Optimization Studies of Photocatalytic Tungsten-Doped Titania Coatings Deposited by Reactive Magnetron Co-Sputtering
Abstract
:1. Introduction
2. Experimental Section
2.1. Sample Preparation
2.2. Coating Characterization
2.3. Evaluation of Photocatalytic Activity
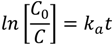
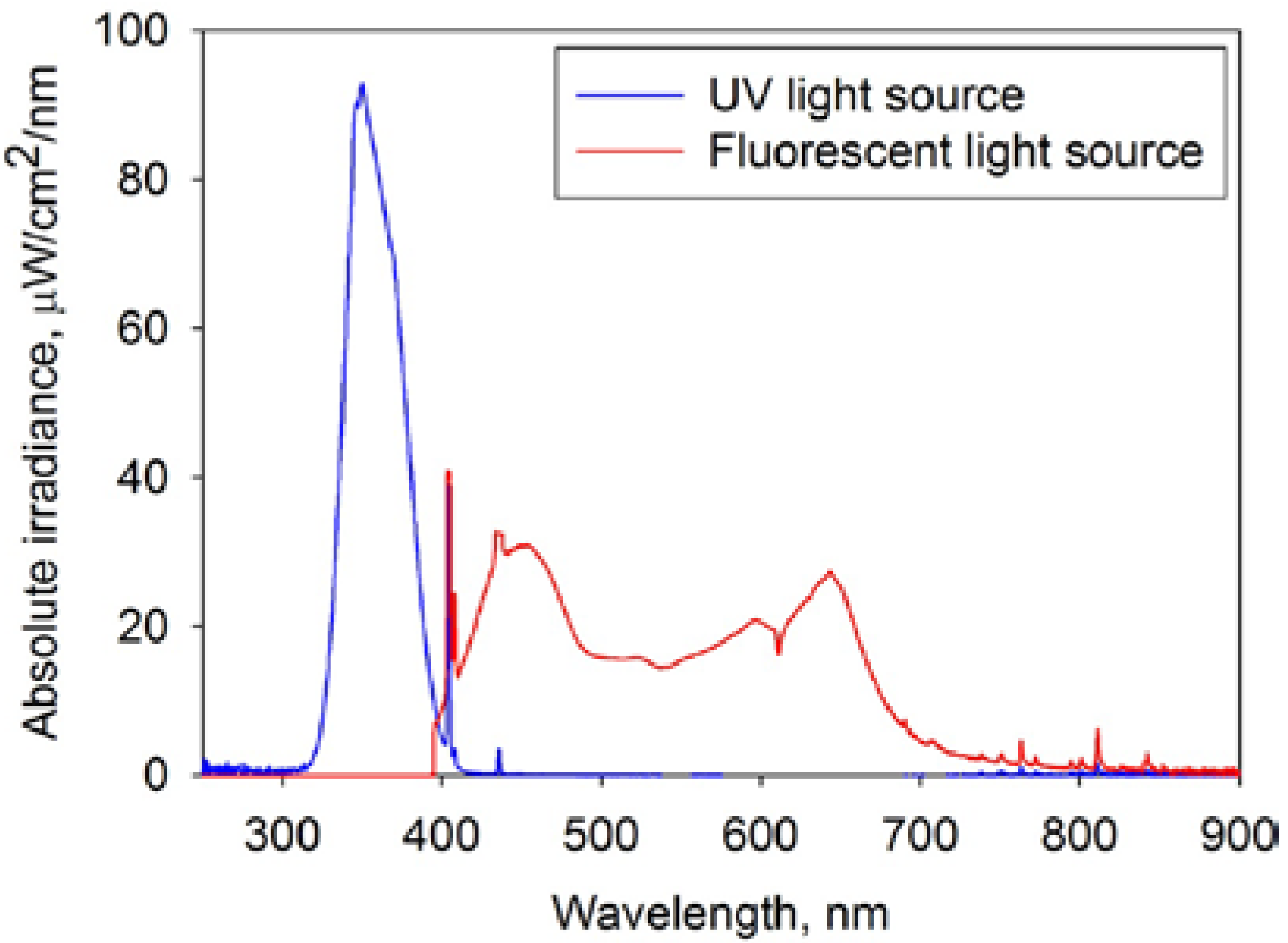
3. Results and Discussion
3.1. Coating Composition and Thickness
| Coating ID | Power applied on tungsten target, W | Tungsten content, at.% | Coating thickness, nm |
|---|---|---|---|
| W60 | 60 | 3.83 | 702 |
| W70 | 70 | 4.64 | 746 |
| W80 | 80 | 5.89 | 758 |
| W90 | 90 | 7.09 | 793 |
| W100 | 100 | 10.03 | 814 |
| W150 | 150 | 13.87 | 889 |
| W180 | 180 | 15.84 | 896 |
3.2. Raman Spectroscopy Results

3.3. XRD Results
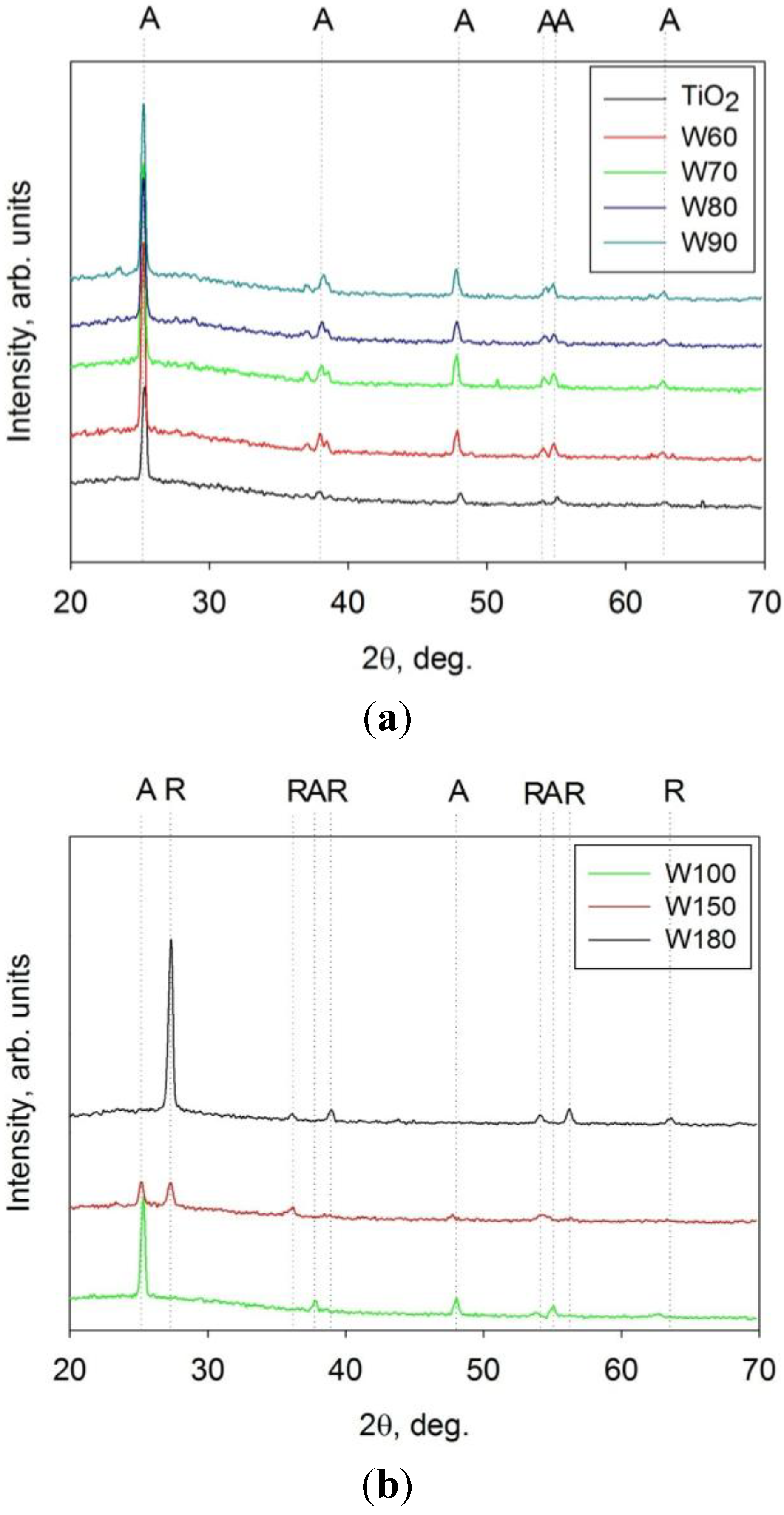
3.4. Band Gap Calculation
| Coating ID | Band gap, eV | ka × 105, s−1 UV light | ka × 105, s−1 Fluorescent light | Surface roughness, μm | Surface area, μm2 | Crystal structure |
|---|---|---|---|---|---|---|
| TiO2 | 3.16 | 1.7 | 0.6 | 0.0114 | 5523 | anatase |
| W60 | 3.12 | 4.0 | 1.0 | 0.0491 | 5620 | anatase |
| W70 | 3.09 | 5.6 | 1.2 | 0.0309 | 5614 | anatase |
| W80 | 3.09 | 9.9 | 2.7 | 0.0218 | 5602 | anatase |
| W90 | 3.05 | 6.4 | 2.1 | 0.0161 | 5554 | anatase |
| W100 | 3.02 | 2.2 | 1.6 | 0.0144 | 5535 | anatase |
| W150 | 3.00 | 1.4 | 0.8 | 0.0138 | 5525 | anatase/rutile |
| W180 | 2.98 | 0.9 | 0.6 | 0.0152 | 5520 | rutile |
3.5. Morphological Properties

3.6. TEM Results
3.7. Photocatalytic Activity Measurements

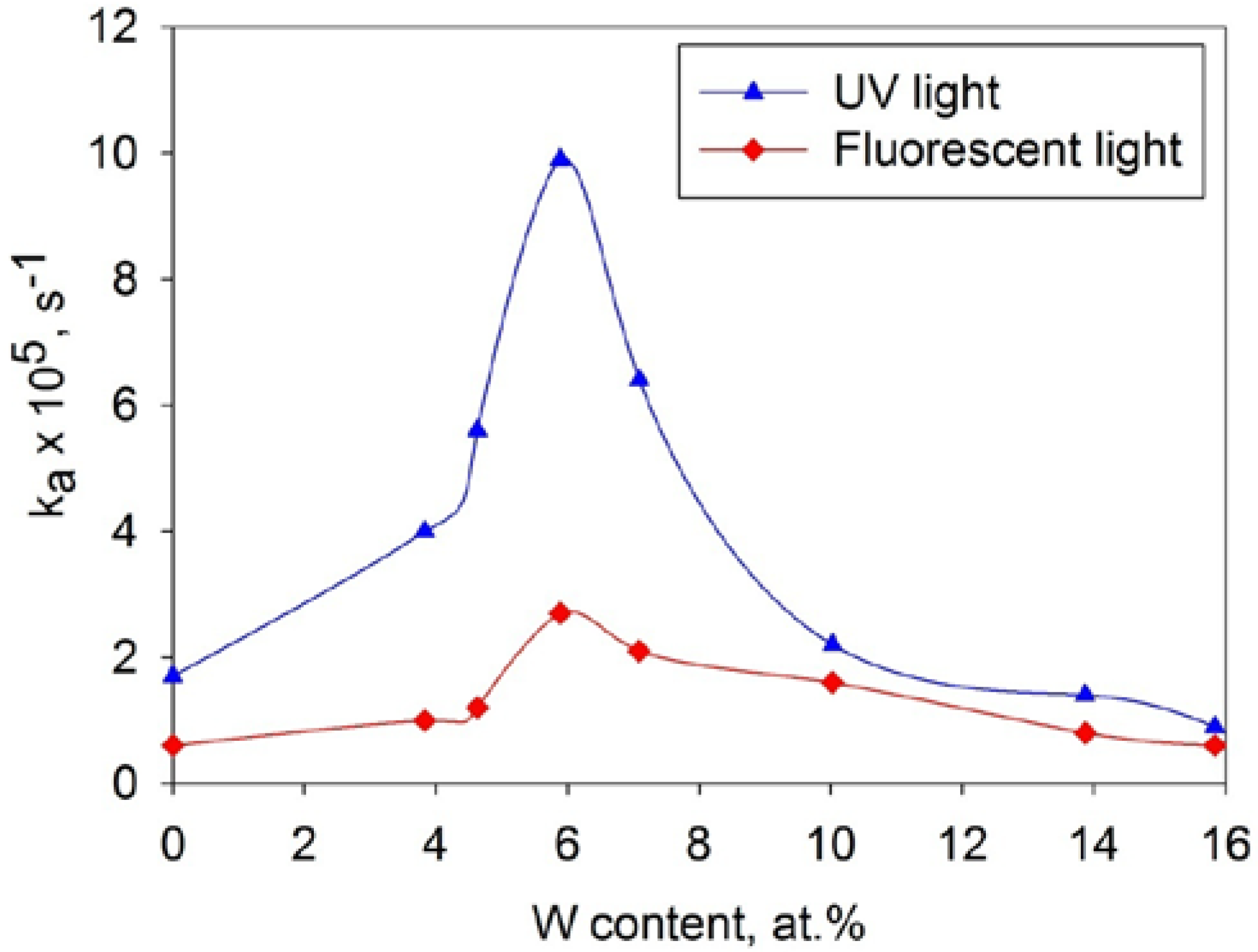

4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Rauf, M.A.; Meetani, M.A.; Hisaindee, S. An overview on the photocatalytic degradation of azo dyes in the presence of TiO2 doped with selective transition metals. Desalination 2011, 276, 13–27. [Google Scholar] [CrossRef]
- Ratova, M.; Kelly, P.J.; West, G.T.; Iordanova, I. Enhanced properties of magnetron sputtered photocatalytic coatings via transition metal doping. Surf. Coat. Technol. 2013, 228, S544–S549. [Google Scholar] [CrossRef]
- Farahani, N.; Kelly, P.J.; West, G.; Ratova, M.; Hill, C.; Vishnyakov, V. Photocatalytic activity of reactively sputtered and directly sputtered titania coatings. Thin Solid Films 2011, 520, 1464–1469. [Google Scholar] [CrossRef]
- Yao, M.; Chen, J.; Zhao, C.; Chen, Y. Photocatalytic activities of ion doped TiO2 thin films when prepared on different substrates. Thin Solid Films 2009, 517, 5994–5999. [Google Scholar] [CrossRef]
- Lorret, O.; Francová, D.; Waldner, G.; Stelzer, N. W-doped titania nanoparticles for UV and visible-light photocatalytic reactions. Appl. Cat. B Environ. 2009, 91, 39–46. [Google Scholar] [CrossRef]
- Kelly, P.J.; Beevers, C.F.; Henderson, P.S.; Arnell, R.D.; Bradley, J.W.; Bäcker, H. A comparison of the properties of titanium-based films produced by pulsed and continuous DC magnetron sputtering. Surf. Coat. Technol. 2003, 174–175, 795–800. [Google Scholar] [CrossRef]
- Kubacka, A.; Colón, G.; Fernández-García, M. Cationic (V, Mo, Nb, W) doping of TiO2-anatase: A real alternative for visible light-driven photocatalysts. Cat. Today 2009, 143, 286–292. [Google Scholar] [CrossRef]
- Abdullah, M.A.; Chong, F.K. Dual-effects of adsorption and photodegradation of methylene blue by tungsten-loaded titanium dioxide. Chem. Eng. J. 2010, 158, 418–425. [Google Scholar] [CrossRef]
- Rampaul, A.; Parkin, I.P.; O’Neill, S.A.; DeSouza, J.; Mills, A.; Elliott, N. Titania and tungsten doped titania thin films on glass; active photocatalysts. Polyhedron 2003, 22, 35–44. [Google Scholar] [CrossRef]
- Li, X.Z.; Li, F.B.; Yang, C.L.; Ge, W.K. Photocatalytic activity of WOx-TiO2 under visible light irradiation. J. Photochem. Photobiol. A Chem. 2001, 141, 209–217. [Google Scholar] [CrossRef]
- ISO10678, Fine Ceramics, Advanced Technical Ceramics—Determination of Photocatalytic Activity of Surfaces in an Aqueous Medium by Degradation of Methylene Blue; ISO: Geneva, Switzerland, 2010.
- Stengl, V.; Bakardjieva, S.; Murafa, N. Preparation and photocatalytic activity of rare earth doped TiO2 nanoparticles. Mat. Chem. Phys. 2009, 114, 217–226. [Google Scholar] [CrossRef]
- Abdullah, M.A.; Chong, F.K. Preparation and characterization of tungsten-loaded titanium dioxide photocatalyst for enhanced dye degradation. J. Hazard. Mat. 2010, 176, 451–458. [Google Scholar] [CrossRef]
- Fuerte, A.; Hernandez-Alonso, M.D.; Maira, A.J.; Martinez-Arias, A.; Fernandez-Garcia, M.; Conesa, J.C.; Soria, J. Visible light-activated nanosized doped-TiO2 photocatalysts. Chem. Commun. 2001, 2718–2719. [Google Scholar]
- Tauc, J.; Grigorovici, R.; Vancu, A. Optical properties and electronic structure of amorphous germanium. Phys. Stat. Solid. B 1966, 15, 627–637. [Google Scholar] [CrossRef]
- Kiriakidou, F.; Kondarides, D.I.; Verykios, X.E. The effect of operational parameters and TiO2-doping on the photocatalytic degradation of azo-dyes. Cat. Today 1999, 54, 119–130. [Google Scholar] [CrossRef]
- Imai, M.; Kikuchi, M.; Oka, N.; Shigesato, Y. Visible light-induced photocatalytic properties of WO3 films deposited by DC reactive magnetron sputtering. J. Vac. Sci. Technol. A Vac. Surf. Films 2012, 30, 031503. [Google Scholar] [CrossRef]
- Hathway, T.; Rockafellow, E.M.; Oh, Y.-C.; Jenks, W.S. Photocatalytic degradation using tungsten-modified TiO2 and visible light: Kinetic and mechanistic effects using multiple catalyst doping strategies. J. Photochem. Photobiol. A Chem. 2009, 207, 197–203. [Google Scholar] [CrossRef]
- Biswas, S.; Hossain, M.F.; Shahjahan, M.; Takahashi, K.; Takahashi, T.; Fujishima, A. Investigation of photocatalytic activity of TiO2/WO3 bilayered thin films with various amounts of WO3 exposed surface. Am. Vac. Soc. 2009, 27, 880–884. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ratova, M.; West, G.T.; Kelly, P.J. Optimization Studies of Photocatalytic Tungsten-Doped Titania Coatings Deposited by Reactive Magnetron Co-Sputtering. Coatings 2013, 3, 194-207. https://doi.org/10.3390/coatings3040194
Ratova M, West GT, Kelly PJ. Optimization Studies of Photocatalytic Tungsten-Doped Titania Coatings Deposited by Reactive Magnetron Co-Sputtering. Coatings. 2013; 3(4):194-207. https://doi.org/10.3390/coatings3040194
Chicago/Turabian StyleRatova, Marina, Glen T. West, and Peter J. Kelly. 2013. "Optimization Studies of Photocatalytic Tungsten-Doped Titania Coatings Deposited by Reactive Magnetron Co-Sputtering" Coatings 3, no. 4: 194-207. https://doi.org/10.3390/coatings3040194







