Improving the Design of a MscL-Based Triggered Nanovalve
Abstract
:1. Introduction
2. Experimental Section
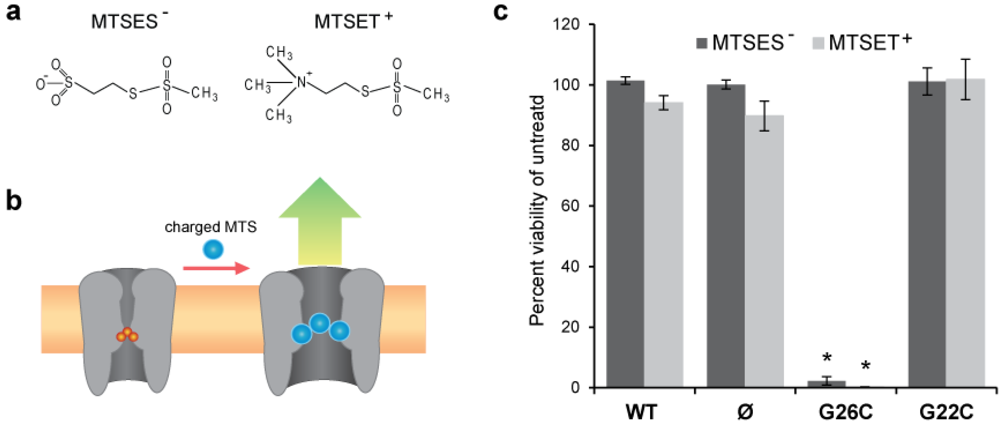
2.1. In Vivo Viability Experiments
2.2. Electrophysiology
2.3. Calcein Efflux Assay
| MscL | Protein concentration, µg/mL | SEM | n |
|---|---|---|---|
| WT | 2,308.3 | 431.8 | 4 |
| G22C | 1,990.1 | 324.1 | 4 |
| G26C | 454.7 | 104.7 | 4 |
| G26C (Δ110-136) | 918.6 | 231.8 | 4 |
3. Results and Discussion
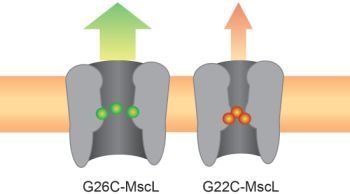
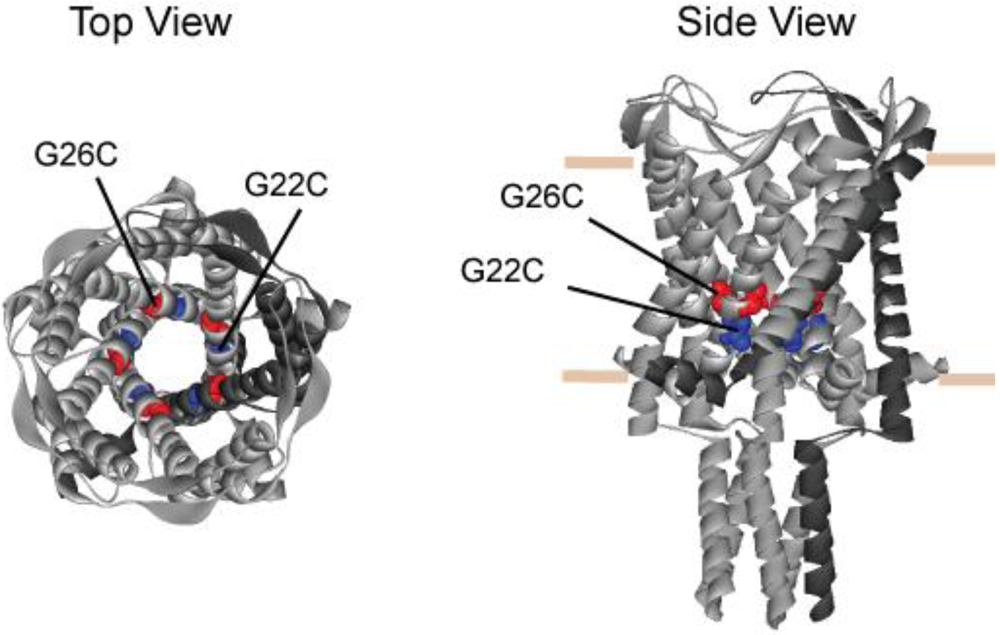
3.1. Comparing Site Accessibility of the MscL Nanovalve in Vivo
3.2. Single Channel Analysis of the Effects MTS Modifications in G26C MscL Activity

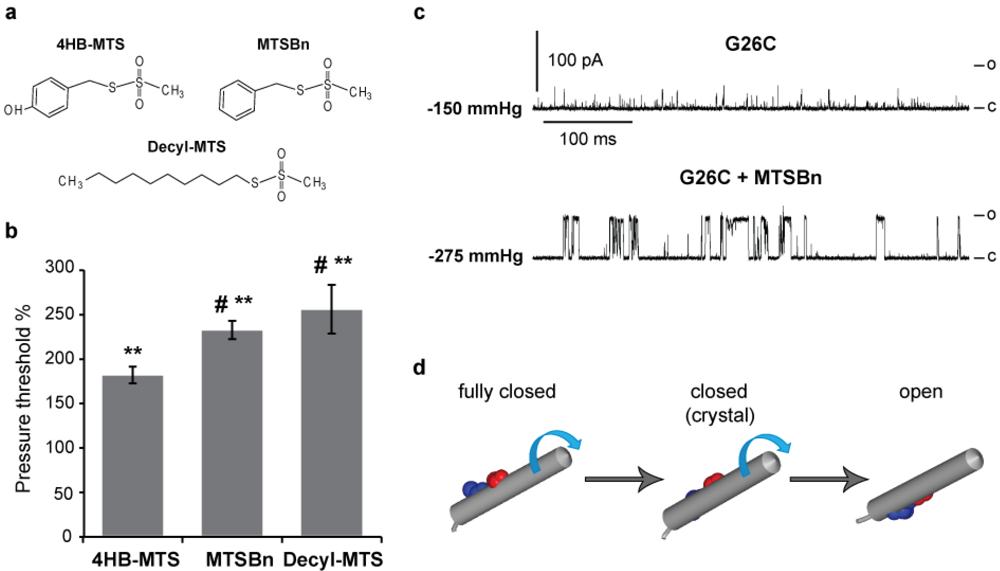
3.3. Hydrophobic MTS Modifications in G26C MscL, Closing MscL Nanovalve
3.4. Calcein Release through MscL Nanovalves from Liposomes: A Model for Drug-Delivery

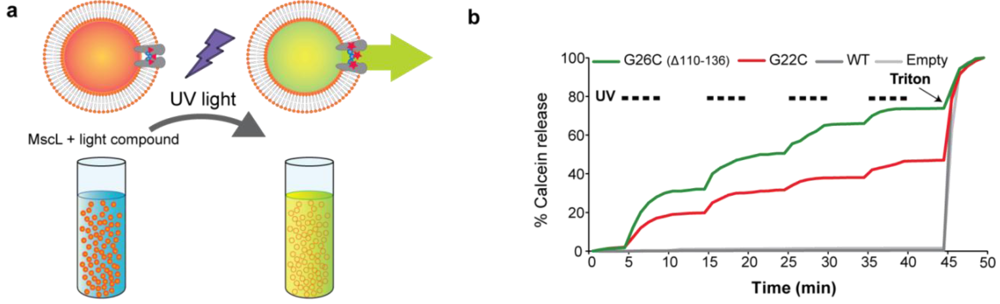
3.5. The Many Dimensions for Designing Device-Specific MscL-Based Triggered Nanovalves
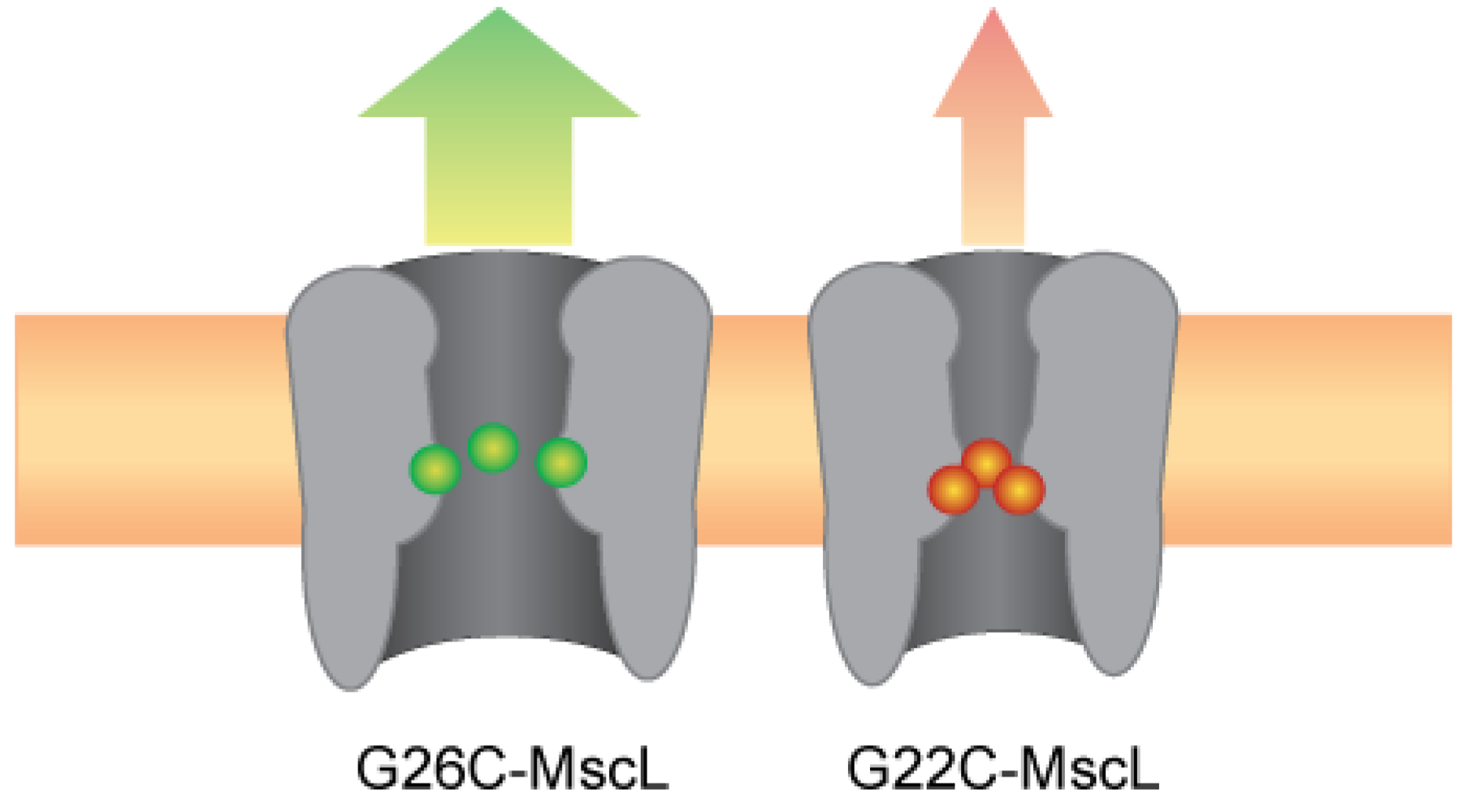
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Blount, P.; Sukharev, S.I.; Moe, P.; Kung, C. Mechanosensitive channels of E. coli: A genetic and molecular dissection. Biol. Bull, 1997; 192, 126–127. [Google Scholar]
- Blount, P.; Iscla, I.; Moe, P.C.; Li, Y. MscL: The bacterial mechanosensitive channel of large conductance. In Mechanosensitive Ion Channels (A Volume in the Current Topics in Membranes Series); Hamill, O.P., Ed.; Elsievier Press: St. Louis, MO, USA, 2007; Volume 58, pp. 202–233. [Google Scholar]
- Levina, N.; Totemeyer, S.; Stokes, N.R.; Louis, P.; Jones, M.A.; Booth, I.R. Protection of Escherichia coli cells against extreme turgor by activation of MscS and MscL mechanosensitive channels: Identification of genes required for MscS activity. EMBO J. 1999, 18, 1730–1737. [Google Scholar] [CrossRef]
- Cruickshank, C.C.; Minchin, R.F.; Le Dain, A.C.; Martinac, B. Estimation of the pore size of the large-conductance mechanosensitive ion channel of Escherichia coli. Biophys. J. 1997, 73, 1925–1931. [Google Scholar] [CrossRef]
- Chang, G.; Spencer, R.H.; Lee, A.T.; Barclay, M.T.; Rees, D.C. Structure of the MscL homolog from Mycobacterium tuberculosis: A gated mechanosensitive ion channel. Science 1998, 282, 2220–2226. [Google Scholar] [CrossRef]
- Steinbacher, S.; Bass, R.; Strop, P.; Rees, D.C. Structures of the prokaryotic mechanosensitive channels MscL and MscS. In Mechanosensitive Ion Channels (A Volume in the Current Topics in Membranes Series); Hamill, O.P., Ed.; Elsievier Press: St. Louis, MO, USA, 2007; Volume 58, pp. 1–20. [Google Scholar]
- Iscla, I.; Levin, G.; Wray, R.; Reynolds, R.; Blount, P. Defining the physical gate of a mechanosensitive channel, MscL, by engineering metal-binding sites. Biophys. J. 2004, 87, 3172–3180. [Google Scholar] [CrossRef]
- Levin, G.; Blount, P. Cysteine scanning of MscL transmembrane domains reveals residues critical for mechanosensitive channel gating. Biophys. J. 2004, 86, 2862–2870. [Google Scholar] [CrossRef]
- Ou, X.; Blount, P.; Hoffman, R.J.; Kung, C. One face of a transmembrane helix is crucial in mechanosensitive channel gating. Proc. Natl. Acad. Sci. USA 1998, 95, 11471–11475. [Google Scholar] [CrossRef]
- Yoshimura, K.; Batiza, A.; Schroeder, M.; Blount, P.; Kung, C. Hydrophilicity of a single residue within MscL correlates with increased channel mechanosensitivity. Biophys. J. 1999, 77, 1960–1972. [Google Scholar] [CrossRef]
- Blount, P.; Moe, P.C. Bacterial mechanosensitive channels: Integrating physiology, structure and function. Trends Microbiol. 1999, 7, 420–424. [Google Scholar] [CrossRef]
- Bartlett, J.L.; Levin, G.; Blount, P. An in vivo assay identifies changes in residue accessibility on mechanosensitive channel gating. Proc. Natl. Acad. Sci. USA 2004, 101, 10161–10165. [Google Scholar] [CrossRef]
- Bartlett, J.L.; Li, Y.; Blount, P. Mechanosensitive channel gating transitions resolved by functional changes upon pore modification. Biophys. J. 2006, 91, 3684–3691. [Google Scholar] [CrossRef]
- Batiza, A.F.; Kuo, M.M.; Yoshimura, K.; Kung, C. Gating the bacterial mechanosensitive channel MscL invivo. Proc. Natl. Acad. Sci. USA 2002, 99, 5643–5648. [Google Scholar] [CrossRef]
- Yoshimura, K.; Batiza, A.; Kung, C. Chemically charging the pore constriction opens the mechanosensitive channel MscL. Biophys. J. 2001, 80, 2198–2206. [Google Scholar] [CrossRef]
- Blount, P.; Sukharev, S.I.; Moe, P.C.; Martinac, B.; Kung, C. Mechanosensitive channels of bacteria. In Method Enzymol; Academic Press: San Diego, CA, USA, 1999; Volume 294, pp. 458–482. [Google Scholar]
- Berrier, C.; Guilvout, I.; Bayan, N.; Park, K.H.; Mesneau, A.; Chami, M.; Pugsley, A.P.; Ghazi, A. Coupled cell-free synthesis and lipid vesicle insertion of a functional oligomeric channel MscL MscL does not need the insertase YidC for insertion in vitro. Biochim. Biophys. Acta 2011, 1808, 41–46. [Google Scholar] [CrossRef]
- Clayton, D.; Shapovalov, G.; Maurer, J.A.; Dougherty, D.A.; Lester, H.A.; Kochendoerfer, G.G. Total chemical synthesis and electrophysiological characterization of mechanosensitive channels from Escherichia coli and Mycobacterium tuberculosis. Proc. Natl. Acad. Sci. USA 2004, 101, 4764–4769. [Google Scholar] [CrossRef]
- Koçer, A.; Walko, M.; Bulten, E.; Halza, E.; Feringa, B.; Meijberg, W. Rationally designed chemical modulators convert a bacterial channel protein into a pH-sensory valve. Angew. Chem. 2006, 45, 3126–3130. [Google Scholar] [CrossRef]
- Koçer, A.; Walko, M.; Feringa, B.L. Synthesis and utilization of reversible and irreversible light-activated nanovalves derived from the channel protein MscL. Nat. Protoc. 2007, 2, 1426–1437. [Google Scholar] [CrossRef]
- Koçer, A.; Walko, M.; Meijberg, W.; Feringa, B.L. A light-actuated nanovalve derived from a channel protein. Science 2005, 309, 755–758. [Google Scholar] [CrossRef]
- Yang, L.M.; Blount, P. Manipulating the permeation of charged compounds through the MscL nanovalve. FASEB J. 2011, 25, 428–434. [Google Scholar] [CrossRef]
- Yang, L.M.; Wray, R.; Parker, J.; Wilson, D.; Duran, R.S.; Blount, P. Three routes to modulate the pore size of the MscL channel/nanovalve. ACS Nano 2012, 6, 1134–1141. [Google Scholar] [CrossRef]
- Iscla, I.; Wray, R.; Blount, P. On the structure of the N-terminal domain of the MscL channel: Helical bundle or membrane interface. Biophys. J. 2008, 95, 2283–2291. [Google Scholar] [CrossRef]
- Powl, A.M.; East, J.M.; Lee, A.G. Anionic phospholipids affect the rate and extent of flux through the mechanosensitive channel of large conductance MscL. Biochemistry 2008, 47, 4317–4328. [Google Scholar] [CrossRef]
- Blount, P.; Sukharev, S.I.; Schroeder, M.J.; Nagle, S.K.; Kung, C. Single residue substitutions that change the gating properties of a mechanosensitive channel in Escherichia coli. Proc. Natl. Acad. Sci. USA 1996, 93, 11652–11657. [Google Scholar] [CrossRef]
- Moe, P.; Blount, P. Assessment of potential stimuli for mechano-dependent gating of MscL: Effects of pressure, tension, and lipid headgroup. Biochemistry 2005, 44, 12239–12244. [Google Scholar] [CrossRef]
- Blount, P.; Sukharev, S.I.; Moe, P.C.; Schroeder, M.J.; Guy, H.R.; Kung, C. Membrane topology and multimeric structure of a mechanosensitive channel protein of Escherichia coli. EMBO J. 1996, 15, 4798–4805. [Google Scholar]
- Li, Y.; Wray, R.; Eaton, C.; Blount, P. An open-pore structure of the mechanosensitive channel MscL derived by determining transmembrane domain interactions upon gating. FASEB J. 2009, 23, 2197–2204. [Google Scholar] [CrossRef]
- Dorwart, M.R.; Wray, R.; Brautigam, C.A.; Jiang, Y.; Blount, P. S. aureus MscL is a pentamer in vivo but of variable stoichiometries in vitro: Implications for detergent-solubilized membrane proteins. PLoS Biol. 2010, 8. [Google Scholar] [CrossRef]
- Sukharev, S.I.; Blount, P.; Martinac, B.; Blattner, F.R.; Kung, C. A large-conductance mechanosensitive channel in E. coli encoded by MscL alone. Nature 1994, 368, 265–268. [Google Scholar] [CrossRef]
- Doerner, J.F.; Febvay, S.; Clapham, D.E. Controlled delivery of bioactive molecules into live cells using the bacterial mechanosensitive channel MscL. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef]
- Iscla, I.; Wray, R.; Blount, P. The oligomeric state of the truncated mechanosensitive channel of large conductance shows no variance in vivo. Protein Sci. 2011, 20, 1638–1642. [Google Scholar] [CrossRef]
- Liu, Z.; Gandhi, C.S.; Rees, D.C. Structure of a tetrameric MscL in an expanded intermediate state. Nature 2009, 461, 120–124. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Iscla, I.; Eaton, C.; Parker, J.; Wray, R.; Kovács, Z.; Blount, P. Improving the Design of a MscL-Based Triggered Nanovalve. Biosensors 2013, 3, 171-184. https://doi.org/10.3390/bios3010171
Iscla I, Eaton C, Parker J, Wray R, Kovács Z, Blount P. Improving the Design of a MscL-Based Triggered Nanovalve. Biosensors. 2013; 3(1):171-184. https://doi.org/10.3390/bios3010171
Chicago/Turabian StyleIscla, Irene, Christina Eaton, Juandell Parker, Robin Wray, Zoltán Kovács, and Paul Blount. 2013. "Improving the Design of a MscL-Based Triggered Nanovalve" Biosensors 3, no. 1: 171-184. https://doi.org/10.3390/bios3010171
APA StyleIscla, I., Eaton, C., Parker, J., Wray, R., Kovács, Z., & Blount, P. (2013). Improving the Design of a MscL-Based Triggered Nanovalve. Biosensors, 3(1), 171-184. https://doi.org/10.3390/bios3010171