Investigating the Synthesis and Characterization of a Novel “Green” H2O2-Assisted, Water-Soluble Chitosan/Polyvinyl Alcohol Nanofiber for Environmental End Uses
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Water Soluble Chitosan (WSCHT)
2.3. Solution Preparation for Electrospinning
2.4. Green Cross-Linking
2.5. Characterization
2.6. Swelling Ratio
2.7. Water Contact Angle
2.8. UV Protection
2.9. In Vitro Drug Release Study
2.10. Dye Removal Efficiency
3. Results and Discussion
3.1. Synthesis of Water Soluble Chitosan (WSCHT)
3.2. Solution Properties and Morphological Behaviour
3.3. Green Cross-Linking
3.4. Fourier Transform Infrared Spectroscopy (FT-IR)
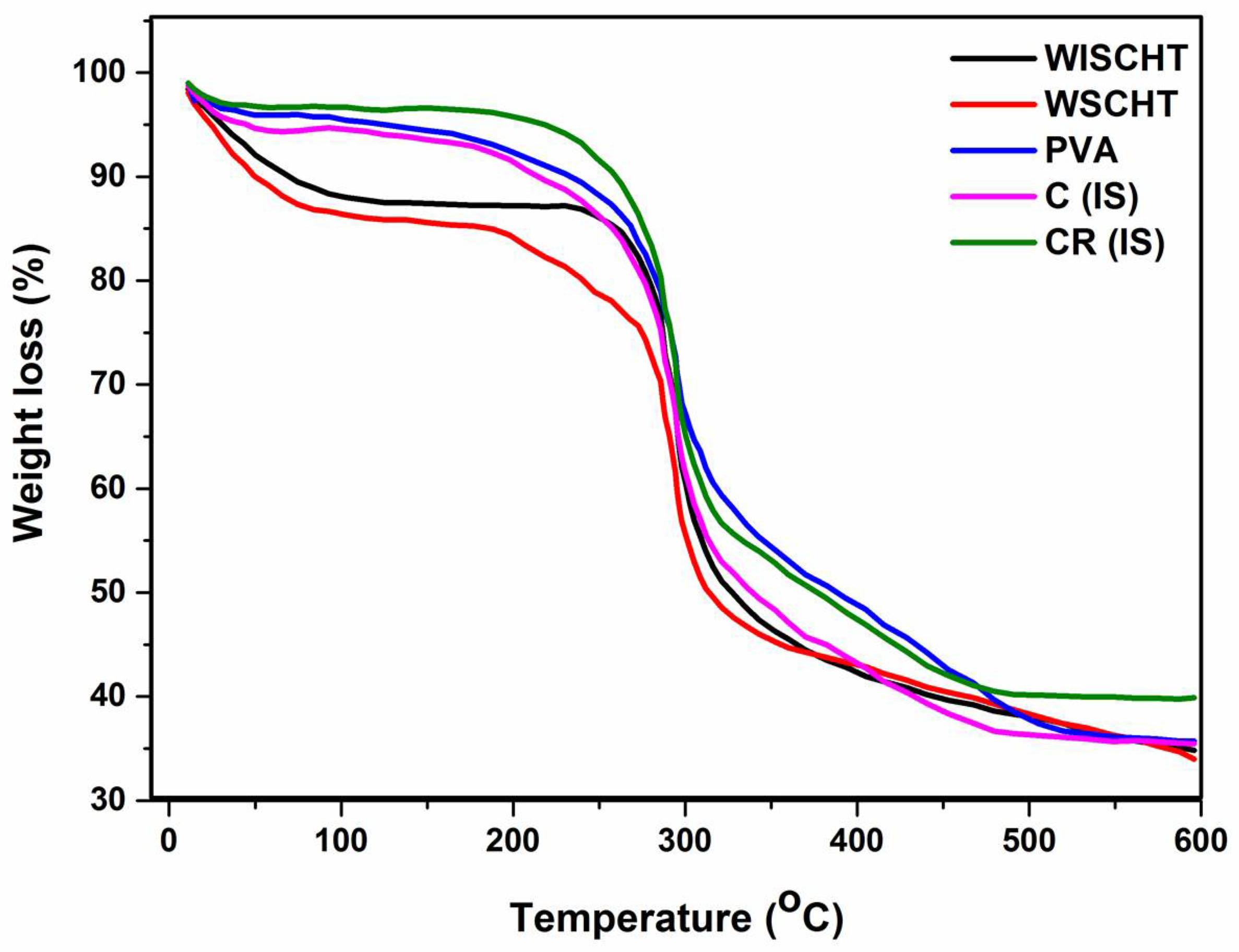
3.5. Thermogravimetric Analysis (TGA)
3.6. Differential Scanning Calorimetry (DSC)
3.7. X-ray Diffraction (XRD)
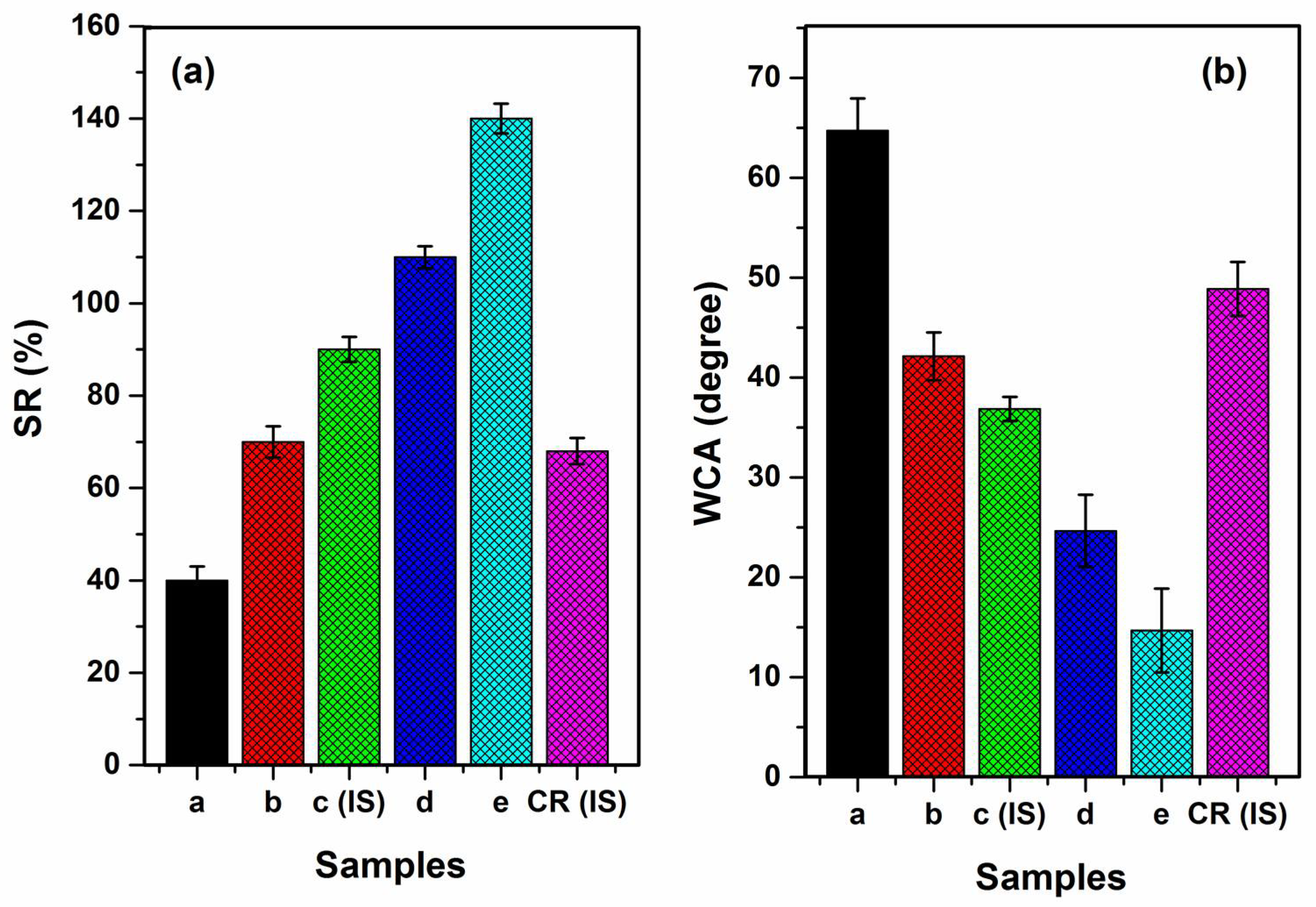
3.8. Swelling Ratio (SR)
3.9. Water Contact Angle (WCA)
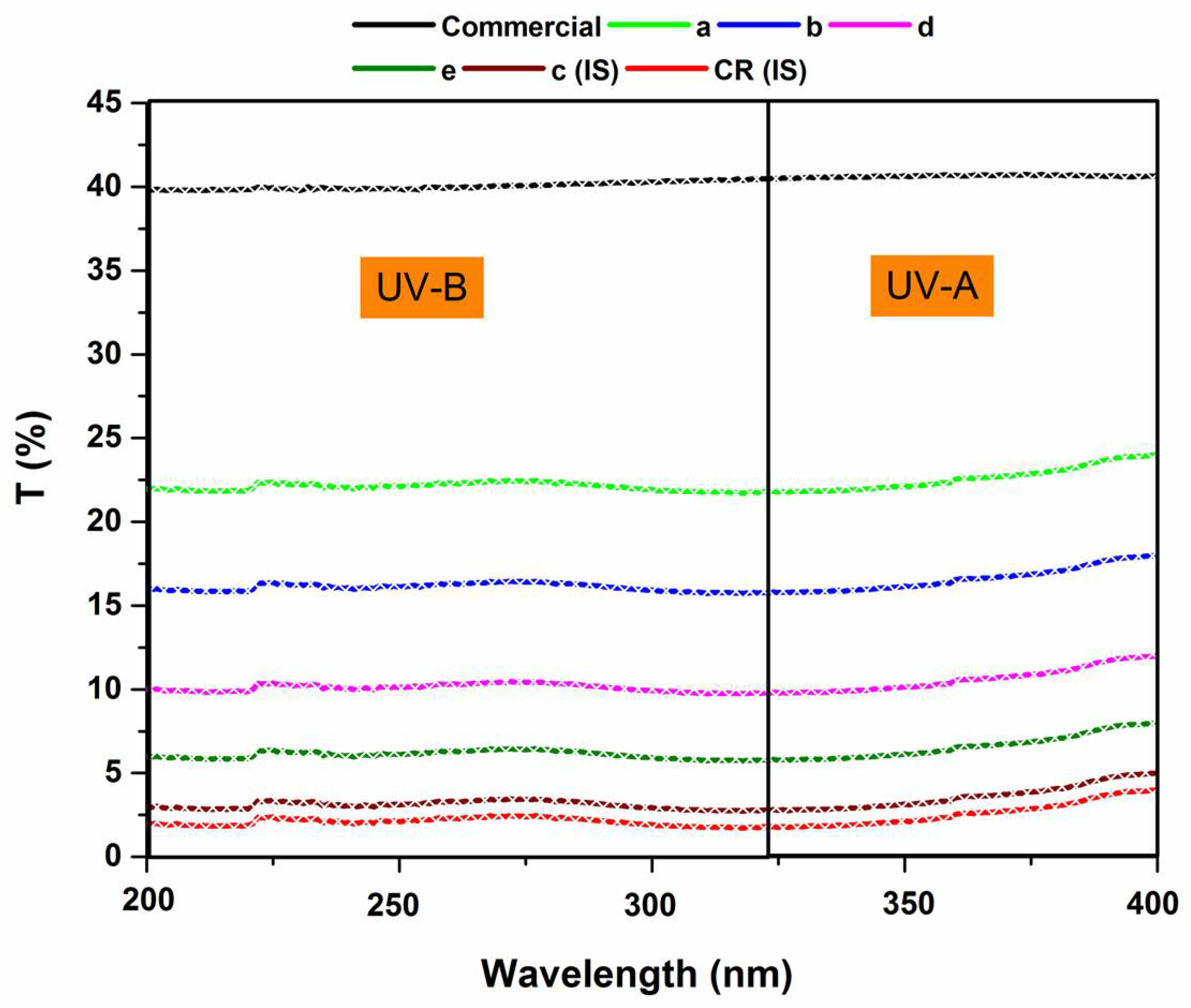
3.10. UV Protection
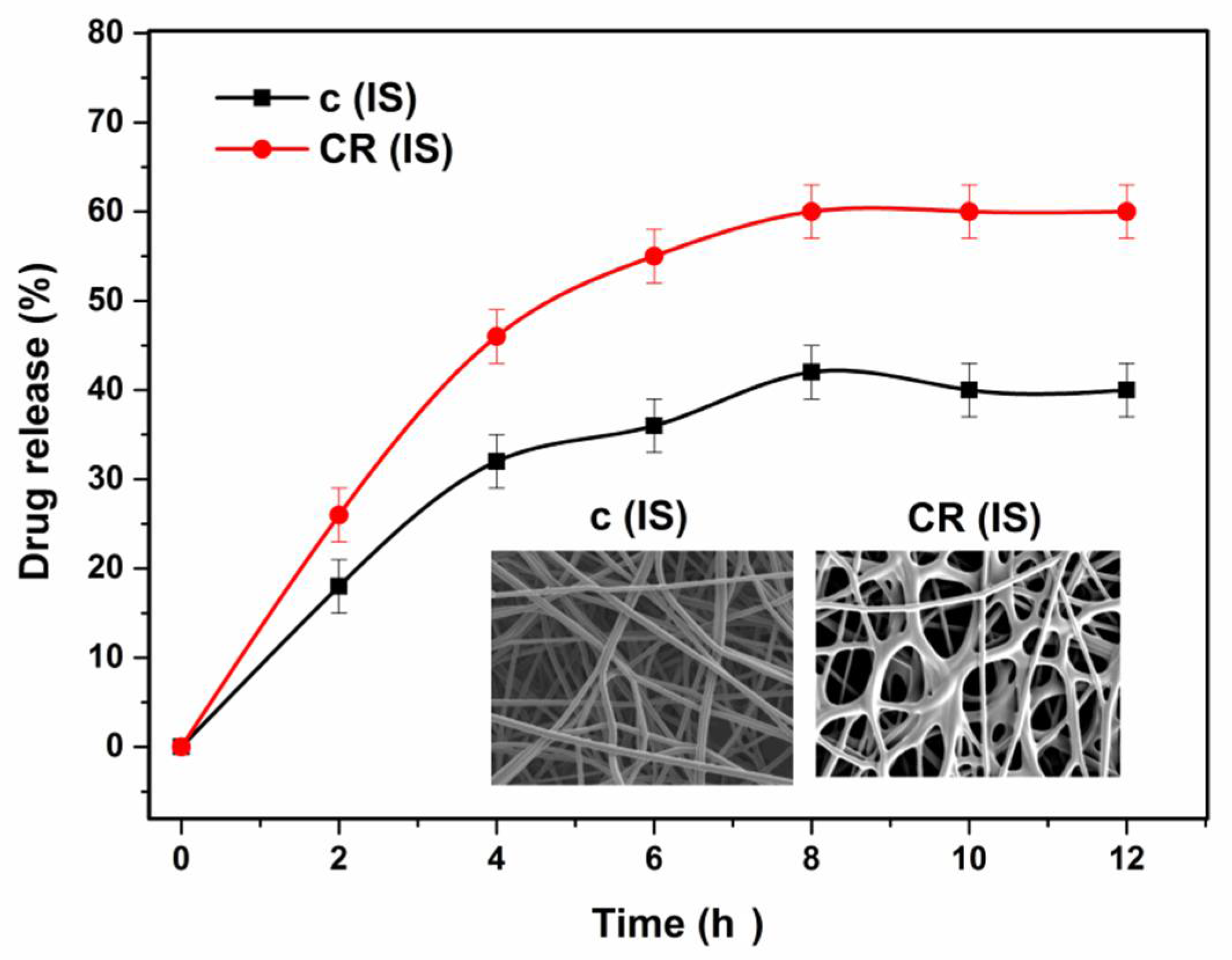
3.11. In Vitro Drug Release Study
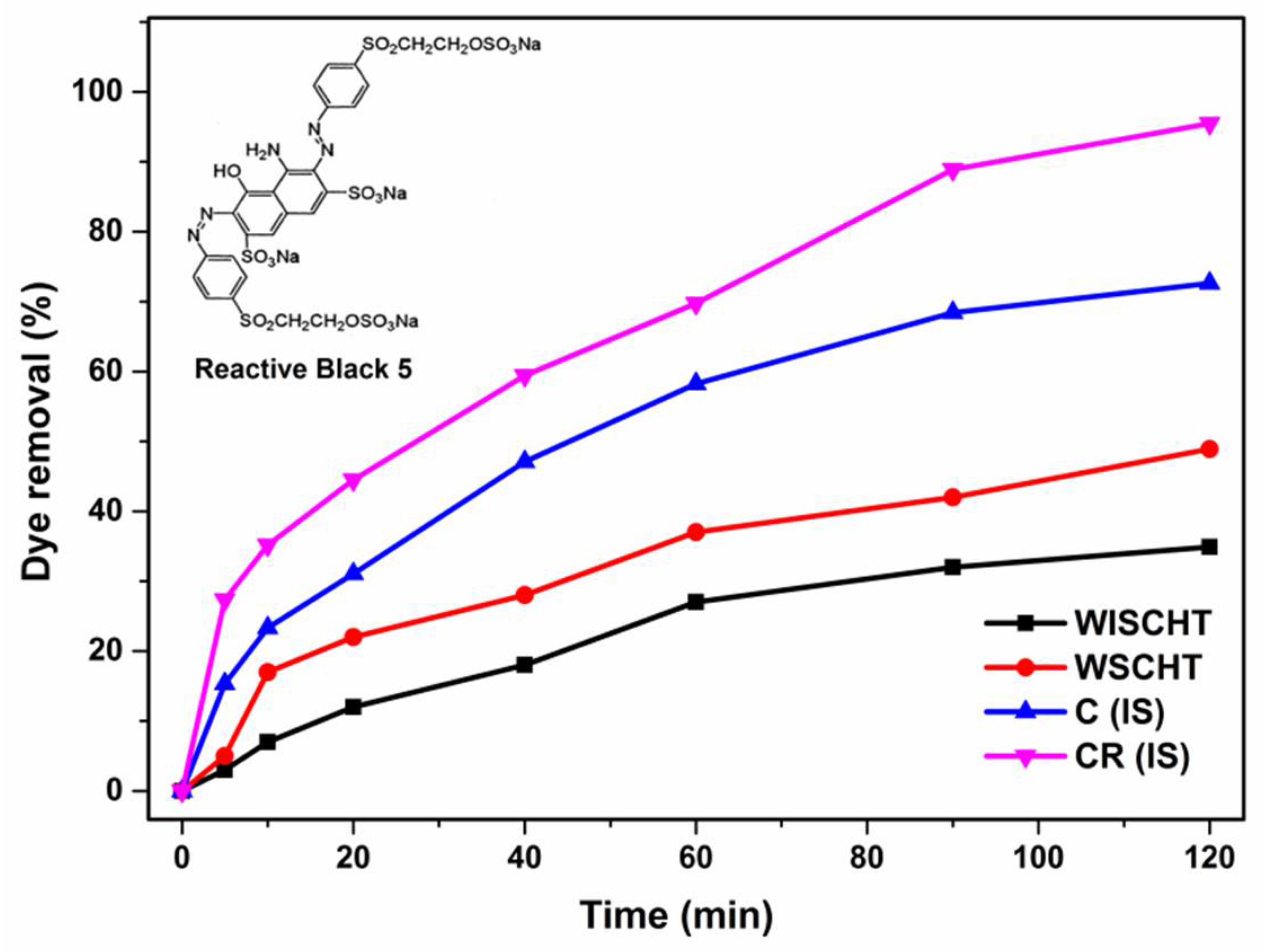
3.12. Adsorption Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tiwari, J.N.; Tiwari, R.N.; Kim, K.S. Zero-dimensional, one-dimensional, two-dimensional and three-dimensional nanostructured materials for advanced electrochemical energy devices. Prog. Mater. Sci. 2012, 57, 724–803. [Google Scholar] [CrossRef]
- Lee, S.J.; Yoo, J.J.; Lim, G.J.; Atala, A.; Stitzel, J. In vitro evaluation of electrospun nanofiber scaffolds for vascular graft application. J. Biomed. Mater. Res. A 2007, 83, 999–1008. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-J.; Wang, X.-M.; Cui, W.-J.; Dou, Y.-Q.; Zhao, D.-Y.; Xia, Y.-Y. Highly ordered mesoporous carbon nanofiber arrays from a crab shell biological template and its application in supercapacitors and fuel cells. J. Mater. Chem. 2010, 20, 4223–4230. [Google Scholar] [CrossRef]
- Wu, S.; Li, F.; Wu, Y.; Xu, R.; Li, G. Preparation of novel poly (vinyl alcohol)/SiO2 composite nanofiber membranes with mesostructure and their application for removal of Cu2+ from waste water. Chem. Commun. 2010, 46, 1694–1696. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.-M.; Zhang, Y.; Ramakrishna, S.; Lim, C. Electrospinning and mechanical characterization of gelatin nanofibers. Polymer 2004, 45, 5361–5368. [Google Scholar] [CrossRef]
- Bazbouz, M.B.; Stylios, G.K. Alignment and optimization of nylon 6 nanofibers by electrospinning. J. Appl. Polym. Sci 2008, 107, 3023–3032. [Google Scholar] [CrossRef]
- Yan, X.; Gevelber, M. Electrospinning of nanofibers: Characterization of jet dynamics and humidity effects. Part. Sci. Technol. 2017, 35, 139–149. [Google Scholar] [CrossRef]
- Kenry; Lim, C.T. Nanofiber technology: Current status and emerging developments. Prog. Polym. Sci. 2017, 70, 1–17. [Google Scholar] [CrossRef]
- Agarwal, S.; Greiner, A. On the way to clean and safe electrospinning—Green electrospinning: Emulsion and suspension electrospinning. Polym. Adv. Technol. 2011, 22, 372–378. [Google Scholar] [CrossRef]
- Fortunati, E.; Peltzer, M.; Armentano, I.; Torre, L.; Jiménez, A.; Kenny, J. Effects of modified cellulose nanocrystals on the barrier and migration properties of pla nano-biocomposites. Carbohydr. Polym. 2012, 90, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Goffin, A.-L.; Raquez, J.-M.; Duquesne, E.; Siqueira, G.; Habibi, Y.; Dufresne, A.; Dubois, P. From interfacial ring-opening polymerization to melt processing of cellulose nanowhisker-filled polylactide-based nanocomposites. Biomacromolecules 2011, 12, 2456–2465. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Martinová, L.; Lubasová, D. Electrospun chitosan based nanofibers. Res. J. Text. Appl. 2008, 12, 72–79. [Google Scholar] [CrossRef]
- Park, Y.J.; Lee, Y.M.; Park, S.N.; Sheen, S.Y.; Chung, C.P.; Lee, S.J. Platelet derived growth factor releasing chitosan sponge for periodontal bone regeneration. Biomaterials 2000, 21, 153–159. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, M. Calcium phosphate/chitosan composite scaffolds for controlled in vitro antibiotic drug release. J. Biomed. Mater. Res. 2002, 62, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Hu, H.; Luo, A.; Wang, Y.; Huang, X.; Song, P. Effect of carboxymethylation on the absorption and chelating properties of chitosan fibers. J. Appl. Polym. Sci. 2006, 99, 3110–3115. [Google Scholar] [CrossRef]
- Qian, L.; Zhang, H. Green synthesis of chitosan-based nanofibers and their applications. Green Chem. 2010, 12, 1207–1214. [Google Scholar] [CrossRef]
- Bedolla-Cázares, F.; Hernández-Marcelo, P.E.; Gómez-Hurtado, M.A.; Rodríguez-García, G.; Rosa, E.; López-Castro, Y.; García-Merinos, J.P.; Torres-Valencia, J.M.; González-Campos, J.B. Silver nanoparticles from AgNO3–affinin complex synthesized by an ecofriendly route: Chitosan-based electrospun composite production. Clean Technol. Environ. Policy 2017, 19, 897–906. [Google Scholar] [CrossRef]
- Bhattarai, N.; Edmondson, D.; Veiseh, O.; Matsen, F.A.; Zhang, M. Electrospun chitosan-based nanofibers and their cellular compatibility. Biomaterials 2005, 26, 6176–6184. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Su, B.; Ramakrishna, S.; Lim, C. Chitosan nanofibers from an easily electrospinnable uhmwpeo-doped chitosan solution system. Biomacromolecules 2007, 9, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Sargazi, G.; Afzali, D.; Mostafavi, A.; Ebrahimipour, S.Y. Synthesis of CS/PVA biodegradable composite nanofibers as a microporous material with well controllable procedure through electrospinning. J. Polym. Environ. 2017, 1–14. [Google Scholar] [CrossRef]
- Duan, B.; Yuan, X.; Zhu, Y.; Zhang, Y.; Li, X.; Zhang, Y.; Yao, K. A nanofibrous composite membrane of PLGA–chitosan/PVA prepared by electrospinning. Eur. Polym. J. 2006, 42, 2013–2022. [Google Scholar] [CrossRef]
- Esmaeili, A.; Beni, A.A. A novel fixed-bed reactor design incorporating an electrospun PVA/chitosan nanofiber membrane. J. Hazard. Mater. 2014, 280, 788–796. [Google Scholar] [CrossRef] [PubMed]
- Majd, S.A.; Khorasgani, M.R.; Moshtaghian, S.J.; Talebi, A.; Khezri, M. Application of chitosan/PVA nano fiber as a potential wound dressing for streptozotocin-induced diabetic rats. Int. J. Biol. Macromol. 2016, 92, 1162–1168. [Google Scholar] [CrossRef] [PubMed]
- Koosha, M.; Mirzadeh, H. Electrospinning, mechanical properties, and cell behavior study of chitosan/PVA nanofibers. J. Biomed. Mater. Res. A 2015, 103, 3081–3093. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Liu, Y.; Hu, K.; Zhao, B. The depolymerization mechanism of chitosan by hydrogen peroxide. J. Mater. Sci. 2003, 38, 4709–4712. [Google Scholar] [CrossRef]
- Yuan, Y.; Chesnutt, B.; Utturkar, G.; Haggard, W.; Yang, Y.; Ong, J.; Bumgardner, J. The effect of cross-linking of chitosan microspheres with genipin on protein release. Carbohydr. Polym. 2007, 68, 561–567. [Google Scholar] [CrossRef]
- Barrientos, I.J.H.; Paladino, E.; Brozio, S.; Passarelli, M.K.; Moug, S.; Black, R.A.; Wilson, C.G.; Lamprou, D.A. Fabrication and characterisation of drug-loaded electrospun polymeric nanofibers for controlled release in hernia repair. Int. J. Pharm. 2017, 517, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Elwakeel, K.Z.; El-Kousy, S.; El-Shorbagy, H.G.; El-Ghaffar, M.A. Comparison between the removal of reactive black 5 from aqueous solutions by 3-amino-1, 2, 4 triazole, 5-thiol and melamine grafted chitosan prepared through four different routes. J. Environ. Chem. Eng. 2016, 4, 733–745. [Google Scholar] [CrossRef]
- Ma, Z.; Wang, W.; Wu, Y.; He, Y.; Wu, T. Oxidative degradation of chitosan to the low molecular water-soluble chitosan over peroxotungstate as chemical scissors. PLoS ONE 2014, 9, e100743. [Google Scholar] [CrossRef] [PubMed]
- Provencher, S.W. Estimation of metabolite concentrations from localized in vivo proton nmr spectra. Magn. Reson. Med. 1993, 30, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Fu, C.; Wu, S.; Liu, G.; Guo, J.; Su, Z. Determination of the deacetylation degree of chitooligosaccharides. Mar. Drugs 2017, 15, 332. [Google Scholar] [CrossRef] [PubMed]
- Kasaai, M.R. A review of several reported procedures to determine the degree of N-acetylation for chitin and chitosan using infrared spectroscopy. Carbohydr. Polym. 2008, 71, 497–508. [Google Scholar] [CrossRef]
- Agarwal, S.; Greiner, A.; Wendorff, J.H. Functional materials by electrospinning of polymers. Prog. Polym. Sci. 2013, 38, 963–991. [Google Scholar] [CrossRef]
- Reneker, D.H.; Yarin, A.L.; Fong, H.; Koombhongse, S. Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J. Appl. Phys. 2000, 87, 4531–4547. [Google Scholar] [CrossRef]
- Briscoe, B.; Luckham, P.; Zhu, S. The effects of hydrogen bonding upon the viscosity of aqueous poly (vinyl alcohol) solutions. Polymer 2000, 41, 3851–3860. [Google Scholar] [CrossRef]
- Homayoni, H.; Ravandi, S.A.H.; Valizadeh, M. Electrospinning of chitosan nanofibers: Processing optimization. Carbohydr. Polym. 2009, 77, 656–661. [Google Scholar] [CrossRef]
- Jia, Z.; Li, Q.; Liu, J.; Yang, Y.; Wang, L.; Guan, Z. Preparation and properties of poly (vinyl alcohol) nanofibers by electrospinning. J. Polym. Eng. 2008, 28, 87–100. [Google Scholar] [CrossRef]
- Zhang, L.; Hsieh, Y.-L. Ultra-fine cellulose acetate/poly (ethylene oxide) bicomponent fibers. Carbohydr. Polym. 2008, 71, 196–207. [Google Scholar] [CrossRef]
- Dastidar, T.G.; Netravali, A.N. ‘Green’crosslinking of native starches with malonic acid and their properties. Carbohydr. Polym. 2012, 90, 1620–1628. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.; Cao, Z.; Hu, Y.; Song, Y.; Yu, L.; Yang, B.; Mu, J.; Huang, Z.; Han, Y. Effects of different cross-linking conditions on the properties of genipin-cross-linked chitosan/collagen scaffolds for cartilage tissue engineering. J. Mater. Sci. Mater. Med. 2011, 22, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Bao, J.; Wu, X.; Wu, Q.; Li, Y.; Zhou, Y.; Li, L.; Bu, H. Genipin crosslinking reduced the immunogenicity of xenogeneic decellularized porcine whole-liver matrices through regulation of immune cell proliferation and polarization. Sci. Rep. 2016, 6, 24779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butler, M.F.; Ng, Y.F.; Pudney, P.D. Mechanism and kinetics of the crosslinking reaction between biopolymers containing primary amine groups and genipin. J. Polym. Sci. A 2003, 41, 3941–3953. [Google Scholar] [CrossRef]
- Peniche, C.; Argüelles-Monal, W.; Davidenko, N.; Sastre, R.; Gallardo, A.; San Román, J. Self-curing membranes of chitosan/PAA IPNs obtained by radical polymerization: Preparation, characterization and interpolymer complexation. Biomaterials 1999, 20, 1869–1878. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.; Tanfani, F.; Emanuelli, M.; Mariotti, S. N-(carboxymethylidene) chitosans and N-(carboxymethyl) chitosans: Novel chelating polyampholytes obtained from chitosan glyoxylate. Carbohydr. Res. 1982, 107, 199–214. [Google Scholar] [CrossRef]
- Mansur, H.S.; Sadahira, C.M.; Souza, A.N.; Mansur, A.A. FTIR spectroscopy characterization of poly (vinyl alcohol) hydrogel with different hydrolysis degree and chemically crosslinked with glutaraldehyde. Mater. Sci. Eng. C 2008, 28, 539–548. [Google Scholar] [CrossRef]
- Alhosseini, S.N.; Moztarzadeh, F.; Mozafari, M.; Asgari, S.; Dodel, M.; Samadikuchaksaraei, A.; Kargozar, S.; Jalali, N. Synthesis and characterization of electrospun polyvinyl alcohol nanofibrous scaffolds modified by blending with chitosan for neural tissue engineering. Int. J. Nanomed. 2012, 7, 25. [Google Scholar] [CrossRef] [Green Version]
- Mathew, J.; Kodama, M. Study of blood compatible polymers I. Modification of poly (vinyl alcohol). Polym. J. 1992, 24, 31–41. [Google Scholar] [CrossRef]
- Khan, Y.H.; Islam, A.; Sarwar, A.; Gull, N.; Khan, S.M.; Munawar, M.A.; Zia, S.; Sabir, A.; Shafiq, M.; Jamil, T. Novel green nano composites films fabricated by indigenously synthesized graphene oxide and chitosan. Carbohydr. Polym. 2016, 146, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, A.; Ikoma, T.; Kobayashi, H.; Tanaka, J. Preparation and mechanical property of core-shell type chitosan/calcium phosphate composite fiber. Mater. Sci. Eng. C 2004, 24, 723–728. [Google Scholar] [CrossRef]
- Qin, C.; Du, Y.; Xiao, L. Effect of hydrogen peroxide treatment on the molecular weight and structure of chitosan. Polym. Degrad. Stab. 2002, 76, 211–218. [Google Scholar] [CrossRef]
- Santos, C.; Silva, C.J.; Büttel, Z.; Guimarães, R.; Pereira, S.B.; Tamagnini, P.; Zille, A. Preparation and characterization of polysaccharides/PVA blend nanofibrous membranes by electrospinning method. Carbohydr. Polym. 2014, 99, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-P.; Chen, S.-H.; Lai, G.-J. Preparation and characterization of biomimetic silk fibroin/chitosan composite nanofibers by electrospinning for osteoblasts culture. Nanoscale Res. Lett. 2012, 7, 170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Semnani, D.; Afrashi, M.; Alihosseini, F.; Dehghan, P.; Maherolnaghsh, M. Investigating the performance of drug delivery system of fluconazole made of nano–micro fibers coated on cotton/polyester fabric. J. Mater. Sci. Mater. Med. 2017, 28, 175. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.H.; Wei, G.S.; Peng, J. Radiation degradation of chitosan in the presence of H2O2. J. Polym. Sci. 2004, 22, 439–444. [Google Scholar]
- Shuai, C.; Mao, Z.; Lu, H.; Nie, Y.; Hu, H.; Peng, S. Fabrication of porous polyvinyl alcohol scaffold for bone tissue engineering via selective laser sintering. Biofabrication 2013, 5, 015014. [Google Scholar] [CrossRef] [PubMed]
- Islam, A.; Yasin, T.; Rafiq, M.A.; Shah, T.H.; Sabir, A.; Khan, S.M.; Jamil, T. In-situ crosslinked nanofiber mats of chitosan/poly (vinyl alcohol) blend: Fabrication, characterization and mtt assay with cancerous bone cells. Fibers Polym. 2015, 16, 1853–1860. [Google Scholar] [CrossRef]
- Hideko, O.T.; Yoshikazu, T.; Ayako, K. Rapid controlled hydrolytic degradation of poly(l-lactic acid) by blending with poly(aspartic acid-co-l-lactide). Polym. Degrad. Stab. 2009, 94, 1419–1426. [Google Scholar] [CrossRef]
- Tighzert, W.; Habi, A.; Ajji, A.; Sadoun, T.; Daoud, F.B.-O. Fabrication and characterization of nanofibers based on poly (lactic acid)/chitosan blends by electrospinning and their functionalization with phospholipase a1. Fibers Polym. 2017, 18, 514–524. [Google Scholar] [CrossRef]
- Wali, A.; Zhang, Y.; Sengupta, P.; Higaki, Y.; Takahara, A.; Badiger, M.V. Electrospinning of non-ionic cellulose ethers/polyvinyl alcohol nanofibers: Characterization and applications. Carbohydr. Polym. 2018, 181, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Donawho, C.K.; Muller, H.K.; Bucana, C.D.; Kripke, M.L. Enhanced growth of murine melanoma in ultraviolet-irradiated skin is associated with local inhibition of immune effector mechanisms. J. Immunol. 1996, 157, 781–786. [Google Scholar] [PubMed]
- Ananthaswamy, H.N.; Pierceall, W.E. Molecular mechanisms of ultraviolet radiation carcinogenesis. Photochem. Photobiol. 1990, 52, 1119–1136. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, S.; Ghoranneviss, M.; Dalalsharifi, S. Preparation of multifunctional wool fabric using chitosan after plasma treatment. J. Text. Inst. 2015, 106, 1127–1134. [Google Scholar] [CrossRef]
- Zhang, H.-L.; Wu, S.-H.; Tao, Y.; Zang, L.-Q.; Su, Z.-Q. Preparation and characterization of water-soluble chitosan nanoparticles as protein delivery system. J. Nanomater. 2010, 2010, 1. [Google Scholar] [CrossRef]
- Hu, W.; Huang, Z.-M.; Liu, X.-Y. Development of braided drug-loaded nanofiber sutures. Nanotechnology 2010, 21, 315104. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Chen, J.; Yang, H.; Wang, W.; Liu, Y.; Zou, D.; Liu, W.; Men, G. Preparation and application of aliquat 336 functionalized chitosan adsorbent for the removal of Pb (II). Chem. Eng. J. 2013, 232, 372–379. [Google Scholar] [CrossRef]
- Jawad, A.H.; Islam, M.A.; Hameed, B. Cross-linked chitosan thin film coated onto glass plate as an effective adsorbent for adsorption of reactive orange 16. Int. J. Biol. Macromol. 2017, 95, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Guibal, E. Heterogeneous catalysis on chitosan-based materials: A review. Prog. Polym. Sci. 2005, 30, 71–109. [Google Scholar] [CrossRef]
- Mahmoodi, N.M. Dendrimer functionalized nanoarchitecture: Synthesis and binary system dye removal. J. Taiwan. Inst. Chem. Eng. 2014, 45, 2008–2020. [Google Scholar] [CrossRef]
- Mirmohseni, A.; Dorraji, M.S.; Figoli, A.; Tasselli, F. Chitosan hollow fibers as effective biosorbent toward dye: Preparation and modeling. Bioresour. Technol. 2012, 121, 212–220. [Google Scholar] [CrossRef] [PubMed]






| Sample Code | WSCHT:PVA Mass Ratio | Viscosity (cP) | Conductivity (mS/cm) | Surface Tension (mN/m) |
|---|---|---|---|---|
| a | 10:90 | 3.26 | 1.26 | 41.89 |
| b | 15:85 | 3.02 | 1.32 | 42.66 |
| c | 20:80 | 2.89 | 1.53 | 43.57 |
| d | 25:75 | 2.63 | 1.62 | 45.77 |
| e | 30:70 | 2.42 | 1.88 | 46.56 |
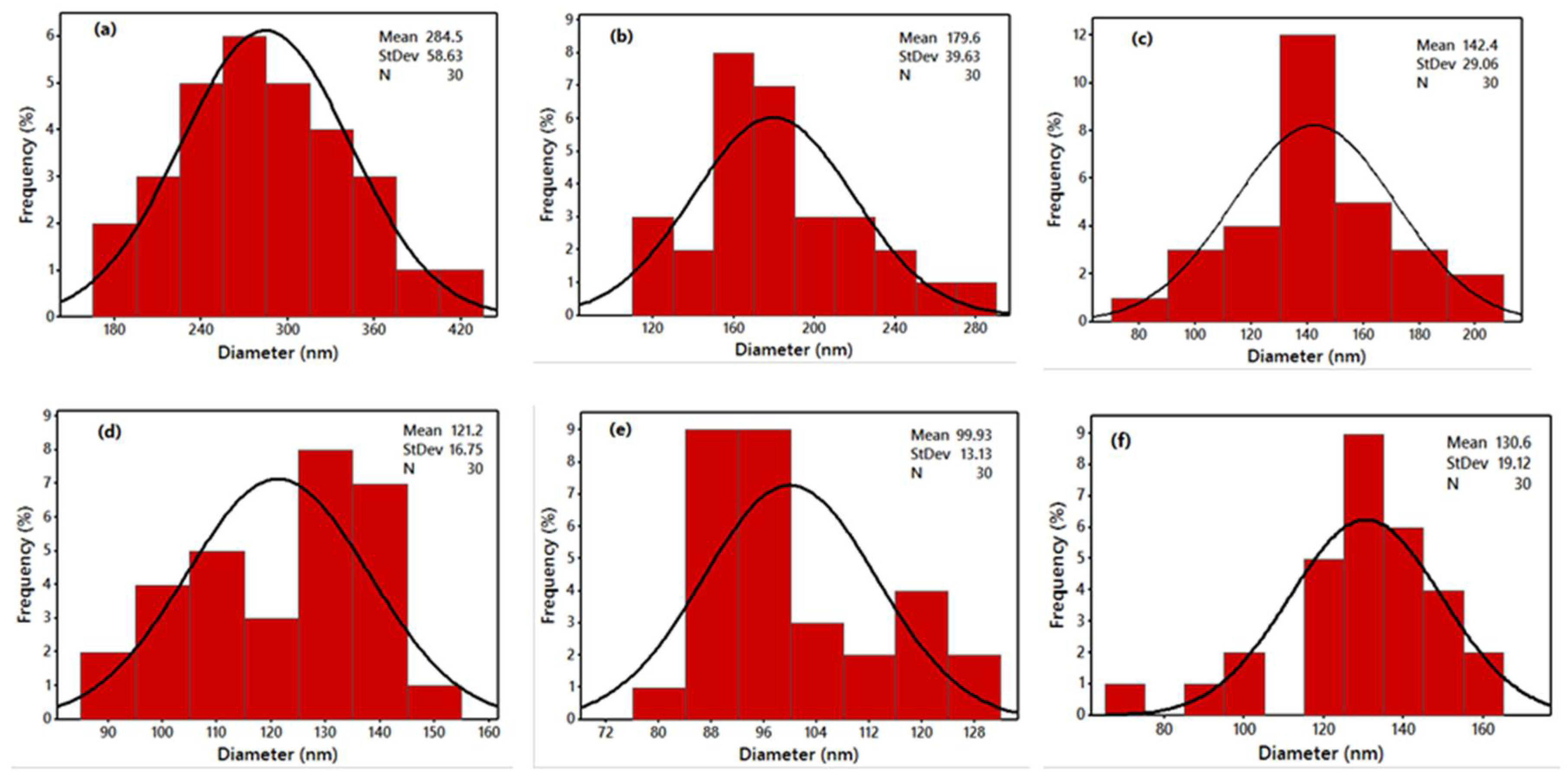
| Sample Code | WSCHT:PVA Mass Ratio | Diameter (nm) |
|---|---|---|
| a | 10:90 | 284.5 |
| b | 15:85 | 179.6 |
| c | 20:80 | 142.4 |
| d | 25:75 | 121.2 |
| e | 30:70 | 99.93 |
| f | 20:80 | 130.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pervez, M.N.; Stylios, G.K. Investigating the Synthesis and Characterization of a Novel “Green” H2O2-Assisted, Water-Soluble Chitosan/Polyvinyl Alcohol Nanofiber for Environmental End Uses. Nanomaterials 2018, 8, 395. https://doi.org/10.3390/nano8060395
Pervez MN, Stylios GK. Investigating the Synthesis and Characterization of a Novel “Green” H2O2-Assisted, Water-Soluble Chitosan/Polyvinyl Alcohol Nanofiber for Environmental End Uses. Nanomaterials. 2018; 8(6):395. https://doi.org/10.3390/nano8060395
Chicago/Turabian StylePervez, Md. Nahid, and George K. Stylios. 2018. "Investigating the Synthesis and Characterization of a Novel “Green” H2O2-Assisted, Water-Soluble Chitosan/Polyvinyl Alcohol Nanofiber for Environmental End Uses" Nanomaterials 8, no. 6: 395. https://doi.org/10.3390/nano8060395






