Supported Bimetallic AuPd Nanoparticles as a Catalyst for the Selective Hydrogenation of Nitroarenes
Abstract
:1. Introduction
2. Experimental
2.1. Catalyst Preparation
2.2. Catalytic Testing
3. Results and Discussion
4. Conclusions
- (1)
- Through systematic catalyst screening, 1% AuPd/TiO2 catalyst prepared by modified impregnation method is the most active and stable catalyst for the solvent-free hydrogenation of nitrobenzene to aniline. After a 15 h reaction, a 99% yield of aniline was obtained at 90 °C. The activation energy of nitrobenzene reduction to aniline was calculated to be 37 kJ mol−1.
- (2)
- The 1% AuPd/TiO2 (MIm) catalyst also showed a good selectivity for the chemoselective hydrogenation of 4-chloronitrobenzene to 4-chloroaniline. Using the kinetic studies, we found that the activation energy of CNB transformation to CAN was calculated to be 25 kJ mol−1, which was lower than that of CNB to AN (31 kJ mol−1). Thus, by decreasing the reaction temperature from 60 °C to 25 °C, a 92% yield of CAN was achieved.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wang, J.H.; Yuan, Z.L.; Nie, R.F.; Hou, Z.Y.; Zheng, X.M. Hydrogenation of nitrobenzene to aniline over silica gel supported nickel catalysts. Ind. Eng. Chem. Res. 2010, 49, 4664–4669. [Google Scholar] [CrossRef]
- Lara, P.; Philippot, K. The hydrogenation of nitroarenes mediated by platinum nanoparticles: An overview. Catal. Sci. Technol. 2014, 4, 2445–2465. [Google Scholar] [CrossRef]
- Wei, H.S.; Liu, X.Y.; Wang, A.Q.; Zhang, L.L.; Qiao, B.T.; Yang, X.F.; Huang, Y.Q.; Miao, S.; Liu, J.Y.; Zhang, T. FeOx-supported platinum single-atom and pseudo-single-atom catalysts for chemoselective hydrogenation of functionalized nitroarenes. Nat. Commun. 2014, 5, 5634. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.Y.; Zhao, Y.F.; Xie, Y.; Tao, R.T.; Zhang, H.Y.; Huang, C.L.; Liu, Z.M. The solvent-free selective hydrogenation of nitrobenzene to aniline: An unexpected catalytic activity of ultrafine Pt nanoparticles deposited on carbon nanotubes. Green Chem. 2010, 12, 1007–1011. [Google Scholar] [CrossRef]
- Rylander, P.N. Hydrogenation Methods; Academic Press: New York, NY, USA, 1990. [Google Scholar]
- Nishimura, S. Handbook of Heterogeneous Catalytic Hydrogenation for Organic Synthesis; Wiley-VCH: New York, NY, USA, 2001. [Google Scholar]
- Turáková, M.; Salmi, T.; Eränen, K.; Wärnå, J.; Murzin, D.Y.; Králik, M. Liquid phase hydrogenation of nitrobenzene. Appl. Catal. A 2015, 499, 66–76. [Google Scholar] [CrossRef]
- Hu, Y.B.; Tao, K.; Wu, C.Z.; Zhou, C.; Yin, H.F.; Zhou, S.H. Size-controlled synthesis of highly stable and active Pd@SiO2 core-shell nanocatalysts for hydrogenation of nitrobenzene. J. Phys. Chem. C 2013, 117, 8974–8982. [Google Scholar] [CrossRef]
- Torres, C.; Campos, C.; Fierro, J.; Oportus, M.; Reyes, P. Nitrobenzene hydrogenation on Au/TiO2 and Au/SiO2 catalyst: Synthesis, characterization and catalytic activity. Catal. Lett. 2013, 143, 763–771. [Google Scholar] [CrossRef]
- Du, W.C.; Chen, G.Z.; Nie, R.F.; Li, Y.W.; Hou, Z.Y. Highly dispersed Pt in MIL-101: An efficient catalyst for the hydrogenation of nitroarenes. Catal. Commun. 2013, 41, 56–59. [Google Scholar] [CrossRef]
- Raj, K.J.A.; Prakash, M.G.; Mahalakshmy, R.; Elangovan, T.; Viswanathan, B. Liquid phase hydrogenation of nitrobenzene over nickel supported on Titania. Chin. J. Catal. 2012, 33, 1299–1305. [Google Scholar] [CrossRef]
- Manzoli, M.; Shetti, V.N.; Blaine, J.A.L.; Zhu, L.; Isrow, D.; Yempally, V.; Captain, B.; Coluccia, S.; Raja, R.; Gianotti, E. RuxPtySnz cluster-derived nanoparticle catalysts: Spectroscopic investigation into the nature of active multinuclear single sites. Dalton Trans. 2012, 41, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Q.; Li, Y.J.; Li, Y.J.; Zhou, H.L.; Duan, X.F.; Huang, Y. Synthesis of PtPd bimetal nanocrystals with controllable shape, composition, and their tunable catalytic properties. Nano Lett. 2012, 12, 4265–4270. [Google Scholar] [CrossRef] [PubMed]
- Turáková, M.; Králik, M.; Lehocký, P.; Pikna, L.; Smrčová, M.; Remeteiová, D.; Hudák, A. Influence of preparation method and palladium content on Pd/C catalysts activity in the liquid phase hydrogenation of nitrobenzene to aniline. Appl. Catal. A 2014, 476, 103–112. [Google Scholar] [CrossRef]
- Easterday, R.; Sanchez-Felix, O.; Losovyj, Y.; Pink, M.; Stein, B.D.; Morgan, D.G.; Rakitin, M.; Doluda, V.Y.; Sulman, M.G.; Mahmoud, W.E.; et al. Design of ruthenium/iron oxide nanoparticle mixtures for hydrogenation of nitrobenzene. Catal. Sci. Technol. 2015, 5, 1902–1910. [Google Scholar] [CrossRef]
- Sheldon, R.A. The E factor 25 years on: The rise of green chemistry and sustainability. Green Chem. 2017, 19, 18–43. [Google Scholar] [CrossRef]
- Huang, H.; Wang, X.; Tan, M.; Chen, C.; Zou, X.; Ding, W.; Lu, X. Solvent-free selective hydrogenation of nitroarenes using nanoclusters of palladium supported on nitrogen-doped ordered mesoporous carbon. ChemCatChem 2016, 8, 1485–1489. [Google Scholar] [CrossRef]
- Bianchi, C.L.; Canton, P.; Dimitratos, N.; Porta, F.; Prati, L. Selective oxidation of glycerol with oxygen using mono and bimetallic catalysts based on Au, Pd and Pt metals. Catal. Today 2005, 102, 203–212. [Google Scholar] [CrossRef]
- Enache, D.I.; Edwards, J.K.; Landon, P.; Solsona-Espriu, B.; Carley, A.F.; Herzing, A.A.; Watanabe, M.; Kiely, C.J.; Knight, D.W.; Hutchings, G.J. Solvent-free oxidation of primary alcohols to aldehydes using AuPd/TiO2 catalysts. Science 2006, 311, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Sankar, M.; Beale, A.M.; He, Q.; Kiely, C.J.; Bruijnincx, P.C.A.; Weckhuysen, B.M. High performing and stable supported nano-alloys for the catalytic hydrogenation of levulinic acid to γ-valerolactone. Nat. Commun. 2015, 6, 6540. [Google Scholar] [CrossRef] [PubMed]
- Freakley, S.J.; He, Q.; Harrhy, J.H.; Lu, L.; Crole, D.A.; Morgan, D.J.; Ntainjua, E.N.; Edwards, J.K.; Carley, A.F.; Borisevich, A.Y.; et al. Palladium-tin catalysts for the direct synthesis of H2O2 with high selectivity. Science 2016, 351, 965–968. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, N.; Freakley, S.J.; McVicker, R.U.; Althahban, S.M.; Dimitratos, N.; He, Q.; Morgan, D.J.; Jenkins, R.L.; Willock, D.J.; Taylor, S.H.; et al. Aqueous Au-Pd colloids catalyze selective CH4 oxidation to CH3OH with O2 under mild conditions. Science 2017, 358, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Sankar, M.; Dimitratos, N.; Miedziak, P.J.; Wells, P.P.; Kiely, C.J.; Hutchings, G.J. Designing bimetallic catalysts for a green and sustainable future. Chem. Soc. Rev. 2012, 41, 8099–8139. [Google Scholar] [CrossRef] [PubMed]
- Hutchings, G.J.; Kiely, C.J. Strategies for the synthesis of supported gold palladium nanoparticles with controlled morphology and composition. Acc. Chem. Res. 2013, 46, 1759–1772. [Google Scholar] [CrossRef] [PubMed]
- Tafesh, A.M.; Weiguny, J. A review of the selective catalytic reduction of aromatic nitro compounds into aromatic amines, isocyanates, carbamates, and ureas using CO. Chem. Rev. 1996, 96, 2035–2052. [Google Scholar] [CrossRef] [PubMed]
- Iihama, S.; Furukawa, S.; Komatsu, T. Efficient catalytic system for chemoselective hydrogenation of halonitrobenzene to haloaniline using PtZn intermetallic compound. ACS Catal. 2016, 6, 742–746. [Google Scholar] [CrossRef]
- Lyu, J.H.; Wang, J.G.; Lu, C.S.; Ma, L.; Zhang, Q.F.; He, X.B.; Li, X.N. Size-dependent halogenated nitrobenzene hydrogenation selectivity of Pd nanoparticles. J. Phys. Chem. C 2014, 118, 2594–2601. [Google Scholar] [CrossRef]
- Wei, Q.; Shi, Y.S.; Sun, K.Q.; Xu, B.Q. Pd-on-Si catalysts prepared via galvanic displacement for the selective hydrogenation of para-chloronitrobenzene. Chem. Commun. 2016, 52, 3026–3029. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, Y.; Ji, H.; Wei, Y.; Wu, N.; Zuo, B.; Wang, Q. Magnetic nanocomposite catalysts with high activity and selectivity for selective hydrogenation of ortho-chloronitrobenzene. J. Catal. 2005, 229, 114–118. [Google Scholar] [CrossRef]
- Cárdenas-Lizana, F.; Gómez-Quero, S.; Keane, M.A. Clean production of chloroanilines by selective gas phase hydrogenation over supported Ni catalysts. Appl. Catal. A 2008, 334, 199–206. [Google Scholar] [CrossRef]
- Sankar, M.; He, Q.; Morad, M.; Pritchard, J.; Freakley, S.J.; Edwards, J.K.; Taylor, S.H.; Morgan, D.J.; Carley, A.F.; Knight, D.W.; et al. Synthesis of stable ligand-free gold–palladium nanoparticles using a simple excess anion method. ACS Nano 2012, 6, 6600–6613. [Google Scholar] [CrossRef] [PubMed]
- Peneau, V.; He, Q.; Shaw, G.; Kondrat, S.A.; Davies, T.E.; Miedziak, P.; Forde, M.; Dimitratos, N.; Kiely, C.J.; Hutchings, G.J. Selective catalytic oxidation using supported gold-platinum and palladium-platinum nanoalloys prepared by sol-immobilisation. Phys. Chem. Chem. Phys. 2013, 15, 10636–10644. [Google Scholar] [CrossRef] [PubMed]
- Kesavan, L.; Tiruvalam, R.; Ab Rahim, M.H.; bin Saiman, M.I.; Enache, D.I.; Jenkins, R.L.; Dimitratos, N.; Lopez-Sanchez, J.A.; Taylor, S.H.; Knight, D.W.; et al. Solvent-free oxidation of primary carbon-hydrogen bonds in toluene using Au-Pd alloy nanoparticles. Science 2011, 331, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Landon, P.; Collier, P.J.; Carley, A.F.; Chadwick, D.; Papworth, A.J.; Burrows, A.; Kiely, C.J.; Hutchings, G.J. Direct synthesis of hydrogen peroxide from H2 and O2 using Pd and Au catalysts. Phys. Chem. Chem. Phys. 2003, 5, 1917–1923. [Google Scholar] [CrossRef]
- Morad, M.; Sankar, M.; Cao, E.H.; Nowicka, E.; Davies, T.E.; Miedziak, P.J.; Morgan, D.J.; Knight, D.W.; Bethell, D.; Gavriilidis, A.; et al. Solvent-free aerobic oxidation of alcohols using supported gold palladium nanoalloys prepared by a modified impregnation method. Catal. Sci. Technol. 2014, 4, 3120–3128. [Google Scholar] [CrossRef] [Green Version]
- Peureux, J.; Torres, M.; Mozzanega, H.; Giroir-Fendler, A.; Dalmon, J.A. Nitrobenzene liquid-phase hydrogenation in a membrane reactor. Catal. Today 1995, 25, 409–415. [Google Scholar] [CrossRef]
- Serpell, C.J.; Cookson, J.; Ozkaya, D.; Beer, P.D. Core@shell bimetallic nanoparticle synthesis via anion coordination. Nat. Chem. 2011, 3, 478–483. [Google Scholar] [CrossRef] [PubMed]

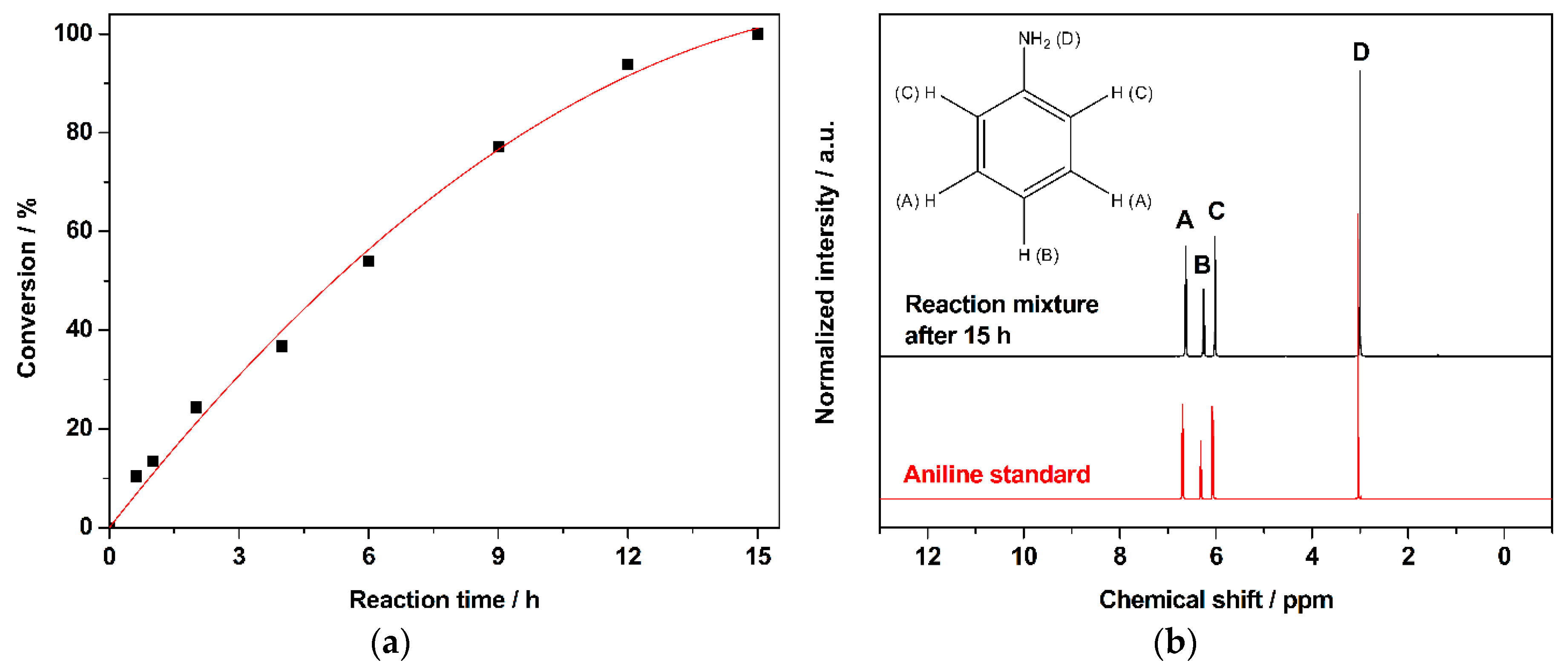
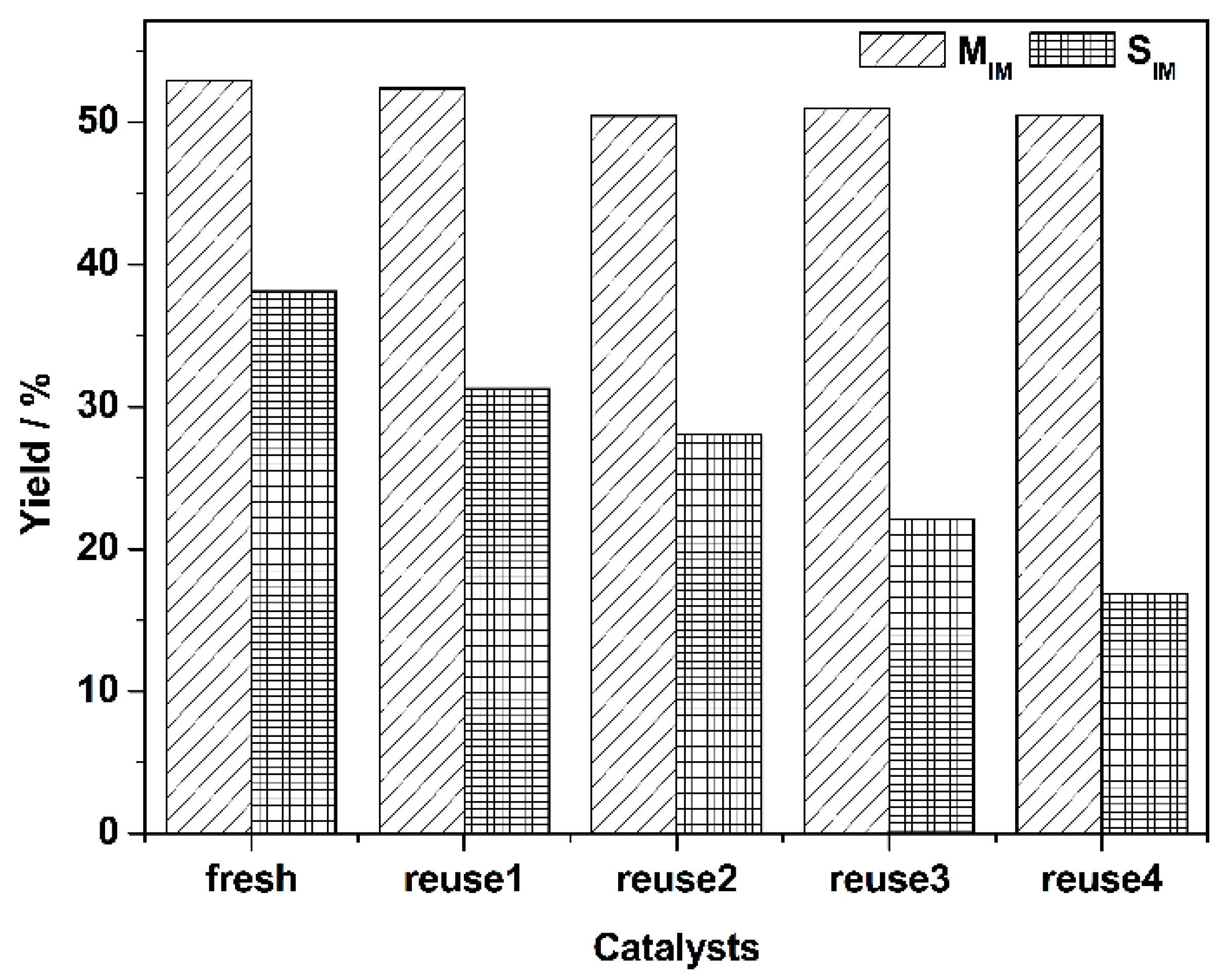


| Catalysts | Reaction Temperature (°C) | Nitrobenzene (NB) Conversion (%) | AN Selectivity (%) |
|---|---|---|---|
| 1% AuPd/TiO2 (MIm) | 30 | 1 | >99 |
| 60 | 11 | 97 | |
| 90 | 24 | 98 |
| Entry | Catalysts | Conversion (%) | Selectivity (%) | ||
|---|---|---|---|---|---|
| Aniline | Azobenzene | Azoxybenzene | |||
| 1 | AuPd/TiO2 (MIm) | 54 | 98 | 0.5 | 0.9 |
| 2 | Au/TiO2 (MIm) | 3 | 98 | 0 | 0 |
| 3 | Pd/TiO2 (MIm) | 41 | 97 | 0.2 | 0 |
| 4 | AuPd/MgO (MIm) | 36 | 94 | 0 | 0.5 |
| 5 | AuPd/C (MIm) | 14 | 76 | 0 | 0 |
| 6 | AuPd/TiO2 (CIm) | 39 | 99 | 0.4 | 0.3 |
| 7 | AuPd/TiO2 (SIm) | 38 | 99 | 0 | 0 |
| Entry | Catalysts | Reaction Time (h) | Conversion (%) | Selectivity (%) | |
|---|---|---|---|---|---|
| CAN | AN | ||||
| 1 | AuPd/TiO2 (CIm) | 1 | 96 | 83 | 17 |
| 2 | AuPd/TiO2 (SIm) | 1 | 100 | 70 | 30 |
| 3 | AuPd/TiO2 (MIm) | 1.5 | 100 | 85 | 15 |
| 4 | AuPd/TiO2 (MIm) b | 8 | 100 | 92 | 8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, R.; Macino, M.; Iqbal, S.; Gao, X.; He, Q.; Hutchings, G.J.; Sankar, M. Supported Bimetallic AuPd Nanoparticles as a Catalyst for the Selective Hydrogenation of Nitroarenes. Nanomaterials 2018, 8, 690. https://doi.org/10.3390/nano8090690
Qu R, Macino M, Iqbal S, Gao X, He Q, Hutchings GJ, Sankar M. Supported Bimetallic AuPd Nanoparticles as a Catalyst for the Selective Hydrogenation of Nitroarenes. Nanomaterials. 2018; 8(9):690. https://doi.org/10.3390/nano8090690
Chicago/Turabian StyleQu, Ruiyang, Margherita Macino, Sarwat Iqbal, Xiang Gao, Qian He, Graham John Hutchings, and Meenakshisundaram Sankar. 2018. "Supported Bimetallic AuPd Nanoparticles as a Catalyst for the Selective Hydrogenation of Nitroarenes" Nanomaterials 8, no. 9: 690. https://doi.org/10.3390/nano8090690







