. The Effects of Dietary Pattern during Intensified Training on Stool Microbiota of Elite Race Walkers. Nutrients 2019, 11, 14. [Google Scholar] [CrossRef] [Green Version]
- Clark, A.; Mach, N. Exercise-induced stress behavior, gut-microbiota-brain axis and diet: A systematic review for athletes. J. Int. Soc. Sports Nutr. 2016, 13, 43. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, C.M.; Davy, B.M.; Hulver, M.W.; Neilson, A.P.; Bennett, B.J.; Davy, K.P. Does Exercise Alter Gut Microbial Composition? A Systematic Review. Med. Sci. Sports Exerc. 2019, 51, 160–167. [Google Scholar] [CrossRef]
- Clemente, J.C.; Manasson, J.; Scher, J.U. The role of the gut microbiome in systemic inflammatory disease. BMJ 2018, 360, j5145. [Google Scholar] [CrossRef] [PubMed]
- Houghton, D.; Hardy, T.; Stewart, C.; Errington, L.; Day, C.P.; Trenell, M.I.; Avery, L. Systematic review assessing the effectiveness of dietary intervention on gut microbiota in adults with type 2 diabetes. Diabetologia 2018, 61, 1700–1711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frampton, J.; Murphy, K.G.; Frost, G.; Chambers, E.S. Short-chain fatty acids as potential regulators of skeletal muscle metabolism and function. Nat. Metab. 2020, 2, 840–848. [Google Scholar] [CrossRef]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef] [Green Version]
- Cani, P.D.; de Vos, W.M. Next-Generation Beneficial Microbes: The Case of Akkermansia muciniphila. Front. Microbiol. 2017, 8, 1765. [Google Scholar] [CrossRef]
- Karlsson, C.L.; Onnerfält, J.; Xu, J.; Molin, G.; Ahrné, S.; Thorngren-Jerneck, K. The microbiota of the gut in preschool children with normal and excessive body weight. Obesity 2012, 20, 2257–2261. [Google Scholar] [CrossRef] [PubMed]
- Depommier, C.; Everard, A.; Druart, C.; Plovier, H.; Van Hul, M.; Vieira-Silva, S.; Falony, G.; Raes, J.; Maiter, D.; Delzenne, N.M.; et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: A proof-of-concept exploratory study. Nat. Med. 2019, 25, 1096–1103. [Google Scholar] [CrossRef]
- Barton, W.; Penney, N.C.; Cronin, O.; Garcia-Perez, I.; Molloy, M.G.; Holmes, E.; Shanahan, F.; Cotter, P.D.; O’Sullivan, O. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 2018, 67, 625–633. [Google Scholar] [CrossRef]
- Bressa, C.; Bailen-Andrino, M.; Perez-Santiago, J.; Gonzalez-Soltero, R.; Perez, M.; Montalvo-Lominchar, M.G.; Mate-Munoz, J.L.; Dominguez, R.; Moreno, D.; Larrosa, M. Differences in gut microbiota profile between women with active lifestyle and sedentary women. PLoS ONE 2017, 12, e0171352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soltys, K.; Lendvorsky, L.; Hric, I.; Baranovicova, E.; Penesova, A.; Mikula, I.; Bohmer, M.; Budis, J.; Vavrova, S.; Grones, J.; et al. Strenuous Physical Training, Physical Fitness, Body Composition and Bacteroides to Prevotella Ratio in the Gut of Elderly Athletes. Front. Physiol. 2021, 12, 12. [Google Scholar] [CrossRef]
- Ottman, N.; Geerlings, S.Y.; Aalvink, S.; de Vos, W.M.; Belzer, C. Action and function of Akkermansia muciniphila in microbiome ecology, health and disease. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 637–642. [Google Scholar] [CrossRef] [Green Version]
- McKenna, C.F.; Salvador, A.F.; Hughes, R.L.; Scaroni, S.E.; Alamilla, R.A.; Askow, A.T.; Paluska, S.A.; Dilger, A.C.; Holscher, H.D.; De Lisio, M.; et al. Higher protein intake during resistance training does not potentiate strength, but modulates gut microbiota, in middle-aged adults: A randomized control trial. Am. J. Physiol.-Endocrinol. Metab. 2021, 320, E900–E913. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, S.; Quigley, M.E.; Hopkins, M.J.; Newton, D.F.; Macfarlane, G.T. Polysaccharide degradation by human intestinal bacteria during growth under multi-substrate limiting conditions in a three-stage continuous culture system. FEMS Microbiol. Ecol. 1998, 26, 231–243. [Google Scholar] [CrossRef]
- Scheiman, J.; Luber, J.M.; Chavkin, T.A.; MacDonald, T.; Tung, A.; Pham, L.-D.; Wibowo, M.C.; Wurth, R.C.; Punthambaker, S.; Tierney, B.T.; et al. Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nat. Med. 2019, 25, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Estaki, M.; Pither, J.; Baumeister, P.; Little, J.P.; Gill, S.K.; Ghosh, S.; Ahmadi-Vand, Z.; Marsden, K.R.; Gibson, D.L. Cardiorespiratory fitness as a predictor of intestinal microbial diversity and distinct metagenomic functions. Microbiome 2016, 4, 42. [Google Scholar] [CrossRef] [Green Version]
- Kulecka, M.; Fraczek, B.; Mikula, M.; Zeber-Lubecka, N.; Karczmarski, J.; Paziewska, A.; Ambrozkiewicz, F.; Jagusztyn-Krynicka, K.; Cieszczyk, P.; Ostrowski, J. The composition and richness of the gut microbiota differentiate the top Polish endurance athletes from sedentary controls. Gut Microbes 2020, 11, 1374–1384. [Google Scholar] [CrossRef]
- Morishima, S.; Aoi, W.; Kawamura, A.; Kawase, T.; Takagi, T.; Naito, Y.; Tsukahara, T.; Inoue, R. Intensive, prolonged exercise seemingly causes gut dysbiosis in female endurance runners. J. Clin. Biochem. Nutr. 2021, 68, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef]
- Petriz, B.A.; Castro, A.P.; Almeida, J.A.; Gomes, C.P.; Fernandes, G.R.; Kruger, R.H.; Pereira, R.W.; Franco, O.L. Exercise induction of gut microbiota modifications in obese, non-obese and hypertensive rats. BMC Genom. 2014, 15, 511. [Google Scholar] [CrossRef] [Green Version]
- Grosicki, G.J.; Durk, R.P.; Bagley, J.R. Rapid gut microbiome changes in a world-class ultramarathon runner. Physiol. Rep. 2019, 7, 7. [Google Scholar] [CrossRef] [Green Version]
- Allen, J.M.; Mailing, L.J.; Niemiro, G.M.; Moore, R.; Cook, M.D.; White, B.A.; Holscher, H.D.; Woods, J.A. Exercise Alters Gut Microbiota Composition and Function in Lean and Obese Humans. Med. Sci. Sports Exerc. 2018, 50, 747–757. [Google Scholar] [CrossRef]
- Munukka, E.; Rintala, A.; Toivonen, R.; Nylund, M.; Yang, B.; Takanen, A.; Hanninen, A.; Vuopio, J.; Huovinen, P.; Jalkanen, S.; et al. Faecalibacterium prausnitzii treatment improves hepatic health and reduces adipose tissue inflammation in high-fat fed mice. ISME J. 2017, 11, 1667–1679. [Google Scholar] [CrossRef] [Green Version]
- Taniguchi, H.; Tanisawa, K.; Sun, X.M.; Kubo, T.; Hoshino, Y.; Hosokawa, M.; Takeyama, H.; Higuchi, M. Effects of short-term endurance exercise on gut microbiota in elderly men. Physiol. Rep. 2018, 6, 16. [Google Scholar] [CrossRef]
- Kern, T.; Blond, M.B.; Hansen, T.H.; Rosenkilde, M.; Quist, J.S.; Gram, A.S.; Ekstrom, C.T.; Hansen, T.; Stallknecht, B. Structured exercise alters the gut microbiota in humans with overweight and obesity-A randomized controlled trial. Int. J. Obes. 2020, 44, 125–135. [Google Scholar] [CrossRef]
- Hampton-Marcell, J.T.; Eshoo, T.W.; Cook, M.D.; Gilbert, J.A.; Horswill, C.A.; Poretsky, R. Comparative Analysis of Gut Microbiota Following Changes in Training Volume Among Swimmers. Int. J. Sports Med. 2020, 41, 292–299. [Google Scholar] [CrossRef]
- Wang, R.; Cai, Y.; Li, J.; Yau, S.Y.; Lu, W.; Stubbs, B.; Su, K.P.; Xu, G.; So, K.F.; Lin, K.; et al. Effects of aerobic exercise on gut microbiota in adolescents with subthreshold mood syndromes and healthy adolescents: A 12-week, randomized controlled trial. J. Affect. Disord. 2021, 293, 363–372. [Google Scholar] [CrossRef]
- Moitinho-Silva, L.; Wegener, M.; May, S.; Schrinner, F.; Akhtar, A.; Boysen, T.J.; Schaeffer, E.; Hansen, C.; Schmidt, T.; Ruhlemann, M.C.; et al. Short-term physical exercise impacts on the human holobiont obtained by a randomised intervention study. BMC Microbiol. 2021, 21, 14. [Google Scholar] [CrossRef] [PubMed]
- Cronin, O.; Barton, W.; Skuse, P.; Penney, N.C.; Garcia-Perez, I.; Murphy, E.F.; Woods, T.; Nugent, H.; Fanning, A.; Melgar, S.; et al. A Prospective Metagenomic and Metabolomic Analysis of the Impact of Exercise and/or Whey Protein Supplementation on the Gut Microbiome of Sedentary Adults. mSystems 2018, 3, e00044-18. [Google Scholar] [CrossRef] [Green Version]
- Cronin, O.; Barton, W.; Moran, C.; Sheehan, D.; Whiston, R.; Nugent, H.; McCarthy, Y.; Molloy, C.B.; O’Sullivan, O.; Cotter, P.D.; et al. Moderate-intensity aerobic and resistance exercise is safe and favorably influences body composition in patients with quiescent Inflammatory Bowel Disease: A randomized controlled cross-over trial. BMC Gastroenterol. 2019, 19, 29. [Google Scholar] [CrossRef] [PubMed]
- Pasini, E.; Corsetti, G.; Assanelli, D.; Testa, C.; Romano, C.; Dioguardi, F.S.; Aquilani, R. Effects of chronic exercise on gut microbiota and intestinal barrier in human with type 2 diabetes. Minerva Med. 2019, 110, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Huber, Y.; Pfirrmann, D.; Gebhardt, I.; Labenz, C.; Gehrke, N.; Straub, B.K.; Ruckes, C.; Bantel, H.; Belda, E.; Clement, K.; et al. Improvement of non-invasive markers of NAFLD from an individualised, web-based exercise program. Aliment. Pharm. Ther. 2019, 50, 930–939. [Google Scholar] [CrossRef]
- Morita, E.; Yokoyama, H.; Imai, D.; Takeda, R.; Ota, A.; Kawai, E.; Hisada, T.; Emoto, M.; Suzuki, Y.; Okazaki, K. Aerobic Exercise Training with Brisk Walking Increases Intestinal Bacteroides in Healthy Elderly Women. Nutrients 2019, 11, 868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, F.; Wen, X.; Yang, M.; Lai, H.Y.; Momma, H.; Cheng, L.; Sun, X.M.; Nagatomi, R.; Huang, C. Effect of an 8-week Exercise Training on Gut Microbiota in Physically Inactive Older Women. Int. J. Sports Med. 2021, 42, 610–623. [Google Scholar] [PubMed]
- Quiroga, R.; Nistal, E.; Estebanez, B.; Porras, D.; Juarez-Fernandez, M.; Martinez-Florez, S.; Garcia-Mediavilla, M.V.; de Paz, J.A.; Gonzalez-Gallego, J.; Sanchez-Campos, S.; et al. Exercise training modulates the gut microbiota profile and impairs inflammatory signaling pathways in obese children. Exp. Mol. Med. 2020, 52, 1048–1061. [Google Scholar] [CrossRef] [PubMed]
- Motiani, K.K.; Collado, M.C.; Eskelinen, J.J.; Virtanen, K.A.; Loyttyniemi, E.; Salminen, S.; Nuutila, P.; Kalliokoski, K.K.; Hannukainen, J.C. Exercise Training Modulates Gut Microbiota Profile and Improves Endotoxemia. Med. Sci. Sports Exerc. 2020, 52, 94–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rettedal, E.A.; Cree, J.M.E.; Adams, S.E.; MacRae, C.; Skidmore, P.M.L.; Cameron-Smith, D.; Gant, N.; Blenkiron, C.; Merry, T.L. Short-term high-intensity interval training exercise does not affect gut bacterial community diversity or composition of lean and overweight men. Exp. Physiol. 2020, 105, 1268–1279. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Y.; Ni, Y.Q.; Cheung, C.K.Y.; Lam, K.S.L.; Wang, Y.; Xia, Z.Y.; Ye, D.W.; Guo, J.; Tse, M.A.; et al. Gut Microbiome Fermentation Determines the Efficacy of Exercise for Diabetes Prevention. Cell Metab. 2020, 31, 77–91.e5. [Google Scholar] [CrossRef] [PubMed]
- Karl, J.P.; Margolis, L.M.; Madslien, E.H.; Murphy, N.E.; Castellani, J.W.; Gundersen, Y.; Hoke, A.V.; Levangie, M.W.; Kumar, R.; Chakraborty, N.; et al. Changes in intestinal microbiota composition and metabolism coincide with increased intestinal permeability in young adults under prolonged physiological stress. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 312, G559–G571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keohane, D.M.; Woods, T.; O’Connor, P.; Underwood, S.; Cronin, O.; Whiston, R.; O’Sullivan, O.; Cotter, P.; Shanahan, F.; Molloy, M.G.M. Four men in a boat: Ultra-endurance exercise alters the gut microbiome. J. Sci. Med. Sport 2019, 22, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Quevrain, E.; Maubert, M.A.; Michon, C.; Chain, F.; Marquant, R.; Tailhades, J.; Miquel, S.; Carlier, L.; Bermudez-Humaran, L.G.; Pigneur, B.; et al. Identification of an anti-inflammatory protein from Faecalibacterium prausnitzii, a commensal bacterium deficient in Crohn’s disease. Gut 2016, 65, 415–425. [Google Scholar] [CrossRef] [Green Version]
- Rossi, O.; van Berkel, L.A.; Chain, F.; Tanweer Khan, M.; Taverne, N.; Sokol, H.; Duncan, S.H.; Flint, H.J.; Harmsen, H.J.; Langella, P.; et al. Faecalibacterium prausnitzii A2-165 has a high capacity to induce IL-10 in human and murine dendritic cells and modulates T cell responses. Sci. Rep. 2016, 6, 18507. [Google Scholar] [CrossRef] [Green Version]
- Sokol, H.; Pigneur, B.; Watterlot, L.; Lakhdari, O.; Bermudez-Humaran, L.G.; Gratadoux, J.J.; Blugeon, S.; Bridonneau, C.; Furet, J.P.; Corthier, G.; et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc. Natl. Acad. Sci. USA 2008, 105, 16731–16736. [Google Scholar] [CrossRef] [Green Version]
- Munukka, E.; Ahtiainen, J.P.; Puigbó, P.; Jalkanen, S.; Pahkala, K.; Keskitalo, A.; Kujala, U.M.; Pietilä, S.; Hollmén, M.; Elo, L.; et al. Six-Week Endurance Exercise Alters Gut Metagenome that is not Reflected in Systemic Metabolism in Over-weight Women. Front. Microbiol. 2018, 9, 2323. [Google Scholar] [CrossRef]
- Cerqueira, E.; Marinho, D.A.; Neiva, H.P.; Lourenco, O. Inflammatory Effects of High and Moderate Intensity Exercise-A Systematic Review. Front. Physiol. 2020, 10, 14. [Google Scholar] [CrossRef]
- Shin, N.-R.; Whon, T.W.; Bae, J.-W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015, 33, 496–503. [Google Scholar] [CrossRef]
- Fujisawa, T.; Mulligan, K.; Wada, L.; Schumacher, L.; Riby, J.; Kretchmer, N. The effect of exercise on fructose absorption. Am. J. Clin. Nutr. 1993, 58, 75–79. [Google Scholar] [CrossRef]
- Krajmalnik-Brown, R.; Ilhan, Z.E.; Kang, D.W.; DiBaise, J.K. Effects of gut microbes on nutrient absorption and energy regulation. Nutr. Clin. Pract. 2012, 27, 201–214. [Google Scholar] [CrossRef] [Green Version]
- De Schryver, A.M.; Keulemans, Y.C.; Peters, H.P.; Akkermans, L.M.; Smout, A.J.; De Vries, W.R.; van Berge-Henegouwen, G.P. Effects of regular physical activity on defecation pattern in middle-aged patients complaining of chronic constipation. Scand. J. Gastroenterol. 2005, 40, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Obata, Y.; Castaño, Á.; Boeing, S.; Bon-Frauches, A.C.; Fung, C.; Fallesen, T.; de Agüero, M.G.; Yilmaz, B.; Lopes, R.; Huseynova, A.; et al. Neuronal programming by microbiota regulates intestinal physiology. Nature 2020, 578, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Schenk, A.; Joisten, N.; Walzik, D.; Koliamitra, C.; Schoser, D.; Bloch, W.; Zimmer, P. Acute exercise impacts AhR and PD-1 levels of CD8(+) T-cells-Exploratory results from a randomized cross-over trial comparing endurance versus resistance exercise. Eur. J. Appl. Physiol. 2021, 121, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Phillips, B.E.; Williams, J.P.; Gustafsson, T.; Bouchard, C.; Rankinen, T.; Knudsen, S.; Smith, K.; Timmons, J.A.; Atherton, P.J. Molecular Networks of Human Muscle Adaptation to Exercise and Age. PLoS Genet. 2013, 9, e1003389. [Google Scholar] [CrossRef]
- Marinelli, L.; Martin-Gallausiaux, C.; Bourhis, J.-M.; Béguet-Crespel, F.; Blottière, H.M.; Lapaque, N. Identification of the novel role of butyrate as AhR ligand in human intestinal epithelial cells. Sci. Rep. 2019, 9, 643. [Google Scholar] [CrossRef]
- Karlund, A.; Gomez-Gallego, C.; Turpeinen, A.M.; Palo-Oja, O.M.; El-Nezami, H.; Kolehmainen, M. Protein Supplements and Their Relation with Nutrition, Microbiota Composition and Health: Is More Protein Always Better for Sportspeople? Nutrients 2019, 11, 829. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Shi, Y.; Wiklund, P.; Tan, X.; Wu, N.; Zhang, X.; Tikkanen, O.; Zhang, C.; Munukka, E.; Cheng, S. The Association between Cardiorespiratory Fitness and Gut Microbiota Composition in Premenopausal Women. Nutrients 2017, 9, 792. [Google Scholar] [CrossRef]
- Guevara-Cruz, M.; Flores-López, A.G.; Aguilar-López, M.; Sánchez-Tapia, M.; Medina-Vera, I.; Díaz, D.; Tovar, A.R.; Torres, N. Improvement of Lipoprotein Profile and Metabolic Endotoxemia by a Lifestyle Intervention That Modifies the Gut Microbiota in Subjects with Metabolic Syndrome. J. Am. Heart Assoc. 2019, 8, e012401. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Nakayama, J. Development of the gut microbiota in infancy and its impact on health in later life. Allergol. Int. 2017, 66, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Petersen, L.M.; Bautista, E.J.; Nguyen, H.; Hanson, B.M.; Chen, L.; Lek, S.H.; Sodergren, E.; Weinstock, G.M. Community characteristics of the gut microbiomes of competitive cyclists. Microbiome 2017, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Antushevich, H. Interplays between inflammasomes and viruses, bacteria (pathogenic and probiotic), yeasts and parasites. Immunol. Lett. 2020, 228, 1–14. [Google Scholar] [CrossRef]
- Castaner, O.; Goday, A.; Park, Y.M.; Lee, S.H.; Magkos, F.; Shiow, S.T.E.; Schroder, H. The Gut Microbiome Profile in Obesity: A Systematic Review. Int. J. Endocrinol. 2018, 2018, 4095789. [Google Scholar] [CrossRef]
- Tuovinen, E.; Keto, J.; Nikkila, J.; Matto, J.; Lahteenmaki, K. Cytokine response of human mononuclear cells induced by intestinal Clostridium species. Anaerobe 2013, 19, 70–76. [Google Scholar] [CrossRef]
- Bouchard, C.; Rankinen, T. Individual differences in response to regular physical activity. Med. Sci. Sports Exerc. 2001, 33, S446–S451, discussion S452–S453. [Google Scholar] [CrossRef] [Green Version]
- Tang, Q.; Jin, G.; Wang, G.; Liu, T.; Liu, X.; Wang, B.; Cao, H. Current Sampling Methods for Gut Microbiota: A Call for More Precise Devices. Front. Cell. Infect. Microbiol. 2020, 10, 151. [Google Scholar] [CrossRef]
- Panek, M.; Čipčić Paljetak, H.; Barešić, A.; Perić, M.; Matijašić, M.; Lojkić, I.; Vranešić Bender, D.; Krznarić, Ž.; Verbanac, D. Methodology challenges in studying human gut microbiota—Effects of collection, storage, DNA extraction and next generation sequencing technologies. Sci. Rep. 2018, 8, 5143. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Zhang, Z.; Hu, B.; Huang, W.; Yuan, C.; Zou, L. Response of Gut Microbiota to Metabolite Changes Induced by Endurance Exercise. Front. Microbiol. 2018, 9, 765. [Google Scholar] [CrossRef] [PubMed]
- Clavel, T.; Desmarchelier, C.; Haller, D.; Gerard, P.; Rohn, S.; Lepage, P.; Daniel, H. Intestinal microbiota in metabolic diseases: From bacterial community structure and functions to species of pathophysiological relevance. Gut Microbes 2014, 5, 544–551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, S.K.; Cook, D.; Meyer, J.; Vernon, S.D.; Le, T.; Clevidence, D.; Robertson, C.E.; Schrodi, S.J.; Yale, S.; Frank, D.N. Changes in Gut and Plasma Microbiome following Exercise Challenge in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS). PLoS ONE 2015, 10, e0145453. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Berk, M.; Galecki, P.; Walder, K.; Maes, M. The Neuro-Immune Pathophysiology of Central and Peripheral Fatigue in Systemic Immune-Inflammatory and Neuro-Immune Diseases. Mol. Neurobiol. 2016, 53, 1195–1219. [Google Scholar] [CrossRef]
- Lundgren-Kownacki, K.; Dahl, M.; Gao, C.; Jakobsson, K.; Linninge, C.; Song, D.; Kuklane, K. Exploring how a traditional diluted yoghurt drink may mitigate heat strain during medium-intensity intermittent work: A multidisciplinary study of occupational heat strain. Ind. Health 2018, 56, 106–121. [Google Scholar] [CrossRef]
- Lee, M.C.; Hsu, Y.J.; Chuang, H.L.; Hsieh, P.S.; Ho, H.H.; Chen, W.L.; Chiu, Y.S.; Huang, C.C. In Vivo Ergogenic Properties of the Bifidobacterium longum OLP-01 Isolated from a Weightlifting Gold Medalist. Nutrients 2019, 11, 2003. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.C.; Hsu, Y.J.; Ho, H.H.; Hsieh, S.H.; Kuo, Y.W.; Sung, H.C.; Huang, C.C. Lactobacillus salivarius Subspecies salicinius SA-03 is a New Probiotic Capable of Enhancing Exercise Performance and Decreasing Fatigue. Microorganisms 2020, 8, 545. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.L.; Hsu, Y.J.; Ho, H.H.; Chang, Y.C.; Kuo, Y.W.; Yeh, Y.T.; Tsai, S.Y.; Chen, C.W.; Chen, J.F.; Huang, C.C.; et al. Bifidobacterium longum subsp. longum OLP-01 Supplemention during Endurance Running Training Improves Exercise Performance in Middle- and Long-Distance Runners: A Double-Blind Controlled Trial. Nutrients 2020, 12, 1972. [Google Scholar] [CrossRef]
- Blutt, S.E.; Conner, M.E. The gastrointestinal frontier: IgA and viruses. Front. Immunol. 2013, 4, 402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jager, R.; Mohr, A.E.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Moussa, A.; Townsend, J.R.; Lamprecht, M.; West, N.P.; Black, K.; et al. International Society of Sports Nutrition Position Stand: Probiotics. J. Int. Soc. Sports Nutr. 2019, 16, 62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calero, C.D.Q.; Rincon, E.O.; Marqueta, P.M. Probiotics, prebiotics and synbiotics: Useful for athletes and active individuals? A systematic review. Benef. Microbes 2020, 11, 135–149. [Google Scholar] [CrossRef] [PubMed]

| Training Mode | Specification and Duration of Training | Study Population | n of Subjects | Outcome | Reference in the List of References |
|---|---|---|---|---|---|
| ENDURANCE TRAINING | |||||
| Aerobic endurance training, increasing in duration (min) and from 60 to 75% of VO2max | 3x per wk, 40–60 min for 6 wks, either ergometer cycling or treadmill running; post-training washout period for 6 wks | lean and obese, physically inactive women and men | n = 14 obese (of which n = 11 female), n = 18 lean (of which n = 9 female) | All subjects VO2max ↑, Fat % ↓ In lean Bacteroides ↓ Faecalibacterium ↑, In obese Bacteroides ↑, Faecalibacterium ↓; SCFA ↑ in feces of lean following exercise. Effects of exercise were mostly reversed during the washout. Diet was followed up and subjects were asked to maintain it; Results of diet follow-up not reported | Allen et al., 2018, [42] |
| Aerobic endurance training | ergometer cycling 3x per wk, for 6 wks, additional control period included for study subjects | overweighed and physically inactive women | n = 17 (served as own controls) | VO2max ↑, Android fat ↓, Proteobacteria ↓, Akkermansia ↑ Food records were collected throughout, diet remained mainly unaffected during exercise intervention | Munukka et al., 2018, [43] |
| Aerobic endurance training; cross-over | ergometer cycling 3x per wk, starting 30 min at 60% of VO2max level, gradually increasing in duration and intensity, for 5 wks | >60 yrs old, healthy Japanese men | n = 31 having both training and control periods | Clostridium difficile ↓, Oscillospira ↑ and VO2max ↑ and HDL ↑, hepatic fat ↓ during exercise Of the metagenomic functions of GM, genetic information processing and metabolism of nucleotides ↑ Diet followed up with questionnaires and food records, changes in diet but they were similar during control and exercise periods | Taniguchi et al., 2018, [44] |
| Aerobic endurance-type, at different intensity levels | 5x per wk, for 6 months; BIKE = commuting by bike, MOD, moderate exercise (50% of VO2 peak); VIG, vigorous exercise (70% of VO2peak); CON, continued habitual sedentary living | overweight or obese, inactive 20–45 yrs old women and men | BIKE, n = 19; MOD, n = 31; VIG, n = 24; CON, n = 14 | Physical performance ↑ and Fat % ↓ in all exercise groups, Exercise ineffective on individual GM abundances VIG: GM α-diversity (Shannon index) ↑ after 3 months MOD: Functional capacity of GM ↑ after 3 moths Food diaries were collected, and changes occurred in exercising groups (intake of fat) | Kern et al., 2020, [45] |
| Endurance training (decreasing training load) | 2 wk swimming 32.4 ± 4.8 km/wk, then 2 wk 19.6 ± 8.2 km/wk, then 2 wk 11.3 ± 8.1 km/wk | 18–24 yrs old, collegiate swimmers | n = 13 | GM α-diversity ↓ as well as abundance of Coprococcus and Faecalibacterium ↓ along the training volume, no changes in changes in weight, fat mass or fat-free mass | Hampton-Marcell et al., 2020 [46] |
| Moderate-intensity aerobic training | 4x per wk, running (at level 50–70% of max. heart rate) 30 min for 12 wks | 12–14 yrs old, healthy or having subthreshold mood symptoms | n = 49 mood syndrome and n = 142 healthy of which n = 96 were training | Exercise was ineffective No information about the diet | Wang et al., 2021, [47] |
| COMBINED TRAINING or STUDIES WITH VARIOUS TRAINING MODES | |||||
| Endurance/Strength/Elite athletes | Endurance: running 4x per wk >30 min (of which 1 supervised); Strength: at gym 4x per wk (of which 2 supervised). Training loads were ↑, total duration 6 wks | Inactive 20–45 yrs old, BMI 20–35 kg/m2, healthy women and men + elite athletes | n = 42 men and women → n = 13 endurance, n = 12 strength, n = 11 ctrl and additional group of elite athletes (n = 13) | Different types of exercise induced some moderate health-related effects, but no systematic effects on GM Diet changed in strength group, but other groups remained unchanged. Diet was accounted in PCA-models. | Moitinho-Silva et al., 2021, [48] |
| Combined resistance and aerobic training, increasing load (with or without whey supplement) | 3x per wk, for 8 wks, each session included moderate aerobic training (18–32 min) + progressive, machine-based repetitive resistance exercises (starting ~70% of one-repetition max. level) | overweight and physically inactive women and men, ~35 yrs old | n = 25 (having exercise training only, we did not include whey groups) | VO2max ↑, Fat % ↓, Lean mass ↑, No significant effect on the GM α-diversity or metabolic pathways In exercise group diet remained unchanged | Cronin et al., 2018, [49] |
| Combined resistance and aerobic training, increasing load | 3x per wk for 8 wks; session included moderate aerobic training (18–32 min) + progressive, machine-based repetitive resistance exercises (starting ~70% of one-repetition max. level) | overweight, ~25 yrs old inactive males and females diagnosed with inflammatory bowel disease (IBD) | n = 13 exercise group, n = 7 control group | In exercise group: Fat % ↓, Lean mass ↑, α-diversity of Archaea species ↑, no effects on IBD activity scores, mood or inflammation No significant effect on GM α-diversity or metabolic pathways No information about diet | Cronin et al., 2019, [50] |
| Combined strength/endurance/stretching training | 3x per wk, 90 min per session for 6 months | type 2 diabetic males | n = 30 | VO2max ↑, weight and fat-% ↓, glycemia ↑ Intestinal mycetes overgrowth ↓, zonulin (intestinal leakage) ↓, systemic inflammation ↓ No information about diet | Pasini et al., 2019, [51] |
| Combined strength and endurance training | 3–5x per wk, for 8 wks, 10 free-weight or rubber band strength exercises and individualized endurance training (first walking/jogging then interval training) | non-alcoholic fatty liver disease patients, who completed >70% of training, age 18–70, BMI 18.5–45 kg/m2 | n = 41 | VO2max ↑, Fat % and weight ↓, hepatic illness scores ↑, inflammation ↓ Metagenomic richness ↑, Bacteroidetes and Euryarchaeota ↑, Actinobacteria ↓ Diet was not controlled/followed up | Huber et al., 2019, [52] |
| Trunk muscle strength or aerobic training | 1 h per day for 12 wks; Aerobic = brisk walking (≥3 METs); strength = trunk muscle training (free-weight gymnastics) | healthy >60 yrs old women | n = 14 strength, n = 17 aerobic training | Both training modes: physical performance ↑, strength and elasticity ↑ Aerobic training: Bacteroides ↑, Clostridium subcluster XIVa ↓ Strength training: Clostridium cluster IX ↑ Diet was followed up with questionnaires and no differences during the study between groups | Morita et al., 2019, [53] |
| Combined aerobic and resistance training | 4x per wk, ~60 min per session for 8 wks, gymnastics (= aerobic) and rubber band (= resistance) training with increase in training load | previously inactive women, ≥60 yrs old | n = 6 sedentary controls, n = 6 training | Firmicutes, Phascolarctobacterium, Mitsuokella ↑ after training, no effect on alpha diversity of GM, exercise improved physical performance No information about diet | Zhong et al., 2021 [54] |
| Combined strength and aerobic exercise | 2x per wk, for 12 wks, bicycle ergometer, strength exercises | pediatric obese patients and non-obese, age 7–12 | n = 39 obese → n = 25 exercise, n = 14 control, n = 14 non-obese | Plasma glucose ↓, Dynamic strength ↑ Proteobacteria and Gammaproteobacteria ↓ Fecal branched chain amino acids, formate, alanine and glucose ↓ | Quiroga et al., 2020, [55] |
| HIGH-INTENSITY TRAINING | |||||
| Increasing sprint interval training or moderate-intensity aerobic training (both used cycling) | Intervals: 4–6x 30 work, 4 min rest; 3x per wk for 2 wks Endurance: 3x per wk for 2 wks, 40–60 min | obese and sedentary men and women who were diabetic or prediabetic, age 40–55 yrs | n = 16 (n = 9 type 2 diabetic, n = 17 prediabetic) | Interval: VO2max ↑, Fat %↓, Both training modes: Firmicutes/Bacteroidetes ↓, Clostridium ↓, Blautia ↓ Moderate intensity: Faecalibacterium ↑ No information about diet, it was asked to maintain unchanged | Motiani et al., 2020, [56] |
| High intensity interval ergometer training, short-term | 3x per wk for 3 wks using ergometer; 8–12 bouts repeated at VO2max level, 1 bout = 60 s work + 75 s rest | lean and obese men, sedentary or less than 3 hrs endurance activity weekly | n = 14 lean, n = 15 obese | Insulin sensitivity and cardiovascular fitness ↑, no changes in GM, some microbes associated with insulin sensitivity among obese Diet differed between the groups before, but was not followed-up during intervention | Rettedal et al., 2020, [57] |
| High intensity interval training: combined strength and endurance with increasing load | 3x per wk for 12 wks, supervised 70 min. sessions containing high-intensity running/biking sessions ~80–95% of VO2max or HRmax level, high-intensity resistance and calisthenics exercises (e.g., kettle ball, squats), warm-up, cooldown and stretching | non-smoking, overweight/obese men, prediabetic | n = 39 → n = 19 sedentary controls, n = 14 exercise responsive and n = 6 non-responsive, validation study with n = 30 obese men | Following HIIT, insulin sensitivity ↑ in responders but not in non-responders, GM α- or β-diversity unaffected, abundances of species among Firmicutes, Bacteroidetes, Proteobacteria changed in response to exercise, e.g., Alistipes putredinis ↓, Bactroides xylanisolvens ↓, Lachnospiraceae bacterium ↑ in responders By machine-learning algorithms GM signatures were shown to predict exercise responses in a validation study Diet was monitored with questionnaires and groups did not differ | Liu et al., 2020, [58] |
| STRENOUS TRAINING | |||||
| Strenous, high-intensity exercise | 4 days, 51-km cross-country ski march | healthy soldiers | n = 73 | Gut permeability ↑, GM diversity ↑ Verrucomicrobia, Tenericutes, Spirochaetes, Lentisphera, Fusobacteria and Firmicutes ↑ Euarchaeota ↓ Fecal metabolism of phenylalanine, tryptophan and tyrosine ↓ Fecal metabolism of carbohydrates, fatty acids and secondary bile acids ↓ | Karl et al., 2017 [59] |
| Continuous rowing | 33 days, 5000 km transatlantic rowing, each athletic rowed 395 hrs | healthy, elite male athletes (~26 yrs) | n = 4 | VO2max unchanged, GM α-diversity ↑, butyrate producing GM (Roseburia hominis, Subdoligranulum) ↑, Bacteroides finegoldii ↓; in functional metabolic pathways of GM gene products, synthesis of L-isoleucine, L-lycine ↑, S-adenosyl-L-methionine ↑, long chain fatty acids ↑, fatty acid elongation and glycolysis ↑ Diet was followed up, and macronutrient intake remained constant, but diet changed during rowing | Keohane et al., 2019, [60] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lensu, S.; Pekkala, S. Gut Microbiota, Microbial Metabolites and Human Physical Performance. Metabolites 2021, 11, 716. https://doi.org/10.3390/metabo11110716
Lensu S, Pekkala S. Gut Microbiota, Microbial Metabolites and Human Physical Performance. Metabolites. 2021; 11(11):716. https://doi.org/10.3390/metabo11110716
Chicago/Turabian StyleLensu, Sanna, and Satu Pekkala. 2021. "Gut Microbiota, Microbial Metabolites and Human Physical Performance" Metabolites 11, no. 11: 716. https://doi.org/10.3390/metabo11110716






