Comparison of Brain Natriuretic Peptide Levels to Simultaneously Obtained Right Heart Hemodynamics in Stable Outpatients with Pulmonary Arterial Hypertension
Abstract
:1. Introduction
2. Materials and Methods
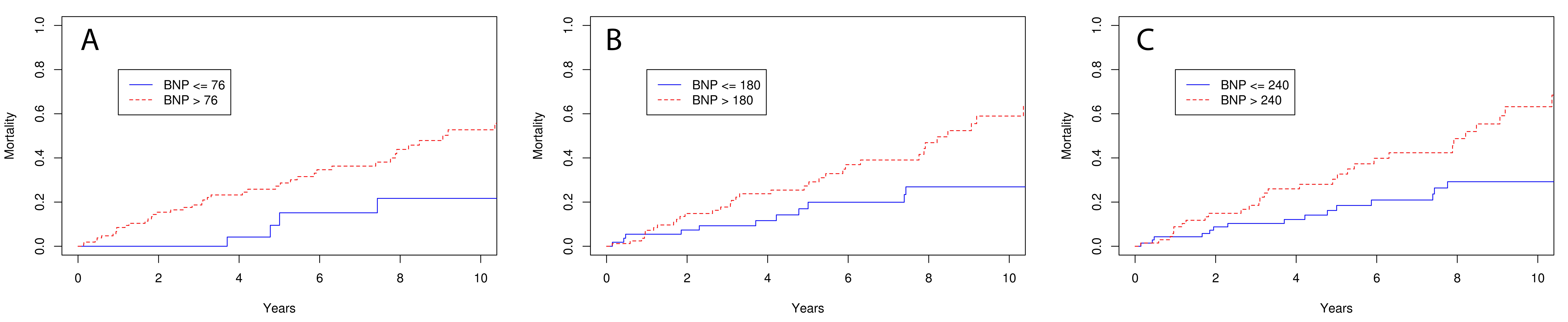
3. Results
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Austin, C.; Burger, C.; Kane, G.; Safford, R.; Blackshear, J.; Ung, R.; Shapiro, B. High-risk echocardiographic features predict mortality in pulmonary arterial hypertension. Am. Heart J. 2017, 189, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Benza, R.L.; Gomberg-Maitland, M.; Miller, D.P.; Frost, A.; Frantz, R.P.; Foreman, A.J.; McGoon, M.D. The REVEAL Registry Risk Score Calculator in Patients Newly Diagnosed With Pulmonary Arterial Hypertension. Chest 2012, 141, 354–362. [Google Scholar] [CrossRef] [PubMed]
- Benza, R.L.; Miller, D.P.; Barst, R.J.; Badesch, D.B.; Frost, A.E.; McGoon, M.D. An Evaluation of Long-term Survival from Time of Diagnosis in Pulmonary Arterial Hypertension from the REVEAL Registry. Chest 2012, 142, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Benza, R.L.; Miller, D.P.; Gomberg-Maitland, M.; Frantz, R.P.; Foreman, A.J.; Coffey, C.S.; McGoon, M.D. Predicting survival in pulmonary arterial hypertension: Insights from the Registry to Evaluate Early and Long-Term Pulmonary Arterial Hypertension Disease Management (REVEAL). Circulation 2010, 122, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Galie, N.; Humbert, M.; Vachiery, J.L.; Gibbs, S.; Lang, I.; Torbicki, A.; Hoeper, M. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur. Respir. J. 2015, 46, 903–975. [Google Scholar] [CrossRef] [PubMed]
- Galiè, N.; Manes, A.; Negro, L.; Palazzini, M.; Bacchi-Reggiani, M.L.; Branzi, A. A meta-analysis of randomized controlled trials in pulmonary arterial hypertension. Eur. Heart J. 2009, 30, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Hunt, S.A.; Abraham, W.T.; Chin, M.H.; Feldman, A.M.; Francis, G.S.; Ganiats, T.G.; Yancy, C.W. 2009 Focused Update Incorporated Into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults. J. Am. Coll. Cardiol. 2009, 53, e1–e90. [Google Scholar] [CrossRef] [PubMed]
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: Guidelines for the six-minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef]
- Leuchte, H.H.; Holzapfel, M.; Baumgartner, R.A.; Ding, I.; Neurohr, C.; Vogeser, M.; Behr, J. Clinical significance of brain natriuretic peptide in primary pulmonary hypertension. J. Am. Coll. Cardiol. 2004, 43, 764–770. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, V.V.; Archer, S.L.; Badesch, D.B.; Barst, R.J.; Farber, H.W.; Lindner, J.R.; Varga, J. ACCF/AHA 2009 Expert Consensus Document on Pulmonary Hypertension. A Report of the American College of Cardiology Foundation Task Force on Expert Consensus Documents and the American Heart Association Developed in Collaboration With the American College of Chest Physicians; American Thoracic Society, Inc.; and the Pulmonary Hypertension Association. J. Am. Coll. Cardiol. 2009, 53, 1573–1619. [Google Scholar] [CrossRef] [PubMed]
- Minai, O.A.; Gudavalli, R.; Mummadi, S.; Liu, X.; McCarthy, K.; Dweik, R.A. Heart rate recovery predicts clinical worsening in patients with pulmonary arterial hypertension. Am. J. Respir. Crit. Care Med. 2012, 185, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Nagaya, N.; Satoh, T.; Kyotani, S.; Sakamaki, F.; Fujita, M.; Miyatake, K. Clinical correlates and prognostic significance of six-minute walk test in patients with primary pulmonary hypertension. Comparison with cardiopulmonary exercise testing. Am. J. Respir. Crit. Care Med. 2000, 161, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Nagaya, N.; Nishikimi, T.; Okano, Y.; Uematsu, M.; Satoh, T.; Kyotani, S.; Kangawa, K. Plasma Brain Natriuretic Peptide Levels Increase in Proportion to the Extent of Right Ventricular Dysfunction in Pulmonary Hypertension. J. Am. Coll. Cardiol. 1998, 31, 202–208. [Google Scholar] [CrossRef]
- Nagaya, N.; Nishikimi, T.; Uematsu, M.; Satoh, T.; Kyotani, S.; Sakamaki, F.; Kangawa, K. Plasma brain natriuretic peptide as a prognostic indicator in patients with primary pulmonary hypertension. Circulation 2000, 102, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Simonneau, G.; Gatzoulis, M.A.; Adatia, I.; Celermajer, D.; Denton, C.; Ghofrani, A.; Souza, R. Updated clinical classification of pulmonary hypertension. J. Am. Coll. Cardiol. 2013, 62 (Suppl. S25), D34–D41. [Google Scholar] [CrossRef] [PubMed]
- Wiese, S.; Breyer, T.; Dragu, A.; Wakili, R.; Burkard, T.; Schmidt-Schweda, S.; Holubarsch, C.J. Gene expression of brain natriuretic peptide in isolated atrial and ventricular human myocardium: Influence of angiotensin II and diastolic fiber length. Circulation 2000, 102, 3074–3079. [Google Scholar] [CrossRef] [PubMed]
- Song, J.W.; Song, J.K.; Kim, D.S. Echocardiography and brain natriuretic peptide as prognostic indicators in idiopathic pulmonary fibrosis. Respir. Med. 2009, 103, 180–186. [Google Scholar] [CrossRef] [PubMed]
| Variables | All Patients (n = 138) | BNP < 240 (n = 68) | BNP ≥ 240 (n = 70) | p-Value for BNP Groups |
|---|---|---|---|---|
| Age (years) | 59.2 ± 13.6 | 59.4 ± 13.3 | 59 ± 13.9 | 0.869 |
| Sex female) | 104 (75.4%) | 47 (69.1%) | 57 (81.4%) | 0.115 |
| Race (n) | 0.513 | |||
| Caucasian | 114 (82.6%) | 56 (82.3%) | 58 (82.9%) | |
| African American | 15 (10.9%) | 9 (13.2%) | 6 (8.6%) | |
| Latino | 5 (3.6%) | 1 (1.5%) | 4 (5.7%) | |
| Asian | 2 (1.4%) | 1 (1.5%) | 1 (1.4%) | |
| Native American | 1 (0.7%) | 0 (0%) | 1 (1.4% | |
| Caribbean | 1 (0.7%) | 1 (1.5%) | 0 (0%) | |
| Body mass index (kg/m2) | 27.9 ± 8.4 | 29.2 ± 10.4 | 26.8 ± 5.7 | 0.098 |
| Creatinine clearance (mL/min) | 84.3 ± 41.8 | 90.8 ± 48.9 | 78 ± 32.7 | 0.071 |
| WHO functional class (n) | 0.007 | |||
| I | 15 (10.97%) | 10 (14.7%) | 5 (7.1%) | |
| II | 25 (18.1%) | 18 (26.5%) | 7 (10%) | |
| III | 70 (50.7%) | 32 (47. 1%) | 38 (54.3%) | |
| IV | 28 (20.3%) | 8 (11.8%) | 20 (28.67%) | |
| Brain natriuretic peptide (BNP) (pg/mL) | 406 ± 443 | 99.4 ± 75.4 | 703.9 ± 448.9 | <0.0001 |
| Hemodynamic parameters | ||||
| RAP (mm Hg) | 9.9 ± 5.7 | 8.7 ± 5 | 11.1 ± 6.2 | 0.021 |
| mPAP (mm Hg) | 47.3 ± 14.7 | 43.1 ± 14.8 | 51.3 ± 13.4 | 0.001 |
| PCWP (mm Hg) | 12.3 ± 5.4 | 13.1 ± 3.8 | 11.1 ± 2.3 | 0.138 |
| CO (L/min) | 4.7 ± 1.9 | 5.2 ± 2.1 | 4.2 ± 1.4 | 0.001 |
| CI (L/min/m2) | 2.5 ± 0.9 | 2.7 ± 0.9 | 2.3 ± 0.7 | 0.004 |
| PVR (dynes) | 681.8 ± 426 | 541.8 ± 361 | 826.4 ± 442.2 | 0.0001 |
| Right heart size by TTE (grade) | ||||
| RAE | 3.2 ± 2.5 | 2.8 ± 2.5 | 3.6 ± 2.4 | 0.051 |
| RVE | 3.4 ± 2.3 | 2.8 ± 2.2 | 4 ± 2 | 0.003 |
| RVD | 3 ± 2.3 | 2.5 ± 2.3 | 3.5 ± 2.2 | 0.013 |
| Six-minute walk test | ||||
| Distance walked (m) | 319.7 ± 119.5 | 388.8 ± 95.9 | 249.6 ± 98.8 | <0.0001 |
| Heart rate recovery (bpm) | 30.1 ± 13.1 | 37.2 ± 12.2 | 23.3 ± 10 | <0.0001 |
| Heart rate recovery (%) | 28.4 ± 10.3 | 33.9 ± 9 | 23.2 ± 8 | <0.0001 |
| Medication use (n) | 0.075 | |||
| Diuretics | 49 (35.5%) | 17 (25%) | 32 (45.7%) | |
| Calcium channel blockers | 25 (18.1%) | 10 (14.7%) | 15 (21.4%) | |
| Oral PAH medication | 46 (33.3%) | 22 (32.4%) | 24 (34.3%) | |
| Inhaled prostacyclin | 12 (8.7%) | 6 (8.8%) | 6 (8.6%) | |
| Intravenous prostacyclin | 11 (7.9%) | 6 (8.8%) | 5 (7.1%) |
| Variable | r | p-Value |
|---|---|---|
| Hemodynamics | ||
| RAP | 0.202 | 0.021 |
| mPAP | 0.253 | 0.003 |
| CO | −0.325 | 0.0002 |
| CI | −0.277 | 0.002 |
| PCWP | −0.207 | 0.76 |
| PVR | 0.305 | 0.0006 |
| Right heart size by TTE | ||
| RAE | 0.181 | 0.04 |
| RVE | 0.186 | 0.03 |
| RVD | 0.151 | 0.08 |
| Six-minute walk test | ||
| Heart rate recovery: absolute | −0.654 | <0.0001 |
| Heart rate recovery: percentage | −0.700 | <0.0001 |
| Distance walked | −0.752 | <0.0001 |
| WHO-FC | ||
| Functional class | 0.257 | 0.002 |
| Variable | Hazard Ratio | 95% CI | p-Value |
|---|---|---|---|
| Brain natriuretic peptide | |||
| lg BNP: continuous | 1.698 | 1.301–2.215 | <0.0001 |
| BNP > 76 pg/mL | 3.674 | 1.317–10.250 | 0.013 |
| BNP >180 pg/mL | 2.540 | 1.287–5.012 | 0.007 |
| BNP >240 pg/mL | 2.401 | 1.305–4.417 | 0.005 |
| Hemodynamics | |||
| RAP | 1.055 | 1.003–1.110 | 0.039 |
| mPAP | 1.013 | 0.992–1.034 | 0.227 |
| Cardiac index | 0.750 | 0.522–1.076 | 0.118 |
| Right heart size by TTE | |||
| RVE | 0.923 | 0.811–1.050 | 0.224 |
| RVD | 0.953 | 0.838–1.084 | 0.466 |
| RAE | 0.857 | 0.754–0.974 | 0.018 |
| WHO-FC | |||
| Functional class | 1.425 | 0.984–2.064 | 0.061 |
| Six minute walk test | |||
| Heart rate recovery–absolute | 0.936 | 0.909–0.964 | <0.0001 |
| Distance walked | 0.992 | 0.990–0.995 | <0.0001 |
| Variable | Hazard Ratio | 95% CI | p-Value |
|---|---|---|---|
| lg BNP | 1.585 | 1.199–2.095 | 0.001 |
| RAP | 1.047 | 0.984–1.113 | 0.15 |
| mPAP | 0.988 | 0.961–1.016 | 0.39 |
| Cardiac index | 0.854 | 0.579–1.258 | 0.42 |
| RVE | 0.879 | 0.658–1.173 | 0.379 |
| RVD | 1.207 | 0.916–1.59 | 0.181 |
| RAE | 0.802 | 0.671–0.96 | 0.016 |
| WHO-FC | 1.303 | 0.895–1.898 | 0.168 |
| Heart rate recovery: absolute | 0.95 | 0.910–0.992 | 0.02 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helgeson, S.A.; Imam, J.S.; Moss, J.E.; Hodge, D.O.; Burger, C.D. Comparison of Brain Natriuretic Peptide Levels to Simultaneously Obtained Right Heart Hemodynamics in Stable Outpatients with Pulmonary Arterial Hypertension. Diseases 2018, 6, 33. https://doi.org/10.3390/diseases6020033
Helgeson SA, Imam JS, Moss JE, Hodge DO, Burger CD. Comparison of Brain Natriuretic Peptide Levels to Simultaneously Obtained Right Heart Hemodynamics in Stable Outpatients with Pulmonary Arterial Hypertension. Diseases. 2018; 6(2):33. https://doi.org/10.3390/diseases6020033
Chicago/Turabian StyleHelgeson, Scott A., J. Saadi Imam, John E. Moss, David O. Hodge, and Charles D. Burger. 2018. "Comparison of Brain Natriuretic Peptide Levels to Simultaneously Obtained Right Heart Hemodynamics in Stable Outpatients with Pulmonary Arterial Hypertension" Diseases 6, no. 2: 33. https://doi.org/10.3390/diseases6020033





