The Role of the Nrf2/ARE Antioxidant System in Preventing Cardiovascular Diseases
Abstract
:1. Introduction
2. The Keap1-Nrf2-ARE Signaling System
2.1. Overview of Nrf2 Signaling
2.2. Effects on Mitochondria
2.3. AMPK Signaling
2.4. Notch Signaling
3. The Role of the Keap1-Nrf2-ARE Signaling System in Cardiovascular Diseases
3.1. Overview
3.2. Inflammation Is Misunderstood in the Past
3.3. Inflammation Is Important in CVD
3.4. The Keap1-Nrf2-ARE System Can Sense Shear Stress
3.5. The Nrf2 System Protects Mitochondria
3.6. Nrf2 Also Activates HO-1
3.7. Nrf2 Also Supports the ER
3.8. The Keap1-Nrf2-ARE Signaling System Is Important in Maintaining the Renewal of Cardiomyocytes
3.9. Nrf2 in Multi-Drug Resistant Cancer
4. Conclusions
Author Contributions
Conflicts of Interest
Abbreviations
| AMP | Adenosine monophosphate |
| AMPK | Adenosine monophosphate kinase |
| ARE | Antioxidant response element |
| ATP | Adenosine triphosphate |
| βTrCP | β-transducin repeat containing protein |
| CAPE | Caffeic acid phenethyl ester |
| Cul3 | Cullin-3 based ligase |
| CVD | Cardiovascular disease |
| DJ-1 | Protein deglycase |
| ECs | endothelial cells |
| EGCG | Epigallocatechin-3-gallate |
| ER | Endoplasmic reticulum |
| GCL | Glutamate cysteine ligase |
| GR | Glutathione reductase |
| Grx | Glutaredoxin |
| GSK-3 | Glycogen synthase kinase-3 |
| GST | Glutathione S-transferase |
| HDACs | histone deacetylases |
| HO-1 | Heme oxygenase-1 |
| Keap1 | Kelch-like enoyl-CoA hydratase-associated protein 1 |
| Maf | Musculo-aponeurotic fibrosarcoma protein |
| MAPK | mitogen-activated protein kinase |
| miRNA | microRNA |
| NAD(P)H | Nicotinamide adenine dinucleotide reduced form, with or without a phosphate |
| NO | nitric oxide |
| NQO1 | NAD(P)H:quinone oxidoreductase-1 |
| Nrf2 | Nuclear erythroid-2 like factor-2 |
| Nrf2+/+ | The wild type of Nrf2, in which both copies of the gene coding for Nrf2 are present |
| Nrf2−/− | The genotype in which neither of the genes coding for Nrf2 are present |
| OS | oscillatory disturbed shear |
| p53 | A tumor suppressor protein with a molecular weight of 53 kiloDaltons |
| PERK | Protein kinase RNA-like endoplasmic reticulum kinase |
| PI3K | Phosphatidylinositol (3,4,5)-trisphosphate |
| PKR | Protein kinase R |
| PtdIns(3,4,5)-P3 | Phosphatidylinositol (3,4,5)-trisphosphate |
| RNA | Ribonucleic acid |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| SRC | Sarcoma |
| SRXN | Sulfiredoxin |
| TCDD | 2,3,7,8-tetrachlorodibenzodioxin |
| TXN | Thioredoxin |
| TXNRD | Thioredoxin reductase |
| µM | micromoles per liter |
| UPR | Unfolded protein response |
| VCAM-1 | Vascular adhesion molecule-1 |
References
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of aging. Proc. Natl. Acad. Sci. 1993, 90, 7915–7922. [Google Scholar] [CrossRef] [PubMed]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, J. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Llorent-Martínez, E.J.; Fernández-de Córdova, M.L.; Ortega-Barrales, P.; Ruiz-Medina, A. Characterization and comparison of the chemical composition of exotic superfoods. Microchem. J. 2013, 110, 444–451. [Google Scholar] [CrossRef]
- Kerimi, A.; Williamson, G. At the interface of antioxidant signalling and cellular function: Key polyphenol effects. Mol. Nutr. Food Res. 2016, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Silva-Palacios, A.; Königsberg, M.; Zazueta, C. Nrf2 signaling and redox homeostasis in the aging heart: A potential target to prevent cardiovascular diseases? Ageing Res. Rev. 2016, 26, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Kensler, T.W.; Wakabayashi, N.; Biswal, B. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Howden, R. Nrf2 and cardiovascular defense. Oxid. Med. Cell. Longev. 2013, 2013, 104308. [Google Scholar] [CrossRef] [PubMed]
- Hybertson, B.M.; Gao, B. Role of the Nrf2 signaling system in health and disease. Clin. Genet. 2014, 86, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Cominacini, L.; Mozzini, C.; Garbin, U.; Pasini, A.; Stranieri, C.; Solani, E.; Vallerio, P.; Tinelli, I.A.; Pasini, A.F. Endoplasmic reticulum stress and Nrf2 signaling in cardiovascular diseases. Free Radic. Biol. Med. 2015, 88, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Stewart, A.J.; Mullen, M.; Burns, J.; Lean, M.E.J.; Brighenti, F.; Crozier, A. HPLC-MSn analysis of phenolic compounds in green and black tea. J. Agric. Food Chem. 2004, 52, 2807–2815. [Google Scholar] [CrossRef] [PubMed]
- Scapagnini, G.; Sonya, V.; Nader, A.G.; Calogero, C.; Zella, D.; Fabio, G. Modulation of Nrf2/ARE pathway by food polyphenols: A nutritional neuroprotective strategy for cognitive and neurodegenerative disorders. Mol. Neurobiol. 2011, 44, 192–201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kode, A.; Rajendrasozhan, S.; Cato, S.; Yang, S.-R.; Megson, I.-L.; Rahman, I. Resveratrol induces glutathione synthesis by activation of Nrf2 and protects against cigarette smoke-mediated oxidative stress in human lung epithelial cells. Lung Cell. Mol. Pathol. 2008, 294, L478–L488. [Google Scholar] [CrossRef] [PubMed]
- Ajit, D.; Simonyi, A.; Li, R.; Chen, Z.; Hannink, M.; Fritsche, K.; Mossine, V.V.; Smith, R.E.; Dobbbs, T.K.; Luo, R.; et al. Phytochemicals and botanical extracts regulate NF-kB and Nrf2/ARE. Neurochem. Int. 2016, 97, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.-T.; Wu, C.H.; Ho, C.Y.; Yen, G.C. Catechin protects against ketoprofen-induced oxidative damage of the gastric mucosa by up-regulating Nrf2 in vitro and in vivo. J. Nutr. Biochem. 2013, 24, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Kim, I.-S.; More, S.V.; Kim, B.-W.; Choi, D.-K. Natural product-derived pharmacological modulators of Nrf2/ARE pathway for chronic diseases. Nat. Prod. Rep. 2014, 31, 109. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Ding, Y.; Zhang, B.; Zhou, K.Y.; Chen, M.C.; Wang, M.M.; Jia, Y.Y.; Song, Y.; Li, Y.W.; Wen, A.D. Dietary ellagic acid improves oxidant-induced endothelial dysfunction and atherosclerosis: Role of Nrf2 activation. Int. J. Cardiol. 2014, 175, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Castellano, J.M.; Guinda, A.; Delgado, T.; Rada, M.; Cayuela, J.A. Biochemical basis of the antidiabetic activity of oleanolic acid and related pentacyclic triterpenes. Diabetes 2013, 62, 1791–1799. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Lee, D.-S.; Kang, Y.; Yao, N.-Q.; An, R.-B.; Kim, Y.-C. Protective effect of ganodermanondiol isolated from the Lingzhi mushroom against tert-butyl hydroperoxide-induced hepatotoxicity through Nrf2-mediated antioxidant enzymes. Food Chem. Toxicol. 2013, 53, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.; Li, W.; Song, W.; Wang, Y.; Liang, W.; Li, K.; Tang, S.; Wang, Q.; Qiao, X.; Zhou, D.; et al. Bioactive constituents of Glycyrrhiza uralensis (licorice): Discovery of the effective components of a traditional herbal medicine. J. Nat. Prod. 2016, 79, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Boettler, U.; Volz, N.; Pahlke, G.; Teller, N.; Kotukza, C.; Somoza, V.; Syiebitz, H.; Bytof, G.; Lantz, I.; Lang, R.; et al. Coffees rich in chlorogenic acid or N-methylpyridinium induce chemopreventive phase II-enzymes via the Nrf2/ARE pathway in vitro and in vivo. Mol. Nutr. Food Res. 2011, 55, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.-Q.; Ding, J.; Zhang, L.; Liu, C.M. Protective effects of ursolic acid in an experimental model of liver fibrosis through Nrf2/ARE pathway. Clin. Res. Hepatol. Gastroenterol. 2015, 39, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Bayram, B.; Ozcelik, B.; Grimm, S.; Roeder, T. A diet rich in olive oil phenolics reduces oxidative stress in the heart of SAMP8 mice by induction of Nrf2-dependent gene expression. Rejuven. Res. 2012, 15, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Fetoni, A.R.; Paciello, F.; Rolesi, R.; Eramo, S.L.M.; Mancuso, C.; Troiani, D. Rosamarinic acid up-regulates the noise-activated Nrf2/HO-1 pathway and protects against noise-induced injury in the rat cochlea. Free Radic. Mol. Biol. Med. 2015, 85, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Varí, R.; D’Archivio, M.; Filesi, C.; Carotenuto, S.; Scazzocchio, B.; Santangelo, C.; Giovannini, C.; Masella, R. Protocatechuic acid induces antioxidant/detoxifying enzyme expression through JNK-mediated Nrf2 activation in murine macrophages. J. Nutr. Biochem. 2011, 22, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Kropat, C.; Mueller, D.; Boettler, U.; Zimmermann, K.; Heiss, E.H.; Dirsch, V.M.; Rogoll, D.; Melcher, R.; Richling, E.; Marko, D. Modulation of Nrf2-dependent gene transcription by bilberry anthocyanins. Mol. Nutr. Food Res. 2013, 57, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.T.; Yen, G.C. Induction of hepatic antioxidant enzymes by phenolic acids in rats is accompanied by increased levels of multidrug resistance associated protein 3 mRNA expression. J. Nutr. 2006, 136, 11–15. [Google Scholar] [PubMed]
- Ma, Z.; Hong, Q.; Wang, Y.; Liang, Q.; Tan, H.; Xiao, C.R.; Tang, X.L.; Shao, S.; Zhou, S.S.; Gao, Y. Ferulic acid induces heme oxygenase-1 via activation of NRK and Nrf2. Drug Discov. Ther. 2011, 5, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Zeraik, M.L.; Yariwake, J.H. Quantification of isoorientin and total flavonoids in Passiflora edulis fruit pulp by HPLC-UV/DAD. Microchem. J. 2010, 96, 86–91. [Google Scholar] [CrossRef]
- Kim, S.R.; Ha, Y.M.; Kim, Y.M.; Park, E.J.; Kim, J.W.; Park, S.W.; Kim, H.J.; Chung, H.T.; Chang, K.C. Ascorbic acid reduces HMGB1 secretion in lipopolysaccharide-activated RAW 264.7 cells and improves survival rate in septic mice by activation of Nrf2/HO-1 signals. Biochem. Pharmacol. 2015, 95, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Velmurugan, K.; Alam, J.; McCord, J.M.; Pugazhenthi, S. Synergistic induction of heme oxygenase-1 by the components of the antioxidant supplement Protandim. Free Radic. Biol. Med. 2009, 46, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Strong, R.; Miller, R.A.; Antebi, A.; Astle, C.A.; Bogue, M.; Denzel, M.S.; Flurkey, K.; Hamilton, K.L.; Lamming, D.W.; Javors, M.A.; et al. Longer lifespan in male mice treated with a weakly-estrogenic agonist, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer. Aging Cell 2016, 15, 872–884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-González, C.; Ciudad, C.J.; Izquierdo-Pulido, M.; Noé, V. Urolithin A causes p21 up-regulation in prostate cancer cells. Eur. J. Nutr. 2016, 55, 1099–1112. [Google Scholar] [CrossRef] [PubMed]
- Grossman, R.; Ram, Z. The dark side of Nrf2. World Neurol. 2013, 80, 284–285. [Google Scholar] [CrossRef] [PubMed]
- Kansanen, E.; Kuosmanen, S.M.; Leinonen, H.; Levonen, A.-L. The Keap1-Nrf2 pathway: Mechanisms of activation and dysregulation in cancer. Redox Biol. 2013, 1, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Cebula, M.; Schmidt, E.E.; Arnér, E.S.J. TrxR1 as a potent regulator of the Nrf2-Keap1 response system. Antioxid. Redox Signal. 2015, 23, 823–853. [Google Scholar] [CrossRef] [PubMed]
- Dodson, M.; Redmann, M.; Rajasekaran, N.S.; Darley-Usmar, V.; Zhang, J. KEAP1–NRF2 signalling and autophagy in protection against oxidative and reductive proteotoxicity. Biochem. J. 2015, 469, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Yamamoto, M. Molecular basis of the Keap1–Nrf2 system. Free Radic. Biol. Med. 2015, 88, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Qu, C.; Niu, T.; Zang, H.; Qi, L.; Lyu, L.; Wang, X.; Nagarkatti, N.; Nagarkatti, P.; Janicki, J.S.; et al. Nrf2-mediated cardiac maladaptive remodeling and dysfunction in a setting of autophagy insufficiency. Hypertension 2016, 67, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Gañán-Gómez, I.; Wei, Y.; Yang, H.; Boyano-Adánez, M.C.; García-Manero, G. Oncogenic functions of the transcription factor Nrf2. Free Radic. Biol. Med. 2013, 65, 750–764. [Google Scholar] [CrossRef] [PubMed]
- Balligand, J.-L. Reducing damage through Nrf-2. Cardiovasc. Res. 2013, 100, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, M.; Rajasekaran, N.S. Reductive potential—A savior turns stressor in protein aggregation cardiomyopathy. Biochim. Biophys. Acta 2015, 1852, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Niture, S.K.; Khatri, R.; Jaiswa, A.K. Regulation of Nrf2—An update. Free Radic. Biol. Med. 2014, 66. [Google Scholar] [CrossRef]
- Mimura, J.; Itoh, K. Role of Nrf2 in the pathogenesis of atherosclerosis. Free Radic. Biol. Med. 2015, 88, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Motohashi, H. Roles of Nrf2 in cell proliferation and differentiation. Free Radic. Biol. Med. 2015, 88, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, W.; Su, Z.-Y.; Kong, A.-N.T. The complexity of the Nrf2 pathway: Beyond the antioxidant response. J. Nutr. Biochem. 2015, 26, 1401–1413. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.K.; Gupta, S.K.; Jacob, M.R.; Khan, S.I.; Ferreira, D. Antioxidant, antimalarial and antimicrobial activities of tannin-rich fractions, ellagitannins and phenolic acids from Punica granatum L. Planta Med. 2007, 53, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Rosillo, M.A.; Sánchez-Hidalgo, M.; Cárdeno, A.; Aparicio-Soto, M.; Sánchez-Fidalgo, S.; Villegas, I.; de la Lastra, C.A. Dietary supplementation of an ellagic acid-enriched pomegranate extract attenuates chronic colonic inflammation in rats. Pharmacol. Res. 2012, 66, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; González-Sarrías, A.; Yáñez-Gascón, M.J.; Selma, M.V.; Azorín-Ortuño, M.; Toti, S.; Tomás-Barberán, F.; Dolara, P.; Espín, J.C. Anti-inflammatory properties of a pomegranate extract and its metabolite urolithin-A in a colitis rat model and the effect of colon inflammation on phenolic metabolism. J. Nutr. Biochem. 2010, 21, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Kaser, A. Gut microbiome, obesity and metabolic dysfunction. J. Clin. Invest. 2011, 121, 2126–2132. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-M.; Hwang, S.-S.; Park, E.-J.; Bae, J.-W. Strict vegetarian diet improves the risk factors associated with metabolic diseases by modulating gut microbiota and reducing intestinal inflammation. Environ. Microbiol. Rep. 2013, 5, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Do Rosario, V.A.; Fernandes, R.; Trindade, E.B. Vegetarian diets and gut microbiota: Important shifts in markers of metabolism and cardiovascular disease. Nutr. Rev. 2016, 74, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.-L.; Zhu, J.-F.; Xu, X.-L.; Zhu, J.; Li, L.; Xi, M.-Y.; Jiang, Z.-Y.; Zhang, M.-Y.; Liu, F.; Bao, Q.-C.; et al. Discovery and modification of in vivo active Nrf2 activators with 1,2,4-oxadiazole core: Hits identification and structure-activity relationship study. J. Med. Chem. 2015, 58, 5419–5436. [Google Scholar] [CrossRef] [PubMed]
- Harder, B.; Jiang, T.; Tao, S.; de la Vega, M.R.; Tian, W.; Chapman, E.; Zhang, D.D. Molecular mechanisms of Nrf2 regulation and how these influence chemical modulation for disease intervention. Biochem. Soc. Trans. 2015, 43, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Ahuja, M.; Kaidery, N.A.; Yang, L.; Calingasasn, L.; Smirnova, N.; Gaisin, A.; Gaisina, I.N.; Gazaryan, I.; Hushpulian, D.M.; Kaddour-Djebbar, I.; et al. Distinct Nrf2 signaling mechanisms of fumaric acid esters and their role in neuroprotection against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced experimental Parkinson’s-like disease. J. Neurosci. 2016, 36, 6332–6351. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.S.; Patel, H.; Allaire, N.; Thai, A.; Cullen, P.; Ryan, S.; Lukashev, M.; Bista, P.; Huang, R.; Rhodes, K.J.; Scannevin, R.H. Pharmacodynamics of dimethyl fumarate are tissue specific and involve Nrf2-dependent and independent mechanisms. Antioxid. Redox Signal. 2016, 24, 1058–1071. [Google Scholar] [CrossRef] [PubMed]
- Schilze-Topphoff, U.; Varrin-Doyer, M.; Pekarek, K.; Spencer, C.M.; Shetty, A.; Sagan, S.A.; Cree, B.A.C.; Sobel, R.A.; Wipke, B.; Steinman, L.; et al. Dimethyl fumarate treatment induces adaptive and innate immune modulation independent of Nrf2. Proc. Natl. Acad. Sci. USA 2016, 113, 4777–4782. [Google Scholar] [CrossRef] [PubMed]
- Kastrati, I.; Siklos, M.I.; Calderon-Gierszal, E.L.; El-Shennawy, L.; Georgieva, G.; Thayer, E.N.; Thatcher, G.R.J.; Frasor, J. Dimethyl fumarate inhibits the nuclear factor NF κB pathway in breast cancer cells by covalent modification of p64 protein. J. Biol. Chem. 2016, 291, 3639–3647. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.C.; Hong, Q.; Wang, Y.G.; Tan, H.L.; Xiao, C.R.; Liang, Q.-D.; Zhang, B.-L.; Gao, Y. Ferulic acid protects human umbilical vein endothelial cells from radiation induced oxidative stress by phosphatidylinositol 3-kinase and extracellular signal-regulated kinase pathways. Biol. Pharm. Bull. 2010, 33, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Wang, H.; Fan, L.; Wu, X.; Xin, A.; Ren, H.; Wang, X.J. Luteolin inhibits Nrf2 leading to negative regulation of the Nrf2/ARE pathway and sensitization of human lung carcinoma A549 cells to therapeutic drugs. Free Radic. Biol. Med. 2011, 50, 1599–1609. [Google Scholar] [CrossRef] [PubMed]
- Kweon, M.H.; Adhami, V.M.; Lee, J.S.; Mukhtar, H. Constitutive overexpression of Nrf2-dependent heme oxygenase-1 in A549 cells contributes to resistance to apoptosis induced by epigallocatechin 3-gallate. J. Biol. Chem. 2006, 281, 33761–33772. [Google Scholar] [CrossRef] [PubMed]
- Tarumoto, T.; Nagai, T.; Ohmine, K.; Miyoshi, T.; Nakamura, M.; Kondo, T.; Mitsugi, K.; Nakano, S.; Muroi, K.; Komatsu, N.; et al. Ascorbic acid restores sensitivity to imatinib via suppression of Nrf2-dependent gene expression in the imatinib-resistant cell line. Exp. Hematol. 2004, 32, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Gao, A.M.; Ke, Z.P.; Wang, J.N.; Yang, J.Y.; Chen, S.Y.; Chen, H. Apigenin sensitizes doxorubicin-resistant hepatocellular carcinoma BEL-7402/ADM cells to doxorubicin via inhibiting PI3K/Akt/Nrf2 pathway. Carcinogen 2013, 34, 1806–1814. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Hayes, J.D.; Henderson, C.J.; Wolf, C.R. Identification of retinoic acid as an inhibitor of transcription factor Nrf2 through activation of retinoic acid receptor alpha. Proc. Natl. Acad. Sci. USA 2007, 104, 19589–19594. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Villeneuve, N.F.; Jiang, T.; Wu, T.; Lau, A.; Toppin, H.I.; Zhang, D.D. Brusatol enhances the efficacy of chemotherapy by inhibiting the Nrf2-mediated defense mechanism. Proc. Natl. Acad. Sci. USA 2011, 108, 1433–1438. [Google Scholar] [CrossRef] [PubMed]
- Arlt, A.; Sebens, S.; Krebs, S.; Geismann, C.; Grossmann, M.; Kruse, M.-L.; Schreiber, S.; Schäfer, H. Inhibition of the Nrf2 transcription factor by the alkaloid trigonelline renders pancreatic cancer cells more susceptible to apoptosis through decreased proteasomal gene expression and proteasome activity. Oncogene 2012, 32, 4825–4835. [Google Scholar] [CrossRef] [PubMed]
- Limonciel, A.; Jennings, P. A review of the evidence that ochratoxin A is an Nrf2 inhibitor: Implications for nephrotoxicity and renal carcinogenicity. Toxins 2014, 6, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-J.; Maliakal, P.; Chen, L.; Meng, X.; Bondoc, F.Y.; Prabhu, S.; Lambert, G.; Mohr, S.; Yang, C.S. Pharmacokinetics of tea catechins after ingestion of green tea and (−)-epigallocatechin-3-gallate by humans: Formation of different metabolites and individual variability. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1025–1032. [Google Scholar]
- Smith, A.; Giunta, B.; Bickford, P.C.; Fountain, M.; Tan, J.; Shytle, R.D. Nanolipidic particles improve the bioavailability and α-secretase inducing ability of epigallocatechin-3-gallate (EGCG) for the treatment of Alzheimer’s disease. Int. J. Pharmaceut. 2010, 389, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Kimura, Y.; Jiang, S.; Harada, K.; Yamashita, Y.; Ashida, H. Luteolin modulates expression of drug-metabolizing enzymes through the AhR and Nrf2 pathways in hepatic cells. Arch. Biochem. Biophys. 2014, 557, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.E.; Boesch-Saadatmandi, C.; Breckwoldt, D.; Schrader, C.; Schmelzer, C.; Döring, F.; Hashida, K.; Hori, O.; Matsugo, S.; Rimbach, G. Ascorbic acid partly antagonizes resveratrol mediated heme oxygenase-1 but not paraoxonase-1 induction in cultured hepatocytes—Role of the redox-regulated transcription factor Nrf2. BMC Complement. Altern. Med. 2011, 11, 8. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, J.; Tozer, T.N.; Rowland, M. Pharmacokinetic perspectives on megadoses of ascorbic acid. Am. J. Clin. Nutr. 1997, 66, 1165–1171. [Google Scholar] [PubMed]
- Duconge, J.; Miranda-Massari, J.R.; Gonzalez, M.J.; Jackson, J.A.; Warnock, W.; Riordan, N.H. Pharmacokinetics of Vitamin C: Insights into the oral and intravenous administration of ascorbate. P. R. Health Sci. J. 2008, 27, 7–19. [Google Scholar] [PubMed]
- Yoh, K.; Itoh, K.; Enomoto, A.; Hirayama, A.; Yamaguchi, N.; Kobayashi, M.; Morito, N.; Koyama, A.; Yamamoto, M.; Takahashi, S. Nrf2-deficient female mice develop lupus-like autoimmune nephritis. Kidney Int. 2001, 60, 1343–1353. [Google Scholar] [CrossRef] [PubMed]
- Tebay, L.E.; Robertson, H.; Durant, S.T.; Vitale, S.R.; Penning, T.M.; Dinkova-Kostova, A.T.; Hayes, J.T. Mechanisms of activation of the transcription factor Nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Mol. Biol. Med. 2015, 88, 108–146. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.I.; Kobayashi, A.; Wakabayashi, N.; Kim, S.G.; Yamamoto, M. Scaffolding of Keap1 to the actin cytoskeleton controls the function of Nrf2 as key regulator of cytoprotective phase 2 genes. Proc. Natl. Acad. Sci. USA 2004, 101, 2046–2051. [Google Scholar] [CrossRef] [PubMed]
- Chapple, S.J.; Siow, R.C.M.; Mann, G.E. Crosstalk between Nrf2 and the proteasome: Therapeutic potential of Nrf2 inducers in vascular disease and aging. Int. J. Biochem. Cell Biol. 2012, 44, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trend. Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef] [PubMed]
- Priestley, J.R.C.; Kautenburg, K.E.; Casati, M.C.; Endres, B.T.; Geurts, A.M.; Lombard, J.H. The NRF2 knockout rat: A new animal model to study endothelial dysfunction, oxidant stress, and microvascular rarefaction. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H478–H487. [Google Scholar] [CrossRef] [PubMed]
- Brigelius-Flohé, R.; Flohé, L. Basic principles and emerging concepts redox transcription factors. Antioxid. Redox Signal. 2011, 15, 2335–2381. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.E. Medicinal Chemistry—Fusion of Traditional and Western Medicine, 3rd ed.; Bentham Science: Sharjah, UAE, 2015; pp. 131–133. [Google Scholar]
- Kruiswijk, F.; Labuschagne, C.F.; Vousden, K.H. p53 in cell survival, death and metabolic health: A lifeguard with a license to kill. Nat. Rev. Mol. Cell Biol. 2015, 16, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Hou, W.; Song, X.; Yu, Y.; Huang, J.; Sun, X.; Kang, R.; Tang, D. Ferroptosis: Process and function. Cell Death Diff. 2016, 23, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.E. Medicinal Chemistry—Fusion of Traditional and Western Medicine, 2nd ed.; Bentham Science: Sharjah, UAE, 2014. [Google Scholar]
- Smith, R.E.; Tran, K.; Richards, K.M. Systems thinking for medicinal chemists. Jacobs J. Med. Chem. 2015, 1, 4. [Google Scholar]
- Dinkova-Kostova, A.T.; Abramov, A.Y. The emerging role of Nrf2 in mitochondrial function. Free Radic. Biol. Med. 2015, 88, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Kell, D.B. Towards a unifying, systems biology understanding of large-scale cellular death and destruction caused by poorly liganded iron: Parkinson’s, Huntington’s, Alzheimer’s, prions, bactericides, chemical toxicology and others as examples. Arch. Toxicol. 2010, 84, 825–889. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, K.; Baldinger, J.; Mayerhofer, B.; Atanasov, A.G.; Dirsch, V.M.; Heiss, E.H. Activated AMPK boosts the Nrf2/HO-1 signaling axis—A role for the unfolded protein response. Free Radic. Biol. Med. 2015, 88, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.Y.H.; Chan, S.H.H. Activation of endogenous antioxidants as a common therapeutic strategy against cancer, neurodegeneration and cardiovascular diseases: A lesson learnt from DJ-1. Pharmacol. Ther. 2015, 156, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, N.; Chartoumpekis, D.V.; Kensler, T.W. Crosstalk between Nrf2 and Notch signaling. Free Radic. Biol. Med. 2015, 88, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.N.; Wason, E.; Edrey, Y.H.; Kristan, D.M.; Nevo, E.; Buffenstein, R. Regulation of Nrf2 signaling and longevity in naturally long-lived rodents. Proc. Natl. Acad. Sci. USA 2015, 112, 3722–3777. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A. Structural and functional characterization of Nrf2 degradation by glycogen synthase kinase 3/β-TrCP. Free Radic. Biol. Med. 2015, 88, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Sies, H. Total antioxidant capacity: Appraisal of a concept. J. Nutr. 2007, 137, 1493–1495. [Google Scholar] [PubMed]
- Libby, P. Inflammation and cardiovascular disease mechanisms. Am. J. Clin. Nutr. 2006, 83, 456S–460S. [Google Scholar] [PubMed]
- McSweeney, S.R.; Warabi, E.; Siow, R.C.M. Nrf2 as an endothelial mechanosensitive transcription factor going with the flow. Hypertension 2016, 67, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Strom, J. A Critical Role of Nrf2 in Protecting Myocytes Against Oxidative Stress and Ischemic Injury. Ph.D. Thesis, University of Arizona, Tucson, AZ, USA, 2014. [Google Scholar]
- Piantadosi, C.A.; Carraway, M.S.; Babiker, A.; Suliman, H.B. Heme oxygenase-1 regulates cardiac mitochondrial biogenesis via Nrf2-mediated transcriptional control of nuclear respiratory factor-1. Circ. Res. 2008, 103, 1232–1240. [Google Scholar] [CrossRef] [PubMed]
- Strom, J.; Xu, B.; Tian, X.; Chen, Q.M. Nrf2 protects mitochondrial decay by oxidative stress. FASEB J. 2016, 30, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Baldelli, S.; Aquilano, K.; Ciriolo, M.R. Punctum on two different transcription factors regulated by PGC-1alpha: Nuclear factor erythroid-derived 2-like 2 and nuclear respiratory factor 2. Biochim. Biophys. Acta 2013, 1830, 4137–4146. [Google Scholar] [CrossRef] [PubMed]
- Diano, S.; Horvath, T.L. Mitochondrial uncoupling protein 2 (UCP2) in glucose and lipid metabolism. Trend. Mol. Med. 2012, 18, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Dhamrait, S.S.; Maubaret, C.; Pedersen-Bjergaard, U.; Brull, D.J.; Gohlke, P.; Payne, J.R.; World, M.; Thorsteinsson, B.; Humphries, S.E.; Montgomery, H.E. Mitochondrial uncoupling proteins regulate angiotensin-converting enzyme expression: Crosstalk between cellular and endocrine metabolic regulators suggested by RNA interference and genetic studies. Inside Cell 2016, 1, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Busiello, R.A.; Savarese, S.; Lombardi, A. Mitochondrial uncoupling proteins and energy metabolism. Front. Physiol. 2015, 6, 36. [Google Scholar] [CrossRef] [PubMed]
- Anedda, A.; López-Bernardo, E.; Acosta-Iborra, B.; Suleiman, M.S.; Landázuri, M.O.; Susana Cadenas, S. The transcription factor Nrf2 promotes survival by enhancing the expression of uncoupling protein 3 under conditions of oxidative stress. Free Radic. Biol. Med. 2013, 61, 395–407. [Google Scholar] [CrossRef] [PubMed]
- Brand, M.D.; Esteves, T.C. Physiological functions of the mitochondrial uncoupling proteins UCP2 and UCP3. Cell Metab. 2005, 2, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Sessions, A.O.; Engler, A.J. Mechanical regulation of cardiac aging in model systems. Circ. Res. 2016, 118, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.R.; Narasimhan, M.; Shanmugam, G.; Hong, J.; Devarajan, A.; Palaniappan, S.; Zhang, J.; Halade, G.V.; Usmar, V.M.; Hoidal, J.R.; et al. Abrogation of Nrf2 impairs antioxidant signaling and promotes atrial hypertrophy in response to high-intensity exercise stress. Transl. Med. 2016, 14. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, Z.; Cai, L. Diabetic cardiomyopathy and its prevention by Nrf2: Current status. Diabetes Metab. J. 2014, 38, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Kannan, S.; Muthusamy, V.R.; Whitehead, K.J.; Wang, L.; Gomes, A.V.; Litwin, S.E.; Kensler, T.W.; Abel, E.D.; Hoidal, J.R.; Rajasekaran, N.S. Nrf2 deficiency prevents reductive stress-induced hypertrophic cardiomyopathy. Cardiovasc. Res. 2013, 100, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Sag, C.M.; Santos, C.X.C.; Shah, A.M. Redox regulation of cardiac hypertrophy. J. Mol. Cell. Cardiol. 2014, 73, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liu, X.; Long, M.; Huang, Y.; Zhang, L.; Zhang, R.; Zheng, Y.; Liao, X.; Yuren Wang, Y.; Liao, Q.; et al. NRF2 activation by antioxidant antidiabetic agents accelerates tumor metastasis. Sci. Transl. Med. 2016, 8, 334ra51. [Google Scholar] [CrossRef] [PubMed]
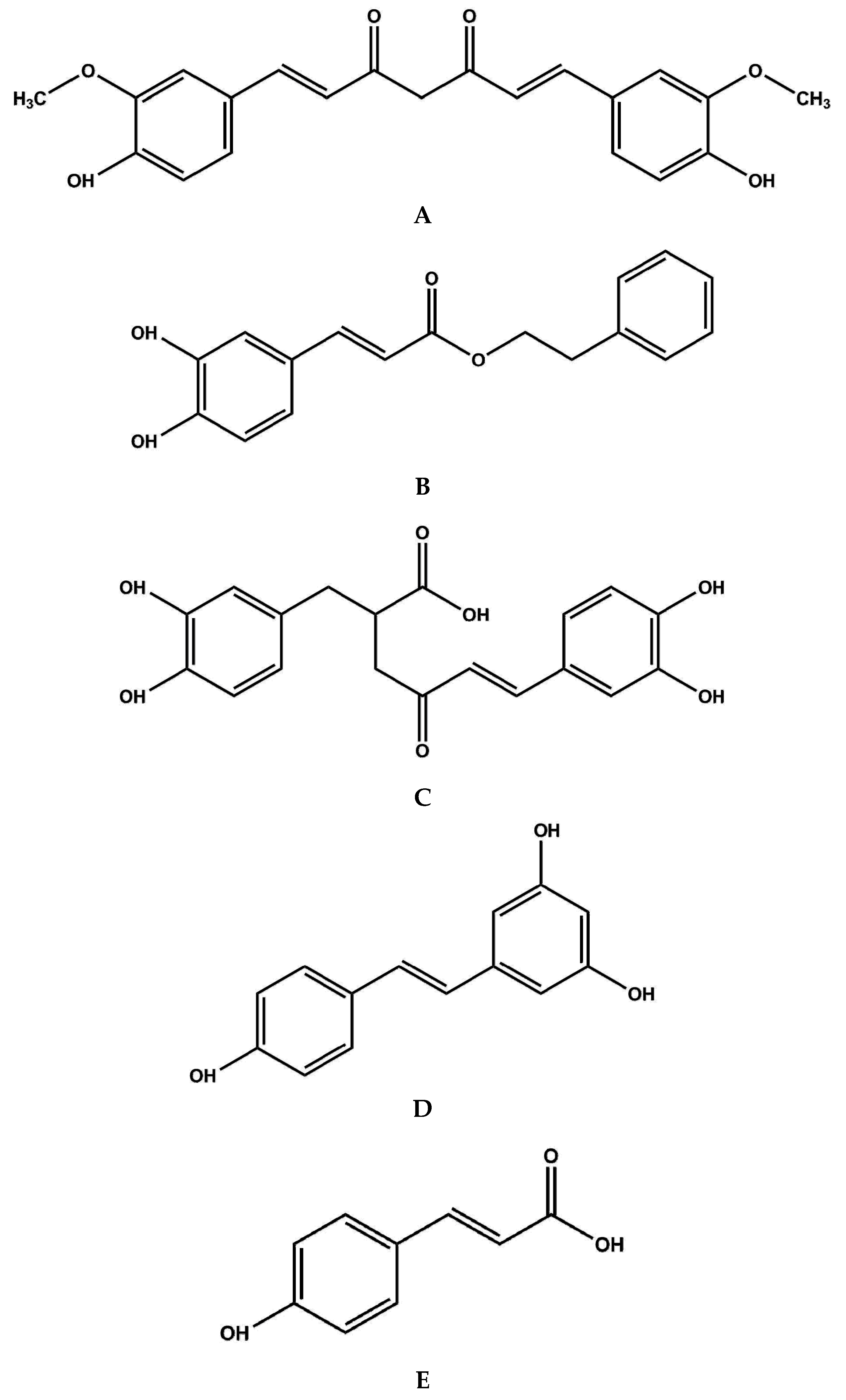
| Compound | Dietary Sources a | References |
|---|---|---|
| Epigallocatechin-3-gallate (EGCG) | Green tea | [11] |
| Curcumin | Turmeric | [12] |
| Carnosol | Rosemary | [12] |
| Zerumbone | Ginger | [12] |
| Caffeic acid phenethyl ester (CAPE) | Honeybee propolis and many plants | [12] |
| Ethyl ferulate | Many plants, including eggplant | [12] |
| Sulphorane | Broccoli and other cruciferous vegetables | [12] |
| Resveratrol | Red wine, Itadori tea | [13] |
| Quercetin | Many foods, including capers | [14] |
| Cyanidin and cyanidin-3-O-glucoside | Many types of fruits and berries | [10] |
| Catechin | Many foods, including cocoa and tea | [15] |
| Epicatechin | Many foods, including cocoa and tea | [16] |
| Kaempferol | Many foods, including green tea and berries | [16] |
| Naringenin-7-O-glucoside | Many foods, including tomatoes | [16] |
| Procyanidin B2 | Many foods, including cocoa and grape juice | [16] |
| Genistein | Soybeans | [16] |
| Butein and phloretin | Fruits, vegetables, nuts, tea, coffee, red wine | [16] |
| Xanthohumol | Comon hop (Humulus lupulus) | [16] |
| Luteolin | Many foods, including celery and broccoli | [17] |
| Tangeretin | Tangerines and other citrus fruits | [17] |
| Ellagic acid | Pomegranates | [18] |
| Oleanolic acid | Many plants, including olive leaves | [19] |
| Ganodermanondiol | Lingzhi mushrooms | [20] |
| Echinatin | Licorice | [21] |
| Chlorogenic acid | Green coffee extract, coffee | [22] |
| N-methylpyridinium | Coffee | [22] |
| Ursolic acid | Apple peels and many other foods and spices | [23] |
| Hydroxytyrosol | Olive oil and olive leaves | [24] |
| Rosmarinic acid | Rosemary | [25] |
| Protocatechuic acid | Raspberries and many other foods | [26] |
| Phloroglucinol aldehyde | Metabolite of anthocyanins | [27] |
| p-coumaric acid | Many foods, including peanut and tomatoes | [28] |
| Ferulic acid | Many herbs used in traditional Chinese medicine | [29] |
| Isoorientin | Açaí, passion fruit, Sasa borealis | [16,30] |
| Ascorbic acid | Vitamin C, citrus fruits | [31] |
| Compound | Dietary Sources a | References |
|---|---|---|
| Ferulic acid | Many plant seeds and cell walls, including Ferula foetida | [30,55] |
| Luteolin | Many foods, including celery and broccoli | [30,56] |
| EGCG | Green tea and green tea extract | [30,57] |
| Ascorbic acid | Vitamin C and citrus fruits | [30,58] |
| Apigenin | Fruits, vegetables | [30,59] |
| All-trans-retinoic acid | From β-carotene | [30,60] |
| Brusatol | Brucea javanica | [30,61] |
| Trigonelline | Fenugreek seeds | [30,62] |
| Ochratoxin A | Aspergillus, Penicillum | [30,63] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, R.E.; Tran, K.; Smith, C.C.; McDonald, M.; Shejwalkar, P.; Hara, K. The Role of the Nrf2/ARE Antioxidant System in Preventing Cardiovascular Diseases. Diseases 2016, 4, 34. https://doi.org/10.3390/diseases4040034
Smith RE, Tran K, Smith CC, McDonald M, Shejwalkar P, Hara K. The Role of the Nrf2/ARE Antioxidant System in Preventing Cardiovascular Diseases. Diseases. 2016; 4(4):34. https://doi.org/10.3390/diseases4040034
Chicago/Turabian StyleSmith, Robert E., Kevin Tran, Cynthia C. Smith, Miranda McDonald, Pushkar Shejwalkar, and Kenji Hara. 2016. "The Role of the Nrf2/ARE Antioxidant System in Preventing Cardiovascular Diseases" Diseases 4, no. 4: 34. https://doi.org/10.3390/diseases4040034






