Ionic Liquids as Surfactants for Layered Double Hydroxide Fillers: Effect on the Final Properties of Poly(Butylene Adipate-Co-Terephthalate)
Abstract
:1. Introduction
2. Results and Discussion
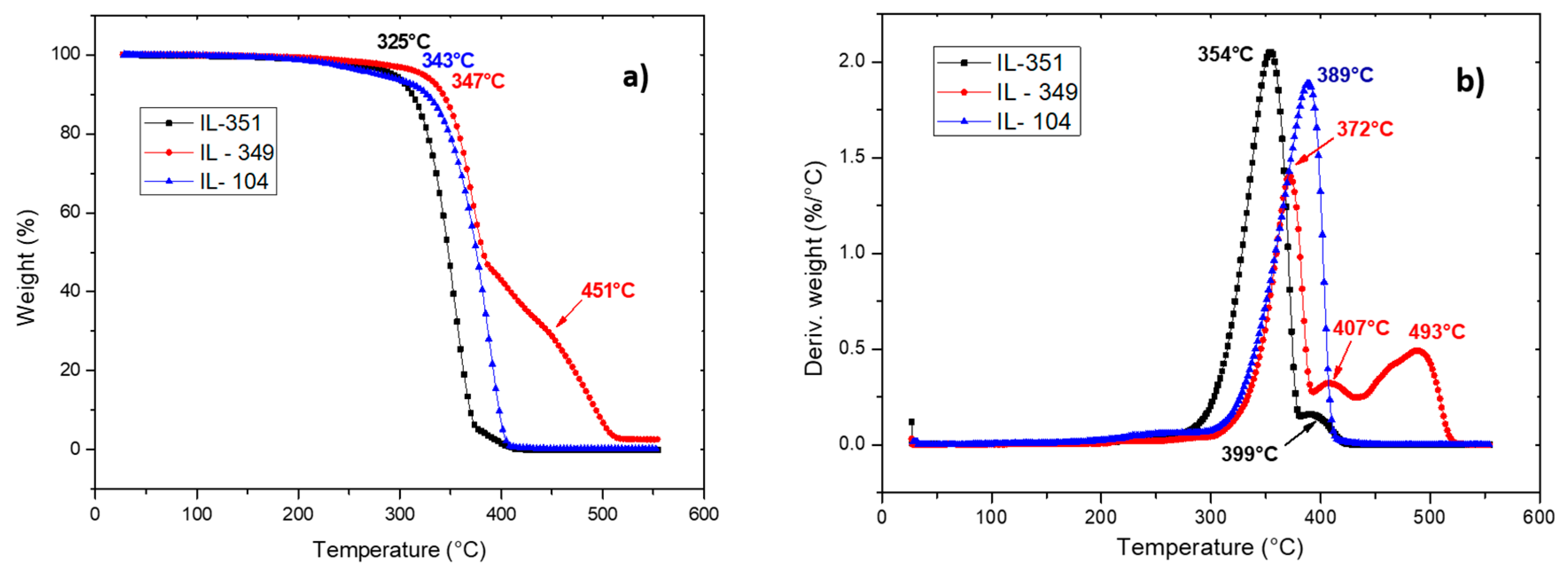
2.1. Characterization of Ionic Liquids
Thermal Behavior
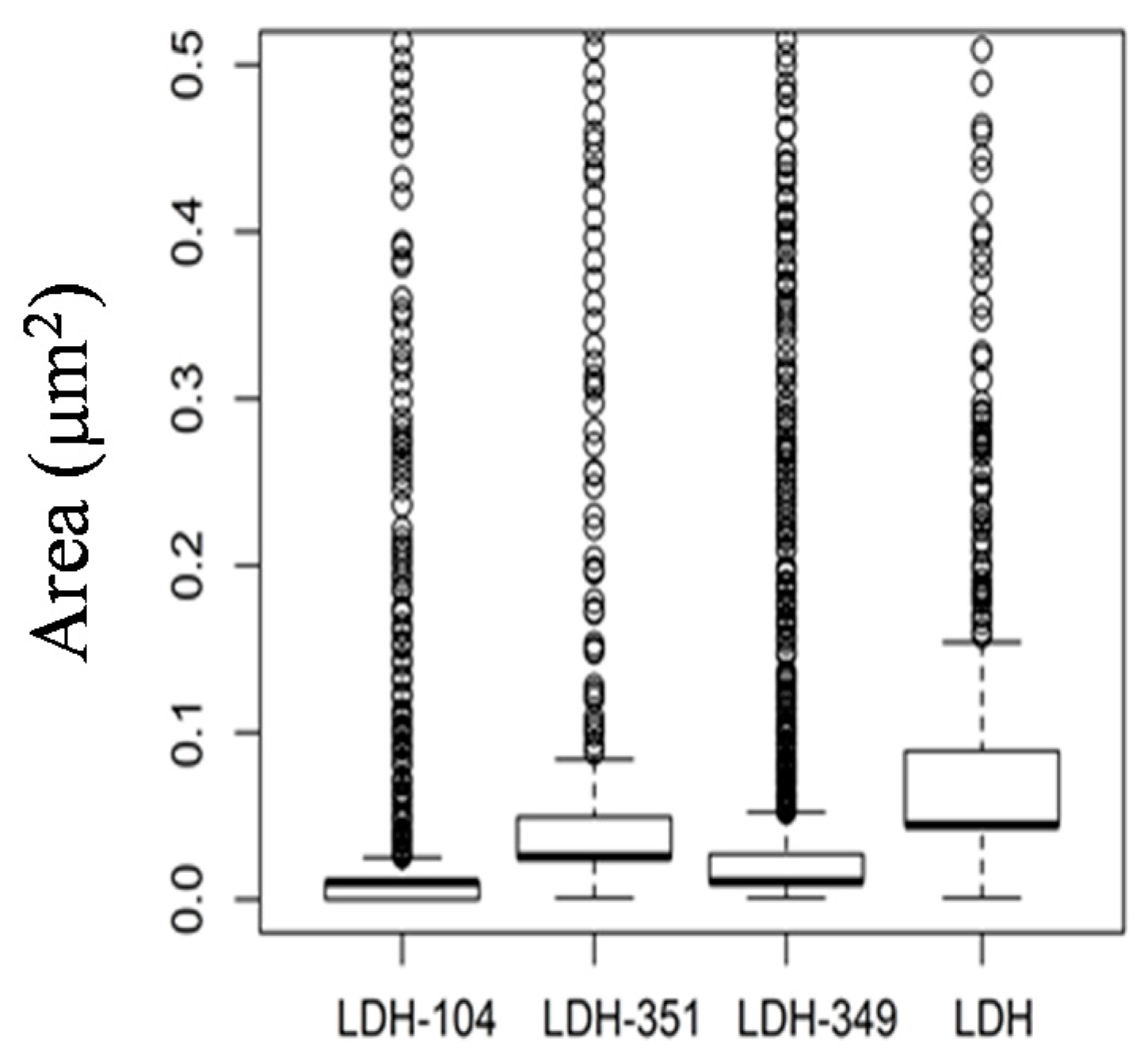
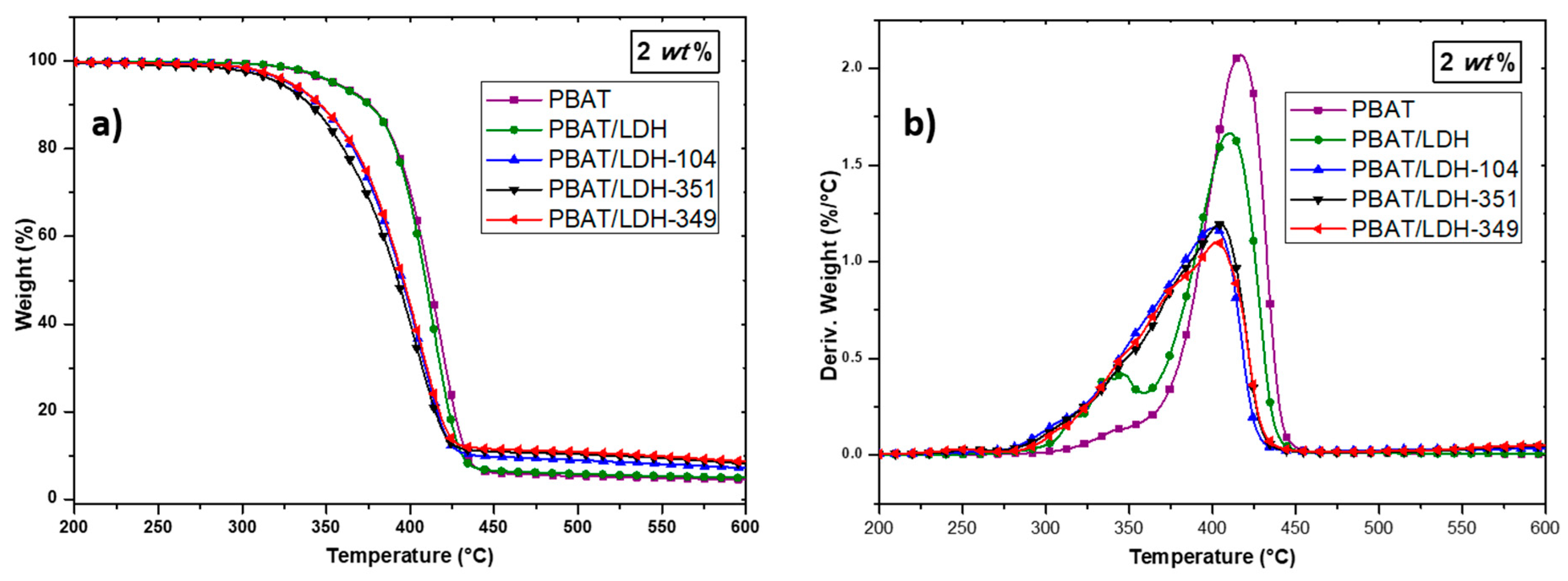
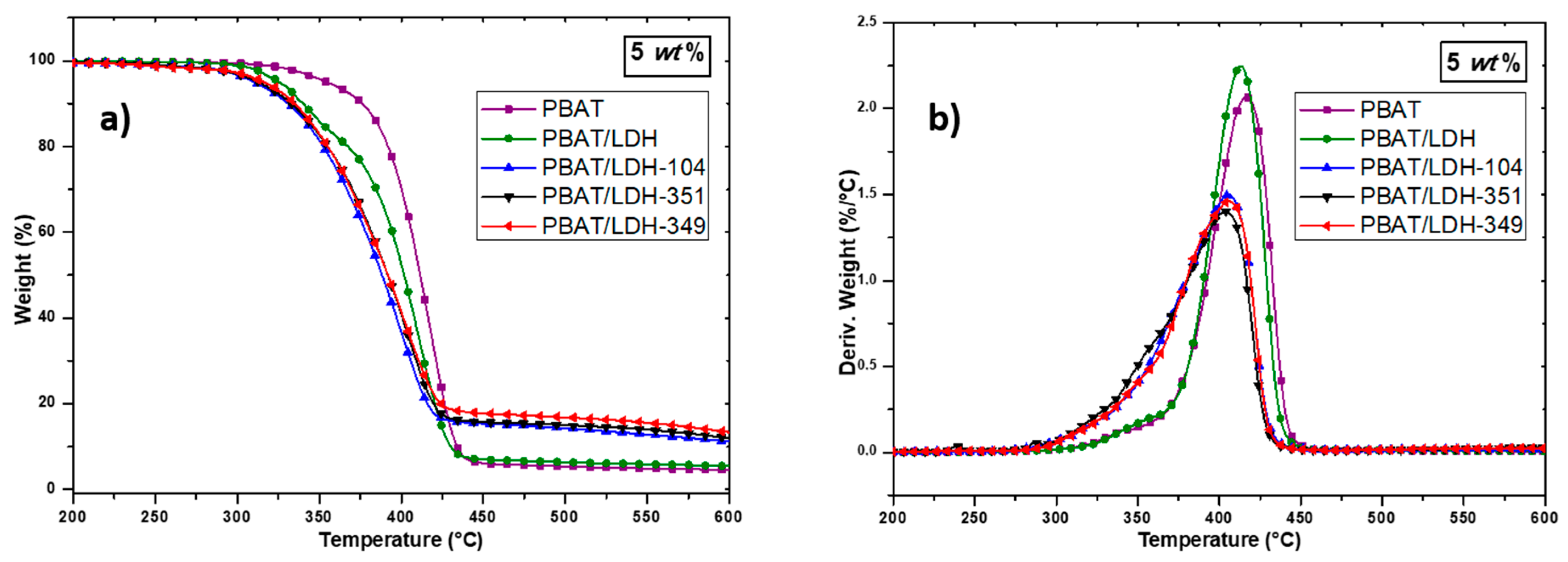
2.2. Characterization of PBAT/Modified-LDH Nanocomposites
2.2.1. Morphology
2.2.2. Thermal Behavior
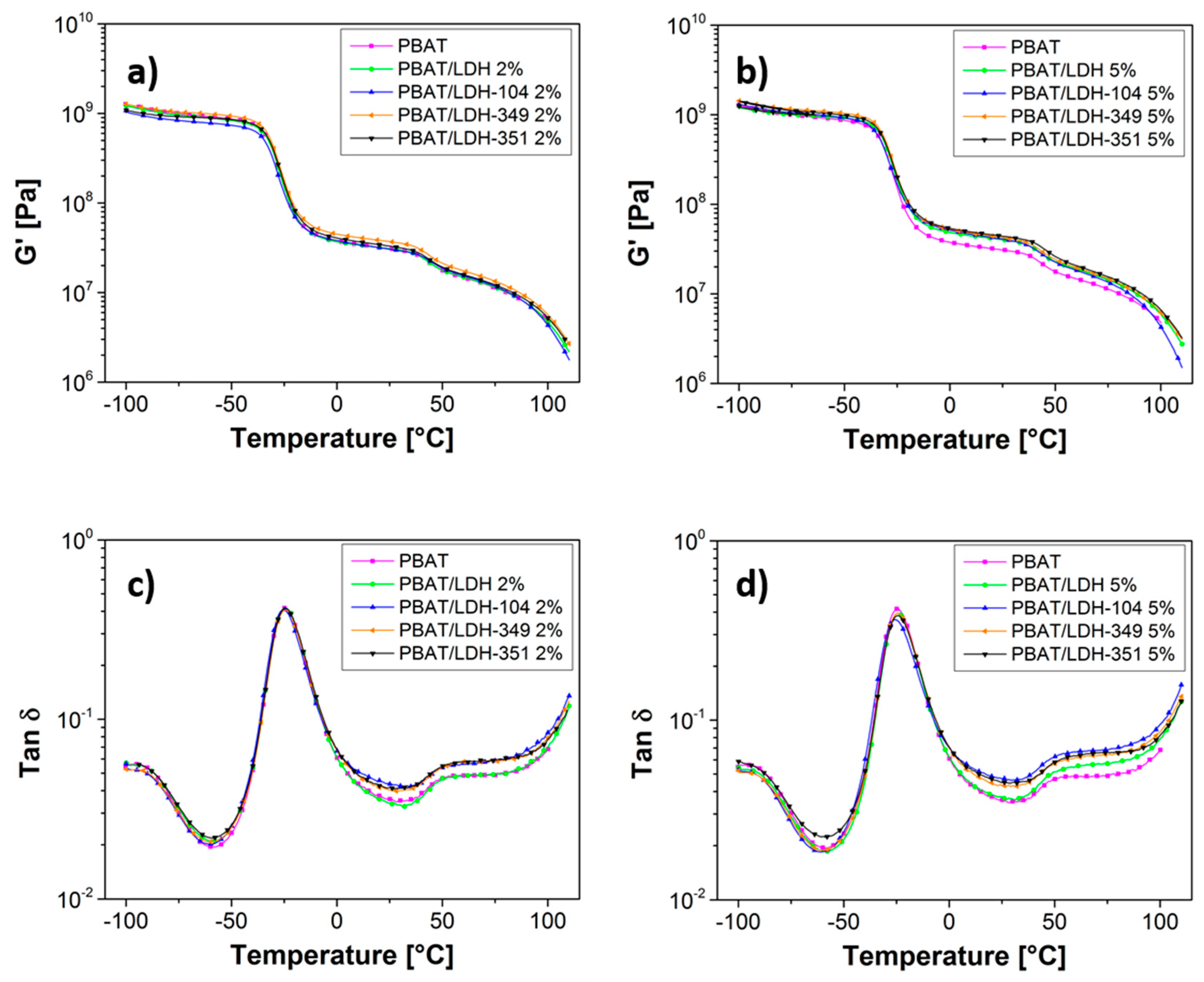
2.2.3. Mechanical Properties
2.2.4. Influence of Modified-LDHs on the Gas Transport Properties of PBAT Matrix
3. Materials and Methods
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Giannelis, E.P. Polymer Layered Silicate Nanocomposites. Adv. Mater. 1996, 8, 29–35. [Google Scholar] [CrossRef]
- Le Baron, P.C.; Zhen, W.; Pinnavaia, J. Polymer-Layered Silicate Nanocomposites: An overview. Appl. Clay Sci. 1999, 15, 11–29. [Google Scholar] [CrossRef]
- Chang, J.H.; An, Y.U.; Cho, D.; Giannelis, E.P. Poly(lactid acid) nanocomposites: Comparison of their properties with montmorillonite and synthetic mica (II). Polymer 2003, 44, 3715–3720. [Google Scholar] [CrossRef]
- Ray, S.S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Polym. Sci. 2003, 28, 1539–1641. [Google Scholar]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, properties and uses of a new class of materials. Mater. Sci. Eng. R Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Lepoittevin, B.; Devalckenaere, M.; Pantoussier, N.; Alexandre, M.; Kubies, D.; Calberg, C.; Jérôme, R.; Dubois, P. Poly(ε-caprolactone)/clay nanocomposites prepared by melt intercalation: Mechanical, thermal and rheological properties. Polymer 2002, 43, 4017–4023. [Google Scholar] [CrossRef]
- Paul, M.-A.; Alexandre, M.; Degée, P.; Henrist, C.; Rulmont, A.; Dubois, P. New nanocomposite materials based on plasticized poly(l-lactide) and organo-modified montmorillonites: Thermal and morphological study. Polymer 2003, 44, 443–450. [Google Scholar] [CrossRef]
- Yang, Z.; Peng, H.; Wang, W.; Liu, T. Crystallization behavior of poly(ε-caprolactone)/layered double hydroxide nanocomposites. J. Appl. Polym. Sci. 2010, 116, 2658–2667. [Google Scholar] [CrossRef]
- Kredatusová, J.; Beneš, H.; Livi, S.; Pop-Georgievski, O.; Ecorchard, P.; Abbrent, S.; Pavlova, E.; Bogdał, D. Influence of ionic liquid-modified LDH on microwave-assisted polymerization of ε-caprolactone. Polymer 2016, 100, 86–94. [Google Scholar] [CrossRef]
- Romeo, V.; Gorrasi, G.; Vittoria, V.; Chronakis, I.S. Encapsulation and Exfoliation of Inorganic Lamellar Fillers into Polycaprolactone by Electrospinning. Biomacromolecules 2007, 8, 3147–3152. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Han, Y.; Liu, T.; Tjiu, W.C.; He, C. Morphology and thermal degradation behavior of highly exfoliated CoAl-layered double hydroxide/polycaprolactone nanocomposites prepared by simple solution intercalation. Thermochim. Acta 2010, 502, 1–7. [Google Scholar] [CrossRef]
- Jager, E.; Donato, R.K.; Perchacz, M.; Jager, A.; Surman, F.; Hocherl, A.; Konefal, R.; Donato, K.Z.; Venturini, C.G.; Bergamo, V.Z.; et al. Biocompatible succinic acid-based polyesters for potential biomedical applications: Fungal biofilm inhibition and mesenchymal stem cell growth. RSC Adv. 2015, 5, 85756–85766. [Google Scholar] [CrossRef]
- Pinheiro, I.F.; Morales, A.R.; Mei, L.H. Polymeric biocomposites of poly (butylene adipate-co-terephthalate) reinforced with natural Munguba fibers. Cellulose 2014, 21, 4381–4391. [Google Scholar] [CrossRef]
- Silva, J.S.P.; Silva, J.M.F.; Soares, B.G.; Livi, S. Fully biodegradable composites based on poly(butylene adipate-co-terephthalate)/peach palm trees fiber. Composites Part B 2017, 129, 117–123. [Google Scholar] [CrossRef]
- Russo, P.; Carfagna, C.; Cimino, F.; Acierno, D.; Persico, P. Biodegradable composites reinforced with kenaf fibers: Thermal, mechanical, and morphological issues. Adv. Polym. Technol. 2013, 32, E313–E322. [Google Scholar] [CrossRef]
- Shen, W.; He, H.; Zhu, J.; Yuan, P.; Frost, R.L. Grafting of montmorillonite with different functional silanes via two different reaction systems. J. Colloid Interface Sci. 2007, 313, 268–273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, H.; Duchet, J.; Galy, J.; Gerard, J.-F. Grafting of swelling clay materials with 3-aminopropyltriethoxysilane. J. Colloid Interface Sci. 2005, 288, 171–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kornmann, X.; Lindberg, H.; Berglund, L.A. Synthesis of epoxy–clay nanocomposites: Influence of the nature of the clay on structure. Polymer 2001, 42, 1303–1310. [Google Scholar] [CrossRef]
- Livi, S.; Duchet-Rumeau, J.; Pham, T.-N.; Gérard, J.-F. A comparative study on different ionic liquids used as surfactants: Effect on thermal and mechanical properties of high-density polyethylene nanocomposites. J. Colloid Interface Sci. 2010, 349, 424–433. [Google Scholar] [CrossRef] [PubMed]
- Heinz, H.; Vaia, R.A.; Krishnamoorti, R.; Farmer, B.L. Self-Assembly of Alkylammonium Chains on Montmorillonite: Effect of Chain Length, Head Group Structure, and Cation Exchange Capacity. Chem. Mater. 2007, 19, 59–68. [Google Scholar] [CrossRef]
- Okamoto, K.; Sasaki, T.; Fujita, T.; Iyi, N. Preparation of highly oriented organic-LDH hybrid films by combining the decarbonation, anion-exchange, and delamination processes. J. Mater. Chem. 2006, 16, 1608–1616. [Google Scholar] [CrossRef]
- Crepaldi, E.L.; Pavan, P.C.; Valim, J.B. A new method of intercalation by anion exchange in layered double hydroxides. Chem. Commun. 1999, 2, 155–156. [Google Scholar] [CrossRef]
- Wang, Q.; O’Hare, D. Recent Advances in the Synthesis and Application of Layered Double Hydroxide (LDH) Nanosheets. Chem. Rev. 2012, 112, 4124–4155. [Google Scholar] [CrossRef] [PubMed]
- Donato, R.K.; Luza, L.; da Silva, R.F.; Moro, C.C.; Guzatto, R.; Samios, D.; Matějka, L.; Dimzoski, B.; Amico, S.C.; Schrekker, H.S. The role of oleate-functionalized layered double hydroxide in the melt compounding of polypropylene nanocomposites. Mater. Sci. Eng. C 2012, 32, 2396–2403. [Google Scholar] [CrossRef]
- Livi, S.; Duchet-Rumeau, J.; Gérard, J.-F. Tailoring of interfacial properties by ionic liquids in a fluorinated matrix based nanocomposites. Eur. Polym. J. 2011, 47, 1361–1369. [Google Scholar] [CrossRef]
- Livi, S.; Duchet-Rumeau, J.; Pham, T.N.; Gérard, J.-F. Synthesis and physical properties of new surfactants based on ionic liquids: Improvement of thermal stability and mechanical behavior of high density polyethylene nanocomposites. J. Colloid Interface Sci. 2011, 354, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Perchacz, M.; Donato, R.K.; Seixas, L.; Zhigunov, A.; Konefał, R.; Serkis-Rodzeń, M.; Beneš, H. Ionic Liquid-Silica Precursors via Solvent-Free Sol–Gel Process and Their Application in Epoxy-Amine Network: A Theoretical/Experimental Study. ACS Appl. Mater. Interfaces 2017, 9, 16474–16487. [Google Scholar] [CrossRef] [PubMed]
- Donato, R.K.; Matejka, L.; Schrekker, H.S.; Plestil, J.; Jigounov, A.; Brus, J.; Slouf, M. The multifunctional role of ionic liquids in the formation of epoxy-silica nanocomposites. J. Mater. Chem. 2011, 21, 13801–13810. [Google Scholar] [CrossRef]
- Donato, R.K.; Lavorgna, M.; Musto, P.; Donato, K.Z.; Jager, A.; Štěpánek, P.; Schrekker, H.S.; Matějka, L. The role of ether-functionalized ionic liquids in the sol–gel process: Effects on the initial alkoxide hydrolysis steps. J. Colloid Interface Sci. 2015, 447, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Donato, R.K.; Perchacz, M.; Ponyrko, S.; Donato, K.Z.; Schrekker, H.S.; Benes, H.; Matejka, L. Epoxy-silica nanocomposite interphase control using task-specific ionic liquids via hydrolytic and non-hydrolytic sol-gel processes. RSC Adv. 2015, 5, 91330–91339. [Google Scholar] [CrossRef]
- Bugatti, V.; Livi, S.; Hayrapetyan, S.; Wang, Y.; Estevez, L.; Vittoria, V.; Giannelis, E.P. Deposition of LDH on plasma treated polylactic acid to reduce water permeability. J. Colloid Interface Sci. 2013, 396, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.K.L.; Livi, S.; Soares, B.G.; Benes, H.; Gérard, J.-F.; Duchet-Rumeau, J. Toughening of Epoxy/Ionic Liquid Networks with Thermoplastics Based on Poly(2,6-dimethyl-1,4-phenylene ether) (PPE). ACS Sustain. Chem. Eng. 2017, 5, 1153–1164. [Google Scholar] [CrossRef]
- Sonnier, R.; Dumazert, L.; Livi, S.; Nguyen, T.K.L.; Duchet-Rumeau, J.; Vahabi, H.; Laheurte, P. Flame retardancy of phosphorus-containing ionic liquid based epoxy networks. Polym. Degrad. Stab. 2016, 134, 186–193. [Google Scholar] [CrossRef]
- Awad, W.H.; Gilman, J.W.; Nyden, M.; Harris, R.H.; Sutto, T.E.; Callahan, J.; Trulove, P.C.; DeLong, H.C.; Fox, D.M. Thermal degradation studies of alkyl-imidazolium salts and their application in nanocomposites. Thermochim. Acta 2004, 409, 3–11. [Google Scholar] [CrossRef]
- Ngo, H.L.; LeCompte, K.; Hargens, L.; McEwen, A.B. Thermal properties of imidazolium ionic liquids. Thermochim. Acta 2000, 357, 97–102. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, F.; Zhang, R.; Evans, D.G.; Duan, X. Preparation of Layered Double-Hydroxide Nanomaterials with a Uniform Crystallite Size Using a New Method Involving Separate Nucleation and Aging Steps. Chem. Mater. 2002, 14, 4286–4291. [Google Scholar] [CrossRef]
- Livi, S.; Bugatti, V.; Estevez, L.; Duchet-Rumeau, J.; Giannelis, E.P. Synthesis and physical properties of new layered double hydroxides based on ionic liquids: Application to a polylactide matrix. J. Colloid Interface Sci. 2012, 388, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Oyarzabal, A.; Mugica, A.; Müller, A.J.; Zubitur, M. Hydrolytic degradation of nanocomposites based on poly(l-lactic acid) and layered double hydroxides modified with a model drug. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Kanezaki, E. Intercalation of naphthalene-2,6-disulfonate between layers of Mg and Al double hydroxide: Preparation, powder X-Ray diffraction, fourier transform infrared spectra and X-Ray photoelectron spectra. Mater. Res. Bull. 1999, 34, 1435–1440. [Google Scholar] [CrossRef]
- Soares, B.G.; Ferreira, S.C.; Livi, S. Modification of anionic and cationic clays by zwitterionic imidazolium ionic liquid and their effect on the epoxy-based nanocomposites. Appl. Clay Sci. 2017, 135, 347–354. [Google Scholar] [CrossRef]
- Ha, J.U.; Xanthos, M. Novel modifiers for layered double hydroxides and their effects on the properties of polylactic acid composites. Appl. Clay Sci. 2010, 47, 303–310. [Google Scholar] [CrossRef]
- Ding, P.; Kang, B.; Zhang, J.; Yang, J.; Song, N.; Tang, S.; Shi, L. Phosphorus-containing flame retardant modified layered double hydroxides and their applications on polylactide film with good transparency. J. Colloid Interface Sci. 2015, 440, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Leroux, F.; Besse, J.-P. Polymer Interleaved Layered Double Hydroxide: A New Emerging Class of Nanocomposites. Chem. Mater. 2001, 13, 3507–3515. [Google Scholar] [CrossRef]
- Aranda, P.; Ruiz-Hitzky, E. Poly(ethylene oxide)-silicate intercalation materials. Chem. Mater. 1992, 4, 1395–1403. [Google Scholar] [CrossRef]
- Greenland, D.J. Adsorption of polyvinyl alcohols by montmorillonite. J. Colloid Sci. 1963, 18, 647–664. [Google Scholar] [CrossRef]
- Livi, S.; Bugatti, V.; Soares, B.G.; Duchet-Rumeau, J. Structuration of ionic liquids in a poly(butylene-adipate-co-terephthalate) matrix: Its influence on the water vapour permeability and mechanical properties. Green Chem. 2014, 16, 3758–3762. [Google Scholar] [CrossRef]
- Park, K.I.; Xanthos, M. A study on the degradation of polylactic acid in the presence of phosphonium ionic liquids. Polym. Degrad. Stab. 2009, 94, 834–844. [Google Scholar] [CrossRef]
- Nayak, S.K. Biodegradable PBAT/starch nanocomposites. Polym. Plast. Technol. Eng. 2010, 49, 1406–1418. [Google Scholar] [CrossRef]
- Livi, S.; Sar, G.; Bugatti, V.; Espuche, E.; Duchet-Rumeau, J. Synthesis and physical properties of new layered silicates based on ionic liquids: Improvement of thermal stability, mechanical behavior and water permeability of PBAT nanocomposites. RSC Adv. 2014, 4, 26452–26461. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, K.; Wu, J.; Ren, G.; Chen, H.; Xu, J. Bio-nanocomposite films reinforced with organo-modified layered double hydroxides: Preparation, morphology and properties. Appl. Clay Sci. 2016, 126, 72–80. [Google Scholar] [CrossRef]
- Du, L.C.; Qu, B.J.; Zhang, M. Thermal properties and combustion characterization of nylon 6/MgAl-LDH nanocomposites via organic modification and melt intercalation. Polym. Degrad. Stab. 2007, 92, 497–502. [Google Scholar] [CrossRef]
- Dagnon, K.L.; Chen, H.H.; Innocentini-Mei, L.H.; Souza, N.A.D. Poly[(3-hydroxybutyrate)-co-(3-hydroxyvalerate)]/layered double hydroxide nanocomposites. Polym. Int. 2009, 58, 133–141. [Google Scholar] [CrossRef]
- Storey, R.F.; Bauch, D.W. Poly(styrene-b-isobutylene-b-styrene) block copolymers and ionomers therefrom: Morphology as determined by small-angle X-ray scattering and transmission electron microscopy. Polymer 2000, 41, 3195–3205. [Google Scholar] [CrossRef]
- Livi, S.; Gerard, J.F.; Duchet-Rumeau, J. Ionic liquids: Structuration agents in a fluorinated matrix. Chem. Commun. 2011, 47, 3589–3591. [Google Scholar]
- Freeman, T.B.; Yampolskii, Y.; Pinnau, I. Materials Science of Membranes for Gas and Vapor Separation; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Gain, O.; Espuche, E.; Pollet, E.; Alexandre, M.; Dubois, P. Gas barrier properties of poly(ε-caprolactone)/clay nanocomposites: Influence of the morphology and polymer/clay interactions. J. Polym. Sci. Part B Polym. Phys. 2005, 43, 205–214. [Google Scholar] [CrossRef]
- Livi, S.; Lins, C.L.; Sar, G.; Gérard, J.-F.; Duchet-Rumeau, J. Supercritical CO2–Ionic Liquids: A Successful Wedding To Prepare Biopolymer Foams. ACS Sustain. Chem. Eng. 2016, 4, 461–470. [Google Scholar] [CrossRef]
- Labruyère, C.; Gorrasi, G.; Monteverde, F.; Alexandre, M.; Dubois, P. Transport properties of organic vapours in silicone/clay nanocomposites. Polymer 2009, 50, 3626–3637. [Google Scholar] [CrossRef]
- Gorrasi, G.; Tortora, M.; Vittoria, V.; Pollet, E.; Lepoittevin, B.; Alexandre, M.; Dubois, P. Vapor barrier properties of polycaprolactone montmorillonite nanocomposites: Effect of clay dispersion. Polymer 2003, 44, 2271–2279. [Google Scholar] [CrossRef]
- Nobuo, I.; Taki, M.; Yoshiro, K.; Kenji, K. A Novel Synthetic Route to Layered Double Hydroxides Using Hexamethylenetetramine. Chem. Lett. 2004, 33, 1122–1123. [Google Scholar]
- Herrera, R.; Franco, L.; Rodríguez-Galán, A.; Puiggalí, J. Characterization and degradation behavior of poly(butylene adipate-co-terephthalate)s. J. Polym. Sci. Part A Polym. Chem. 2002, 40, 4141–4157. [Google Scholar] [CrossRef]
- Rutherford, S.W.; Do, D.D. Review of time lag permeation technique as a method for characterisation of porous media and membranes. Adsorption 1997, 3, 283–312. [Google Scholar] [CrossRef]


| Nomenclature | Young Modulus (Mpa) | Stress (Mpa) | Strain at Break (%) |
|---|---|---|---|
| PBAT | 47 ± 1 | 24 ± 1 | 511 ± 17 |
| PBAT/LDH | 56 ± 1 | 24 ± 1 | 577 ± 18 |
| PBAT/LDH-349 2% | 62 ± 1 | 22 ± 1 | 462 ± 30 |
| PBAT/LDH-349 5% | 72 ± 2 | 22 ± 1 | 400 ± 13 |
| PBAT/LDH-104 2% | 52 ± 2 | 22 ± 1 | 567 ± 7 |
| PBAT/LDH-104 5% | 50 ± 1 | 22 ± 1 | 940 ± 10 |
| PBAT/LDH-351 2% | 57 ± 2 | 21 ± 1 | 455 ± 30 |
| PBAT/LDH-351 5% | 61 ± 1 | 20 ± 2 | 440 ± 15 |
| Materials | Permeability Coefficient (Barrer) a | Ideal Selectivity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| H2 | O2 | N2 | CO2 | H2O | H2/N2 | H2O/O2 | O2/N2 | CO2/H2 | CO2/N2 | |
| PBAT | 4.92 | 1.22 | 0.33 | 12.5 | 2580 | 15.1 | 2110 | 3.7 | 2.5 | 38.3 |
| PBAT/LDH 2% | 4.53 | 1.04 | 0.31 | 11.0 | 1970 | 14.8 | 1900 | 3.4 | 2.4 | 36.1 |
| PBAT/LDH 5% | 4.38 | 0.95 | 0.31 | 10.0 | 2210 | 14.3 | 2320 | 3.1 | 2.3 | 32.7 |
| PBAT/LDH-349 2% | 4.01 | 0.95 | 0.29 | 9.9 | 1630 | 13.8 | 1730 | 3.3 | 2.5 | 33.9 |
| PBAT/LDH-104 2% | 3.74 | 0.93 | 0.27 | 9.5 | 1360 | 13.7 | 1470 | 3.4 | 2.5 | 34.8 |
| PBAT/LDH-349 5% | 4.97 | 1.08 | 0.34 | 11.7 | 1400 | 14.6 | 1300 | 3.2 | 2.4 | 34.5 |
| PBAT/LDH-351 5% | 4.20 | 1.00 | 0.28 | 10.1 | 1520 | 15.2 | 1520 | 3.6 | 2.4 | 36.5 |
| PBAT/LDH-104 5% | 4.27 | 1.05 | 0.32 | 10.4 | 1710 | 13.2 | 1630 | 3.2 | 2.4 | 32.1 |
| Ionic Liquid | Chemical Structure | Designation |
|---|---|---|
| Trihexyl(tetradecyl)phosphonium bis(2,4,4-trimethylpentyl)phosphinate |  | LDH-104 |
| Trihexyl(tetradecyl)phosphonium 2-ethylhexanoate |  | LDH-351 |
| Trihexyl(tetradecyl)phosphonium bis(2-ethylhexyl)phosphate |  | LDH-349 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Livi, S.; Lins, L.C.; Peter, J.; Benes, H.; Kredatusova, J.; Donato, R.K.; Pruvost, S. Ionic Liquids as Surfactants for Layered Double Hydroxide Fillers: Effect on the Final Properties of Poly(Butylene Adipate-Co-Terephthalate). Nanomaterials 2017, 7, 297. https://doi.org/10.3390/nano7100297
Livi S, Lins LC, Peter J, Benes H, Kredatusova J, Donato RK, Pruvost S. Ionic Liquids as Surfactants for Layered Double Hydroxide Fillers: Effect on the Final Properties of Poly(Butylene Adipate-Co-Terephthalate). Nanomaterials. 2017; 7(10):297. https://doi.org/10.3390/nano7100297
Chicago/Turabian StyleLivi, Sébastien, Luanda Chaves Lins, Jakub Peter, Hynek Benes, Jana Kredatusova, Ricardo K. Donato, and Sébastien Pruvost. 2017. "Ionic Liquids as Surfactants for Layered Double Hydroxide Fillers: Effect on the Final Properties of Poly(Butylene Adipate-Co-Terephthalate)" Nanomaterials 7, no. 10: 297. https://doi.org/10.3390/nano7100297








