The Distribution and Identity of Edaphic Fungi in the McMurdo Dry Valleys
Abstract
:1. Introduction
2. Experimental
2.1. Sample Collection
| Valley | Coordinates | Elevation | Sampling Date |
|---|---|---|---|
| Miers Valley | 78°05.486'S, 163°48.539'E | 171 m | December 2006 |
| Beacon Valley | 77°52.321'S, 160°29.725'E | 1376 m | December 2006 |
| Upper Wright Valley | 77°31.122'S, 160°45.813'E | 947 m | January 2008 |
| Battleship Promontory | 76°54.694'S, 160°55.676'E | 1028 m | January 2008 |
| Alatna Valley | 76°54.816'S, 161°02.213'E | 1057 m | November 2010 |
| University Valley | 77°51.668'S, 160°42.736'E | 1680 m | November 2010 |

2.2. Soil Chemistry
2.3. DNA Extraction
2.4. Terminal Restriction Fragment Length Polymorphism Analysis
2.5. 454 Pyrosequencing
2.6. Data Analysis
3. Results and Discussion
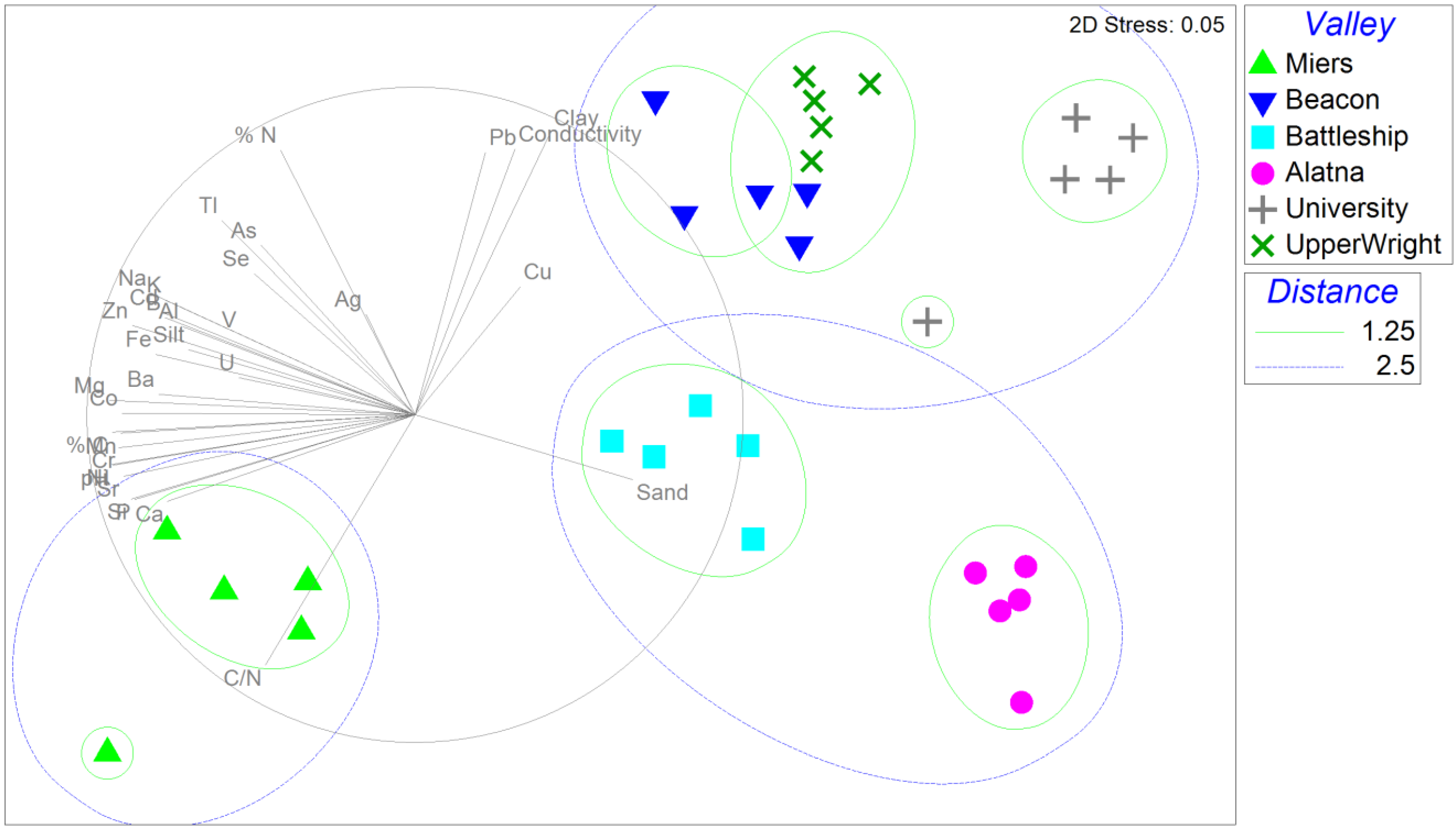
3.1. Soil Geochemistry
3.2. Community Fingerprinting with tRFLP
| Valley | Average Concentration ± S.D. |
|---|---|
| Miers Valley | 48.60 ± 27.79 ng/µL |
| Beacon Valley | 0.48 ± 0.55 ng/µL |
| Battleship Promontory | 20.87 ± 5.61 ng/µL |
| Upper Wright Valley | 3.68 ± 7.57 ng/µL |
| Alatna Valley | 15.84 ± 13.49 ng/µL |
| University Valley | 0.05 ± 0.09 ng/µL |
| Valley | Total PF | Average PF ± S.D. |
|---|---|---|
| Miers Valley | 5 | 1.0 ± 1.2 |
| Beacon Valley | 2 | 0.4 * |
| Battleship Valley | 13 | 2.6 ± 1.5 |
| Wright Valley | 2 | 0.4 * |
| Alatna Valley | 11 | 2.2 ± 3.2 |
| University Valley | 0 | 0 |

3.3. 454 Pyrosequencing
| Read Count | Best Match in GenBank nr Database | |||||||
|---|---|---|---|---|---|---|---|---|
| OTU # | AV_N | BP_D | MV_A | Total | GenBank ID | Identity (%) | Phylum | Organism |
| 3 | 1852 | 407 | 1 | 2283 | AB032673 | 99 | Basidiomycota | Cryptococcus consortionis |
| 4 | 841 | 728 | 191 | 1760 | EF432821 | 93 | Chytridiomycota | Lobulomycetales sp. AF017 |
| 6 | 1058 | 122 | 369 | 1542 | EF060799 | 99 | Ascomycota | Herpotrichiellaceae sp. LM500 |
| 7 | 505 | 68 | 233 | 806 | JF747078 | 99 | Ascomycota | Exophiala equina |
| 10 | 129 | 351 | 61 | 541 | EU480339 | 93 | Unknown | Uncultured clone |
| 11 | 0 | 0 | 372 | 372 | GQ250013 | 92 | Ascomycota | Cordyceps sp. BCC22921 |
| 14 | 246 | 0 | 0 | 246 | EF535204 | 90 | Ascomycota | Candelaria crawfordii strain CHN265 |
| 16 | 179 | 0 | 0 | 179 | FJ827708 | 90 | Chytridiomycota | Powellomyces sp. PL 142 |
| 20 | 0 | 109 | 0 | 109 | EU352772 | 93 | Chytridiomycota | Chytridiales sp. JEL178 |
| 22 | 0 | 0 | 109 | 109 | DQ457086 | 85 | Unknown | Uncultured clone |
| 24 | 0 | 83 | 0 | 83 | AM901700 | 97 | Ascomycota | Ascomycete sp. BF104 |
| 25 | 0 | 0 | 81 | 81 | FJ827708 | 94 | Chytridiomycota | Powellomyces sp. PL 142 |
| 26 | 80 | 0 | 0 | 80 | GU184116 | 96 | Ascomycota | Acarospora rosulata isolate ACABUL_USA2 |
| 28 | 36 | 30 | 0 | 66 | KC222134 | 83 | Ascomycota | Trichoglossum octopartitum |
| 29 | 0 | 0 | 61 | 61 | EF585664 | 83 | Chytridiomycota | Betamyces americaemeridionalis |
| 35 | 0 | 0 | 54 | 54 | EU352770 | 92 | Chytridiomycota | Lobulomyces poculatus |
| 39 | 0 | 47 | 0 | 47 | AF106527 | 91 | Ascomycota | Arthrobotrys arcuata strain CBS 174.89 |
| 40 | 8 | 33 | 1 | 42 | DQ494379 | 94 | Ascomycota | Vermispora fusarina |
| 41 | 12 | 27 | 3 | 42 | JX171180 | 94 | Basidiomycota | Meira sp. ANTCW08-165 |
| 45 | 34 | 1 | 5 | 40 | FJ827741 | 96 | Chytridiomycota | Gaertneriomyces sp. JEL 550 |
| 48 | 29 | 1 | 0 | 30 | HQ634632 | 97 | Ascomycota | Chaetothyriales sp. M-Cre1-2 |
| 49 | 29 | 0 | 0 | 29 | JX124723 | 98 | Ascomycota | Taphrina sp. CCFEE 5198 |
| 51 | 0 | 0 | 28 | 28 | JX036093 | 93 | Ascomycota | Polysporina frigida |
| 54 | 0 | 10 | 17 | 27 | EU352770 | 92 | Chytridiomycota | Lobulomyces poculatus |
| 56 | 0 | 0 | 25 | 25 | JF809853 | 99 | Chytridiomycota | Betamyces sp. PL 173 |
| 59 | 0 | 0 | 23 | 23 | AY373015 | 91 | Unknown | Olpidium brassicae |
| 60 | 0 | 22 | 0 | 22 | JQ936330 | 99 | Unknown | Phaeosphaeriopsis sp. CBP21E |
| 61 | 0 | 0 | 22 | 22 | JX219783 | 91 | Ascomycota | Cortinarius callisteus |
| 62 | 0 | 0 | 22 | 22 | JN416510 | 89 | Basidiomycota | Basidiobolus sp. BCU1 |
| 64 | 1 | 19 | 1 | 21 | JX173100 | 99 | Ascomycota | Cladosporium sp. AF13 |
| 67 | 18 | 0 | 0 | 18 | AY781244 | 89 | Unknown | Ascomycete sp. olrim401 |
| 68 | 0 | 18 | 0 | 18 | AY394892 | 94 | Ascomycota | Mycorrhizal sp. pkc11 |
| 72 | 0 | 0 | 17 | 17 | EF634250 | 80 | Chytridiomycota | Coralloidiomyces digitatus |
| 78 | 0 | 15 | 0 | 15 | EU480016 | 90 | Unknown | Uncultured clone |
| 101 | 0 | 0 | 11 | 11 | JN882333 | 94 | Chytridiomycota | Monoblepharis hypogyna |
| 102 | 0 | 0 | 11 | 11 | DQ485612 | 93 | Chytridiomycota | Rhizophydium carpophilum |
| 105 | 0 | 0 | 10 | 10 | JQ711836 | 99 | Basidiomycota | Russula nigricans |

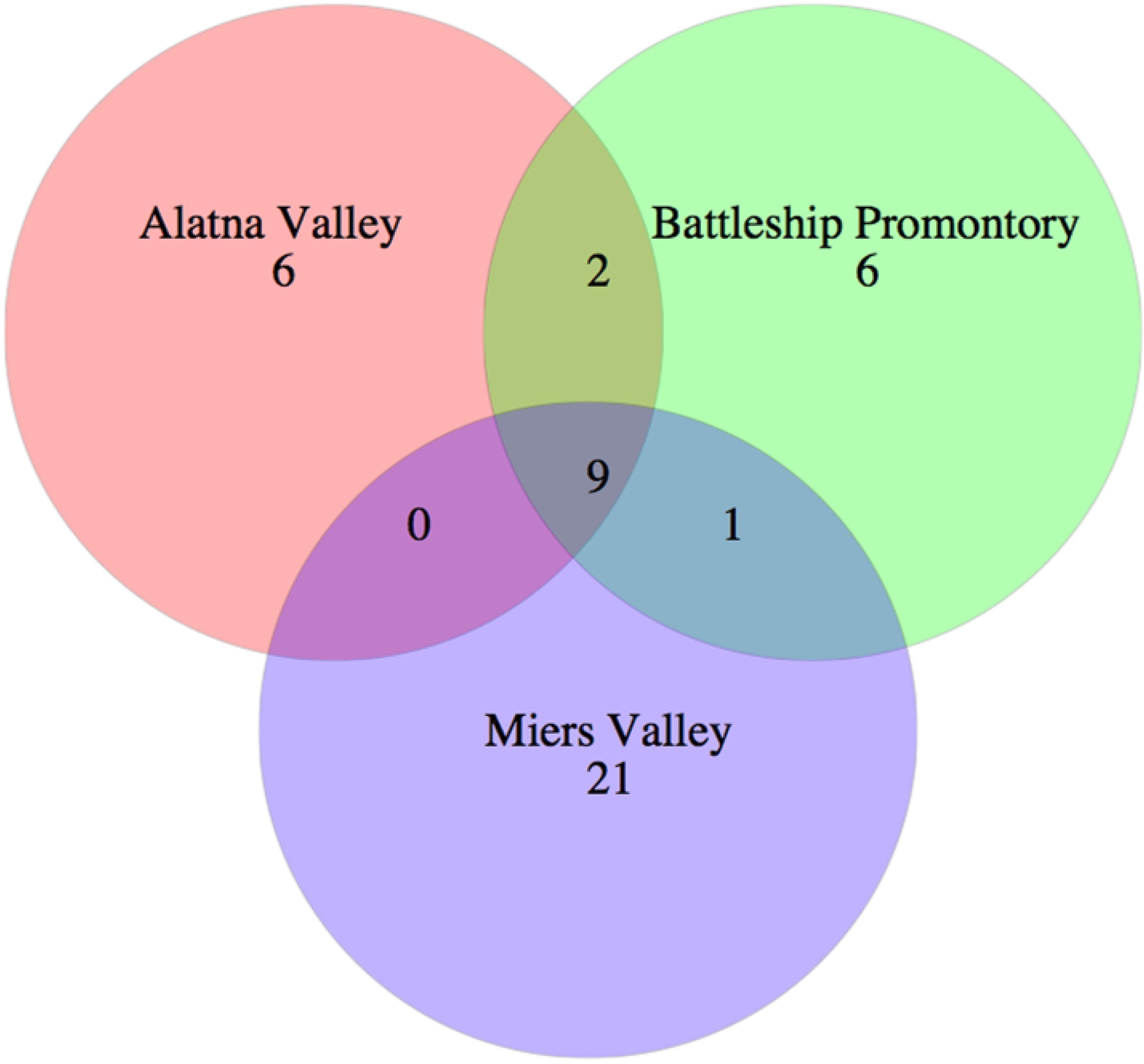
3.4. Biogeography and Local Adaptation
3.5. Environmental Drivers of Fungal Distribution
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflict of Interest
References
- Cary, S.C.; McDonald, I.R.; Barrett, J.E.; Cowan, D.A. On the rocks: The microbiology of Antarctic Dry Valley soils. Nat. Rev. Microbiol. 2010, 8, 129–138. [Google Scholar] [CrossRef]
- Vishniac, H.S. The microbiology of Antarctic soils. In Antarctic Microbiology; Friedmann, E.I., Ed.; Wiley-Liss: New York, NY, USA, 1993; pp. 297–341. [Google Scholar]
- Horowitz, N.H.; Cameron, R.E.; Hubbard, J.S. Microbiology of the Dry Valleys of Antarctica. Science 1972, 176, 242–245. [Google Scholar]
- Wynn-Williams, D.D. Ecological aspects of Antarctic microbiology. In Advances in Microbial Ecology; Marshall, K.C., Ed.; Springer US: New York, NY, USA, 1990; Volume 11, pp. 71–146. [Google Scholar]
- Doran, P.T.; Priscu, J.C.; Lyons, W.B.; Walsh, J.E.; Fountain, A.G.; McKnight, D.M.; Moorhead, D.L.; Virginia, R.A.; Wall, D.H.; Clow, G.D.; et al. Antarctic climate cooling and terrestrial ecosystem response. Nature 2002, 415, 517–520. [Google Scholar] [CrossRef]
- Claridge, G.G.C.; Campbell, I.B. The salts in Antarctic soils, their distribution and relationship to soil processes. Soil Sci. 1977, 123, 377–384. [Google Scholar] [CrossRef]
- Bockheim, J.G. Properties and classification of cold desert soils from Antarctica. Soil Sci. Soc. Am. J. 1997, 61, 224–231. [Google Scholar] [CrossRef]
- Treonis, A.M.; Wall, D.H.; Virginia, R.A. The use of anhydrobiosis by soil nematodes in the Antarctic Dry Valleys. Funct. Ecol. 2000, 14, 460–467. [Google Scholar] [CrossRef]
- Vincent, W.F. Microbial Ecosystems of Antarctica; Cambridge University Press: Cambridge, UK, 1988; p. 59. [Google Scholar]
- Aislabie, J.M.; Chhour, K.L.; Saul, D.J.; Miyauchi, S.; Ayton, J.; Paetzold, R.F.; Balks, M. Dominant bacteria in soils of Marble Point and Wright Valley, Victoria Land, Antarctica. Soil Biol. Biochem. 2006, 38, 3041–3056. [Google Scholar] [CrossRef]
- Poage, M.A.; Barrett, J.E.; Virginia, R.A.; Wall, D.H. The influence of soil geochemistry on nematode distribution, McMurdo Dry Valleys, Antarctica. Arct. Antarct. Alp. Res. 2008, 40, 119–128. [Google Scholar] [CrossRef]
- Priscu, J.C. Ecosystem Dynamics in A Polar Desert: The McMurdo Dry Valleys, Antarctica, 1st ed.; American Geophysical Union: Washington, DC, USA, 1998; Volume 72, p. 369. [Google Scholar]
- Smith, R.C.; Prezelin, B.B.; Baker, K.S.; Bidigare, R.R.; Boucher, N.P.; Coley, T.; Karentz, D.; MacIntyre, S.; Matlick, H.A.; Menzies, D.; et al. Ozone depletion: Ultraviolet radiation and phytoplankton biology in Antarctic waters. Science 1992, 255, 952–959. [Google Scholar]
- Tosi, S.; Brusoni, M.; Zucconi, L.; Vishniac, H. Response of Antarctic soil fungal assemblages to experimental warming and reduction of UV radiation. Polar Biol. 2005, 28, 470–482. [Google Scholar] [CrossRef]
- Lee, C.K.; Barbier, B.A.; Bottos, E.M.; McDonald, I.R.; Cary, S.C. The inter-valley soil comparative survey: The ecology of Dry Valley edaphic microbial communities. ISME J. 2012, 6, 1046–1057. [Google Scholar] [CrossRef]
- Wood, S.A.; Rueckert, A.; Cowan, D.A.; Cary, S.C. Sources of edaphic cyanobacterial diversity in the Dry Valleys of eastern Antarctica. ISME J. 2008, 2, 308–320. [Google Scholar] [CrossRef]
- Niederberger, T.D.; McDonald, I.R.; Hacker, A.L.; Soo, R.M.; Barrett, J.E.; Wall, D.H.; Cary, S.C. Microbial community composition in soils of Northern Victoria Land, Antarctica. Environ. Microbiol. 2008, 10, 1713–1724. [Google Scholar] [CrossRef]
- Pointing, S.B.; Chan, Y.; Lacap, D.C.; Lau, M.C.; Jurgens, J.A.; Farrell, R.L. Highly specialized microbial diversity in hyper-arid polar desert. Proc. Natl. Acad. Sci. USA 2009, 106, 19964–19969. [Google Scholar]
- Wall, D.H.; Virginia, R.A. Controls on soil biodiversity: Insights from extreme environments. Appl. Soil. Ecol. 1999, 13, 137–150. [Google Scholar] [CrossRef]
- Connell, L.; Redman, R.; Craig, S.; Scorzetti, G.; Iszard, M.; Rodriguez, R. Diversity of soil yeasts isolated from South Victoria Land, Antarctica. Microb. Ecol. 2008, 56, 448–459. [Google Scholar] [CrossRef]
- Arenz, B.E.; Blanchette, R.A. Distribution and abundance of soil fungi in Antarctica at sites on the Peninsula, Ross Sea Region and McMurdo Dry Valleys. Soil Biol. Biochem. 2011, 43, 308–315. [Google Scholar] [CrossRef]
- Arenz, B.E.; Held, B.W.; Jurgens, J.A.; Farrell, R.L.; Blanchette, R.A. Fungal diversity in soils and historic wood from the Ross Sea region of Antarctica. Soil Biol. Biochem. 2006, 38, 3057–3064. [Google Scholar] [CrossRef]
- Connell, L.; Redman, R.; Craig, S.; Rodriguez, R. Distribution and abundance of fungi in the soils of Taylor Valley, Antarctica. Soil Biol. Biochem. 2006, 38, 3083–3094. [Google Scholar] [CrossRef]
- Farrell, R.L.; Arenz, B.E.; Duncan, S.M.; Held, B.W.; Jurgens, J.A.; Blanchette, R.A. Introduced and indigenous fungi of the Ross Island historic huts and pristine areas of Antarctica. Polar Biol. 2011, 34, 1669–1677. [Google Scholar] [CrossRef]
- Blanchette, R.A.; Held, B.W.; Arenz, B.E.; Jurgens, J.A.; Baltes, N.J.; Duncan, S.M.; Farrell, R.L. An Antarctic hot spot for fungi at Shackleton's historic hut on Cape Royds. Microb. Ecol. 2010, 60, 29–38. [Google Scholar]
- Duncan, S.M.; Farrell, R.L.; Jordan, N.; Jurgens, J.A.; Blanchette, R.A. Monitoring and identification of airborne fungi at historic locations on Ross Island, Antarctica. Polar Sci. 2010, 4, 275–283. [Google Scholar] [CrossRef]
- Selbmann, L.; de Hoog, G.S.; Mazzaglia, A.; Friedmann, E.I.; Onofri, S. Fungi at the edge of life: Cryptoendolithic black fungi from Antarctic desert. Stud. Mycol. 2005, 51, 1–32. [Google Scholar]
- Coyne, K.J.; Hutchins, D.A.; Hare, C.E.; Cary, S.C. Assessing temporal and spatial variability in Pfiesteria piscicida distributions using molecular probing techniques. Aquat. Microb. Ecol. 2001, 24, 275–285. [Google Scholar] [CrossRef]
- Rueckert, A.; Morgan, H.W. Removal of contaminating DNA from polymerase chain reaction using ethidium monoazide. J. Microbiol. Methods 2007, 68, 596–600. [Google Scholar] [CrossRef]
- Quince, C.; Lanzen, A.; Davenport, R.J.; Turnbaugh, P.J. Removing noise from pyrosequenced amplicons. BMC Bioinform. 2011, 12, 1–18. [Google Scholar] [CrossRef]
- Sun, Y.; Cai, Y.; Liu, L.; Yu, F.; Farrell, M.L.; McKendree, W.; Farmerie, W. Esprit: Estimating species richness using large collections of 16s rRNA pyrosequences. Nucleic Acids Res. 2009, 37, e76. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Kennedy, A.D. Water as a limiting factor in the Antarctic terrestrial environment: A biogeographical synthesis. Arct. Alp. Res. 1993, 25, 308–315. [Google Scholar] [CrossRef]
- McKnight, D.M.; Tate, C.M.; Andrews, E.D.; Niyogi, D.K.; Cozzetto, K.; Welch, K.; Lyons, W.B.; Capone, D.G. Reactivation of a cryptobiotic stream ecosystem in the McMurdo Dry Valleys, Antarctica: A long-term geomorphological experiment. Geomorphology 2007, 89, 186–204. [Google Scholar] [CrossRef]
- Katurji, M.; Zawar-Reza, P.; Zhong, S. Surface layer response to topographic solar shading in Antarctica’s Dry Valleys. J. Geophys. Res. Atmos. 2013, 118, 12332–12344. [Google Scholar] [CrossRef]
- Pollard, W.H.; Lacelle, D.; Davila, A.F.; Andersen, D.; McKay, C.P.; Marinova, M.; Heldmann, J. Ground ice conditions in University Valley, McMurdo Dry Valleys, Antarctica. In Proceedings of the Tenth International Conference on Permafrost (TICOP), Salekhard, Russia, 25–29 June 2012; Volume 1, pp. 305–310.
- Lamsal, K.; Paudyal, G.N.; Saeed, M. Model for assessing impact of salinity on soil water availability and crop yield. Agric. Water Manag. 1999, 41, 57–70. [Google Scholar] [CrossRef]
- Campbell, I.B.; Claridge, G.G.C. Antarctica: Soils, Weathering Processes and Environment; Elsevier Science Publishers: Amsterdam, The Netherlands, 1987; Volume 16, p. 406. [Google Scholar]
- Duncan, S.M.; Farrell, R.L.; Thwaites, J.M.; Held, B.W.; Arenz, B.E.; Jurgens, J.A.; Blanchette, R.A. Endoglucanase-producing fungi isolated from Cape Evans historic expedition hut on Ross Island, Antarctica. Environ. Microbiol. 2006, 8, 1212–1219. [Google Scholar] [CrossRef]
- Lawley, B.; Ripley, S.; Bridge, P.; Convey, P. Molecular analysis of geographic patterns of eukaryotic diversity in Antarctic soils. Appl. Environ. Microbiol. 2004, 70, 5963–5972. [Google Scholar] [CrossRef]
- Vishniac, H.S. Cryptococcus socialis sp. nov. and Cryptococcus consortionis sp. nov., Antarctic Basidioblastomycetes. Int. J. Syst. Bacteriol. 1985, 35, 119–122. [Google Scholar] [CrossRef]
- Simmons, D.R.; James, T.Y.; Meyer, A.F.; Longcore, J.E. Lobulomycetales, a new order in the Chytridiomycota. Mycol. Res. 2009, 113, 450–460. [Google Scholar] [CrossRef]
- De Hoog, G.S.; Vicente, V.A.; Najafzadeh, M.J.; Harrak, M.J.; Badali, H.; Seyedmousavi, S. Waterborne Exophiala species causing disease in cold-blooded animals. Persoonia 2011, 27, 46–72. [Google Scholar] [CrossRef]
- Kantvilas, G.; Seppelt, R.D. Polysporina frigida sp. Nov. from Antarctica. Lichenologist 2006, 38, 109–113. [Google Scholar] [CrossRef]
- Slemmons, C.; Johnson, G.; Connell, L.B. Application of an automated ribosomal intergenic spacer analysis database for identification of cultured Antarctic fungi. Antarct. Sci. 2013, 25, 44–50. [Google Scholar] [CrossRef]
- Kerry, E. Effects of temperature on growth rates of fungi from Subantarctic Macquarie Island and Casey, Antarctica. Polar Biol. 1990, 10, 293–299. [Google Scholar]
- Marshall, W.A. Seasonality in Antarctic airborne fungal spores. Appl. Environ. Microbiol. 1997, 63, 2240–2245. [Google Scholar]
- Adams, B.J.; Bargett, R.D.; Ayres, E.; Wall, D.H.; Aislabie, J.; Bamforth, S.; Bargagli, R.; Cary, C.; Cavacini, P.; Conell, L.; et al. Diversity and distribution of Victoria Land biota. Soil. Biol. Biochem. 2006, 38, 3003–3018. [Google Scholar] [CrossRef]
- Fletcher, L.D.; Kerry, E.J.; Weste, G.M. Microfungi of MacRobertson and Enderby Iands, Antarctica. Polar Biol. 1985, 4, 81–88. [Google Scholar] [CrossRef]
- Vincent, W.F. Evolutionary origins of Antarctic microbiota: Invasion, selection and endemism. Antarct. Sci. 2000, 12, 374–385. [Google Scholar] [CrossRef]
- Marshall, W.A. Biological particles over Antarctica. Nature 1996, 383, 680. [Google Scholar] [CrossRef]
- Rao, S.; Chan, Y.; Lacap, D.C.; Hyde, K.D.; Pointing, S.B.; Farrell, R.L. Low-diversity fungal assemblage in an Antarctic Dry Valleys soil. Polar Biol. 2012, 35, 567–574. [Google Scholar] [CrossRef] [Green Version]
- Green, J.L.; Holmes, A.J.; Westoby, M.; Oliver, I.; Briscoe, D.; Dangerfield, M.; Gillings, M.; Beattie, A.J. Spatial scaling of microbial eukaryote diversity. Nature 2004, 432, 747–750. [Google Scholar] [CrossRef]
- Convey, P. The influence of envrionmental characteristics on life history attributes of Antarctic terrestrial biota. Biol. Rev. 1996, 71, 191–225. [Google Scholar] [CrossRef]
- Hogg, I.D.; Cary, C.S.; Convey, P.; Newsham, K.K.; O’Donnell, A.G.; Adams, B.J.; Aislabie, J.; Frati, F.; Stevens, M.I.; Wall, D.H. Biotic interactions in Antarctic terrestrial ecosystems: Are they a factor? Soil. Biol. Biochem. 2006, 38, 3035–3040. [Google Scholar] [CrossRef]
- Hopkins, D.W.; Sparrow, A.D.; Novis, P.M.; Gregorich, E.G.; Elberling, B.; Greenfield, L.G. Controls on the distribution of productivity and organic resources in Antarctic Dry Valley soils. Proc. R. Soc. Sci. B 2006, 273, 2687–2695. [Google Scholar] [CrossRef]
- Barrett, J.E.; Virginia, R.A.; Wall, D.H.; Cary, S.C.; Adams, B.J.; Hacker, A.L. Co-variation in soil biodiversity and biogeochemistry in Northern and Southern Victoria Land, Antarctica. Antarct. Sci. 2006, 18, 535–548. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dreesens, L.L.; Lee, C.K.; Cary, S.C. The Distribution and Identity of Edaphic Fungi in the McMurdo Dry Valleys. Biology 2014, 3, 466-483. https://doi.org/10.3390/biology3030466
Dreesens LL, Lee CK, Cary SC. The Distribution and Identity of Edaphic Fungi in the McMurdo Dry Valleys. Biology. 2014; 3(3):466-483. https://doi.org/10.3390/biology3030466
Chicago/Turabian StyleDreesens, Lisa L., Charles K. Lee, and S. Craig Cary. 2014. "The Distribution and Identity of Edaphic Fungi in the McMurdo Dry Valleys" Biology 3, no. 3: 466-483. https://doi.org/10.3390/biology3030466
APA StyleDreesens, L. L., Lee, C. K., & Cary, S. C. (2014). The Distribution and Identity of Edaphic Fungi in the McMurdo Dry Valleys. Biology, 3(3), 466-483. https://doi.org/10.3390/biology3030466




