Poly(lactic acid)/Carbon Nanotube Fibers as Novel Platforms for Glucose Biosensors
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Methods
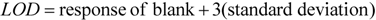
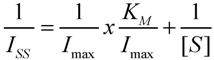
3. Results and Discussion
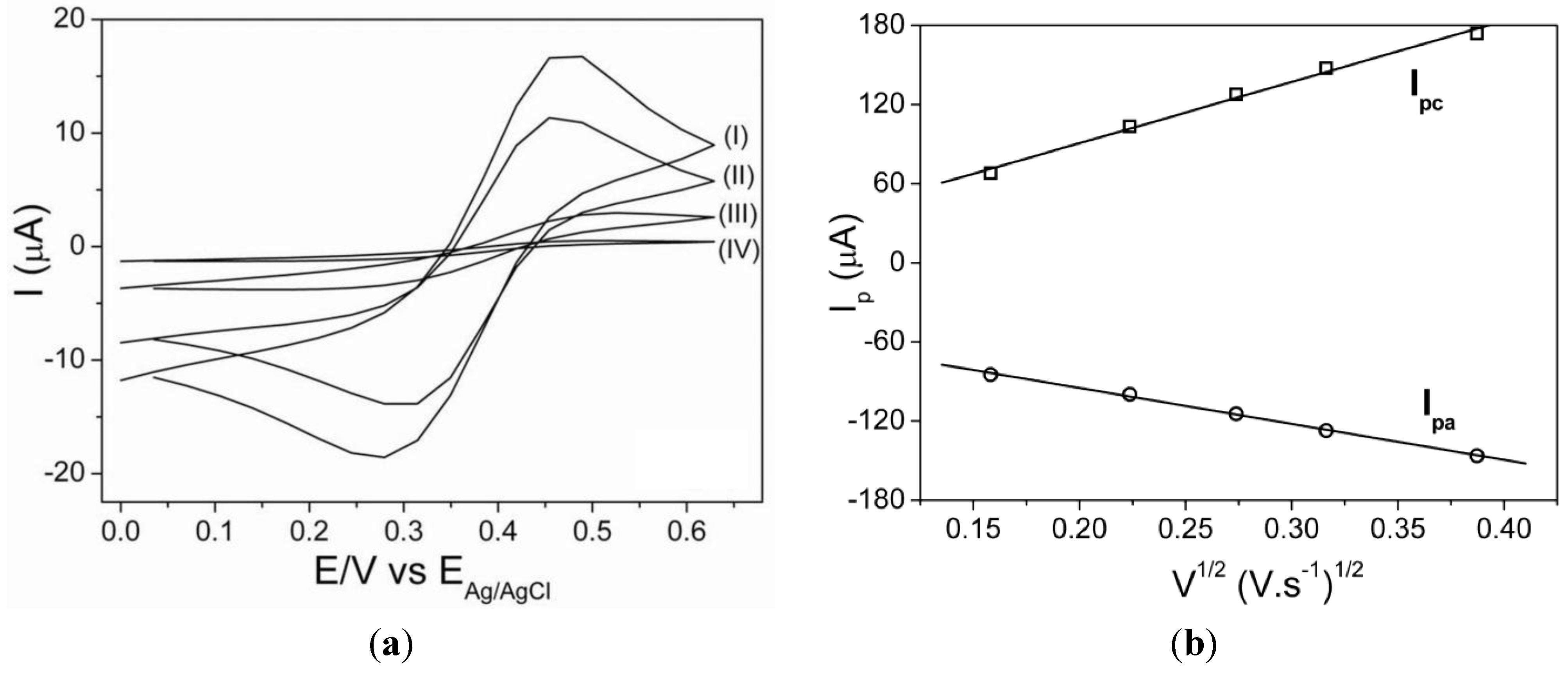
3.1. Electrode Characterization
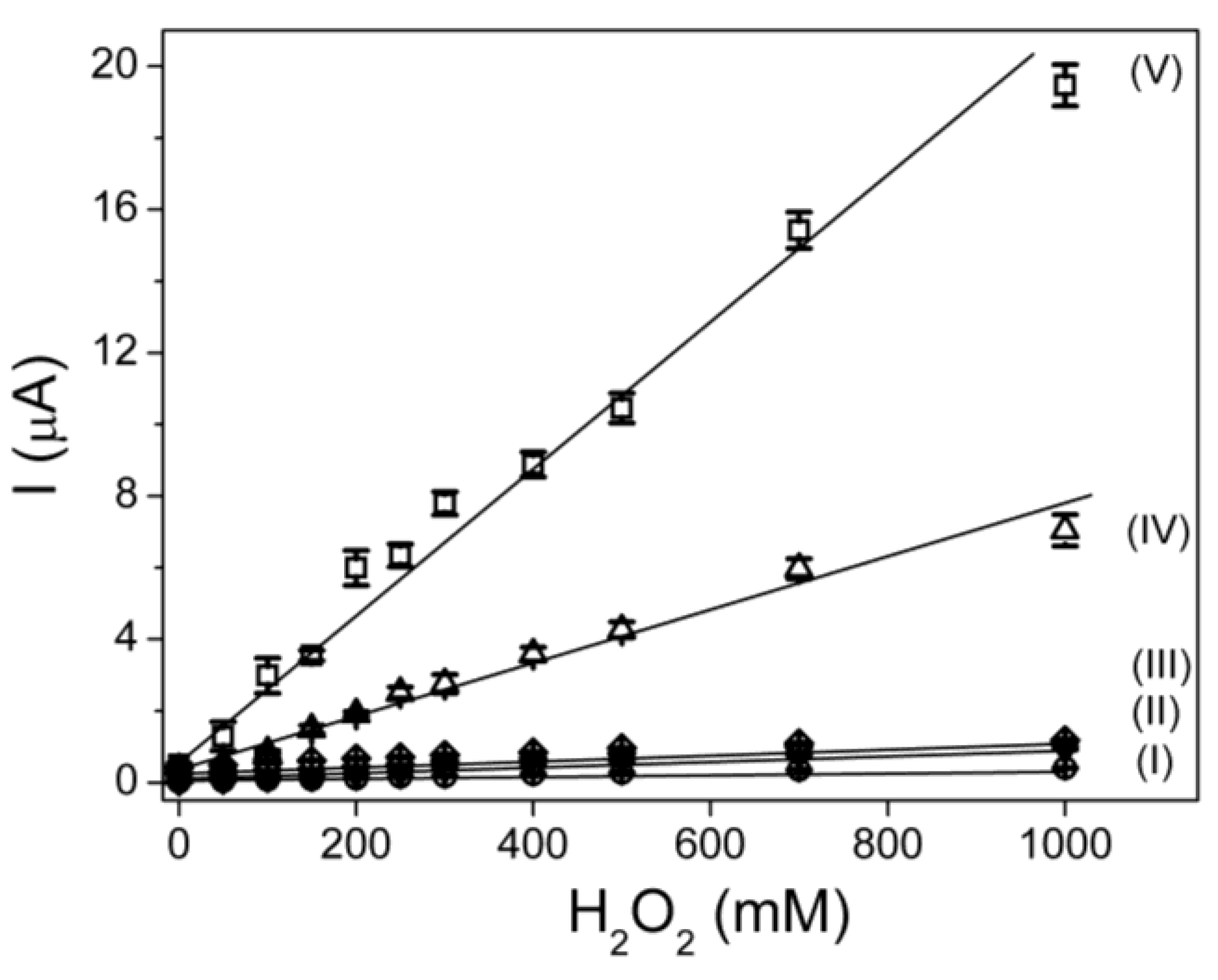
3.2. Influence of Carbon Nanotubes Concentration
| MWCNT (%) | LOD (mM) | Sensitivity (nA·mM−1) |
|---|---|---|
| 0 | 8.3 ± 0.4 | 15 ± 2 |
| 0.1 | 1.01 ±0.3 | 5 ± 3 |
| 0.5 | 0.4 ±0.3 | 22 ± 3 |
| 1 | 0.3 ± 0.1 | 213 ± 4 |
| 3 | 1.4 ± 0.3 | 83 ± 3 |
| Time of deposition (min) | LOD (mM) | Sensitivity (nA·mM−1) |
| 1 | 0.2 ± 0.1 | 208 ± 4 |
| 3 | 1.2 ±0.1 | 36 ± 7 |
| 15 | 1.5 ±0.2 | 2 ± 8 |
3.3. Influence of Deposition Time on Sensor Properties

3.4. Enzyme Immobilization

3.5. Amperometric Determination of Glucose at Poly(lactic acid)/Carbon Nanotubes Modified Electrodes
| Glutaraldehyde (% v/v) | LOD (mM) | Sensitivity (nA·mM−1) |
|---|---|---|
| 0 | 7.6 ± 0.8 | 31 ± 1 |
| 0.125 | 2.8 ± 0.2 | 144 ± 4 |
| 0.25 | 1.2 ± 0.3 | 140 ± 5 |
| 1.25 | 3.1 ± 0.4 | 15 ± 1 |
| 2.5 | 5.5 ± 0.4 | 6 ± 1 |
| Enzyme Concentration(U·µL−1) | LOD (mM) | Sensitivity (nA·mM−1) |
| 0.25 | 2.5 ± 0.4 | 7 ± 2 |
| 0.5 | 2.3 ± 0.5 | 23 ± 4 |
| 0.75 | 1.6 ± 0.3 | 358 ± 9 |
| 1 | 1.4 ± 0.3 | 140 ± 5 |
| 2 | 3.8 ± 0.4 | 26 ± 3 |
| pH | LOD (mM) | Sensitivity (nA·mM−1) |
| 5.4 | 3.5 ± 0.3 | 10 ± 4 |
| 6 | 2.3 ± 0.4 | 22 ± 1 |
| 7 | 1.5 ± 0.2 | 145 ± 6 |
| 8 | 1.5 ± 0.1 | 204 ± 6 |
| 9 | 1.4 ± 0.2 | 147 ± 3 |
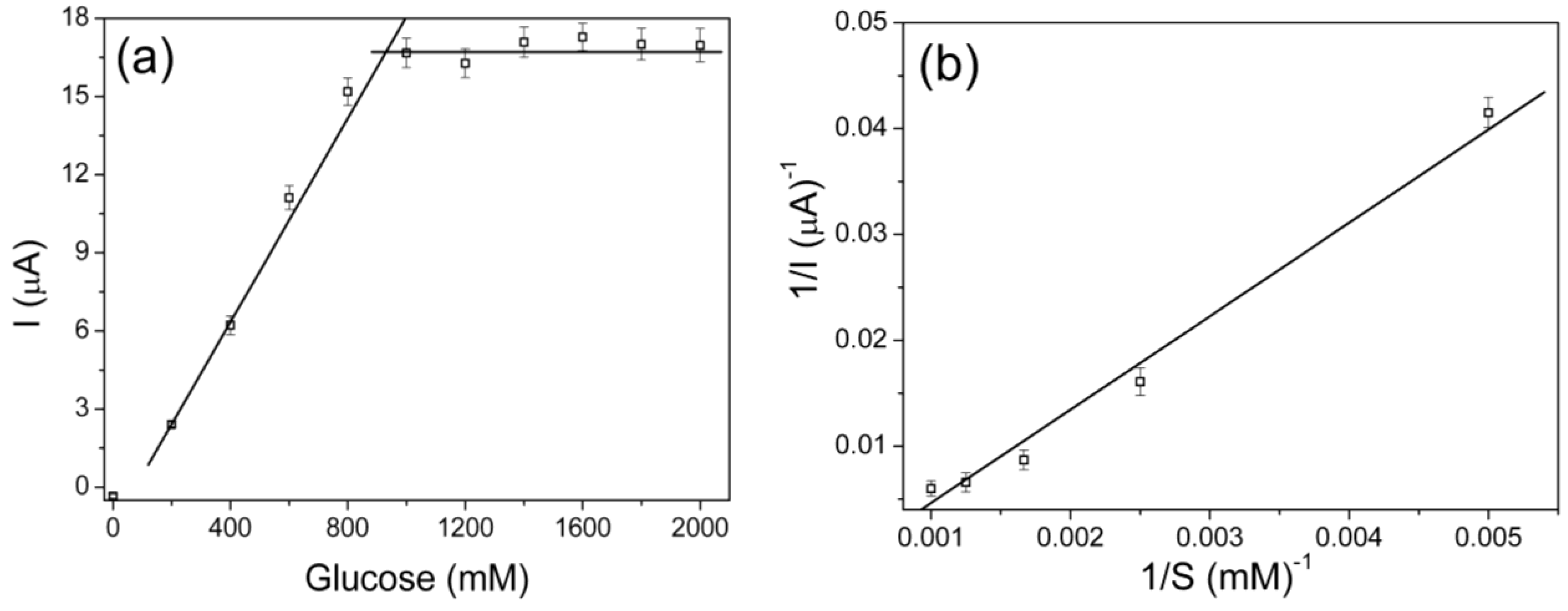
3.6. Kinetic Parameters of the Immobilized Glucose Oxidase on the Nanocomposite Fibers Modified Electrodes
4. Conclusions
Acknowledgments
References
- Rahman, M.M.; Ahammad, A.J.S.; Jin, J.H.; Ahn, S.J.; Lee, J.J. A comprehensive review of glucose biosensors based on nanostructured metal-oxides. Sensors 2010, 10, 4855–4886. [Google Scholar]
- Wang, J. Glucose biosensors: 40 years of advances and challenges. Electroanalysis 2001, 13, 983–988. [Google Scholar] [CrossRef]
- Wilson, G.S.; Gifford, R. Biosensors for real-time in vivo measurements. Biosens. Bioelectron. 2005, 20, 2388–2403. [Google Scholar] [CrossRef]
- Yoo, E.H.; Lee, S.Y. Glucose biosensors: An overview of use in clinical practice. Sensors 2010, 10, 4558–4576. [Google Scholar] [CrossRef]
- Bankar, S.B.; Bule, M.V.; Singhal, R.S.; Ananthanarayan, L. Glucose oxidase—An overview. Biotechnol. Adv. 2009, 27, 489–501. [Google Scholar]
- Gibson, Q.H.; Massey, V.; Swoboda, B.E.P. Kinetics + mechanism of action of glucose oxidase. J. Biol. Chem. 1964, 239, 3927–3934. [Google Scholar]
- Wilson, R.; Turner, A.P.F. Glucose oxidase: An ideal enzyme. Biosens. Bioelectron. 1992, 7, 165–185. [Google Scholar]
- Albareda-Sirvent, M.; Merkoci, A.; Alegret, S. Configurations used in the design of screen-printed enzymatic biosensors. A review. Sens. Actuat. B Chem. 2000, 69, 153–163. [Google Scholar] [CrossRef]
- Pingarron, J.M.; Yanez-Sedeno, P.; Gonzalez-Cortes, A. Gold nanoparticle-based electrochemical biosensors. Electrochim. Acta 2008, 53, 5848–5866. [Google Scholar] [CrossRef]
- Wang, Z.G.; Wan, L.S.; Liu, Z.M.; Huang, X.J.; Xu, Z.K. Enzyme immobilization on electrospun polymer nanofibers: An overview. J. Mol. Catal. B Enzym. 2009, 56, 189–195. [Google Scholar]
- Caseli, L.; Crespilho, F.N.; Nobre, T.M.; Zaniquelli, M.E.D.; Zucolotto, V.; Oliveira, O.N. Using phospholipid langmuir and langmuir-blodgett films as matrix for urease immobilization. J. Colloid. Interface Sci. 2008, 319, 100–108. [Google Scholar]
- Siqueira, J.R.; Gasparotto, L.H.S.; Oliveira, O.N.; Zucolotto, V. Processing of electroactive nanostructured films incorporating carbon nanotubes and phthalocyanines for sensing. J. Phys. Chem. C 2008, 112, 9050–9055. [Google Scholar]
- Arecchi, A.; Scampicchio, M.; Drusch, S.; Mannino, S. Nanofibrous membrane based tyrosinase-biosensor for the detection of phenolic compounds. Anal. Chim. Acta 2010, 659, 133–136. [Google Scholar]
- Li, D.P.; Frey, M.W.; Baeumner, A.J. Electrospun polylactic acid nanofiber membranes as substrates for biosensor assemblies. J. Membr. Sci. 2006, 279, 354–363. [Google Scholar] [CrossRef]
- Manesh, K.M.; Kim, H.T.; Santhosh, P.; Gopalan, A.I.; Lee, K.P. A novel glucose biosensor based on immobilization of glucose oxidase into multiwall carbon nanotubes-polyelectrolyte-loaded electrospun nanofibrous membrane. Biosens. Bioelectron. 2008, 23, 771–779. [Google Scholar] [CrossRef]
- Scampicchio, M.; Arecchi, A.; Bianco, A.; Bulbarello, A.; Bertarelli, C.; Mannino, S. Nylon nanofibrous biosensors for glucose determination. Electroanalysis 2010, 22, 1056–1060. [Google Scholar]
- Shin, Y.J.; Wang, M.; Kameoka, J. Electrospun nanofiber biosensor for measuring glucose concentration. J. Photopolym. Sci. Technol. 2009, 22, 235–237. [Google Scholar]
- Yoon, O.J.; Kim, H.W.; Kim, D.J.; Lee, H.J.; Yun, J.Y.; Noh, Y.H.; Lee, D.Y.; Kim, D.H.; Kim, S.S.; Lee, N.E. Nanocomposites of electrospun poly[(D,L-lactic)-co-(glycolic acid)] and plasma-functionalized single-walled carbon nanotubes for biomedical applications. Plasma Process. Polym. 2009, 6, 101–109. [Google Scholar]
- Badrossamay, M.R.; McIlwee, H.A.; Goss, J.A.; Parker, K.K. Nanofiber assembly by rotary jet-spinning. Nano Lett. 2010, 10, 2257–2261. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar]
- Medeiros, E.S.; Glenn, G.M.; Klamczynski, A.P.; Orts, W.J.; Mattoso, L.H.C. Solution blow spinning: A new method to produce micro- and nanofibers from polymer solutions. J. Appl. Polym. Sci. 2009, 113, 2322–2330. [Google Scholar]
- Sinha-Ray, S.; Yarin, A.L.; Pourdeyhimi, B. Meltblowing: I-basic physical mechanisms and threadline model. J. Appl. Phys. 2010, 108, 034912–1. [Google Scholar]
- Shan, Y.P.; Yang, G.C.; Jia, Y.T.; Gong, J.; Su, Z.M.; Qu, L.Y. ITO electrode modified with chitosan nanofibers loading polyoxometalate by one step self-assembly method and its electrocatalysis. Electrochem. Commun. 2007, 9, 2224–2228. [Google Scholar]
- Tang, H.; Yan, F.; Tai, Q.D.; Chan, H.L.W. The improvement of glucose bioelectrocatalytic properties of platinum electrodes modified with electrospun TiO2 nanofibers. Biosens. Bioelectron. 2010, 25, 1646–1651. [Google Scholar] [CrossRef]
- Jia, H.F.; Zhu, G.Y.; Vugrinovich, B.; Kataphinan, W.; Reneker, D.H.; Wang, P. Enzyme-carrying polymeric nanofibers prepared via electrospinning for use as unique biocatalysts. Biotechnol. Prog. 2002, 18, 1027–1032. [Google Scholar]
- Joshi, P.P.; Merchant, S.A.; Wang, Y.D.; Schmidtke, D.W. Amperometric biosensors based on redox polymer-carbon nanotube-enzyme composites. Anal. Chem. 2005, 77, 3183–3188. [Google Scholar]
- Lee, K.P.; Gopalan, A.I.; Komathi, S. Direct electrochemistry of cytochrome c and biosensing for hydrogen peroxide on polyaniline grafted multi-walled carbon nanotube electrode. Sens. Actuat. B Chem. 2009, 141, 518–525. [Google Scholar]
- Liu, Y.; Chen, J.; Anh, N.T.; Too, C.O.; Misoska, V.; Wallace, G.G. Nanofiber mats from DNA, swnts, and poly(ethylene oxide) and their application in glucose biosensor. J. Electrochem. Soc. 2008, 155, K100–K103. [Google Scholar]
- Liu, Y.; Wang, M.K.; Zhao, F.; Xu, Z.A.; Dong, S.J. The direct electron transfer of glucose oxidase and glucose biosensor based on carbon nanotubes/chitosan matrix. Biosens. Bioelectron. 2005, 21, 984–988. [Google Scholar] [CrossRef]
- Merkoci, A.; Pumera, M.; Llopis, X.; Perez, B.; del Valle, M.; Alegret, S. New materials for electrochemical sensing VI: Carbon nanotubes. Trac-Trends Anal. Chem. 2005, 24, 826–838. [Google Scholar] [CrossRef]
- Trojanowicz, M. Analytical applications of carbon nanotubes: A review. Trac-Trends Anal. Chem. 2006, 25, 480–489. [Google Scholar] [CrossRef]
- Wohlstadter, J.N.; Wilbur, J.L.; Sigal, G.B.; Biebuyck, H.A.; Billadeau, M.A.; Dong, L.W.; Fischer, A.B.; Gudibande, S.R.; Jamieson, S.H.; Kenten, J.H.; et al. Carbon nanotube-based biosensor. Adv. Mater. 2003, 15, 1184–1187. [Google Scholar]
- McCullen, S.D.; Stano, K.L.; Stevens, D.R.; Roberts, W.A.; Monteiro-Riviere, N.A.; Clarke, L.I.; Gorga, R.E. Development, optimization, and characterization of electrospun poly(lactic acid) nanofibers containing multi-walled carbon nanotubes. J. Appl. Polym. Sci. 2007, 105, 1668–1678. [Google Scholar]
- Picciani, P.H.S.; Medeiros, E.S.; Pan, Z.L.; Wood, D.F.; Orts, W.J.; Mattoso, L.H.C.; Soares, B.G. Structural, electrical, mechanical, and thermal properties of electrospun poly(lactic acid)/polyaniline blend fibers. Macromol. Mater. Eng. 2010, 295, 618–627. [Google Scholar] [CrossRef]
- Oliveira, J.E.; Zucolotto, V.; Mattoso, L.H.C.; Medeiros, E.S. Multi-wall carbon nanotube/poly (lactic acid) nanocomposite fibrous membranes obtained by solution blow spinning. J. Nanosci. Nanotechnol. 2011. [Google Scholar] [CrossRef]
- Long, G.L.; Winefordner, J.D. Limit of detection. A closer look at the iupac definition. Anal. Chem. 1983, 55, 712–724. [Google Scholar]
- Lawrence, N.S.; Deo, R.P.; Wang, J. Electrochemical determination of hydrogen sulfide at carbon nanotube modified electrodes. Anal. Chim. Acta 2004, 517, 131–137. [Google Scholar] [CrossRef]
- Martinez, M.A.; Herrero, J.; Gutierrez, M.T. Electrochemical stability of indium tin oxide thin-films. Electrochim. Acta 1992, 37, 2565–2571. [Google Scholar] [CrossRef]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. Interfacial Electrochem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Zhao, Y.-D.; Zhang, W.-D.; Chen, H.; Luo, Q.-M.; Li, S.F.Y. Direct electrochemistry of horseradish peroxidase at carbon nanotube powder microelectrode. Sens. Actuat. B Chem. 2002, 87, 168–172. [Google Scholar] [CrossRef]
- Cai, C.; Chen, J. Direct electron transfer and bioelectrocatalysis of hemoglobin at a carbon nanotube electrode. Anal. Biochem. 2004, 325, 285–292. [Google Scholar]
- Kong, T.; Chen, Y.; Ye, Y.P.; Zhang, K.; Wang, Z.X.; Wang, X.P. An amperometric glucose biosensor based on the immobilization of glucose oxidase on the zno nanotubes. Sens. Actuat. B Chem. 2009, 138, 344–350. [Google Scholar]
- Wang, B.Q.; Li, B.; Deng, Q.; Dong, S.J. Amperometric glucose biosensor based on sol-gel organic-inorganic hybrid material. Anal. Chem. 1998, 70, 3170–3174. [Google Scholar]
- Wang, J.J.; Myung, N.V.; Yun, M.H.; Monbouquette, H.G. Glucose oxidase entrapped in polypyrrole on high-surface-area pt electrodes: A model platform for sensitive electroenzymatic biosensors. J. Electroanal. Chem. 2005, 575, 139–146. [Google Scholar]
- Li, Q.W.; Luo, G.A.; Feng, J.; Zhou, Q.; Zhang, L.; Zhu, Y.F. Amperometric detection of glucose with glucose oxidase absorbed on porous nanocrystalline TiO2 film. Electroanalysis 2001, 13, 413–416. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oliveira, J.E.; Mattoso, L.H.C.; Medeiros, E.S.; Zucolotto, V. Poly(lactic acid)/Carbon Nanotube Fibers as Novel Platforms for Glucose Biosensors. Biosensors 2012, 2, 70-82. https://doi.org/10.3390/bios2010070
Oliveira JE, Mattoso LHC, Medeiros ES, Zucolotto V. Poly(lactic acid)/Carbon Nanotube Fibers as Novel Platforms for Glucose Biosensors. Biosensors. 2012; 2(1):70-82. https://doi.org/10.3390/bios2010070
Chicago/Turabian StyleOliveira, Juliano Elvis, Luiz Henrique Capparelli Mattoso, Eliton Souto Medeiros, and Valtencir Zucolotto. 2012. "Poly(lactic acid)/Carbon Nanotube Fibers as Novel Platforms for Glucose Biosensors" Biosensors 2, no. 1: 70-82. https://doi.org/10.3390/bios2010070
APA StyleOliveira, J. E., Mattoso, L. H. C., Medeiros, E. S., & Zucolotto, V. (2012). Poly(lactic acid)/Carbon Nanotube Fibers as Novel Platforms for Glucose Biosensors. Biosensors, 2(1), 70-82. https://doi.org/10.3390/bios2010070





