Highly Efficient and Stable Organic Solar Cells via Interface Engineering with a Nanostructured ITR-GO/PFN Bilayer Cathode Interlayer
Abstract
:1. Introduction
2. Experimental Section
3. Result and Discussion
3.1. Fundamental Properties of the ITR-GO Template
3.2. Characterization of ITR-GO/PFN Bilayer CILs
3.3. Device Performance of OSCs Based on Various CILs
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kaltenbrunner, M.; Sekitani, T.; Reeder, J.; Yokota, T.; Kuribara, K.; Tokuhara, T.; Drack, M.; Schwodiauer, R.; Graz, I.; Bauer-Gogonea, S.; et al. An ultra-light weight design for imperceptible plastic electronics. Nature 2013, 499, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zheng, Y.; Huang, J. Towards high performance organic photovoltaic cells: A review of recent development in organic photovoltaics. Polymers 2014, 6, 2473–2509. [Google Scholar] [CrossRef]
- Huang, J.; Wang, H.; Yan, K.; Zhang, X.; Chen, H.; Li, C.-Z.; Yu, J. Highly efficient organic solar cells consisting of double bulk heterojunction layers. Adv. Mater. 2017, 29, 1606729. [Google Scholar] [CrossRef] [PubMed]
- Xing, S.; Wang, H.; Kong, T.; Yu, J.; Jiang, J.; Wang, C. Realization of performance enhancement in ternary polymer solar cells by broad absorption, efficient energy transfer, and balanced charge carrier mobility. IEEE J. Photovolt. 2017, 7, 1058–1064. [Google Scholar] [CrossRef]
- Kong, T.; Wang, H.; Liu, X.; Yu, J.; Wang, C. Improving the efficiency of bulk heterojunction polymer solar cells via binary-solvent treatment. IEEE J. Photovolt. 2017, 7, 214–220. [Google Scholar] [CrossRef]
- Darling, S.B.; You, F. The case for organic photovoltaics. RSC Adv. 2013, 3, 17633–17648. [Google Scholar] [CrossRef]
- Ouyang, X.; Peng, R.; Ai, L.; Zhang, X.; Ge, Z. Efficient polymer solar cells employing a non-conjugated small-molecule electrolyte. Nat. Photonics 2015, 9, 520–524. [Google Scholar] [CrossRef]
- Lu, L.Y.; Xu, T.; Chen, W.; Landry, E.S.; Yui, L.P. Ternary blend polymer solar cells with enhanced power conversion efficiency. Nat. Photonics 2014, 8, 716–722. [Google Scholar] [CrossRef]
- Zhao, W.; Qian, D.; Zhang, S.; Li, S.; Inganas, O.; Gao, F.; Hou, J. Fullerene-free polymer solar cells with over 11% efficiency and excellent thermal stability. Adv. Mater. 2016, 28, 4734–4739. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhang, K.; Lu, J.; Zhang, J.; Yip, H.-L.; Huang, F.; Cao, Y. High-efficiency polymer solar cells via the incorporation of an amino-functionalized conjugated metallopolymer as a cathode interlayer. J. Am. Chem. Soc. 2013, 135, 15326–15329. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhong, C.; Huang, X.; Wong, W.-Y.; Wu, H.; Chen, L.; Su, S.; Cao, Y. Simultaneous enhancement of open-circuit voltage, short-circuit current density, and fill factor in polymer solar cells. Adv. Mater. 2011, 23, 4636–4643. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, P.; Wang, Y.; Sarvari, H.; Liu, D.; Wu, J.; Yang, Y.; Wang, Z.; Chen, Z.D. Interface engineering of high efficiency perovskite solar cells based on ZnO nanorods using atomic layer deposition. Nano Res. 2017, 10, 1092–1103. [Google Scholar] [CrossRef]
- Wang, Y.; Li, S.; Zhang, P.; Liu, D.; Gu, X.; Sarvari, H.; Ye, Z.; Wu, J.; Wang, Z.; Chen, Z.D. Solvent annealing of PbI2 for the high-quality crystallization of perovskite films for solar cells with efficiencies exceeding 18%. Nanoscale 2016, 8, 19654–19661. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Cheun, H.; Potscavage, W.J., Jr.; Fuentes-Hernandez, C.; Kim, S.-J.; Kippelen, B. Inverted organic solar cells with ITO electrodes modified with an ultrathin Al2O3 buffer layer deposited by atomic layer deposition. J. Mater. Chem. 2010, 20, 6189–6194. [Google Scholar] [CrossRef]
- Jagadamma, L.K.; Al-Senani, M.; El-Labban, A.; Gereige, I.; Ndjawa, G.O.N.; Faria, J.C.D.; Kim, T.; Zhao, K.; Cruciani, F.; Anjum, D.H. Polymer solar cells with efficiency > 10% enabled via a facile solution-processed Al-doped ZnO electron transporting layer. Adv. Energy Mater. 2015, 5, 1500204. [Google Scholar] [CrossRef]
- Kuwabara, T.; Nakamoto, M.; Kawahara, Y.; Yamaguchi, T.; Takahashi, K. Characterization of ZnS-layer-inserted bulk-heterojunction organic solar cells by ac impedance spectroscopy. J. Appl. Phys. 2009, 105, 124513. [Google Scholar] [CrossRef]
- Zhu, J.-J.; Xu, Z.-Q.; Fan, G.-Q.; Lee, S.-T.; Li, Y.-Q.; Tang, J.-X. Inverted polymer solar cells with atomic layer deposited cds film as an electron collection layer. Org. Electron. 2011, 12, 2151–2158. [Google Scholar] [CrossRef]
- Woo, S.; Kim, W.H.; Kim, H.; Yi, Y.; Lyu, H.-K.; Kim, Y. 8.9% single-stack inverted polymer solar cells with electron-rich polymer nanolayer-modified inorganic electron-collecting buffer layers. Adv. Energy Mater. 2014, 4, 1301692. [Google Scholar] [CrossRef]
- He, Z.; Zhong, C.; Su, S.; Xu, M.; Wu, H.; Cao, Y. Enhanced power-conversion efficiency in polymer solar cells using an inverted device structure. Nat. Photonics 2012, 6, 591–595. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, F.; Wang, J.; An, Q.; Zhao, C.; Li, L.; Teng, F.; Hu, B. A two-step strategy to clarify the roles of a solution processed PFN interfacial layer in highly efficient polymer solar cells. J. Mater. Chem. A 2015, 3, 18432–18441. [Google Scholar] [CrossRef]
- Huang, F.; Wu, H.; Cao, Y. Water/alcohol soluble conjugated polymers as highly efficient electron transporting/injection layer in optoelectronic devices. Chem. Soc. Rev. 2010, 39, 2500–2521. [Google Scholar] [CrossRef] [PubMed]
- Duan, C.; Cai, W.; Hsu, B.B.Y.; Zhong, C.; Zhang, K.; Liu, C.; Hu, Z.; Huang, F.; Bazan, G.C.; Heeger, A.J.; et al. Toward green solvent processable photovoltaic materials for polymer solar cells: The role of highly polar pendant groups in charge carrier transport and photovoltaic behavior. Energy Environ. Sci. 2013, 6, 3022–3034. [Google Scholar] [CrossRef]
- Han, C.; Cheng, Y.; Chen, L.; Qian, L.; Yang, Z.; Xue, W.; Zhang, T.; Yang, Y.; Cao, W. Enhanced performance of inverted polymer solar cells by combining ZnO nanoparticles and poly (9,9-bis(3′-(N,N-dimethylamino)propyl)-2,7-fluorene)-alt-2,7-(9,9-dioctyfluorene) as electron transport layer. ACS Appl. Mater. Interfaces 2016, 8, 3301–3307. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Cheng, X.; Xu, H.; Yang, H.; Liu, H.; Wu, F.; Chen, L.; Chen, Y. Interface-induced face-on orientation of the active layer by self-assembled diblock conjugated polyelectrolytes for efficient organic photovoltaic cells. J. Mater. Chem. A 2016, 4, 18478–18489. [Google Scholar] [CrossRef]
- Paulo, S.; Palomares, E.; Martinez-Ferrero, E. Graphene and carbon quantum dot-based materials in photovoltaic devices: From synthesis to applications. Nanomaterials 2016, 6, 157. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Wu, Z.; Jiang, Y.; Liu, W.; Li, X.; Jiao, B.; Abbas, W.; Hou, X. A flexible and thin graphene/silver nanowires/polymer hybrid transparent electrode for optoelectronic devices. ACS Appl. Mater. Interfaces 2016, 8, 31212–31221. [Google Scholar] [CrossRef] [PubMed]
- Tung, V.C.; Kim, J.; Huang, J. Graphene oxide: Single-walled carbon nanotube-based interfacial layer for all-solution-processed multijunction solar cells in both regular and inverted geometries. Adv. Energy Mater. 2012, 2, 299–303. [Google Scholar] [CrossRef]
- Udorn, J.; Hatta, A.; Furuta, H. Carbon nanotube (CNT) honeycomb cell area-dependent optical reflectance. Nanomaterials 2016, 6, 202. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Huang, W.; Fan, P.; Zheng, Y.; Huang, J.; Yu, J. Preparation of reduced graphene oxide: ZnO hybrid cathode interlayer using in situ thermal reduction/annealing for interconnecting nanostructure and its effect on organic solar cell. ACS Appl. Mater. Interfaces 2017, 9, 4898–4907. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Yang, G.; Zheng, Y.; Fan, P.; Ji, R.; Huang, J.; Zhang, W.; Yu, J. Carbon nano-onions as a functional dopant to modify hole transporting layers for improving stability and performance of planar perovskite solar cells. Electrochim. Acta 2017, 247, 548–557. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Zhang, L.; Lee, S.; Dai, H. Chemically derived, ultrasmooth grapheme nanoribbon semiconductors. Science 2008, 319, 1229–1232. [Google Scholar] [CrossRef] [PubMed]
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, D.; Huang, J.; Zheng, Y.; Yu, J. High performance airbrush spray coated organic solar cells via tuning the surface tension and saturated vapor pressure of different ternary solvent systems. Org. Electron. 2015, 25, 275–282. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved synthesis of graphene oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.C.; Ho, C.C.; Chang, C.Y.; Jao, M.-H.; Darling, S.B.; Su, W.F. Additives for morphology control in high-efficiency organic solar cells. Mater. Today 2013, 16, 326–336. [Google Scholar] [CrossRef]
- Viet Hung, P.; Tran Viet, C.; Nguyen-Phan, T.-D.; Hai Dinh, P.; Kim, E.J.; Hur, S.H.; Shin, E.W.; Kim, S.; Chung, J.S. One-step synthesis of superior dispersion of chemically converted graphene in organic solvents. Chem. Commun. 2010, 46, 4375–4377. [Google Scholar]
- Ding, Z.; Miao, Z.; Xie, Z.; Liu, J. Functionalized graphene quantum dots as a novel cathode interlayer of polymer solar cells. J. Mater. Chem. A 2016, 4, 2413–2418. [Google Scholar] [CrossRef]
- Yang, D.; Velamakanni, A.; Bozoklu, G.; Park, S.; Stoller, M.; Piner, R.D.; Stankovich, S.; Jung, I.; Field, D.A.; Ventrice, C.A.; et al. Chemical analysis of graphene oxide films after heat and chemical treatments by X-ray photoelectron and micro-raman spectroscopy. Carbon 2009, 47, 145–152. [Google Scholar] [CrossRef]
- Yumitori, S. Correlation of C-1s chemical state intensities with the O-1s intensity in the XPS analysis of anodically oxidized glass-like carbon samples. J. Mater. Sci. 2000, 35, 139–146. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Interpretation of Raman spectra of disordered and amorphous carbon. Phys. Rev. B 2000, 61, 14095–14107. [Google Scholar] [CrossRef]
- Graf, D.; Molitor, F.; Ensslin, K.; Stampfer, C.; Jungen, A.; Hierold, C.; Wirtz, L. Spatially resolved raman spectroscopy of single- and few-layer graphene. Nano Lett. 2007, 7, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Heo, S.W.; Han, Y.W.; Moon, D.K. An organic-inorganic hybrid interlayer for improved electron extraction in inverted polymer solar cells. J. Mater. Chem. C 2016, 4, 2463–2469. [Google Scholar] [CrossRef]
- Bi, C.; Wang, Q.; Shao, Y.; Yuan, Y.; Xiao, Z.; Huang, J. Non-wetting surface-driven high-aspect-ratio crystalline grain growth for efficient hybrid perovskite solar cells. Nat. Commun. 2015, 6, 7747. [Google Scholar] [CrossRef] [PubMed]

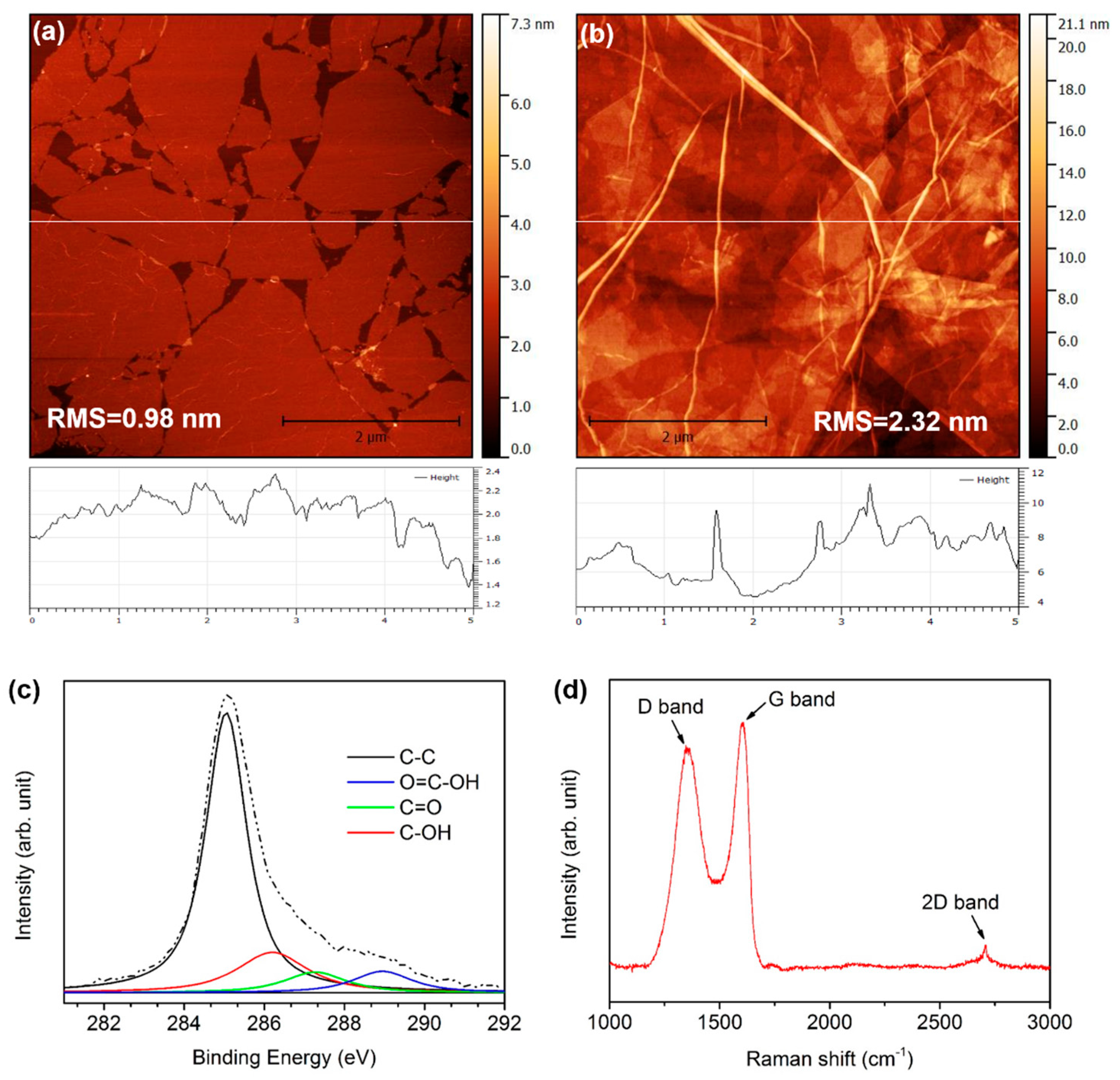
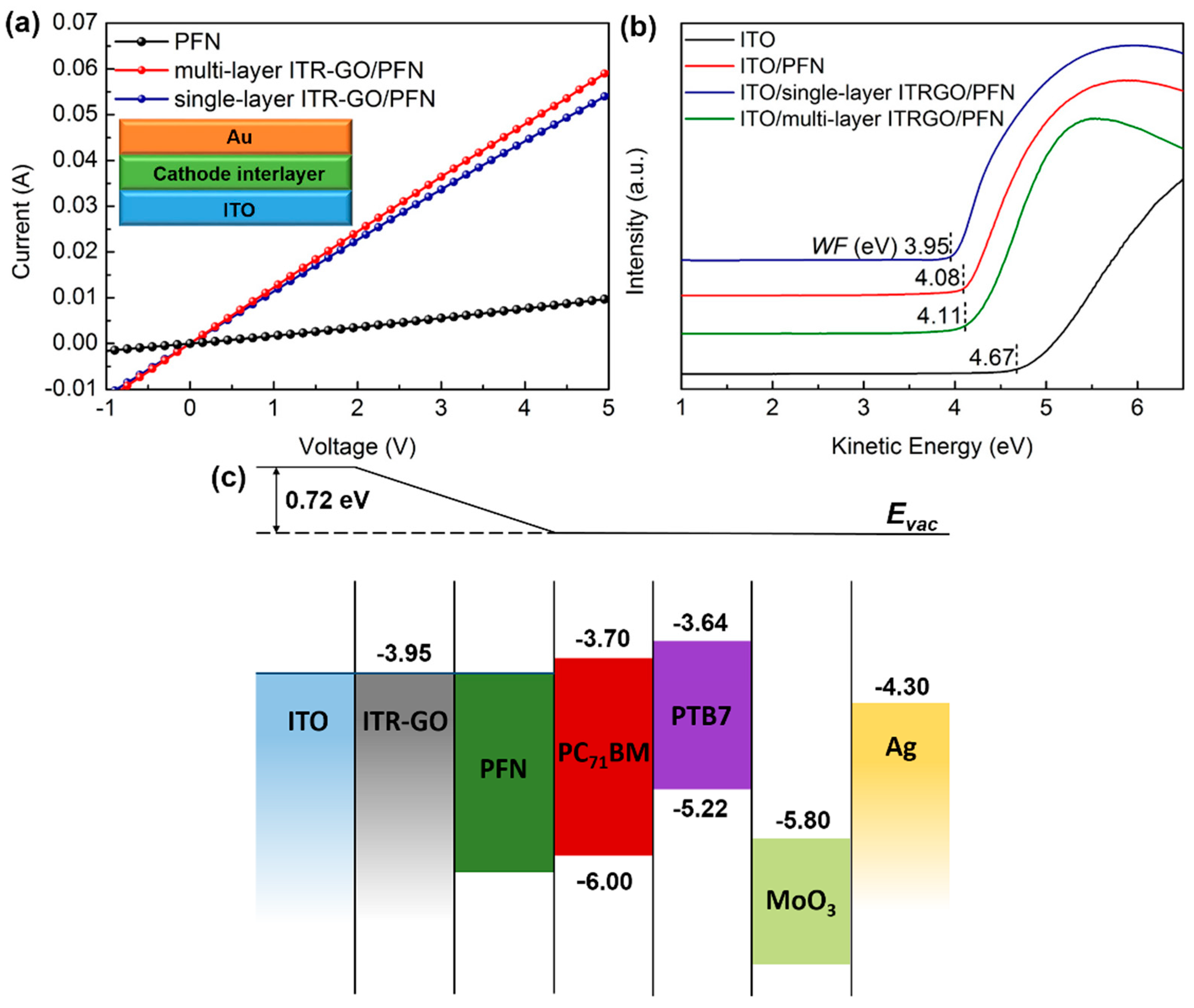

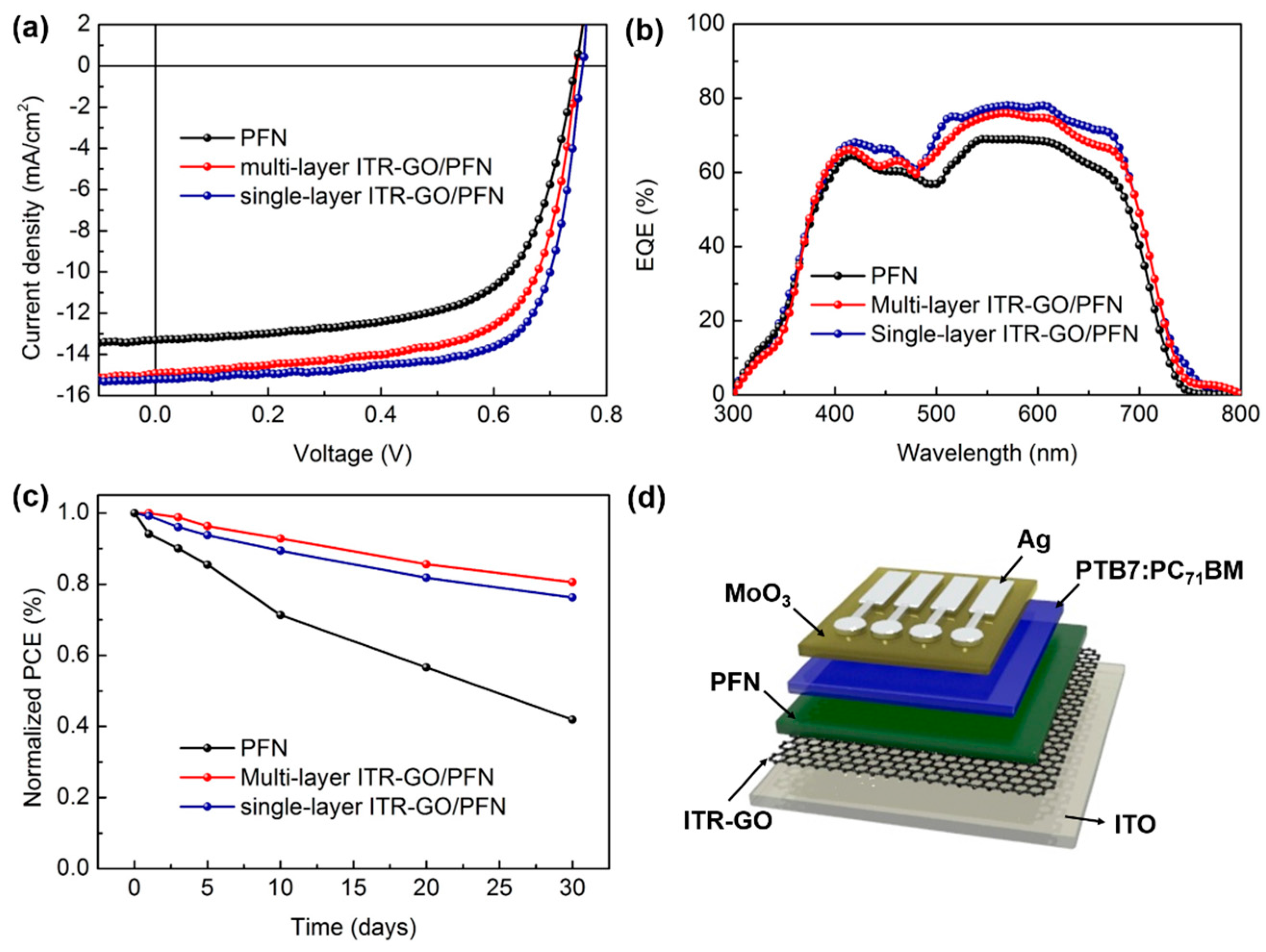
| CILs | Conductivity (S/cm) | WF (eV) | VOC (V) | JSC (mA/cm2) | FF (%) | PCE (%) | |
|---|---|---|---|---|---|---|---|
| ZnO [29] | 0.74 | −13.69 | 65.94 | 6.68 | |||
| PFN | 5.72 × 10−3 | 4.08 | Average | 0.74 | −13.02 | 64.45 | 6.21 ± 0.31 |
| Best | 0.75 | −13.31 | 64.87 | 6.47 | |||
| M-ITR-GO/PFN | 3.60 × 10−2 | 4.11 | Average | 0.75 | −14.54 | 66.36 | 7.24 ± 0.12 |
| Best | 0.75 | −14.92 | 67.90 | 7.59 | |||
| S-ITR-GO/PFN | 3.32 × 10−2 | 3.95 | Average | 0.76 | −15.082 | 69.97 | 8.02 ± 0.24 |
| Best | 0.76 | −15.21 | 72.16 | 8.34 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zheng, D.; Zhao, L.; Fan, P.; Ji, R.; Yu, J. Highly Efficient and Stable Organic Solar Cells via Interface Engineering with a Nanostructured ITR-GO/PFN Bilayer Cathode Interlayer. Nanomaterials 2017, 7, 233. https://doi.org/10.3390/nano7090233
Zheng D, Zhao L, Fan P, Ji R, Yu J. Highly Efficient and Stable Organic Solar Cells via Interface Engineering with a Nanostructured ITR-GO/PFN Bilayer Cathode Interlayer. Nanomaterials. 2017; 7(9):233. https://doi.org/10.3390/nano7090233
Chicago/Turabian StyleZheng, Ding, Lili Zhao, Pu Fan, Ran Ji, and Junsheng Yu. 2017. "Highly Efficient and Stable Organic Solar Cells via Interface Engineering with a Nanostructured ITR-GO/PFN Bilayer Cathode Interlayer" Nanomaterials 7, no. 9: 233. https://doi.org/10.3390/nano7090233





