Quinone-Based Polymers for Label-Free and Reagentless Electrochemical Immunosensors: Application to Proteins, Antibodies and Pesticides Detection
Abstract
:1. General Introduction
1.1. Generalities
1.2. Immunoassays
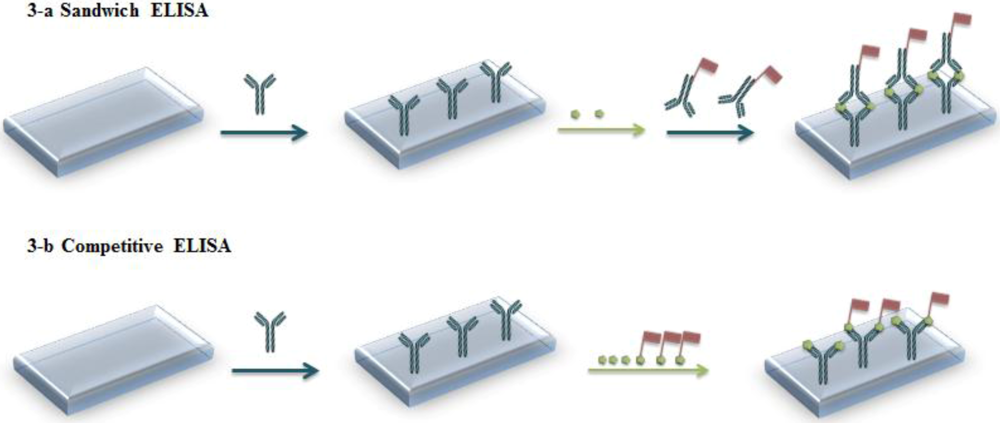
1.3. Immunosensors
1.4. Electrochemical Immunosensors
1.5. Electrochemical Immunosensors Based on Conducting Polymers
1.5.1. Polypyrrole
1.5.2. Other ECPs
2. Recent Advances on Polyquinone-Modified Electrodes for Immunosensing
2.1. General Approach
2.1.1. Principles
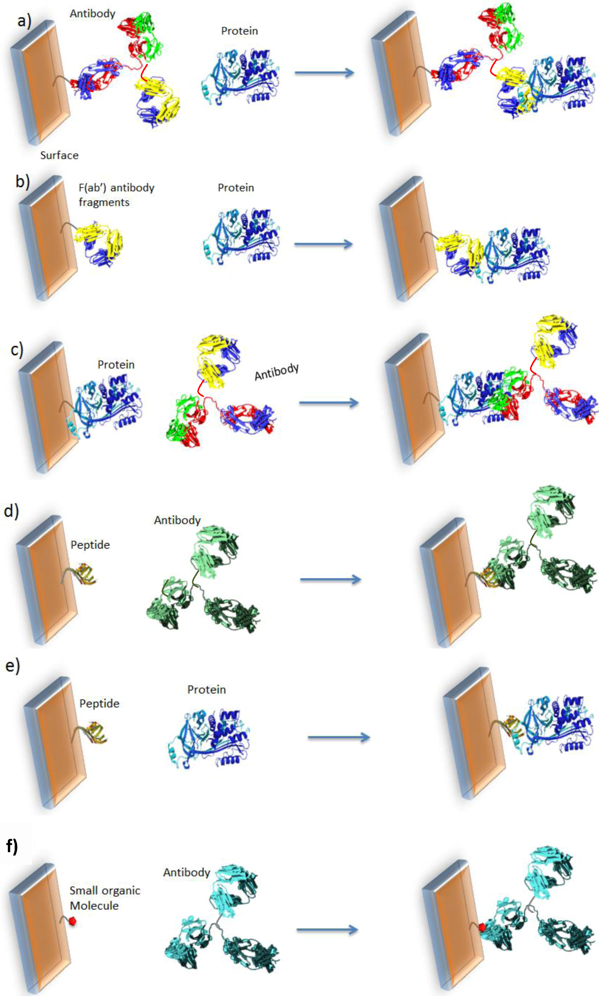
2.1.2. Design

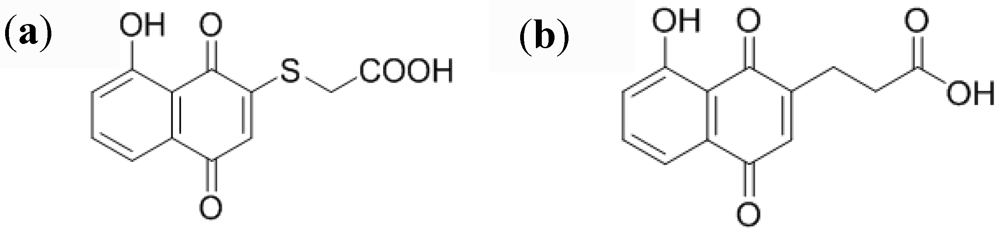
2.2. Applications
2.2.1. Proteins as Probes


2.2.2. Peptides for Biomolecule Grafting


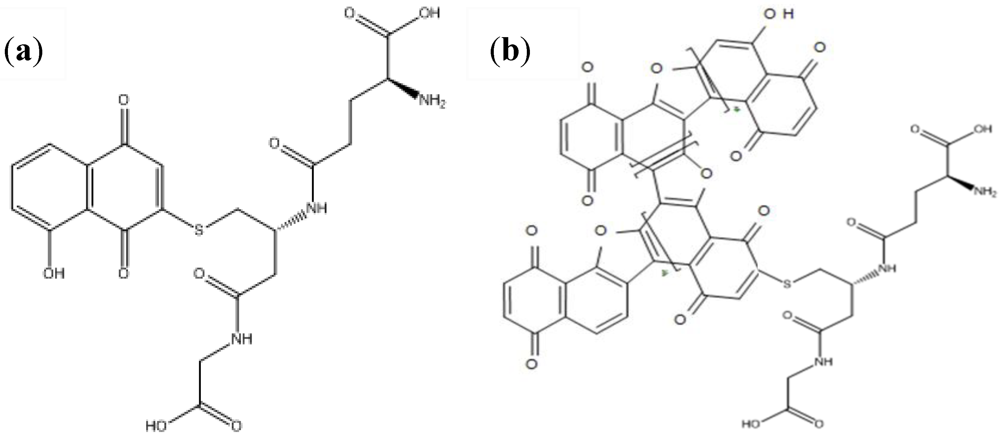
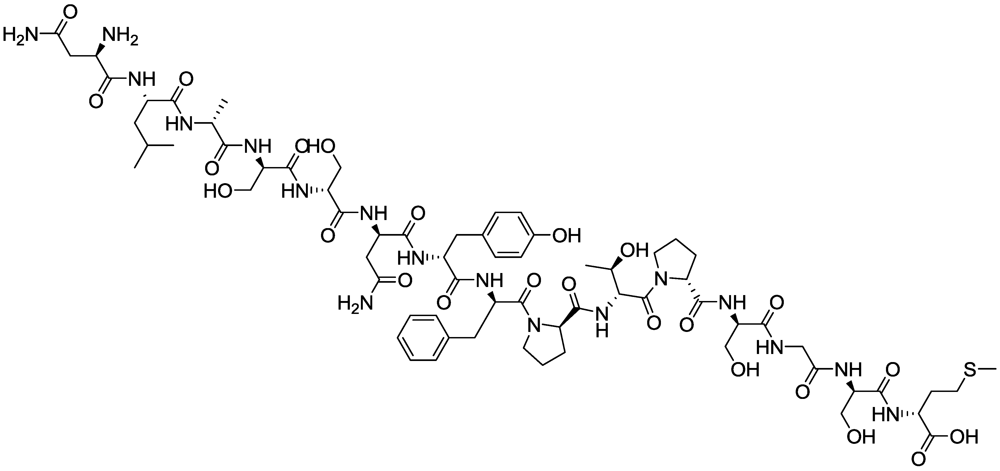
2.2.3. Peptides as Probes


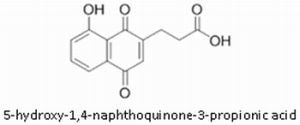
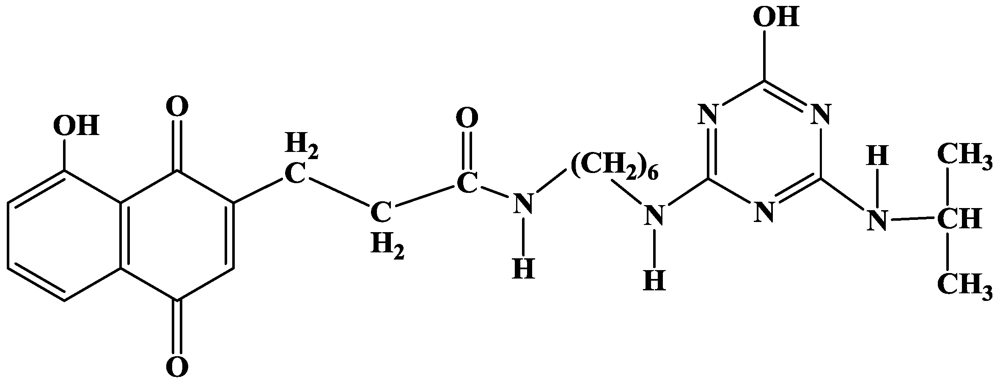
2.2.4. Small Organic Molecules as Probes
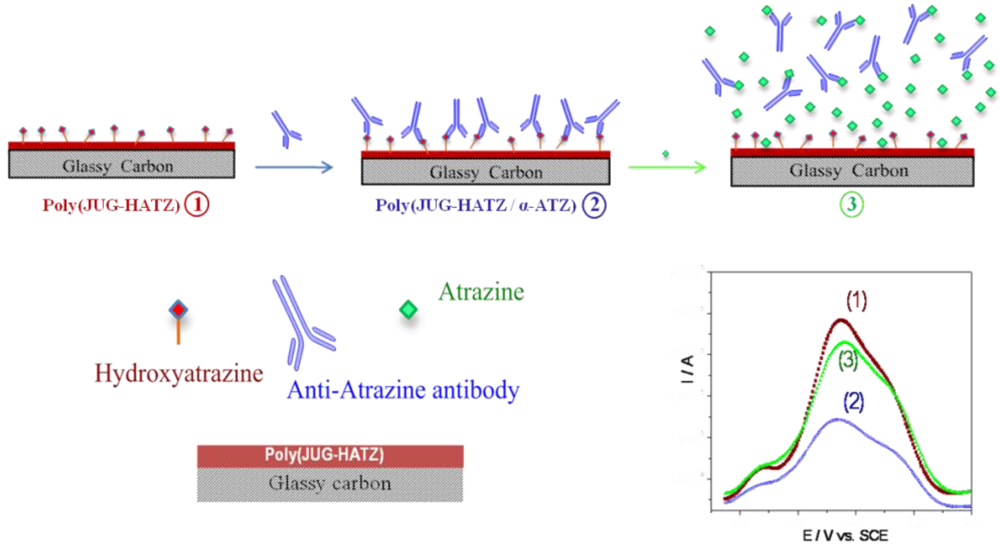

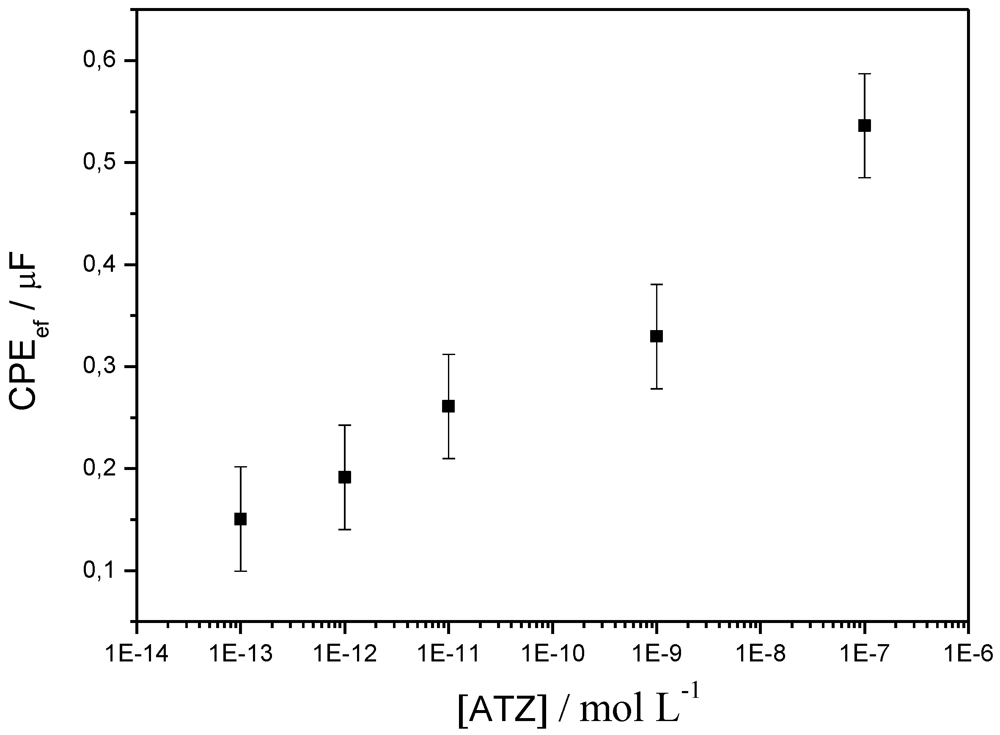
3. Conclusions
Acknowledgments
References
- Sparreboom, A.; Rongen, H.A.H.; van Bennekom, W.P. Assays for interferons and interleukins in biological matrices. Anal. Chim. Acta 1994, 295, 1–26. [Google Scholar] [CrossRef]
- Rongen, H.A.H.; Hoetelmans, R.M.W.; Bult, A.; van Bennekom, W.P. Chemiluminescence and Immunoassays. J. Pharm. Biomed. Appl. 1994, 12, 433–462. [Google Scholar] [CrossRef]
- Gosling, J.P. Enzyme immunoassay. Immunoassay 1996, 287–308. [Google Scholar] [CrossRef]
- Gosling, J.P. Enzyme immunoassay: With and without separation. In Principles and Practice of Immunoassay, 2nd ed.; Price, C.P., Newman, D.J., Eds.; Stockton Press: London, UK, 1997; pp. 349, 351–351. [Google Scholar]
- Porstmann, T.; Kiessig, S.T. Enzyme immunoassay techniques. J. Immunol. Methods 1992, 150, 5–21. [Google Scholar] [CrossRef]
- Tijssen, P. Principles of immunoassays. Enzymes. Methods Immunol. Anal. 1993, 1, 283–297. [Google Scholar]
- Wegner, G.J.; Lee, H.J.; Corn, R.M. Characterization and optimization of peptide arrays for the study of epitope-antibody interactions using surface plasmon resonance imaging. Anal. Chem. 2002, 74, 5161–5168. [Google Scholar] [CrossRef]
- Kanda, V.; Kariuki, J.K.; Harrison, D.J.; McDermott, M.T. Label-free reading of microarray-based immunoassays with surface plasmon resonance imaging. Anal. Chem. 2004, 76, 7257–7262. [Google Scholar]
- Bizet, K.; Gabrielli, C.; Perrot, H.; Therasse, J. Validation of antibody-based recognition by piezoelectric transducers through electroacoustic admittance analysis. Biosens. Bioelectron. 1998, 13, 259–269. [Google Scholar] [CrossRef]
- Jones, V.W.; Kenseth, J.R.; Porter, M.D.; Mosher, C.L.; Henderson, E. Microminiaturized immunoassays using atomic force microscopy and compositionally patterned antigen arrays. Anal. Chem. 1998, 70, 1233–1241. [Google Scholar]
- Jaffrezic-Renault, N.; Martelet, C. Preparation of well-engineered thin molecular layers on semiconductor-based transducers. Sens. Actuator. A 1992, 32, 307–312. [Google Scholar] [CrossRef]
- Lin, P.; Yan, F. Organic thin-film transistors for chemical and biological sensing. Adv. Mater. 2012, 24, 34–51. [Google Scholar] [CrossRef]
- Skládal, P. Advances in electrochemical immunosensors. Electroanalysis 1997, 9, 737–745. [Google Scholar] [CrossRef]
- Prodromidis, M.I. Impedimetric immunosensors—A review. Electrochim. Acta 2010, 55, 4227–4233. [Google Scholar] [CrossRef]
- Sargent, A.; Sadik, O.A. Monitoring antibody-antigen reactions at conducting polymer-based immunosensors using impedance spectroscopy. Electrochim. Acta 1999, 44, 4667–4675. [Google Scholar] [CrossRef]
- Cosnier, S. Biomolecule immobilization on electrode surfaces by entrapment or attachment to electrochemically polymerized films. A review. Biosens. Bioelectron. 1999, 14, 443–456. [Google Scholar] [CrossRef]
- Gerard, M.; Chaubey, A.; Malhotra, B.D. Application of conducting polymers to biosensors. Biosens. Bioelectron. 2002, 17, 345–359. [Google Scholar] [CrossRef]
- Cosnier, S. Biosensors based on electropolymerized films: New trends. Anal. Bioanal. Chem. 2003, 377, 507–520. [Google Scholar] [CrossRef]
- Cosnier, S. Affinity biosensors based on electropolymerized films. Electroanalysis 2005, 17, 1701–1715. [Google Scholar] [CrossRef]
- Ionescu, R.E.; Gondran, C.; Bouffier, L.; Jaffresic-Renault, N.; Martelet, C.; Cosnier, S. Label-free impedimetric immunosensor for sensitive detection of atrazine. Electrochim. Acta 2010, 55, 6228–6232. [Google Scholar] [CrossRef]
- Ramanavičius, A.; Ramanavičienė, A.; Malinauskas, A. Electrochemical sensors based on conducting polymer—Polypyrrole. Electrochim. Acta 2006, 51, 6025–6037. [Google Scholar]
- John, R.; Spencer, M.; Wallace, G.G.; Smyth, M.R. Development of a polypyrrole-based human serum albumin sensor. Anal. Chim. Acta 1991, 249, 381–385. [Google Scholar] [CrossRef]
- Sadik, O.A.; Wallace, G.G. Pulsed amperometric detection of proteins using antibody containing conducting polymers. Anal. Chim. Acta 1993, 279, 209–212. [Google Scholar] [CrossRef]
- Lu, W.; Zhao, H.; Wallace, G.G. Pulsed electrochemical detection of proteins using conducting polymer based sensors. Anal. Chim. Acta 1995, 315, 27–33. [Google Scholar] [CrossRef]
- Sadik, O.A.; van Emon, J.M. Designing immunosensors for environmental monitoring. Chemtech 1997, 27, 38–46. [Google Scholar]
- Brender, S.; Sadik, O.A.; van Emon, J.M. Direct electrochemical immunosensor for polychlorinated biphenyls. Environ. Sci. Technol. 1998, 32, 788–797. [Google Scholar] [CrossRef]
- Sadik, O.A. Bioaffinity sensors based on conducting polymers: A short review. Electroanalysis 1999, 11, 839–844. [Google Scholar] [CrossRef]
- Xu, H.; Masila, M.; Yan, F.; Sadik, O.A. Multiarray sensors for pesticides and toxic metals. Proc. SPIE 1999, 3534, 437–445. [Google Scholar]
- Rahman, M.A.; Shiddiky, M.J.A.; Park, J.-S.; Shim, Y.-B. An impedimetric immunosensor for the label-free detection of bisphenol A. Biosens. Bioelectron. 2007, 22, 2464–2470. [Google Scholar] [CrossRef]
- Khan, R.; Dhayal, M. Chitosan/polyaniline hybrid conducting biopolymer base impedimetric immunosensor to detect Ochratoxin-A. Biosens. Bioelectron. 2009, 24, 1700–1705. [Google Scholar] [CrossRef]
- Lacaze, P.C.; Pham, M.C.; Delamar, M.; Dubois, J.E. Extension of the voltammetric theory to inert, permeable thin-film coated rotating-disk electrodes. Behavior of ferrocene-ferricinium ion and quinone-hydroquinone systems on polymer-coated electrodes. J. Electroanal. Chem. 1980, 108, 9–16. [Google Scholar] [CrossRef]
- Leidner, C.R.; Gater, V.K. Ion and electron-transport within amino-quinone polymer-films. J. Electrochem. Soc. 1987, 134, C502. [Google Scholar] [CrossRef]
- Gater, V.K.; Love, M.D.; Liu, M.D.; Leidner, C.R. Quinone molecular films derived from 1,5-diaminoanthraquinone. J. Electronanal. Chem. 1987, 235, 381–385. [Google Scholar] [CrossRef]
- Gater, V.K.; Liu, M.D.; Love, M.D.; Leidner, C.R. Quinone molecular films derived from aminoquinones. J. Electroanal. Chem. 1988, 257, 133–146. [Google Scholar] [CrossRef]
- Miller, L.L.; Zinger, B.; Degrand, C. The effects of cross-linking and anodic surface roughening on quinone polymer carbon electrodes. J. Electroanal. Chem. 1984, 178, 87–99. [Google Scholar] [CrossRef]
- Pham, M.C.; Piro, B.; Bazzaoui, E.A.; Hedayatullah, M.; Lacroix, J.C.; Novak, P.; Haas, O. Anodic oxidation of 5-amino-1,4-naphthoquinone (ANQ) and synthesis of a conducting polymer (PANQ). Synth. Met. 1998, 92, 197–205. [Google Scholar] [CrossRef]
- Haringer, D.; Novak, P.; Haas, O.; Piro, B.; Pham, M.C. Poly(5-amino-1,4-naphthoquinone), a novel lithium-inserting electroactive polymer with high specific charge. J. Electrochem. Soc. 1999, 146, 2393–2396. [Google Scholar] [CrossRef]
- Reisberg, S.; Piro, B.; Noel, V.; Pham, M.C. Selectivity and sensitivity of a reagentless electrochemical DNA sensor studied by square wave voltammetry and fluorescence. Bioelectrochemistry 2006, 69, 172–179. [Google Scholar] [CrossRef]
- Piro, B.; Reisberg, S.; Noel, V.; Pham, M.C. Investigations of the steric effect on electrochemical transduction in a quinone-based DNA sensor. Biosens. Bioelectron. 2007, 22, 3126–3131. [Google Scholar] [CrossRef]
- Piro, B.; Haccoun, J.; Pham, M.C.; Tran, L.D.; Rubin, A.; Perrot, H.; Gabrielli, C. Study of the DNA hybridization transduction behavior of a quinone-containing electroactive polymer by cyclic voltammetry and electrochemical impedance spectroscopy. J. Electroanal. Chem. 2005, 577, 155–165. [Google Scholar] [CrossRef]
- Reisberg, S.; Acevedo, D.F.; Korovitch, A.; Piro, B.; Noel, V.; Buchet, I.; Tran, L.D.; Barbero, C.A.; Pham, M.C. Design of a new electrogenerated polyquinone film substituted with glutathione. Towards direct electrochemical biosensors. Talanta 2010, 80, 1318–1325. [Google Scholar] [CrossRef]
- Piro, B.; Kapella, A.; Le, V.H.; Anquetin, G.; Zhang, Q.D.; Reisberg, S.; Noel, V.; Tran, L.D.; Duc, H.T.; Pham, M.C. Towards the detection of human papillomavirus infection by a reagentless electrochemical peptide biosensor. Electrochim. Acta 2011, 56, 10688–10693. [Google Scholar]
- Zhang, Q.D.; March, G.; Noel, V.; Piro, B.; Reisberg, S.; Tran, L.D.; Hai, L.V.; Abadia, E.; Nielsen, P.E.; Sola, C.; Pham, M.C. Label-free and reagentless electrochemical detection of PCR fragments using self-assembled quinone derivative monolayer: Application to Mycobacterium tuberculosis. Biosens. Bioelectron. 2012, 32, 163–168. [Google Scholar] [CrossRef]
- Piro, B.; Zhang, Q.D.; Reisberg, S.; Noel, V.; Dang, L.A.; Duc, H.T.; Pham, M.C. Direct and rapid electrochemical immunosensing system based on a conducting polymer. Talanta 2010, 82, 608–612. [Google Scholar] [CrossRef]
- Piro, B.; Reisberg, S.; Noel, V.; Pham, M.C. Investigations of the steric effect on electrochemical transduction in a quinone-based DNA sensor. Biosens. Bioelectron. 2007, 12, 3126–3131. [Google Scholar]
- Reisberg, S.; Piro, B.; Noel, V.; Nguyen, T.D.; Nielsen, P.E.; Pham, M.C. Investigation of the charge effect on the electrochemical transduction in a quinone-based DNA sensor. Electrochim. Acta 2008, 54, 346–351. [Google Scholar] [CrossRef]
- Tran, H.V.; Yougnia, R.; Reisberg, S.; Piro, B.; Serradji, N.; Nguyen, T.D.; Tran, L.D.; Dong, C.Z.; Pham, M.C. A label-free electrochemical immunosensor for direct, signal-on and sensitive pesticide detection. Biosens. Bioelectron. 2012, 31, 62–68. [Google Scholar] [CrossRef]
- Sassolas, A.; Prieto-Simón, B.; Marty, J. Biosensors for pesticide detection: New trends. Am. J. Anal. Chem. 2012, 3, 210–232. [Google Scholar] [CrossRef]
- Thomson, R.H. Studies in the juglone series. 3. Addition reactions. J. Org. Chem. 1951, 16, 1082–1090. [Google Scholar] [CrossRef]
- Bosch, F.X.; Manos, M.M.; Munoz, N.; Sherman, M.; Jansen, A.M.; Peto, J.; Schiffman, M.H.; Moreno, V.; Kurman, R.; Shan, K.V. Prevalence of human papillomavirus in cervical-cancer—A worldwide perspective. J. Nat. Cancer. Inst. 1995, 87, 796–802. [Google Scholar] [CrossRef]
- Munoz, N.; Bosch, F.X.; De Sanjose, S.; Herrero, R.; Castellsague, X.; Shah, K.V.; Snijders, P.J.F.; Meijer, C.J.L.M. Epidemiologic classification of human papillomavirus types associated with cervical cancer. New Engl. J. Med. 2003, 348, 518–527. [Google Scholar] [CrossRef]
- Tyring, S.K. Human papillomavirus infections: Epidemiology, pathogenesis, and host immune response. J. Am. Acad. Dermatol. 2000, 43, S18–S26. [Google Scholar] [CrossRef]
- Dillner, J. The serological response to papillomaviruses. Semin. Cancer Biol. 1999, 9, 423–430. [Google Scholar]
- Yeager, M.D.; Aste-Amezaga, M.; Brown, D.R.; Martin, M.M.; Shah, M.J.; Cook, J.C.; Christensen, N.D.; Ackerson, C.; Lowe, R.S.; Smith, J.F.; Keller, P.; Jansen, K.U. Neutralization of human papillomavirus (HPV) pseudovirions: A novel and efficient approach to detect and characterize HPV neutralizing antibodies. Virology 2000, 278, 570–577. [Google Scholar] [CrossRef]
- Caygill, R.L.; Blair, G.E.; Millner, P.A. A review on viral biosensors to detect human pathogens. Anal. Chim. Acta 2010, 681, 8–15. [Google Scholar] [CrossRef]
- Caygill, R.L.; Hodges, C.S.; Holmes, J.L.; Higson, S.P.J.; Blair, G.E.; Millner, P.A. Novel impedimetric immunosensor for the detection and quantitation of Adenovirus using reduced antibody fragments immobilized onto a conducting copolymer surface. Biosens. Bioelectron. 2012, 32, 104–110. [Google Scholar] [CrossRef]
- Ionescu, R.E.; Cosnier, S.; Herrmann, S.; Marks, R.S. Amperometric immunosensor for the detection of anti-West Nile virus IgG. Anal. Chem. 2007, 79, 8662–8668. [Google Scholar]
- Marty, J.L.; Garcia, D.; Rouillon, R. Biosensors—Potential in pesticide detection. Trends Anal. Chem. 1995, 14, 329–333. [Google Scholar]
- Dennison, M.J.; Turner, A.P.F. Biosensors for environmental monitoring. Biotechnol. Adv. 1995, 13, 1–12. [Google Scholar] [CrossRef]
- Hleli, S.; Martelet, C.; Abdelghani, A.; Burais, N.; Jaffrezic-Renault, N. Atrazine analysis using an impedimetric immunosensor based on mixed biotinylated self-assembled monolayer. Sens. Actuator. B 2006, 113, 711–717. [Google Scholar] [CrossRef]
- Valera, E.; Ramón-Azcón, J.; Barranco, A.; Alfaro, B.; Sánchez-Baeza, F.; Marco, M.-P.; Rodríguez, Á. Determination of atrazine residues in red wine samples. A conductimetric solution. Food Chem. 2010, 122, 888–894. [Google Scholar] [CrossRef]
- Besombes, J.L.; Cosnier, S.; Labbé, P.; Reverdy, G. A biosensor as warning device for the detection of cyanide, chlorophenols, atrazine and carbamate pesticides. Anal. Chim. Acta 1995, 311, 255–263. [Google Scholar] [CrossRef]
- Ionescu, R.E.; Gondran, C.; Bouffier, L.; Jaffrezic-Renault, N.; Martelet, C.; Cosnier, S. Label-free impedimetric immunosensor for sensitive detection of atrazine. Electrochim. Acta 2010, 55, 6228–6232. [Google Scholar] [CrossRef]
- Ballesteros-Gomez, A.; Rubio, S. Recent advances in environmental analysis. Anal. Chem. 2011, 83, 4579–4613. [Google Scholar] [CrossRef]
- Schlaeppi, J.M.; Foery, W.; Ramsteiner, K. Hydroxyatrazine and atrazine determination in soil and water by enzyme-linked immunosorbent-assay using specific monoclonal-antibodies. J. Agric. Food Chem. 1989, 37, 1532–1538. [Google Scholar] [CrossRef]
- Wortberg, M.; Jones, G.B.; Kreissig, S.; Rocke, D.M.; Gee, S.J.; Hammock, B.D. An approach to the construction of an immunoarray for differentiating and quantitating cross reacting analytes. Anal. Chim. Acta 1996, 319, 291–303. [Google Scholar] [CrossRef]
- Charlton, K.; Harris, W.J.; Porter, A.J. The isolation of super-sensitive anti-hapten antibodies from combinatorial antibody libraries derived from sheep. Biosens. Bioelectron. 2001, 16, 639–646. [Google Scholar] [CrossRef]
- Tran, H.V.; Reisberg, S.; Piro, B.; Pham, M.C. Label-free electrochemical immunoaffinity sensor based on impedimetric method for pesticide detection. Electroanalysis 2013, in press. [Google Scholar]
Appendx
Experimentals
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Piro, B.; Reisberg, S.; Anquetin, G.; Duc, H.-T.; Pham, M.-C. Quinone-Based Polymers for Label-Free and Reagentless Electrochemical Immunosensors: Application to Proteins, Antibodies and Pesticides Detection. Biosensors 2013, 3, 58-76. https://doi.org/10.3390/bios3010058
Piro B, Reisberg S, Anquetin G, Duc H-T, Pham M-C. Quinone-Based Polymers for Label-Free and Reagentless Electrochemical Immunosensors: Application to Proteins, Antibodies and Pesticides Detection. Biosensors. 2013; 3(1):58-76. https://doi.org/10.3390/bios3010058
Chicago/Turabian StylePiro, Benoit, Steeve Reisberg, Guillaume Anquetin, Huynh-Thien Duc, and Minh-Chau Pham. 2013. "Quinone-Based Polymers for Label-Free and Reagentless Electrochemical Immunosensors: Application to Proteins, Antibodies and Pesticides Detection" Biosensors 3, no. 1: 58-76. https://doi.org/10.3390/bios3010058