Application of Electro-Technologies in Processing of Flax Fiber
Abstract
:1. Introduction
2. Structure of Natural Agricultural Fibers
| Type of fiber | Cellulose (%) | Lignin (%) | Hemicellulose (%) | Pectin (%) | Ash (%) |
|---|---|---|---|---|---|
| Fiber flax | 71 | 2.2 | 18.6– 20.6 | 2.3 | – |
| Seed flax | 43–47 | 21–23 | 24–26 | – | 5 |
| Kenaf | 31–57 | 15–19 | 21.5–23 | – | 2–5 |
| Jute | 45–71.5 | 12–26 | 13.6–21 | 0.2 | 0.5–2 |
| Hemp | 57–77 | 3.7–13 | 14–22.4 | 0.9 | 0.8 |
| Ramie | 68.6–91 | 0.6–0.7 | 5–16.7 | 1.9 | – |
3. Retting
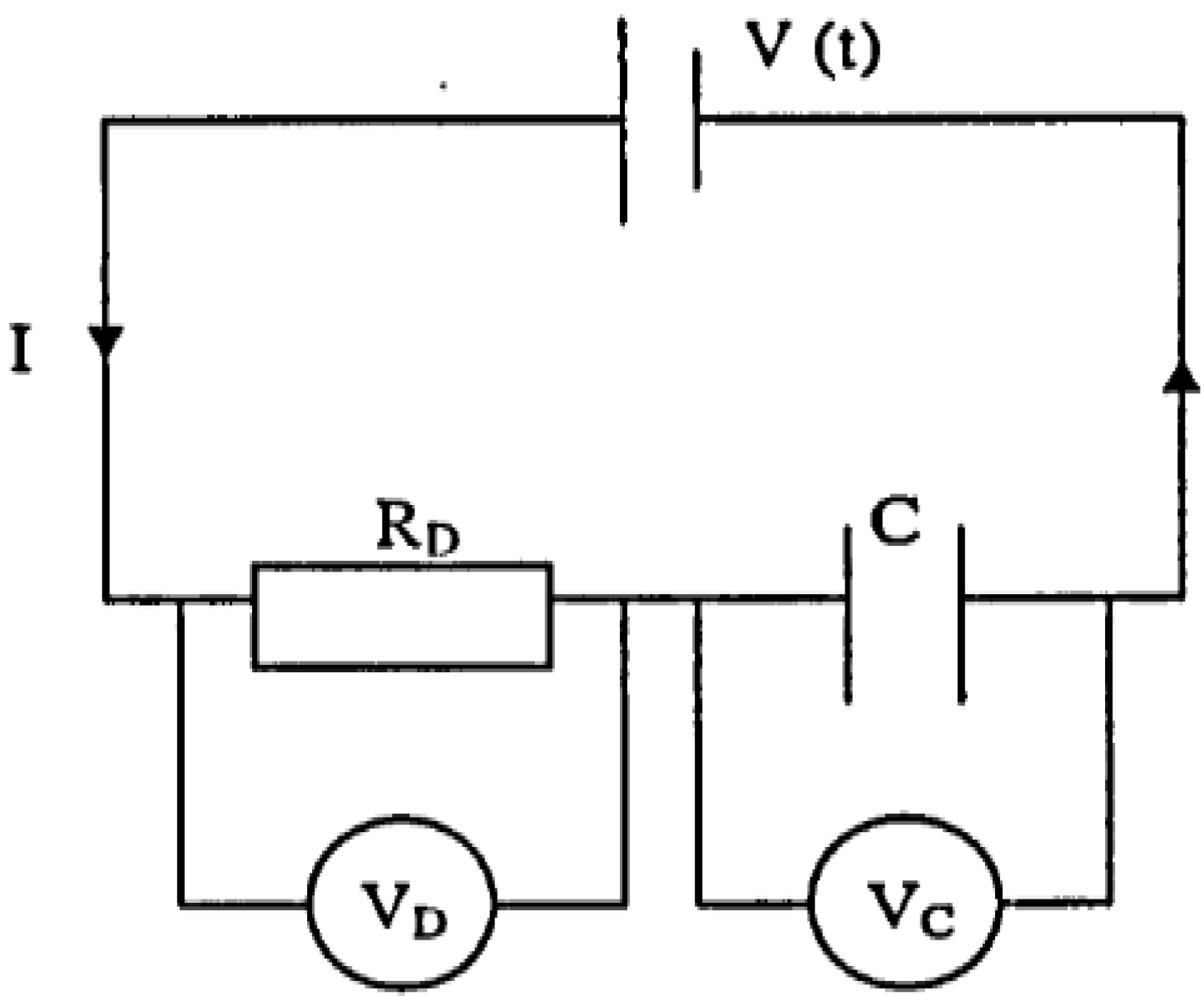
4. Role of Electromagnetic Energy in Retting of Flax Stem
wherej= √ − 1
5. Microwave Assisted Retting Process of Flax
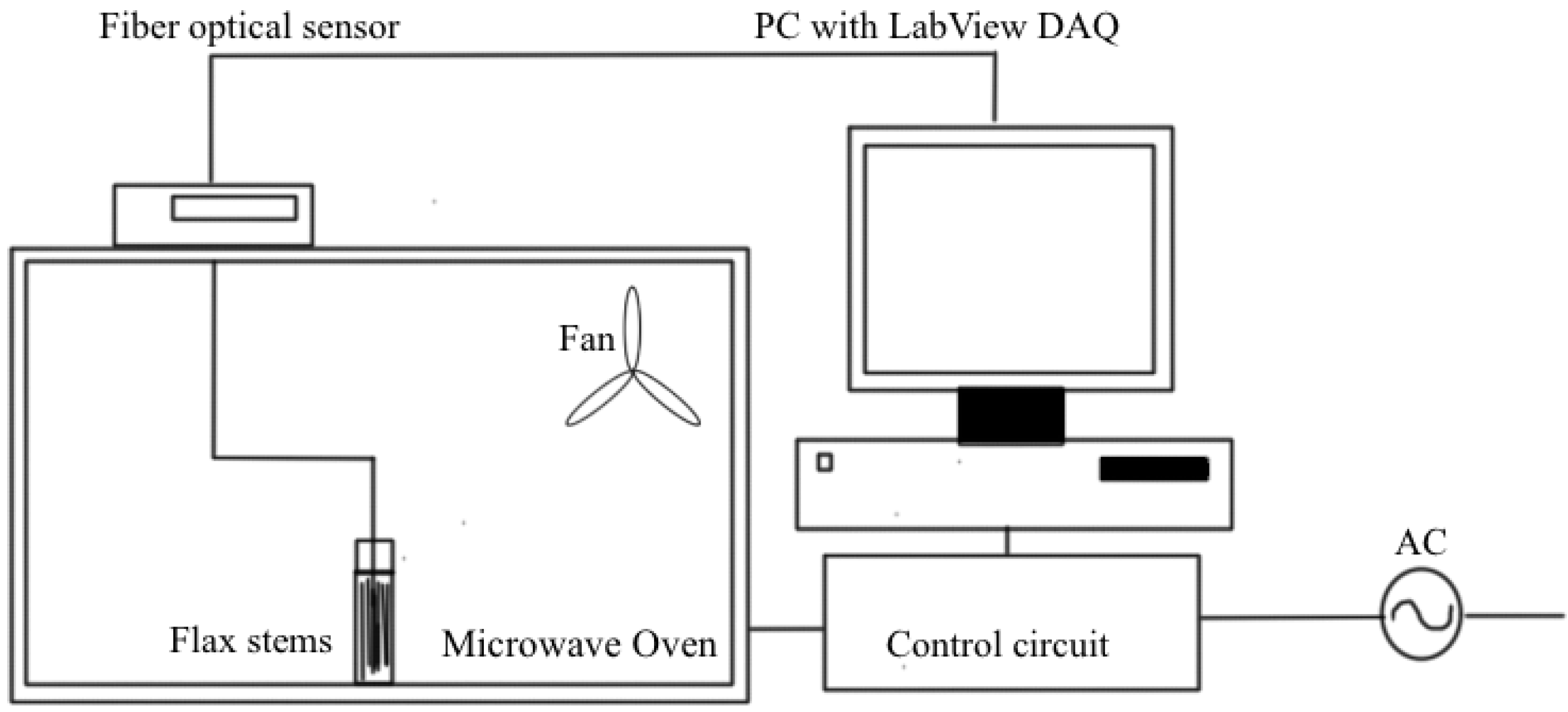
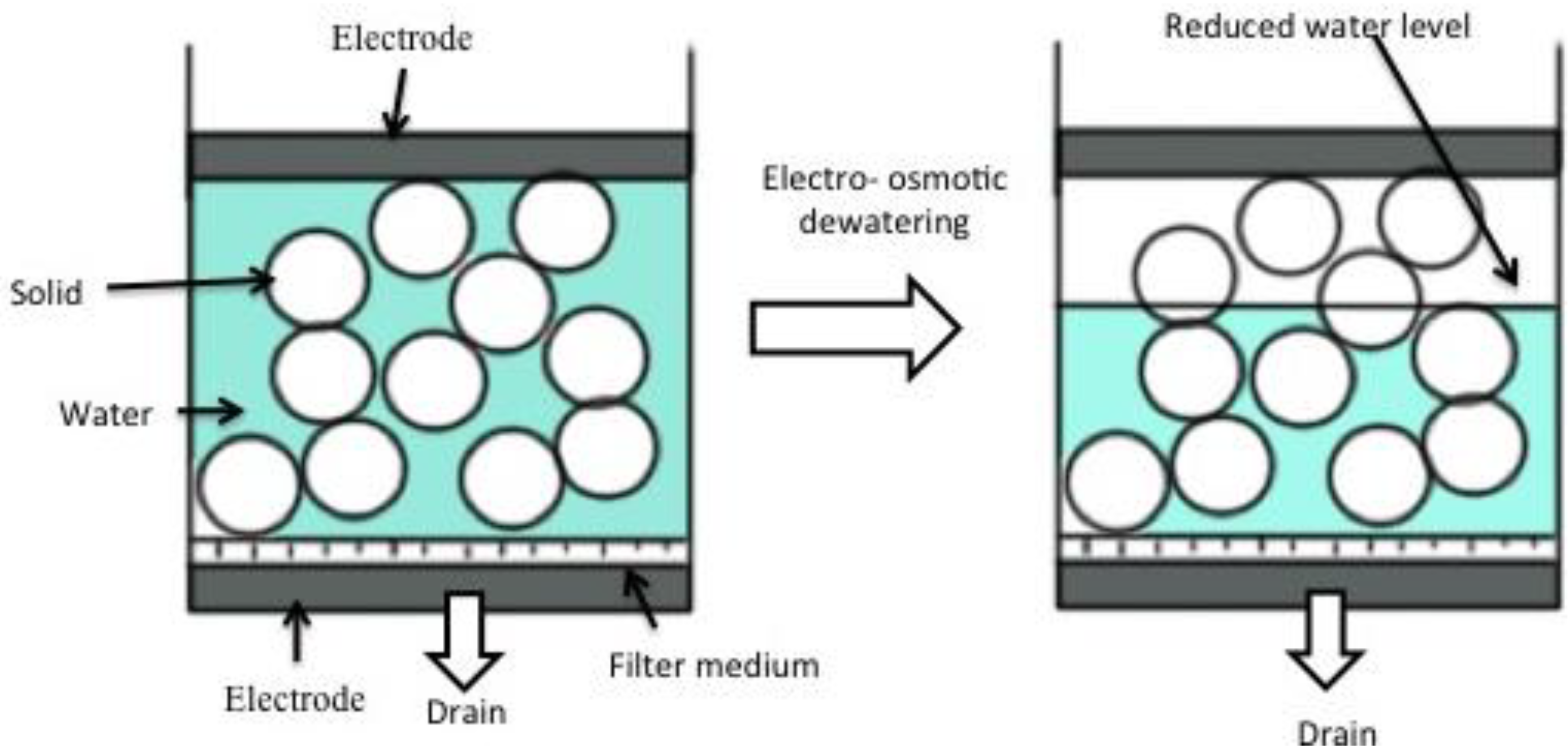
6. Electro-Osmotic Dewatering of Retted Flax Stem
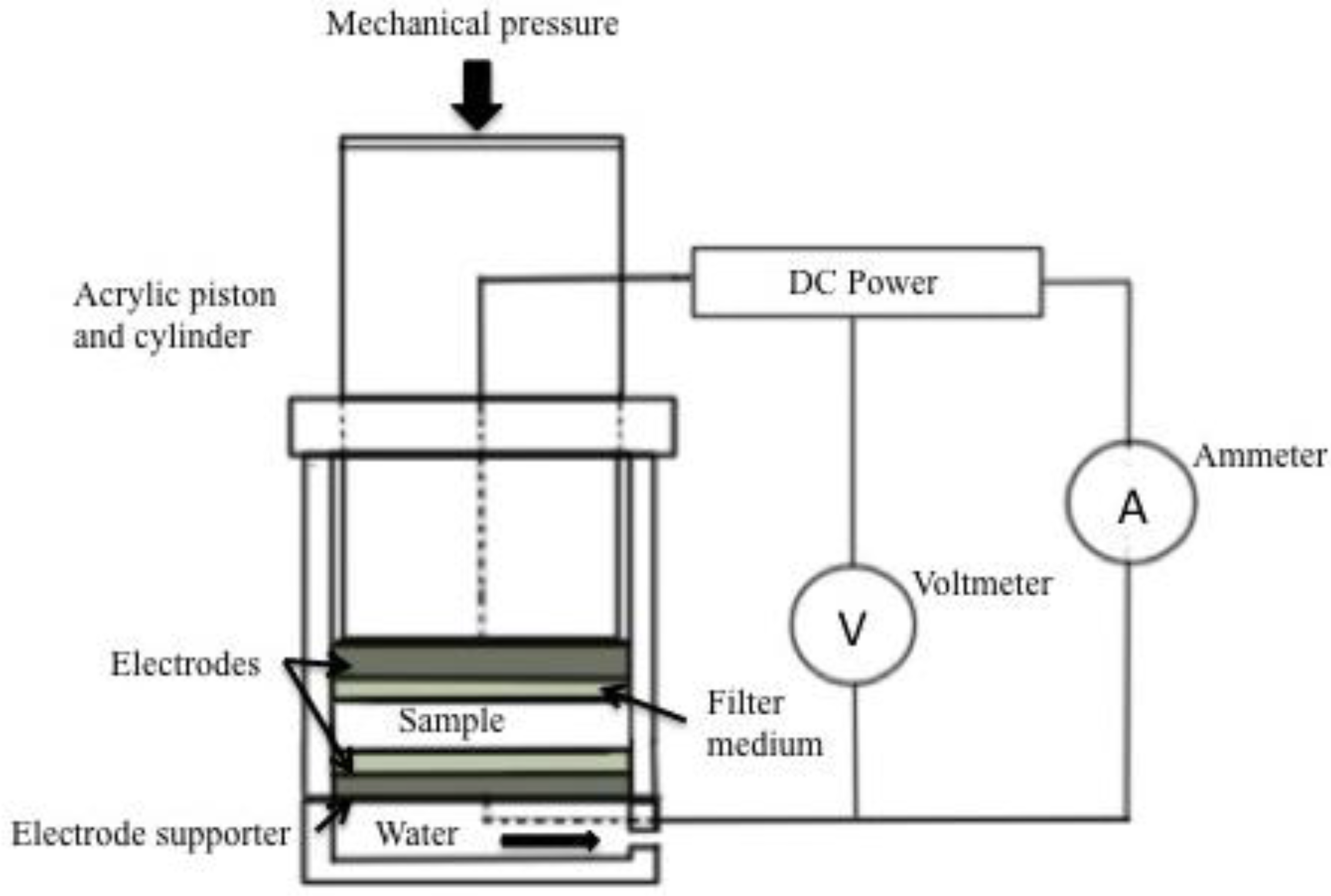
6.1. Design of Electro-Osmotic Dewatering Apparatus
- The quantity of flax to be dewatered.
- Zeta potential of the flax-water mixture (The electric potential between the solid and liquid components, which is a function of the geometry, the pH, and the ion concentration of the substance)
- The pressure applied (combination of pressure and electro-osmosis is proved to be more economical than electro-osmosis alone [34].
- Type of membrane used.
Vol = (SD2−SD1)M/SD2
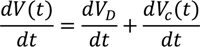
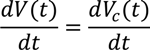
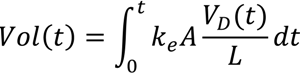
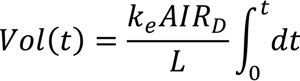
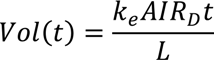
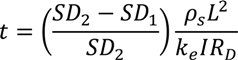
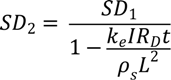
6.2. Design Parameters for Dewatering of Flax
6.2.1. Sample Thickness
6.2.2. Applied Current
6.2.3. Power Supply
6.2.4. Pressure
6.2.5. Type of Electrodes
6.2.6. Microwave-Drying of Flax Straw
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Akin, D.E.; Gamble, G.R.; Morrison, W.H., III; Rigsby, L.L.; Dodd, R.B. Chemical and structural analysis of fiber and core tissues from flax. J. Sci. Food Agric. 1996, 72, 155–165. [Google Scholar] [CrossRef]
- Thomsen, A.B.; Thygesen, A.; Bohnc, V.; Vad Nielsen, K.; Pallesen, B.; Jørgensen, M.S. Effects of chemical–physical pre-treatment processes on hemp fibers for reinforcement of composites and for textiles. Ind. Crops Prod. 2006, 24, 113–118. [Google Scholar] [CrossRef]
- Hegi, G. Illustrierte Flora Von Mitteleuropa; Lehmanns Verlag: Munich, Germany, 1925; Volume 5, pp. 3–38. [Google Scholar]
- Sharma, H.S.S. Chemical retting of flax using chelating compounds. Ann. Appl. Biol. 1988, 113, 159–165. [Google Scholar] [CrossRef]
- Atton, M. Flax Culture from Flower to Fabric; The Ginger Press: Owen Sound, ON, USA, 1989. [Google Scholar]
- Nair, G.R.; Li, Z.; Gariepy, Y.; Raghavan, G.S.V. Microwave drying of corn (Zea mays L. ssp.) for the seed industry. Dry. Technol. 2011, 29, 1291–1296. [Google Scholar] [CrossRef]
- Van Sumere, C.F. Retting of Flax with Special Reference to Enzyme Retting. In The Biology and Processing of Flax; Sharma, H.S.S., Van Sumere, C.F., Eds.; M Publications: Belfast, Northern Ireland, 1992; pp. 157–198. [Google Scholar]
- Baley, C. Analysis of the flax fibres tensile behaviour and analysis of the tensile stiffness increase. Composites A 2002, 33, 939–948. [Google Scholar] [CrossRef]
- Akin, D.E.; Henriksson, G.; Evans, J.D.; Adamsen, A.P.S.; Foulk, J.A.; Dodd, R.B. Progress in enzyme-retting of flax. J. Nat. Fibers 2004, 1, 21–47. [Google Scholar] [CrossRef]
- Thygsen, L.G.; Bilde-Dorensen, J.B.; Hoffmeyer, P. Visualization of dislocations in hemp fibers—A comparison between scanning electron microscopy and polarized light microscopy. Ind. Crops Prod. 2006, 24, 181–185. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Misra, M.; Hinrichsen, G. Biofibers, biodegradable polymers and biocomposites: An overview. Macromol. Mater. Eng 2000, 276–277, 1–24. [Google Scholar] [CrossRef]
- Akin, D.E.; Morrison, H., III; Rigsby, L.L.; Evans, J.D.; Foulk, J.A. Influence of water pre-soak on enzyme-retting of flax. Ind. Crops Prod. 2003, 17, 149–159. [Google Scholar] [CrossRef]
- Uludag, S.; Loha, V.; Prokop, A.; Tanner, R. The effect of fermentation (retting) time and harvest time on kudzu (Pueraria lobata) fiber strength. Appl. Biochem. Biotechnol. 1996, 57–58, 75–84. [Google Scholar] [CrossRef]
- Foulk, J.A.; Akin, D.E.; Dodd, R.B. Processing techniques for improving enzyme-retting of flax. Ind. Crops Prod. 2001, 13, 239–248. [Google Scholar] [CrossRef]
- Pallesen, B.E. The quality of combine-harvested fiber flax for industrials purposes depends on the degree of retting. Ind. Crops Prod. 1996, 5, 65–78. [Google Scholar] [CrossRef]
- Adamsen, A.P.S.; Akin, D.E.; Rigsby, L.L. Chemical retting of flax straw under alkaline conditions. Text. Res. J. 2002, 72, 789–794. [Google Scholar] [CrossRef]
- Adamsen, A.P.S.; Akin, D.E.; Rigsby, L.L. Chelating agents and enzyme retting of flax. Text. Res. J. 2002, 72, 296–302. [Google Scholar] [CrossRef]
- Kessler, R.W.; Becker, U.; Kohler, R.; Goth, B. Steam explosion of flax—A superior technique for upgrading fiber value. Biomass Bioenergy 1998, 14, 237–249. [Google Scholar] [CrossRef]
- Nuchter, M.; Ondruschka, B.; Bonrath, W.; Gum, A. Microwave assisted synthesis—A critical technology overview. Green Chem. 2004, 6, 128–141. [Google Scholar] [CrossRef]
- Galema, S.A. Microwave Chemistry. Chem. Soc. Rev. 1997, 26, 233–238. [Google Scholar] [CrossRef]
- Sridar, V. Microwave radiation as a catalyst for chemical reactions. Curr. Sci. 1998, 74, 446–450. [Google Scholar]
- Steel, B.C.; Bilek, M.M.; McKenzie, D.R.; dos Remedios, C.G. A technique for micro second heating and cooling of a thin biological sample. Eur. Biophys. J. 2002, 31, 378–382. [Google Scholar]
- George, D.F.; Bilek, M.M.; McKenzie, D.R. Non-thermal effects in the microwave induced unfolding of proteins observed by chaperone binding. Bioelectromagnetics 2008, 29, 324–330. [Google Scholar] [CrossRef]
- Sato, J. Observation of the Coronal Hard X-ray Sources. Astrophys. J. 2001, 558, L137–L140. [Google Scholar] [CrossRef]
- Banik, S.; Bandyopadhyay, S.; Ganguly, S. Bioeffects of microwave—A brief review. Bioresour. Technol. 2003, 87, 155–159. [Google Scholar] [CrossRef]
- Oliveira, M.E.C.; Franca, A.S. Microwave heating of foodstuff. J. Food Eng. 2002, 53, 347–359. [Google Scholar] [CrossRef]
- Tsubaki, S.; Azuma, J. Application of Microwave Technology for Utilization of Recalcitrant Biomass. In Advances in Induction and Microwave Heating of Mineral and Organic Materials; InTech: Kyoto University, Japan, 2011; ISBN: 978-953-307-522-8. [Google Scholar]
- Nair, G.R.; Liplap, P.; Gariepy, Y.; Raghavan, G.S.V. Microwave drying of flax fiber at controlled temperature. J. Agric. Sci. Technol. 2011, 1, 1103–1115. [Google Scholar]
- Budarin, V.L.; Clark, J.H.; Lanigan, B.A.; Shuttleworth, P.; Macquarrie, D.J. Microwave assisted decomposition of cellulose: A new thermochemical route for biomass exploitation. Bioresour. Technol. 2010, 101, 3776–3779. [Google Scholar]
- Mohanty, A.K.; Misra, M.; Drzal, L.T. Surface modifications of natural fibers and performance of resulting biocomposite. Compos. Interface 2001, 8, 313–343. [Google Scholar] [CrossRef]
- Schwartzbcrg, H.G.; Roseneau, J.R.; Richardson, G. The Removal of Water by Expression. In Water Removal Processes: Drying and Concentration of Food and Other Materials; AlCHE Symposium Series; King, C.J., Clark, J.P., Eds.; American Institute of Chemical Engineers: New York, NY, USA, 1977; Volume 73. [Google Scholar]
- Lightfoot, D.G.; Raghavan, G.S.V. Combined fields dewatering of seaweed (Nereocystis luetkeana). Trans. ASABE 1994, 37, 899–906. [Google Scholar]
- Orsat, V.; Raghavan, G.S.V.; Sotorinal, S.; Lightfoot, D.G.; Gopalakrishnan, S. Roller press for electro- osmotic dewatering of biomaterials. Dry. Technol. 1999, 17, 523–538. [Google Scholar] [CrossRef]
- Mujumdar, A.S.; Yoshida, H. Electro-Osmotic Dewatering (EOD) of Bio-Materials. In Electro-Technologies for Extraction from Food Plants and Biomaterials; Springer: New York, NY, USA, 2008; pp. 121–154. [Google Scholar]
- Yoshida, H. Practical aspects of dewatering enhanced by electro-osmosis. Dry. Technol. 1993, 11, 787–814. [Google Scholar] [CrossRef]
- Yoshida, H.; Yukawa, H. A theoretical analysis of the electroosmotic dewatering of sludge. Inter. Chem. Eng. 1988, 28, 477–485. [Google Scholar]
- Yoshida, H.; Yukawa, H. Analysis of dewatering processes enhanced by electro-osmosis. Fluid/Part. Sep. J. 1991, 4, 1–7. [Google Scholar]
- Yoshida, H.; Yukawa, H. Analysis of Electro-Osmotically Enhanced Sludge Dewatering. In Advances in Drying; Mujumdar, A.S., Ed.; Hemisphere: Bristol, UK, 1992; Volume 5, pp. 301–323. [Google Scholar]
- Lockhart, N.C. Combined field dewatering: Bridging the science-industry gap. Dry. Technol. 1992, 10, 839–874. [Google Scholar] [CrossRef]
- Rampacek, C. Electro-Osmotic and Electro-Phoretic Dewatering as Applied to Solid Liquid Separation. In Solid-Liquid Separation: A Review and a Bibliography; Poole, J.B., Doyle, D., Eds.; Her Majesty’s Stationery Office: London, UK, 1966; pp. 100–108. [Google Scholar]
- Mitchell, J.K. Conducting Phenomena: From theory to geotechnical practice. Geotechnique 1991, 41, 299–340. [Google Scholar] [CrossRef]
- Mok, C.K. Design and Modelling of Electroosmotic Dewatering. Ph.D. Thesis, University of Newcastle upon Tyne, England, UK, 2006. [Google Scholar]
- Grundl, T.; Michalski, P. Electro-osmotically driven water flow in sediments. Water Res. 1996, 30, 811–818. [Google Scholar] [CrossRef]
- Vijh, A.K.; Novak, J.P. A new theoretical approach to electro-osmotic dewatering based on non-equilibrium thermodynamics. Dry. Technol. 1997, 15, 699–709. [Google Scholar] [CrossRef]
- Keshwani, D.; Cheng, J.J. Switchgrass for bioethanol and other value-added applications: A review. Bioresour. Technol. 2009, 100, 1515–1522. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nair, G.R.; Rho, D.; Raghavan, G.S.V. Application of Electro-Technologies in Processing of Flax Fiber. Fibers 2013, 1, 21-35. https://doi.org/10.3390/fib1020021
Nair GR, Rho D, Raghavan GSV. Application of Electro-Technologies in Processing of Flax Fiber. Fibers. 2013; 1(2):21-35. https://doi.org/10.3390/fib1020021
Chicago/Turabian StyleNair, Gopu R., Denis Rho, and G. S. Vijaya Raghavan. 2013. "Application of Electro-Technologies in Processing of Flax Fiber" Fibers 1, no. 2: 21-35. https://doi.org/10.3390/fib1020021




