Factors Associated with Increased Morbidity and Mortality of Obese and Overweight COVID-19 Patients
Abstract
:1. Introduction
2. Methods
2.1. Protocol
2.2. Search Strategy
2.3. Data Collection
2.4. Quality Evaluation
3. Obesogenic Mechanism
4. Genetic Factors Involved in Obesity
4.1. Adipogenesis
4.2. Hypoxia
5. COVID-19
6. Obesity and COVID-19
6.1. Obesity as a Risk Factor for Infection with COVID-19
6.2. Obesity Mechanisms in COVID-19 Infections
7. Quarantine and COVID-19
8. Key Gaps in Knowledge to Direct Future Research
9. Obesity and Stress
10. Gut Microbiome and Obesity
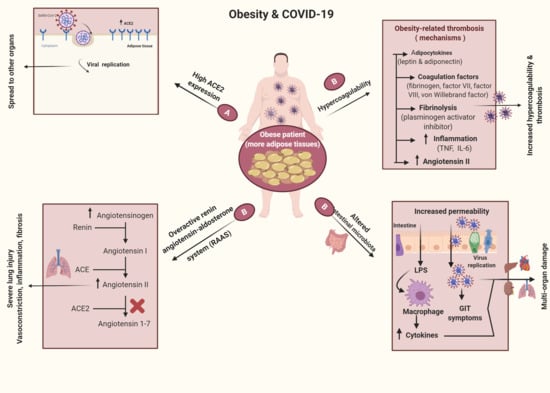
11. Obesity and Thrombosis in COVID-19 Infections
12. Obesity and Immunity
12.1. Obesity and Antiviral Immunity
12.2. Obesity and Atherosclerosis
12.3. Obesity and Psoriasis
13. Obesity and Vaccines
14. Obesity and Pathogens
14.1. Obesity and Respiratory Tract Infections
14.2. Obesity and Viral Infection
14.3. Obesity and Adenovirus
14.4. Obesity and Influenza Virus
14.4.1. Influenza Virus in Obese Mice
14.4.2. Influenza Virus in Human Cells
14.5. Obesity and Spreading of Pathogens
15. Obesity Control
16. Obesity and Exercise
17. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Key Facts; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Aguilar, M.; Bhuket, T.; Torres, S.; Liu, B.; Wong, R.J. Prevalence of the metabolic syndrome in the United States, 2003–2012. JAMA 2015, 313, 1973–1974. [Google Scholar] [CrossRef]
- Abbas, A.M.; Fathy, S.K.; Fawzy, A.T.; Salem, A.S.; Shawky, M.S. The mutual effects of COVID-19 and obesity. Obes. Med. 2020, 100250. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, F.M.; Weschenfelder, J.; Sander, C.; Minkwitz, J.; Thormann, J.; Chittka, T.; Mergl, R.; Kirkby, K.C.; Faßhauer, M.; Stumvoll, M. Inflammatory cytokines in general and central obesity and modulating effects of physical activity. PLoS ONE 2015, 10, e0121971. [Google Scholar] [CrossRef] [PubMed]
- Caër, C.; Rouault, C.; Le Roy, T.; Poitou, C.; Aron-Wisnewsky, J.; Torcivia, A.; Bichet, J.-C.; Clément, K.; Guerre-Millo, M.; André, S. Immune cell-derived cytokines contribute to obesity-related inflammation, fibrogenesis and metabolic deregulation in human adipose tissue. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Honce, R.; Schultz-Cherry, S. Impact of obesity on influenza A virus pathogenesis, immune response, and evolution. Front. Immunol. 2019, 10, 1071. [Google Scholar] [CrossRef]
- Wang, Z.; Nakayama, T. Inflammation, a link between obesity and cardiovascular disease. Med. Inflamm. 2010, 2010. [Google Scholar] [CrossRef] [Green Version]
- Richard, C.; Wadowski, M.; Goruk, S.; Cameron, L.; Sharma, A.M.; Field, C.J. Individuals with obesity and type 2 diabetes have additional immune dysfunction compared with obese individuals who are metabolically healthy. BMJ Open Diabetes Res. Care 2017, 5. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kim, J.-W.; Osborne, O.; Sasik, R.; Schenk, S.; Chen, A.; Chung, H.; Murphy, A.; Watkins, S.M.; Quehenberger, O. Increased adipocyte O2 consumption triggers HIF-1α, causing inflammation and insulin resistance in obesity. Cell 2014, 157, 1339–1352. [Google Scholar] [CrossRef] [Green Version]
- Huh, J.Y.; Park, Y.J.; Ham, M.; Kim, J.B. Crosstalk between adipocytes and immune cells in adipose tissue inflammation and metabolic dysregulation in obesity. Mol. Cells 2014, 37, 365. [Google Scholar] [CrossRef]
- Andersen, C.J.; Murphy, K.E.; Fernandez, M.L. Impact of obesity and metabolic syndrome on immunity. Adv. Nutr. 2016, 7, 66–75. [Google Scholar] [CrossRef] [Green Version]
- Luzi, L.; Radaelli, M.G. Influenza and obesity: Its odd relationship and the lessons for COVID-19 pandemic. Acta Diabetol. 2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Afolabi, H.A.; bin Zakariya, Z.; Shokri, A.B.A.; Hasim, M.N.B.M.; Vinayak, R.; Afolabi-Owolabi, O.T.; Elesho, R.F. The relationship between obesity and other medical comorbidities. Obes. Med. 2020, 17, 100164. [Google Scholar] [CrossRef]
- Sattar, N.; McInnes, I.B.; McMurray, J.J. Obesity a risk factor for severe COVID-19 infection: Multiple potential mechanisms. Circulation 2020. [Google Scholar] [CrossRef] [PubMed]
- Hâncu, A.; Mihălţan, F. Two Pandemics Coalition: Covid-19 and Obesity. Intern. Med. 2020, 17, 37–45. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72 314 cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85–97. [Google Scholar] [CrossRef]
- Ahn, S.-Y.; Sohn, S.-H.; Lee, S.-Y.; Park, H.-L.; Park, Y.-W.; Kim, H.; Nam, J.-H. The effect of lipopolysaccharide-induced obesity and its chronic inflammation on influenza virus-related pathology. Environ. Toxicol. Pharmacol. 2015, 40, 924–930. [Google Scholar] [CrossRef]
- Karlsson, E.A.; Hertz, T.; Johnson, C.; Mehle, A.; Krammer, F.; Schultz-Cherry, S. Obesity outweighs protection conferred by adjuvanted influenza vaccination. mBio 2016, 7. [Google Scholar] [CrossRef] [Green Version]
- Xue, K.S.; Stevens-Ayers, T.; Campbell, A.P.; Englund, J.A.; Pergam, S.A.; Boeckh, M.; Bloom, J.D. Parallel evolution of influenza across multiple spatiotemporal scales. eLife 2017, 6, e26875. [Google Scholar] [CrossRef]
- Zhang, A.J.; To, K.K.; Li, C.; Lau, C.C.; Poon, V.K.; Chan, C.C.; Zheng, B.-J.; Hung, I.F.; Lam, K.S.; Xu, A. Leptin mediates the pathogenesis of severe 2009 pandemic influenza A (H1N1) infection associated with cytokine dysregulation in mice with diet-induced obesity. J. Infect. Dis. 2013, 207, 1270–1280. [Google Scholar] [CrossRef]
- Park, S.; Jeon, J.-H.; Min, B.-K.; Ha, C.-M.; Thoudam, T.; Park, B.-Y.; Lee, I.-K. Role of the pyruvate dehydrogenase complex in metabolic remodeling: Differential pyruvate dehydrogenase complex functions in metabolism. Diabetes Metabol. J. 2018, 42, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Cui, G.; Chen, J.; Gao, H.; Wei, Y.; Uede, T.; Chen, Z.; Diao, H. Regular exercise enhances the immune response against microbial antigens through up-regulation of toll-like receptor signaling pathways. Cell. Physiol. Biochem. 2015, 37, 735–746. [Google Scholar] [CrossRef] [PubMed]
- Reidy, P.T.; Yonemura, N.M.; Madsen, J.H.; McKenzie, A.I.; Mahmassani, Z.S.; Rondina, M.T.; Lin, Y.K.; Kaput, K.; Drummond, M.J. An accumulation of muscle macrophages is accompanied by altered insulin sensitivity after reduced activity and recovery. Acta Physiol. 2019, 226, e13251. [Google Scholar] [CrossRef] [PubMed]
- Na, H.-N.; Nam, J.-H. Adenovirus 36 as an obesity agent maintains the obesity state by increasing MCP-1 and inducing inflammation. J. Infect. Dis. 2012, 205, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Na, H.; Kim, J.-A.; Nam, J.-H. What we know and what we need to know about adenovirus 36-induced obesity. Int. J. Obes. 2020. [Google Scholar] [CrossRef]
- Trayhurn, P. Hypoxia and adipose tissue function and dysfunction in obesity. Physiol. Rev. 2013, 93, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Jang, M.-K.; Son, Y.; Jung, M.H. ATF3 plays a role in adipocyte hypoxia-mediated mitochondria dysfunction in obesity. Biochem. Biophys. Res. Commun. 2013, 431, 421–427. [Google Scholar] [CrossRef]
- Na, H.; Kim, H.; Nam, J. Novel genes and cellular pathways related to infection with adenovirus-36 as an obesity agent in human mesenchymal stem cells. Int. J. Obes. 2012, 36, 195–200. [Google Scholar] [CrossRef] [Green Version]
- Na, H.; Kim, J.; Lee, H.; Shim, K.; Kimm, H.; Jee, S.; Jo, I.; Nam, J. Association of human adenovirus-36 in overweight Korean adults. Int. J. Obes. 2012, 36, 281–285. [Google Scholar] [CrossRef] [Green Version]
- Mizumoto, K.; Kagaya, K.; Zarebski, A.; Chowell, G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance 2020, 25, 2000180. [Google Scholar] [CrossRef] [Green Version]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- Guan, W.-J.; Ni, Z.-Y.; Hu, Y.; Liang, W.-H.; Ou, C.-Q.; He, J.-X.; Liu, L.; Shan, H.; Lei, C.-L.; Hui, D.S. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- WHO Characterizes COVID-19 as a Pandemic (11 March 2020). Available online: https://bit.ly/2wNmAGc (accessed on 23 August 2020).
- Zhang, Y.; Chen, C.; Zhu, S.; Shu, C.; Wang, D.; Song, J.; Song, Y.; Zhen, W.; Feng, Z.; Wu, G. Isolation of 2019-nCoV from a stool specimen of a laboratory-confirmed case of the coronavirus disease 2019 (COVID-19). China CDC Wkly. 2020, 2, 123–124. [Google Scholar] [CrossRef]
- Guo, Y.-R.; Cao, Q.-D.; Hong, Z.-S.; Tan, Y.-Y.; Chen, S.-D.; Jin, H.-J.; Tan, K.-S.; Wang, D.-Y.; Yan, Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak–an update on the status. Milit. Med. Res. 2020, 7, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Batiha, G.E.; Tayebwa, D.S.; Beshbishy, A.M.; N’Da, D.D.; Yokoyama, N.; Igarashi, I. Inhibitory effects of novel ciprofloxacin derivatives on the growth of four Babesia species and Theileria equi. Parasitol. Res. 2020, 119, 3061–3073. [Google Scholar] [CrossRef]
- Shereen, M.A.; Khan, S.; Kazmi, A.; Bashir, N.; Siddique, R. COVID-19 infection: Origin, transmission, and characteristics of human coronaviruses. J. Adv. Res. 2020. [Google Scholar] [CrossRef]
- Perrier, A.; Bonnin, A.; Desmarets, L.; Danneels, A.; Goffard, A.; Rouillé, Y.; Dubuisson, J.; Belouzard, S. The C-terminal domain of the MERS coronavirus M protein contains a trans-Golgi network localization signal. J. Biol. Chem. 2019, 294, 14406–14421. [Google Scholar] [CrossRef] [Green Version]
- Alanagreh, L.A.; Alzoughool, F.; Atoum, M. The human coronavirus disease COVID-19: Its origin, characteristics, and insights into potential drugs and its mechanisms. Pathogens 2020, 9, 331. [Google Scholar] [CrossRef]
- Magdy Beshbishy, A.; Alghamdi, S.; Onyiche, T.E.; Zahoor, M.; Rivero-Perez, N.; Zaragoza-Bastida, A.; Ghorab, M.A.; Meshaal, A.K.; El-Esawi, M.A.; Hetta, H.F.; et al. Biogenesis, biologic function and clinical potential of exosomes in different diseases. Appl. Sci. 2020, 10, 4428. [Google Scholar] [CrossRef]
- Ogden, C.L.; Fakhouri, T.H.; Carroll, M.D.; Hales, C.M.; Fryar, C.D.; Li, X.; Freedman, D.S. Prevalence of obesity among adults, by household income and education—United States, 2011–2014. MMWR. Morb. Mortal. Wkl. Rep. 2017, 66, 1369. [Google Scholar] [CrossRef]
- Onder, G.; Rezza, G.; Brusaferro, S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA 2020, 323, 1775–1776. [Google Scholar] [CrossRef]
- Lighter, J.; Phillips, M.; Hochman, S.; Sterling, S.; Johnson, D.; Francois, F.; Stachel, A. Obesity in patients younger than 60 years is a risk factor for Covid-19 hospital admission. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [Green Version]
- Caspard, H.; Jabbour, S.; Hammar, N.; Fenici, P.; Sheehan, J.J.; Kosiborod, M. Recent trends in the prevalence of type 2 diabetes and the association with abdominal obesity lead to growing health disparities in the USA: An analysis of the NHANES surveys from 1999 to 2014. Diabetes Obes. Metabol. 2018, 20, 667–671. [Google Scholar] [CrossRef] [Green Version]
- Simonnet, A.; Chetboun, M.; Poissy, J.; Raverdy, V.; Noulette, J.; Duhamel, A.; Labreuche, J.; Mathieu, D.; Pattou, F.; Jourdain, M. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity 2020. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020. [Google Scholar] [CrossRef]
- Cai, Q.; Chen, F.; Wang, T.; Luo, F.; Liu, X.; Wu, Q.; He, Q.; Wang, Z.; Liu, Y.; Liu, L. Obesity and COVID-19 severity in a designated hospital in Shenzhen, China. Diabetes Care 2020. [Google Scholar] [CrossRef]
- Mehra, M.R.; Desai, S.S.; Ruschitzka, F.; Patel, A.N. Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: A multinational registry analysis. Lancet 2020. [Google Scholar] [CrossRef]
- Petrilli, C.M.; Jones, S.A.; Yang, J.; Rajagopalan, H.; O’Donnell, L.F.; Chernyak, Y.; Tobin, K.; Cerfolio, R.J.; Francois, F.; Horwitz, L.I. Factors associated with hospitalization and critical illness among 4,103 patients with COVID-19 disease in New York City. MedRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Dietz, W.; Santos-Burgoa, C. Obesity and its Implications for COVID-19 Mortality. Obesity 2020, 28, 1005. [Google Scholar] [CrossRef] [Green Version]
- Petersen, A.; Bressem, K.; Albrecht, J.; Thiess, H.-M.; Vahldiek, J.; Hamm, B.; Makowski, M.; Niehues, A.; Niehues, S.; Adams, L.C. Obesity and COVID-19: The role of visceral adipose tissue. medRxiv 2020. [Google Scholar] [CrossRef]
- Bornstein, S.R.; Dalan, R.; Hopkins, D.; Mingrone, G.; Boehm, B.O. Endocrine and metabolic link to coronavirus infection. Nat. Rev. Endocrinol. 2020, 16, 297–298. [Google Scholar] [CrossRef] [Green Version]
- Bourgeois, C.; Gorwood, J.; Barrail-Tran, A.; Lagathu, C.; Capeau, J.; Desjardins, D.; Le Grand, R.; Damouche, A.; Béréziat, V.; Lambotte, O. Specific biological features of adipose tissue, and their impact on HIV persistence. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef] [Green Version]
- Rao, S.; Lau, A.; So, H.-C. Exploring diseases/traits and blood proteins causally related to expression of ACE2, the putative receptor of SARS-CoV-2: A Mendelian Randomization analysis highlights tentative relevance of diabetes-related traits. Diabetes Care 2020. [Google Scholar] [CrossRef]
- Zheng, K.I.; Gao, F.; Wang, X.-B.; Sun, Q.-F.; Pan, K.-H.; Wang, T.-Y.; Ma, H.-L.; Liu, W.-Y.; George, J.; Zheng, M.-H. Obesity as a risk factor for greater severity of COVID-19 in patients with metabolic associated fatty liver disease. Metabolism 2020, 154244. [Google Scholar] [CrossRef]
- Hall, K. A review of the carbohydrate–insulin model of obesity. Eur. J. Clin. Nutr. 2017, 71, 323–326. [Google Scholar] [CrossRef]
- Schulte, E.M.; Avena, N.M.; Gearhardt, A.N. Which foods may be addictive? The roles of processing, fat content, and glycemic load. PLoS ONE 2015, 10, e0117959. [Google Scholar] [CrossRef]
- Petrakis, D.; Margină, D.; Tsarouhas, K.; Tekos, F.; Stan, M.; Nikitovic, D.; Kouretas, D.; Spandidos, D.A.; Tsatsakis, A. Obesity—A risk factor for increased COVID-19 prevalence, severity and lethality (Review). Mol. Med. Rep. 2020, 22, 9–19. [Google Scholar] [CrossRef]
- Driggin, E.; Madhavan, M.V.; Bikdeli, B.; Chuich, T.; Laracy, J.; Biondi-Zoccai, G.; Brown, T.S.; Der Nigoghossian, C.; Zidar, D.A.; Haythe, J. Cardiovascular considerations for patients, health care workers, and health systems during the COVID-19 pandemic. J. Am. Coll. Cardiol. 2020, 75, 2352–2371. [Google Scholar] [CrossRef]
- Rodríguez, M.Á.; Crespo, I.; Olmedillas, H. Exercising in times of COVID-19: What do experts recommend doing within four walls? Rev. Espanola Cardiol. (Engl. Ed.) 2020. [Google Scholar] [CrossRef]
- Isasi, C.R.; Parrinello, C.M.; Jung, M.M.; Carnethon, M.R.; Birnbaum-Weitzman, O.; Espinoza, R.A.; Penedo, F.J.; Perreira, K.M.; Schneiderman, N.; Sotres-Alvarez, D. Psychosocial stress is associated with obesity and diet quality in Hispanic/Latino adults. Ann. Epidemiol. 2015, 25, 84–89. [Google Scholar] [CrossRef] [Green Version]
- Liang, T. Handbook of COVID-19 prevention and treatment. First Affil. Hosp. Zhejiang Univ. Sch. Med. Compil. Accord. Clin. Exp. 2020, 1–59. Available online: https://covid-19.alibabacloud.com/ (accessed on 23 August 2020).
- Castaner, O.; Goday, A.; Park, Y.-M.; Lee, S.-H.; Magkos, F.; Shiow, S.-A.T.E.; Schröder, H. The gut microbiome profile in obesity: A systematic review. Int. J. Endocrinol. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Ding, H.; Wang, T.; Hooper, L.V.; Koh, G.Y.; Nagy, A.; Semenkovich, C.F.; Gordon, J.I. The gut microbiota as an environmental factor that regulates fat storage. Proc. Natl. Acad. Sci. USA 2004, 101, 15718–15723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027. [Google Scholar] [CrossRef]
- Koliada, A.; Syzenko, G.; Moseiko, V.; Budovska, L.; Puchkov, K.; Perederiy, V.; Gavalko, Y.; Dorofeyev, A.; Romanenko, M.; Tkach, S. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017, 17, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Hernández, A.; Beneit, N.; Díaz-Castroverde, S.; Escribano, Ó. Differential role of adipose tissues in obesity and related metabolic and vascular complications. Int. J. Endocrinol. 2016, 2016. [Google Scholar] [CrossRef] [Green Version]
- Winer, D.A.; Luck, H.; Tsai, S.; Winer, S. The intestinal immune system in obesity and insulin resistance. Cell Metab. 2016, 23, 413–426. [Google Scholar] [CrossRef] [Green Version]
- Boucard-Jourdin, M.; Kugler, D.; Ahanda, M.-L.E.; This, S.; De Calisto, J.; Zhang, A.; Mora, J.R.; Stuart, L.M.; Savill, J.; Lacy-Hulbert, A. β8 integrin expression and activation of TGF-β by intestinal dendritic cells are determined by both tissue microenvironment and cell lineage. J. Immunol. 2016, 197, 1968–1978. [Google Scholar] [CrossRef]
- Wang, T.; Chen, R.; Liu, C.; Liang, W.; Guan, W.; Tang, R.; Tang, C.; Zhang, N.; Zhong, N.; Li, S. Attention should be paid to venous thromboembolism prophylaxis in the management of COVID-19. Lancet Haematol. 2020, 7, e362–e363. [Google Scholar] [CrossRef]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef] [Green Version]
- Lorenzet, R.; Napoleone, E.; Cutrone, A.; Donati, M.B. Thrombosis and obesity: Cellular bases. Thromb. Res. 2012, 129, 285–289. [Google Scholar] [CrossRef]
- Campello, E.; Zabeo, E.; Radu, C.M.; Spiezia, L.; Gavasso, S.; Fadin, M.; Woodhams, B.; Vettor, R.; Simioni, P. Hypercoagulability in overweight and obese subjects who are asymptomatic for thrombotic events. Thromb. Haemost. 2015, 113, 85–96. [Google Scholar] [PubMed]
- Samad, F.; Ruf, W. Inflammation, obesity, and thrombosis. Blood J. Am. Soc. Hematol. 2013, 122, 3415–3422. [Google Scholar] [CrossRef] [PubMed]
- Sanchis-Gomar, F.; Lavie, C.J.; Mehra, M.R.; Henry, B.M.; Lippi, G. Obesity and outcomes in COVID-19: When an epidemic and pandemic collide. Mayo Clin. Proc. 2020, 95, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Milner, J.J.; Beck, M.A. The impact of obesity on the immune response to infection. Proc. Nutr. Soc. 2012, 71, 298–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trim, W.; Turner, J.E.; Thompson, D. Parallels in immunometabolic adipose tissue dysfunction with ageing and obesity. Front. Immunol. 2018, 9, 169. [Google Scholar] [CrossRef] [Green Version]
- Laddu, D.R.; Lavie, C.J.; Phillips, S.A.; Arena, R. Physical activity for immunity protection: Inoculating populations with healthy living medicine in preparation for the next pandemic. Prog. Cardiovasc. Dis. 2020. [Google Scholar] [CrossRef]
- Tanaka, S.I.; Isoda, F.; Ishihara, Y.; Kimura, M.; Yamakawa, T. T lymphopaenia in relation to body mass index and TNF-α in human obesity: Adequate weight reduction can be corrective. Clin. Endocrinol. 2001, 54, 347–354. [Google Scholar] [CrossRef]
- Meliopoulos, V.; Livingston, B.; Van de Velde, L.-A.; Honce, R.; Schultz-Cherry, S. Absence of β6 integrin reduces influenza disease severity in highly susceptible obese mice. J. Virol. 2019, 93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Saber Batiha, G.; Magdy Beshbishy, A.; Stephen Adeyemi, O.; Nadwa, E.; Rashwan, E.; Yokoyama, N.; Igarashi, I. Safety and efficacy of hydroxyurea and eflornithine against most blood parasites Babesia and Theileria. PLoS ONE 2020, 15, e0228996. [Google Scholar] [CrossRef] [PubMed]
- Kass, D.A.; Duggal, P.; Cingolani, O. Obesity could shift severe COVID-19 disease to younger ages. Lancet (Lond. Engl.) 2020. [Google Scholar] [CrossRef]
- Maier, H.E.; Lopez, R.; Sanchez, N.; Ng, S.; Gresh, L.; Ojeda, S.; Burger-Calderon, R.; Kuan, G.; Harris, E.; Balmaseda, A. Obesity increases the duration of influenza A virus shedding in adults. J. Infect. Dis. 2018, 218, 1378–1382. [Google Scholar] [CrossRef] [Green Version]
- Smith, A.G.; Sheridan, P.A.; Harp, J.B.; Beck, M.A. Diet-induced obese mice have increased mortality and altered immune responses when infected with influenza virus. J. Nutr. 2007, 137, 1236–1243. [Google Scholar] [CrossRef] [Green Version]
- Karlsson, E.A.; Sheridan, P.A.; Beck, M.A. Diet-induced obesity impairs the T cell memory response to influenza virus infection. J. Immunol. 2010, 184, 3127–3133. [Google Scholar] [CrossRef] [Green Version]
- Honce, R.; Karlsson, E.A.; Wohlgemuth, N.; Estrada, L.D.; Meliopoulos, V.A.; Yao, J.; Schultz-Cherry, S. Obesity-related microenvironment promotes emergence of virulent influenza virus strains. mBio 2020, 11. [Google Scholar] [CrossRef] [Green Version]
- Easterbrook, J.D.; Dunfee, R.L.; Schwartzman, L.M.; Jagger, B.W.; Sandouk, A.; Kash, J.C.; Memoli, M.J.; Taubenberger, J.K. Obese mice have increased morbidity and mortality compared to non-obese mice during infection with the 2009 pandemic H1N1 influenza virus. Influenza Other Respir. Viruses 2011, 5, 418–425. [Google Scholar] [CrossRef]
- Karlsson, E.A.; Meliopoulos, V.A.; van de Velde, N.C.; van de Velde, L.-A.; Mann, B.; Gao, G.; Rosch, J.; Tuomanen, E.; McCullers, J.; Vogel, P. A perfect storm: Increased colonization and failure of vaccination leads to severe secondary bacterial infection in influenza virus-infected obese mice. mBio 2017, 8. [Google Scholar] [CrossRef] [Green Version]
- Paich, H.A.; Sheridan, P.A.; Handy, J.; Karlsson, E.A.; Schultz-Cherry, S.; Hudgens, M.G.; Noah, T.L.; Weir, S.S.; Beck, M.A. Overweight and obese adult humans have a defective cellular immune response to pandemic H1N1 influenza A virus. Obesity 2013, 21, 2377–2386. [Google Scholar] [CrossRef]
- O’Shea, D.; Corrigan, M.; Dunne, M.R.; Jackson, R.; Woods, C.; Gaoatswe, G.; Moynagh, P.N.; O’Connell, J.; Hogan, A. Changes in human dendritic cell number and function in severe obesity may contribute to increased susceptibility to viral infection. Int. J. Obes. 2013, 37, 1510–1513. [Google Scholar] [CrossRef] [Green Version]
- Radigan, K.A.; Morales-Nebreda, L.; Soberanes, S.; Nicholson, T.; Nigdelioglu, R.; Cho, T.; Chi, M.; Hamanaka, R.B.; Misharin, A.V.; Perlman, H. Impaired clearance of influenza A virus in obese, leptin receptor deficient mice is independent of leptin signaling in the lung epithelium and macrophages. PLoS ONE 2014, 9, e108138. [Google Scholar] [CrossRef]
- Rebeles, J.; Green, W.D.; Alwarawrah, Y.; Nichols, A.G.; Eisner, W.; Danzaki, K.; MacIver, N.J.; Beck, M.A. Obesity-induced changes in T-cell metabolism are associated with impaired memory T-cell response to influenza and are not reversed with weight loss. J. Infect. Dis. 2019, 219, 1652–1661. [Google Scholar] [CrossRef] [PubMed]
- Rocha, V.Z.; Libby, P. Obesity, inflammation, and atherosclerosis. Nat. Rev. Cardiol. 2009, 6, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Batiha, G.E.; Beshbishy, A.M.; Tayebwa, D.S.; Adeyemi, O.S.; Yokoyama, N.; Igarashi, I. Anti-piroplasmic potential of the methanolic Peganum harmala seeds and ethanolic Artemisia absinthium leaf extracts. J. Protoz. Res. 2019, 29, 8–25. [Google Scholar]
- Shoelson, S.E.; Herrero, L.; Naaz, A. Obesity, inflammation, and insulin resistance. Gastroenterology 2007, 132, 2169–2180. [Google Scholar] [CrossRef]
- Lee, H.; Lee, I.S.; Choue, R. Obesity, inflammation and diet. Pediatr. Gastroenterol. Hepatol. Nutr. 2013, 16, 143–152. [Google Scholar] [CrossRef] [Green Version]
- Setty, A.R.; Curhan, G.; Choi, H.K. Obesity, waist circumference, weight change, and the risk of psoriasis in women: Nurses’ Health Study II. Arch. Intern. Med. 2007, 167, 1670–1675. [Google Scholar] [CrossRef] [Green Version]
- Manzel, A.; Muller, D.N.; Hafler, D.A.; Erdman, S.E.; Linker, R.A.; Kleinewietfeld, M. Role of “Western diet” in inflammatory autoimmune diseases. Curr. Allergy Asthma Rep. 2014, 14, 404. [Google Scholar] [CrossRef] [Green Version]
- Nakamizo, S.; Honda, T.; Adachi, A.; Nagatake, T.; Kunisawa, J.; Kitoh, A.; Otsuka, A.; Dainichi, T.; Nomura, T.; Ginhoux, F. High fat diet exacerbates murine psoriatic dermatitis by increasing the number of IL-17-producing γδ T cells. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Neimann, A.L.; Shin, D.B.; Wang, X.; Margolis, D.J.; Troxel, A.B.; Gelfand, J.M. Prevalence of cardiovascular risk factors in patients with psoriasis. J. Am. Acad. Dermatol. 2006, 55, 829–835. [Google Scholar] [CrossRef]
- Cohen, A.; Sherf, M.; Vidavsky, L.; Vardy, D.; Shapiro, J.; Meyerovitch, J. Association between psoriasis and the metabolic syndrome. Dermatology 2008, 216, 152–155. [Google Scholar] [CrossRef]
- Paules, C.I.; Sullivan, S.G.; Subbarao, K.; Fauci, A.S. Chasing seasonal influenza—The need for a universal influenza vaccine. N. Engl. J. Med. 2018, 378, 7–9. [Google Scholar] [CrossRef]
- Smit, M.A.; Wang, H.-L.; Kim, E.; Barragan, N.; Aldrovandi, G.M.; El Amin, A.N.; Mascola, L.; Pannaraj, P.S. Influenza vaccine is protective against laboratory-confirmed influenza in obese children. Pediatr. Infect. Dis. J. 2016, 35, 440–445. [Google Scholar] [CrossRef] [Green Version]
- Sheridan, P.A.; Paich, H.A.; Handy, J.; Karlsson, E.A.; Hudgens, M.G.; Sammon, A.B.; Holland, L.A.; Weir, S.; Noah, T.L.; Beck, M.A. Obesity is associated with impaired immune response to influenza vaccination in humans. Int. J. Obes. 2012, 36, 1072–1077. [Google Scholar] [CrossRef] [Green Version]
- Moser, J.A.S.; Galindo-Fraga, A.; Ortiz-Hernández, A.A.; Gu, W.; Hunsberger, S.; Galán-Herrera, J.F.; Guerrero, M.L.; Ruiz-Palacios, G.M.; Beigel, J.H.; Group, L.R.I.S. Underweight, overweight, and obesity as independent risk factors for hospitalization in adults and children from influenza and other respiratory viruses. Influenza Other Respir. Viruses 2019, 13, 3–9. [Google Scholar] [CrossRef]
- Torres, L.; Martins, V.D.; Faria, A.M.C.; Maioli, T.U. The intriguing relationship between obesity and infection. J. Infectiol. 2018, 1, 5. [Google Scholar] [CrossRef]
- Dhurandhar, N.; Bailey, D.; Thomas, D. Interaction of obesity and infections. Obes. Rev. 2015, 16, 1017–1029. [Google Scholar] [CrossRef]
- Louie, J.K.; Acosta, M.; Samuel, M.C.; Schechter, R.; Vugia, D.J.; Harriman, K.; Matyas, B.T. A novel risk factor for a novel virus: Obesity and 2009 pandemic influenza A (H1N1). Clin. Infect. Dis. 2011, 52, 301–312. [Google Scholar] [CrossRef] [Green Version]
- Zulkipli, M.S.; Dahlui, M.; Peramalah, D.; Wai, V.H.C.; Bulgiba, A.; Rampal, S. The association between obesity and dengue severity among pediatric patients: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2018, 12, e0006263. [Google Scholar] [CrossRef] [Green Version]
- El-Saber Batiha, G.; Alqahtani, A.; Ilesanmi, O.B.; Saati, A.A.; El-Mleeh, A.; Hetta, H.F.; Magdy Beshbishy, A. Avermectin derivatives, pharmacokinetics, therapeutic and toxic dosages, mechanism of action, and their biological effects. Pharmaceuticals 2020, 13, 196. [Google Scholar] [CrossRef]
- Birnbaum, H.G.; Morley, M.; Greenberg, P.E.; Colice, G.L. Economic burden of respiratory infections in an employed population. Chest 2002, 122, 603–611. [Google Scholar] [CrossRef] [Green Version]
- Harpsøe, M.C.; Nielsen, N.M.; Friis-Møller, N.; Andersson, M.; Wohlfahrt, J.; Linneberg, A.; Nohr, E.A.; Jess, T. Body mass index and risk of infections among women in the Danish National Birth Cohort. Am. J. Epidemiol. 2016, 183, 1008–1017. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-Y.; Forno, E.; Gogna, M.; Celedón, J.C. Obesity and rhinitis in a nationwide study of children and adults in the United States. J. Allergy Clin. Immunol. 2016, 137, 1460–1465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cocoros, N.M.; Lash, T.L.; DeMaria, A., Jr.; Klompas, M. Obesity as a risk factor for severe influenza-like illness. Influenza Other Respir. Viruses 2014, 8, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, L.; Weber, S.; Elgizouli, M.; Stoehlker, A.-S.; Geist, I.; Peter, H.-H.; Vach, W.; Nieters, A. Obesity and risk of respiratory tract infections: Results of an infection-diary based cohort study. BMC Public Health 2018, 18, 271. [Google Scholar] [CrossRef]
- Lee, Y.L.; Chen, Y.C.; Chen, Y.A. Obesity and the occurrence of bronchitis in adolescents. Obesity 2013, 21, E149–E153. [Google Scholar] [CrossRef]
- Short, K.R.; Kedzierska, K.; van de Sandt, C.E. Back to the future: Lessons learned from the 1918 influenza pandemic. Front. Cell. Infect. Microbiol. 2018, 8, 343. [Google Scholar] [CrossRef]
- Martin, E.T.; Archer, C.; McRoberts, J.; Kulik, J.; Thurston, T.; Lephart, P.; Kaye, K.S. Epidemiology of severe influenza outcomes among adult patients with obesity in Detroit, Michigan, 2011. Influenza Other Respir. Viruses 2013, 7, 1004–1007. [Google Scholar] [CrossRef]
- He, J.; Liu, Z.-W.; Lu, Y.-P.; Li, T.-Y.; Liang, X.-J.; Arck, P.C.; Huang, S.-M.; Hocher, B.; Chen, Y.-P. A systematic review and meta-analysis of influenza a virus infection during pregnancy associated with an increased risk for stillbirth and low birth weight. Kidney Blood Press. Res. 2017, 42, 232–243. [Google Scholar] [CrossRef]
- Nakajima, N.; Hata, S.; Sato, Y.; Tobiume, M.; Katano, H.; Kaneko, K.; Nagata, N.; Kataoka, M.; Ainai, A.; Hasegawa, H. The first autopsy case of pandemic influenza (A/H1N1pdm) virus infection in Japan: Detection of a high copy number of the virus in type II alveolar epithelial cells by pathological and virological examination. Jpn. J. Infect. Dis. 2010, 63, 67–71. [Google Scholar]
- Al-Tawfiq, J.A.; Auwaerter, P.G. Healthcare-associated infections: The hallmark of Middle East respiratory syndrome coronavirus with review of the literature. J. Hosp. Infect. 2019, 101, 20–29. [Google Scholar] [CrossRef] [Green Version]
- Alserehi, H.; Wali, G.; Alshukairi, A.; Alraddadi, B. Impact of Middle East Respiratory Syndrome coronavirus (MERS-CoV) on pregnancy and perinatal outcome. BMC Infect. Dis. 2016, 16, 1–4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montes-Galindo, D.A.; Espiritu-Mojarro, A.C.; Melnikov, V.; Moy-López, N.A.; Soriano-Hernandez, A.D.; Galvan-Salazar, H.R.; Guzman-Muñiz, J.; Guzman-Esquivel, J.; Martinez-Fierro, M.L.; Rodriguez-Sanchez, I.P. Adenovirus 5 produces obesity and adverse metabolic, morphological, and functional changes in the long term in animals fed a balanced diet or a high-fat diet: A study on hamsters. Arch. Virol. 2019, 164, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Pasarica, M.; Loiler, S.; Dhurandhar, N.V. Acute effect of infection by adipogenic human adenovirus Ad36. Arch. Virol. 2008, 153, 2097. [Google Scholar] [CrossRef] [Green Version]
- Pasarica, M.; Shin, A.C.; Yu, M.; Yang, H.M.O.; Rathod, M.; Jen, K.L.C.; Kumar, S.M.; MohanKumar, P.S.; Markward, N.; Dhurandhar, N.V. Human adenovirus 36 induces adiposity, increases insulin sensitivity, and alters hypothalamic monoamines in rats. Obesity 2006, 14, 1905–1913. [Google Scholar] [CrossRef]
- Atkinson, R.; Dhurandhar, N.; Allison, D.; Bowen, R.; Israel, B.; Albu, J.; Augustus, A. Human adenovirus-36 is associated with increased body weight and paradoxical reduction of serum lipids. Int. J. Obes. 2005, 29, 281–286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vangipuram, S.; Yu, M.; Tian, J.; Stanhope, K.; Pasarica, M.; Havel, P.J.; Heydari, A.; Dhurandhar, N. Adipogenic human adenovirus-36 reduces leptin expression and secretion and increases glucose uptake by fat cells. Int. J. Obes. 2007, 31, 87–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vangipuram, S.D.; Sheele, J.; Atkinson, R.L.; Holland, T.C.; Dhurandhar, N.V. A human adenovirus enhances preadipocyte differentiation. Obes. Res. 2004, 12, 770–777. [Google Scholar] [CrossRef]
- Sang, Y.; Shields, L.E.; Sang, E.R.; Si, H.; Pigg, A.; Blecha, F. Ileal transcriptome analysis in obese rats induced by high-fat diets and an adenoviral infection. Int. J. Obes. 2019, 43, 2134–2142. [Google Scholar] [CrossRef]
- Morgan, O.W.; Bramley, A.; Fowlkes, A.; Freedman, D.S.; Taylor, T.H.; Gargiullo, P.; Belay, B.; Jain, S.; Cox, C.; Kamimoto, L. Morbid obesity as a risk factor for hospitalization and death due to 2009 pandemic influenza A (H1N1) disease. PLoS ONE 2010, 5, e9694. [Google Scholar] [CrossRef]
- Venkata, C.; Sampathkumar, P.; Afessa, B. Hospitalized patients with 2009 H1N1 influenza infection: The Mayo Clinic experience. Mayo Clin. Proc. 2010, 85, 798–805. [Google Scholar] [CrossRef]
- Martín, V.; Castilla, J.; Godoy, P.; Delgado-Rodríguez, M.; Soldevila, N.; Fernández-Villa, T.; Molina, A.J.; Astray, J.; Castro, A.; González-Candelas, F. High Body Mass Index as a Risk Factor for Hospitalization Due to Influenza: A Case–Control Study. Arch. Bronconeumol. (Engl. Ed.) 2016, 52, 299–307. [Google Scholar] [CrossRef]
- Taubenberger, J.K.; Morens, D.M. The pathology of influenza virus infections. Annu. Rev. Pathol. Mech. Dis. 2008, 3, 499–522. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.B.; Trapnell, C.; Bloom, J.D. Extreme heterogeneity of influenza virus infection in single cells. eLife 2018, 7, e32303. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.B.; Vogel, P.; Duan, S.; Govorkova, E.A.; Webby, R.J.; McCullers, J.A.; Schultz-Cherry, S. Impaired wound healing predisposes obese mice to severe influenza virus infection. J. Infect. Dis. 2012, 205, 252–261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klinkhammer, J.; Schnepf, D.; Ye, L.; Schwaderlapp, M.; Gad, H.H.; Hartmann, R.; Garcin, D.; Mahlakõiv, T.; Staeheli, P. IFN-λ prevents influenza virus spread from the upper airways to the lungs and limits virus transmission. eLife 2018, 7, e33354. [Google Scholar] [CrossRef] [PubMed]
- Yamane, K.; Indalao, I.L.; Chida, J.; Yamamoto, Y.; Hanawa, M.; Kido, H. Diisopropylamine dichloroacetate, a novel pyruvate dehydrogenase kinase 4 inhibitor, as a potential therapeutic agent for metabolic disorders and multiorgan failure in severe influenza. PLoS ONE 2014, 9, e98032. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Kim, J.-K.; Kim, D.-J.; Nam, J.-H.; Shim, S.-M.; Choi, Y.-K.; Lee, C.-H.; Poo, H. Diet-induced obesity dramatically reduces the efficacy of a 2009 pandemic H1N1 vaccine in a mouse model. J. Infect. Dis. 2012, 205, 244–251. [Google Scholar] [CrossRef] [Green Version]
- Ritter, J.B.; Wahl, A.S.; Freund, S.; Genzel, Y.; Reichl, U. Metabolic effects of influenza virus infection in cultured animal cells: Intra-and extracellular metabolite profiling. BMC Syst. Biol. 2010, 4, 61. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Russell, M.L.; Fonseca, K.; Earn, D.J.; Horsman, G.; Van Caeseele, P.; Chokani, K.; Vooght, M.; Babiuk, L.; Walter, S.D. Predictors of influenza a molecular viral shedding in Hutterite communities. Influenza Other Respir. Viruses 2017, 11, 254–262. [Google Scholar] [CrossRef] [Green Version]
- Yan, J.; Grantham, M.; Pantelic, J.; De Mesquita, P.J.B.; Albert, B.; Liu, F.; Ehrman, S.; Milton, D.K.; Consortium, E. Infectious virus in exhaled breath of symptomatic seasonal influenza cases from a college community. Proc. Natl. Acad. Sci. USA 2018, 115, 1081–1086. [Google Scholar] [CrossRef] [Green Version]
- Carrat, F.; Vergu, E.; Ferguson, N.M.; Lemaitre, M.; Cauchemez, S.; Leach, S.; Valleron, A.-J. Time lines of infection and disease in human influenza: A review of volunteer challenge studies. Am. J. Epidemiol. 2008, 167, 775–785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahmati-Ahmadabad, S.; Hosseini, F. Exercise against SARS-CoV-2 (COVID-19): Does workout intensity matter?(A mini review of some indirect evidence related to obesity). Obes. Med. 2020, 100245. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nutrition and Your Health: 2015–2020 Dietary Guidelines for Americans, 8th ed.; US Government Printing Office: Washington, DC, USA, 2015.
- World Health Organization. Diet, Nutrition, and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Blanck, H.M.; Collins, J. CDC’s Winnable Battles: Improved Nutrition, Physical Activity, and Decreased Obesity; Mary Ann Liebert, Inc.: New Rochelle, NY, USA, 2013. [Google Scholar]
- Fogli-Cawley, J.J.; Dwyer, J.T.; Saltzman, E.; McCullough, M.L.; Troy, L.M.; Meigs, J.B.; Jacques, P.F. The 2005 dietary guidelines for Americans and insulin resistance in the Framingham offspring cohort. Diabetes Care 2007, 30, 817–822. [Google Scholar] [CrossRef] [Green Version]
- Klein, S.; Sheard, N.F.; Pi-Sunyer, X.; Daly, A.; Wylie-Rosett, J.; Kulkarni, K.; Clark, N.G. Weight management through lifestyle modification for the prevention and management of type 2 diabetes: Rationale and strategies. A statement of the American Diabetes Association, the North American Association for the Study of Obesity, and the American Society for Clinical Nutrition. Am. J. Clin. Nutr. 2004, 80, 257–263. [Google Scholar]
- Qatanani, M.; Lazar, M.A. Mechanisms of obesity-associated insulin resistance: Many choices on the menu. Genes Dev. 2007, 21, 1443–1455. [Google Scholar] [CrossRef] [Green Version]
- Choon Lim Wong, G.; Narang, V.; Lu, Y.; Camous, X.; Nyunt, M.S.Z.; Carre, C.; Tan, C.; Xian, C.H.; Chong, J.; Chua, M. Hallmarks of improved immunological responses in the vaccination of more physically active elderly females. Exerc. Immunol. Rev. 2019, 25, 20–33. [Google Scholar]
- Warren, K.J.; Olson, M.M.; Thompson, N.J.; Cahill, M.L.; Wyatt, T.A.; Yoon, K.J.; Loiacono, C.M.; Kohut, M.L. Exercise improves host response to influenza viral infection in obese and non-obese mice through different mechanisms. PLoS ONE 2015, 10, e0129713. [Google Scholar] [CrossRef] [Green Version]
- Codella, R.; Luzi, L.; Inverardi, L.; Ricordi, C. The anti-inflammatory effects of exercise in the syndromic thread of diabetes and autoimmunity. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 3709–3722. [Google Scholar]
- Hagman, E.; Besor, O.; Hershkop, K.; Santoro, N.; Pierpont, B.; Mata, M.; Caprio, S.; Weiss, R. Relation of the degree of obesity in childhood to adipose tissue insulin resistance. Acta Diabetol. 2019, 56, 219–226. [Google Scholar] [CrossRef] [Green Version]



| Databases | Search Query |
|---|---|
| Research Gate | (Obesogenic OR obese OR obesity OR overweight OR BMI) and (SARS-CoV-2 OR COVID-19 OR coronavirus) and (quarantine OR hospital care OR hospitalization OR mortality OR morbidity) |
| PubMed | (Obesogenic OR obese OR obesity OR overweight OR BMI) and (morbidity OR mortality OR mortality rate OR death rate) and (influenza OR COVID-19 OR coronavirus OR SARS-CoV-2 OR health care) |
| Web of Science | (Coronavirus OR COVID-19 OR SARS-CoV-2 OR influenza) and (thrombosis vaccine OR WHO OR hospitalization OR intense care) and (obesity OR BMI OR overweight OR care episode OR morbidity OR mortality) |
| Google Scholar | (SARS-CoV-2 OR COVID-19 OR coronavirus) and (obesogenic OR obese OR obesity OR overweight OR BMI OR stress OR control) and (hospitalization OR hospital stay OR mortality OR morbidity) |
| Hand searching | Obesity and COVID-19 and immunity |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magdy Beshbishy, A.; Hetta, H.F.; Hussein, D.E.; Saati, A.A.; C. Uba, C.; Rivero-Perez, N.; Zaragoza-Bastida, A.; Shah, M.A.; Behl, T.; Batiha, G.E.-S. Factors Associated with Increased Morbidity and Mortality of Obese and Overweight COVID-19 Patients. Biology 2020, 9, 280. https://doi.org/10.3390/biology9090280
Magdy Beshbishy A, Hetta HF, Hussein DE, Saati AA, C. Uba C, Rivero-Perez N, Zaragoza-Bastida A, Shah MA, Behl T, Batiha GE-S. Factors Associated with Increased Morbidity and Mortality of Obese and Overweight COVID-19 Patients. Biology. 2020; 9(9):280. https://doi.org/10.3390/biology9090280
Chicago/Turabian StyleMagdy Beshbishy, Amany, Helal F. Hetta, Diaa E. Hussein, Abdullah A. Saati, Christian C. Uba, Nallely Rivero-Perez, Adrian Zaragoza-Bastida, Muhammad Ajmal Shah, Tapan Behl, and Gaber El-Saber Batiha. 2020. "Factors Associated with Increased Morbidity and Mortality of Obese and Overweight COVID-19 Patients" Biology 9, no. 9: 280. https://doi.org/10.3390/biology9090280