Structural Regulation and Electroconductivity Change of Nitrogen-Doping Reduced Graphene Oxide Prepared Using p-Phenylene Diamine as Modifier
Abstract
:1. Introduction
2. Experimental Section
2.1. Raw Materials and Reagents
2.2. Sample Preparation
2.3. Sample Characterization
3. Results and Discussion
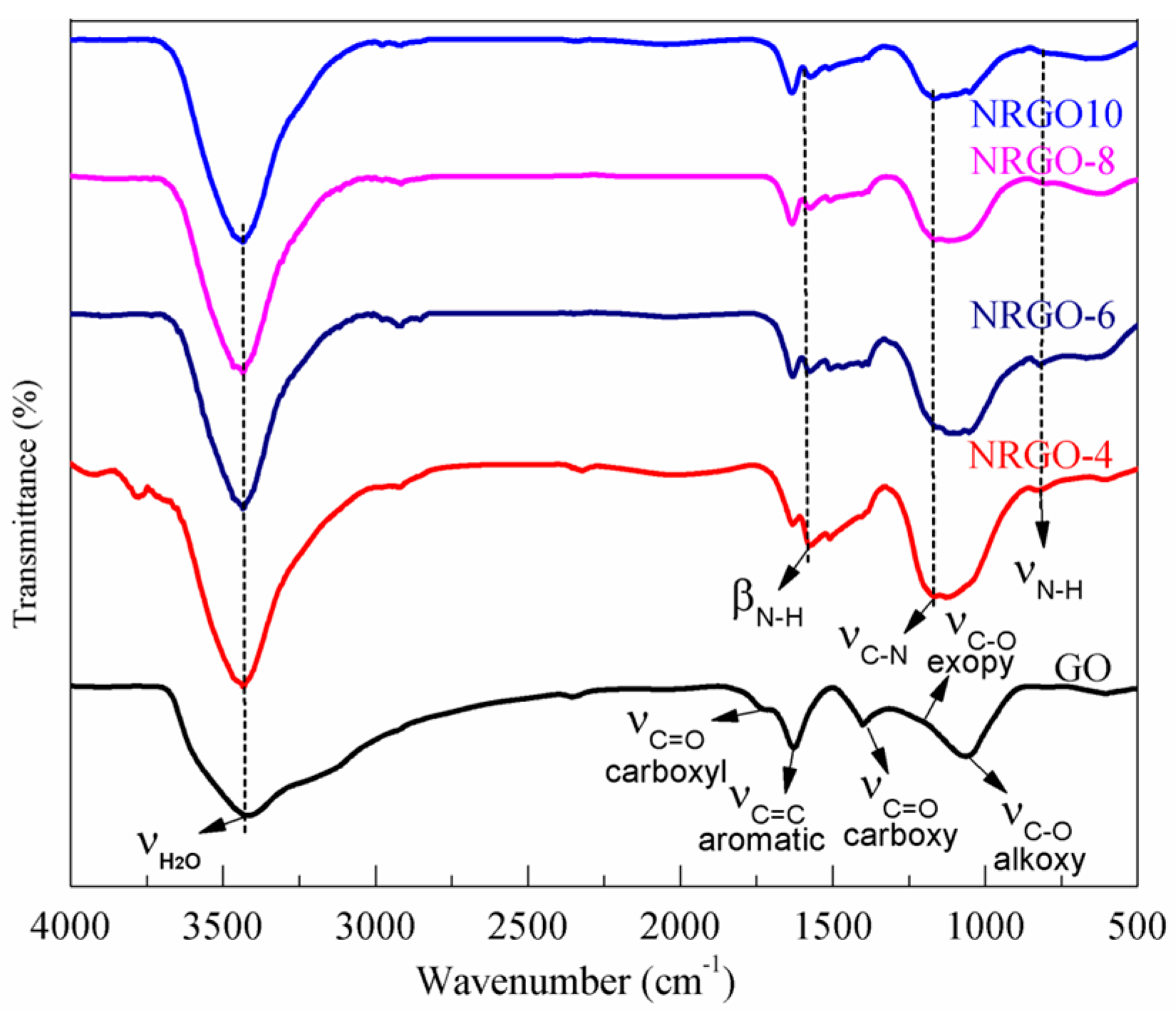
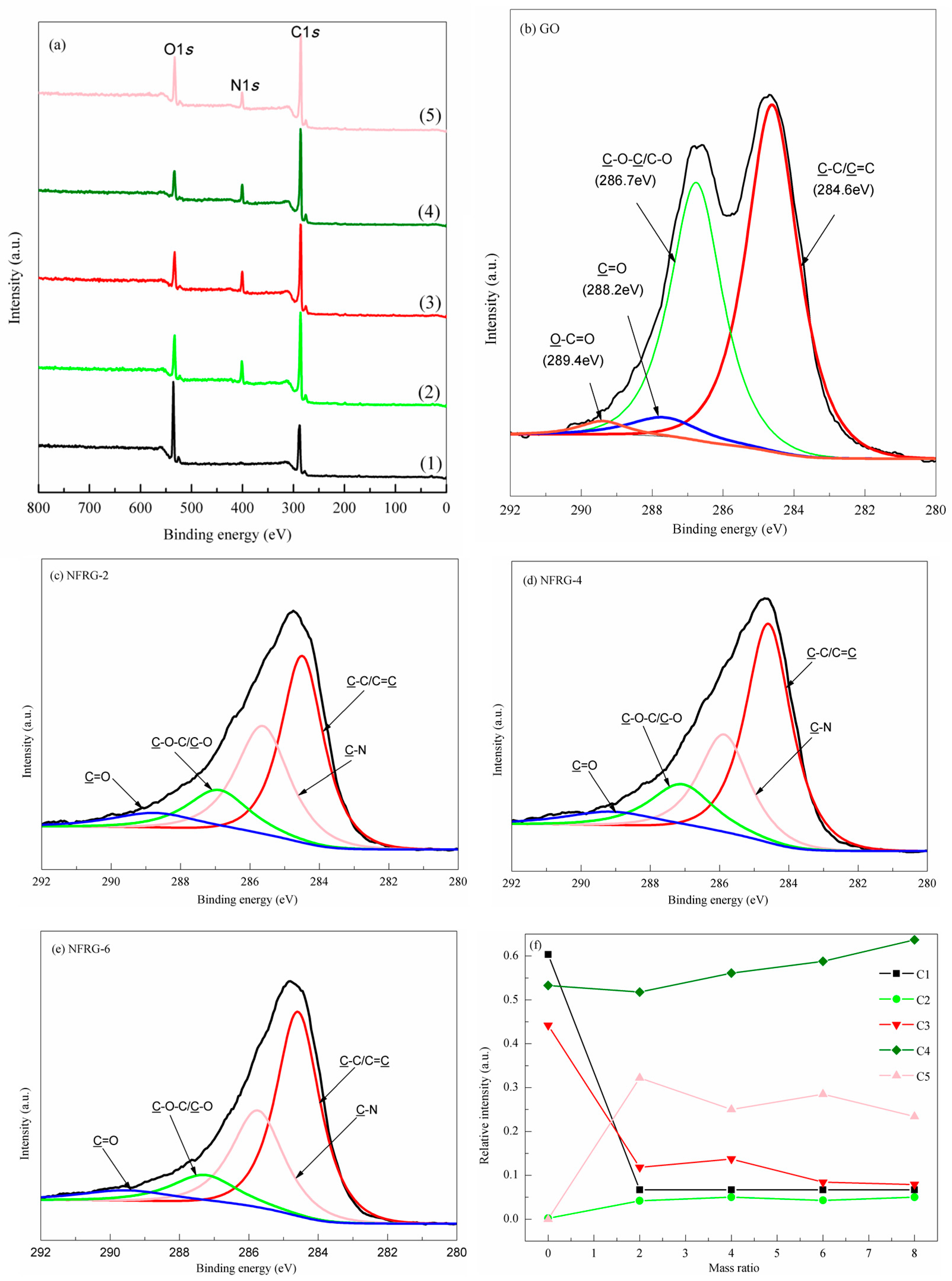
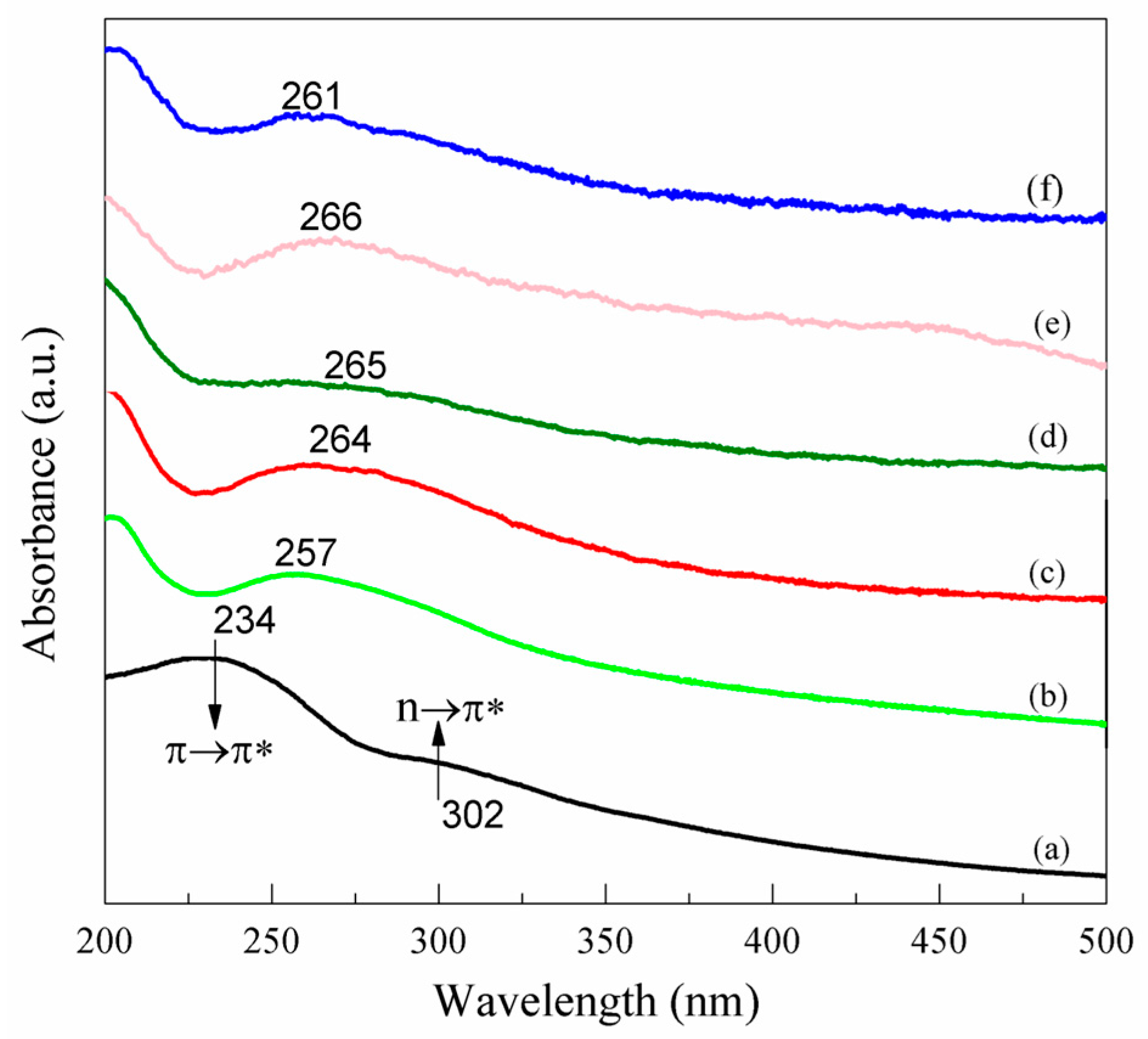
3.1. Change of Surface Functional Groups before and after Functionalization
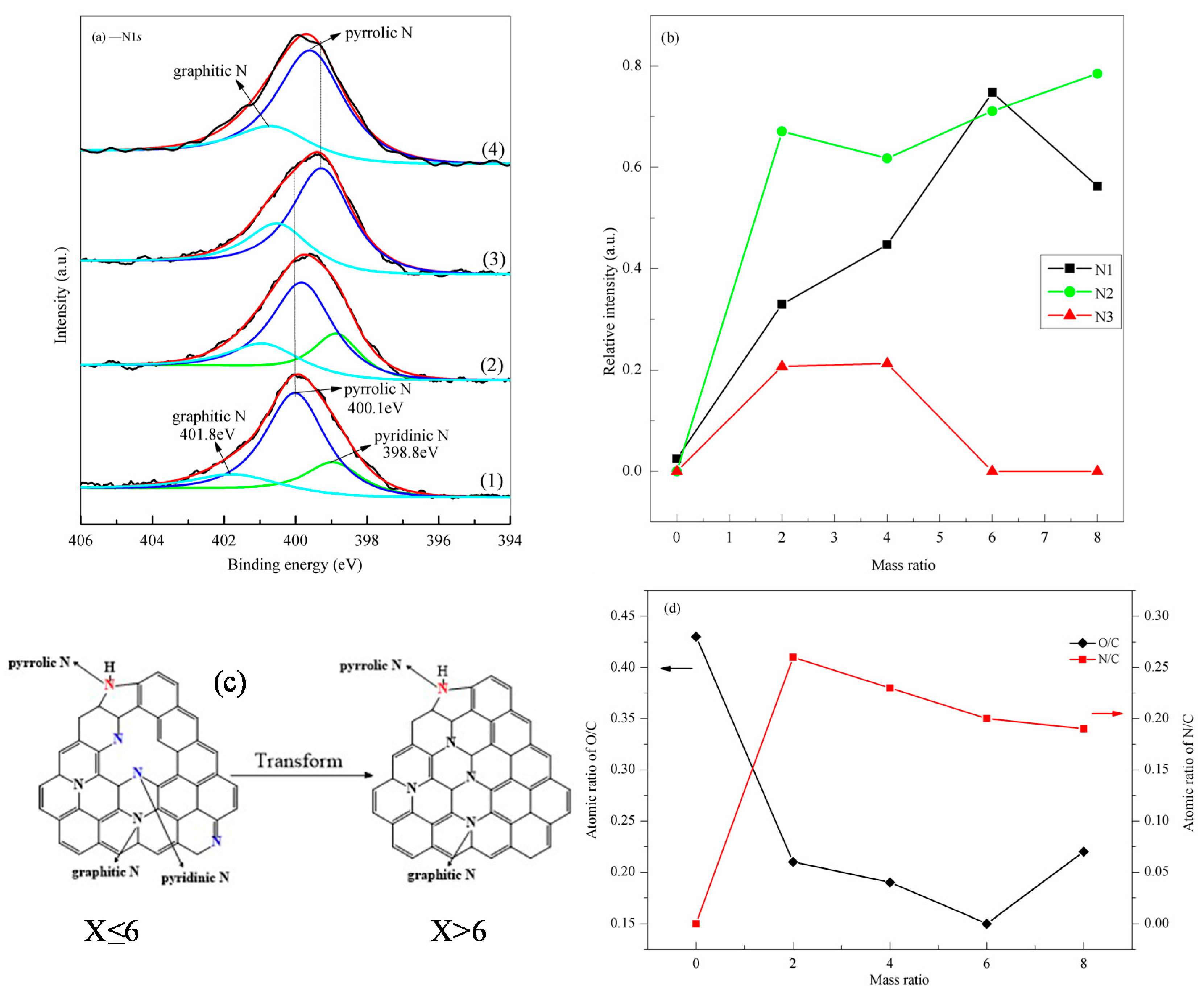
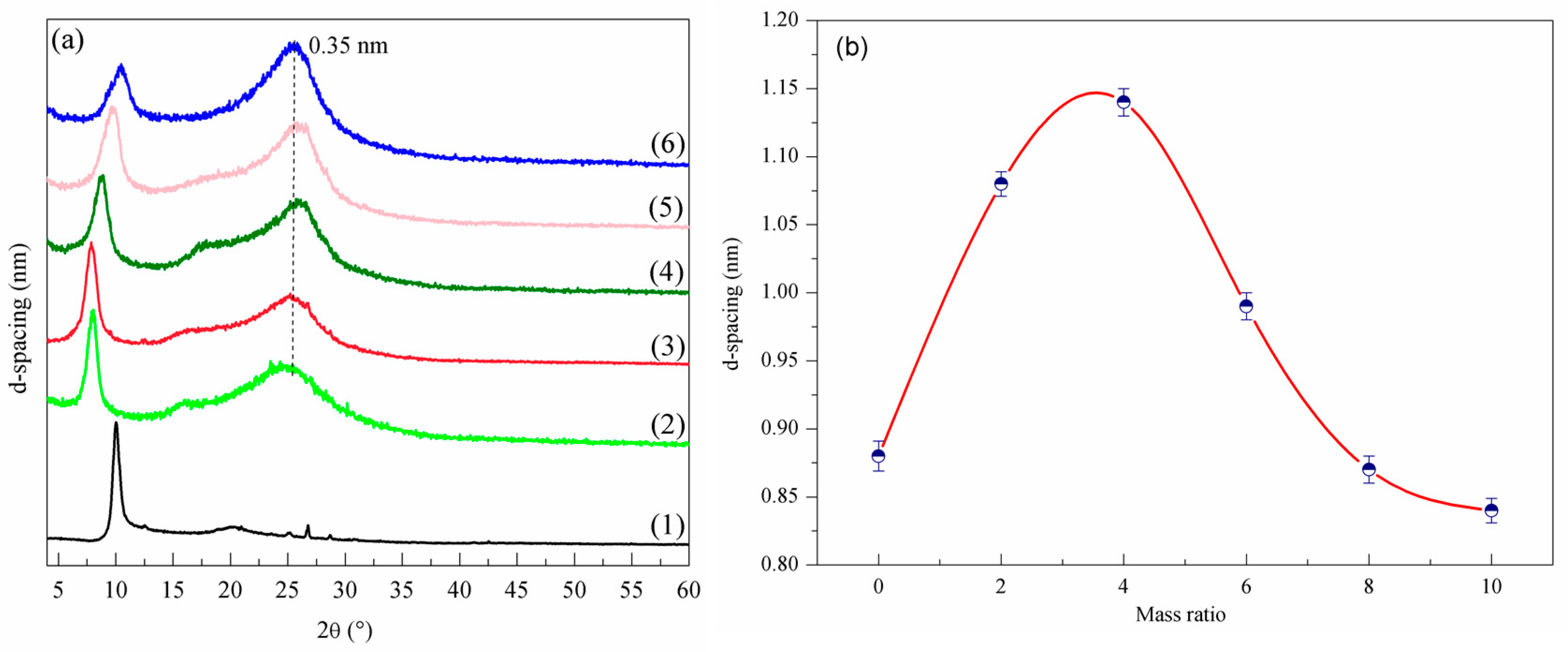
3.2. Layer Structure Changes at Different Ratios of X
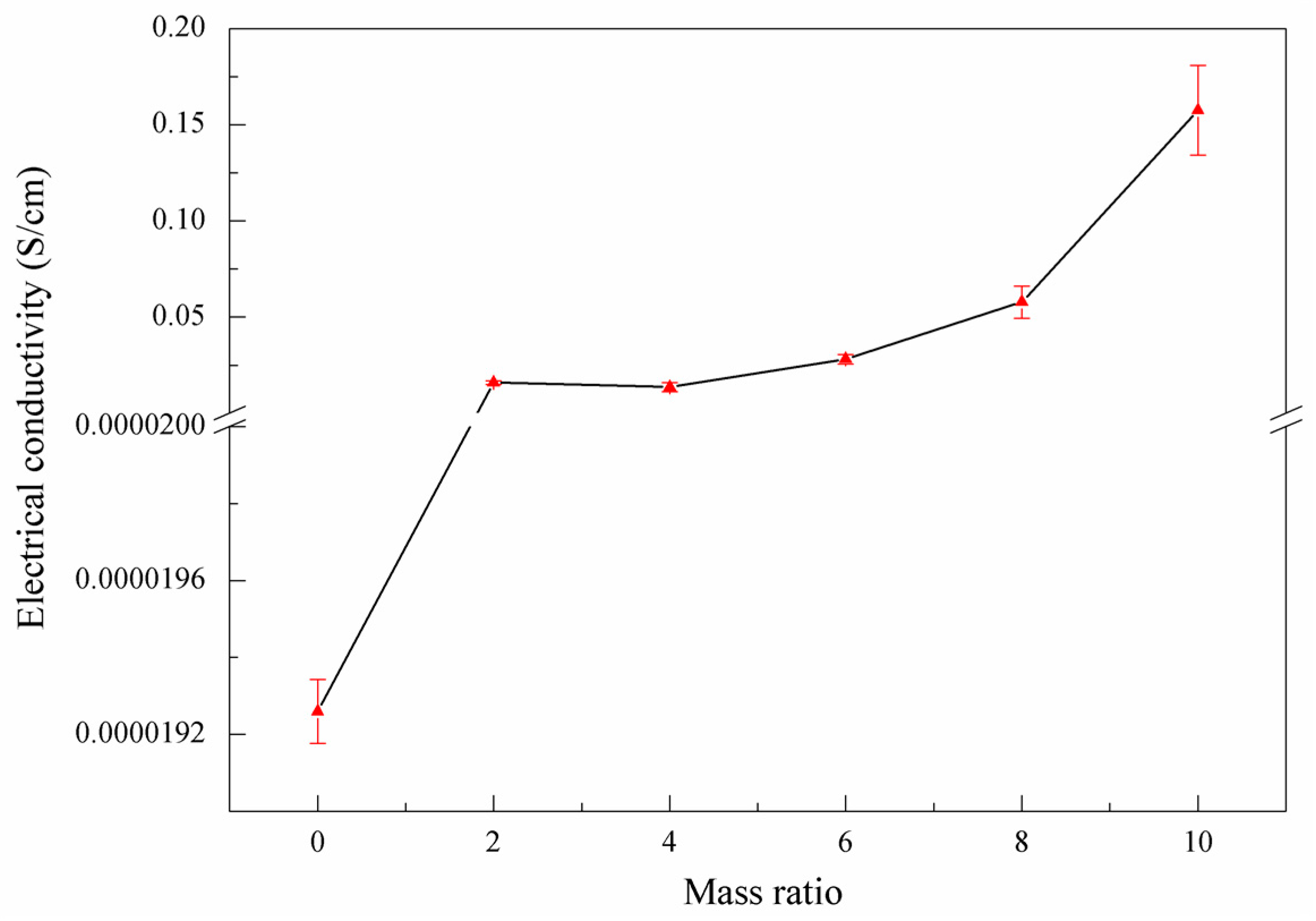
3.3. Conjugate Structure, Electrical Conductivity Change, and Morphology
4. Conclusions
- (1)
- PPD was selected as a nitrogen source and modifier. During the reaction, GO can be reduced and doped with N. The reduction of GO as well as the type and extent of N doping can be regulated by changing the mass ratio, X. At X = 2, the N/C atomic ratio can reach 0.26, while the O/C ratio decreased from 0.43 to 0.15.
- (2)
- When X ≤ 2, the changes of relative strengths of different C species were not the same. When X > 2, the relative strength of C species followed the order: C–C/C=C > C–N > C–O–C/C–O > O–C=O > C=O. When X < 6, there were three types of N atomic configurations in NRGO-X, i.e., pyrrole N, pyridine N, and graphite carbon skeleton N. When X ≥ 6, there were only two configuration types, that is, pyridine N and graphite carbon skeleton N existing in the NRGO-X structure. The relative strength of graphite skeleton N reached the highest value only when X = 6. At other mass ratios of X, the relative strength followed the order: pyrrole N > graphite skeleton N > pyridine N.
- (3)
- With the increase of X, the layer spacing D value of NRGO-X first increased and then decreased, generally in a parabolic distribution, while the electrical conductivity increased gradually.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kim, H.; Miura, Y.; Macosko, C.W. Graphene/polyurethane nanocomposites for improved gas barrier and electrical conductivity. Chem. Mater. 2010, 22, 3441–3450. [Google Scholar] [CrossRef]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Zhan, H.; Zhang, Y.; Bell, J.M.; Mai, Y.-W.; Gu, Y. Structure-mediated thermal transport of monolayer graphene allotropes nanoribbons. Carbon 2014, 77, 416–423. [Google Scholar] [CrossRef]
- Zhan, H.; Zhang, G.; Bell, J.M.; Gu, Y. Graphene with patterned fluorination: Morphology modulation and implications. J. Phys. Chem. C 2015, 119, 27562–27568. [Google Scholar] [CrossRef] [Green Version]
- Shi, X.; Ruan, W.; Hu, J.; Fan, M.; Cao, R.; Wei, X. Optimizing the removal of rhodamine B in aqueous solutions by reduced graphene oxide-supported nanoscale zerovalent iron (NZVI/RGO) using an artificial neural network-genetic algorithm (ANN-GA). Nanomaterials 2017, 7, 134. [Google Scholar] [CrossRef] [PubMed]
- Báez, D.; Pardo, H.; Laborda, I.; Marco, J.; Yáñez, C.; Bollo, S. Reduced graphene oxides: Influence of the reduction method on the electrocatalytic effect towards nucleic acid oxidation. Nanomaterials 2017, 7, 168. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Shi, J.; Zhao, Y.; Song, N.-J.; Wang, Y. A novel porous reduced microcrystalline graphene oxide supported Fe3O4@C nanoparticle composite as anode material with excellent lithium storage performances. Chem. Eng. J. 2017, 326, 507–517. [Google Scholar] [CrossRef]
- Yao, S.; Xu, L.; Gao, Q.; Wang, X.; Kong, N.; Li, W.; Wang, J.; Li, G.; Pu, X. Enhanced photocatalytic degradation of rhodamine B by reduced graphene oxides wrapped-Cu2SnS3 flower-like architectures. J. Alloys Compd. 2017, 704, 469–477. [Google Scholar] [CrossRef]
- Cao, N.; Lyu, Q.; Li, J.; Wang, Y.; Yang, B.; Szunerits, S.; Boukherroub, R. Facile synthesis of fluorinated polydopamine/chitosan/reduced graphene oxide composite aerogel for efficient oil/water separation. Chem. Eng. J. 2017, 326, 17–28. [Google Scholar] [CrossRef]
- Xu, X.; Zheng, Q.; Bai, G.; Song, L.; Yao, Y.; Cao, X.; Liu, S.; Yao, C. Polydopamine induced in-situ growth of Au nanoparticles on reduced graphene oxide as an efficient biosensing platform for ultrasensitive detection of bisphenol A. Electrochim. Acta 2017, 242, 56–65. [Google Scholar] [CrossRef]
- Yuan, Y.-J.; Chen, D.-Q.; Shi, X.-F.; Tu, J.-R.; Hu, B.; Yang, L.-X.; Yu, Z.-T.; Zou, Z.-G. Facile fabrication of “green” SnS2 quantum dots/reduced graphene oxide composites with enhanced photocatalytic performance. Chem. Eng. J. 2017, 313, 1438–1446. [Google Scholar] [CrossRef]
- Susi, T.; Kotakoski, J.; Arenal, R.; Kurasch, S.; Jiang, H.; Skakalova, V.; Stephan, O.; Krasheninnikov, A.V.; Kauppinen, E.I.; Kaiser, U.; et al. Correction to atomistic description of electron beam damage in nitrogen-doped graphene and single-walled carbon nanotubes. ACS Nano 2013, 7, 7436. [Google Scholar] [CrossRef]
- Zhang, L.; Xia, Z. Mechanisms of oxygen reduction reaction on nitrogen-doped graphene for fuel cells. J. Phys. Chem. C 2011, 115, 11170–11176. [Google Scholar] [CrossRef]
- Wang, H.; Maiyalagan, T.; Wang, X. Review on recent progress in nitrogen-doped graphene: Synthesis, characterization, and its potential applications. ACS Catal. 2012, 2, 781–794. [Google Scholar] [CrossRef]
- Panomsuwan, G.; Saito, N.; Ishizaki, T. Electrocatalytic oxygen reduction on nitrogen-doped carbon nanoparticles derived from cyano-aromatic molecules via a solution plasma approach. Carbon 2016, 98, 411–420. [Google Scholar] [CrossRef]
- Li, J.; Ren, Z.; Zhou, Y.; Wu, X.; Xu, X.; Qi, M.; Li, W.; Bai, J.; Wang, L. Scalable synthesis of pyrrolic N-doped graphene by atmospheric pressure chemical vapor deposition and its terahertz response. Carbon 2013, 62, 330–336. [Google Scholar] [CrossRef]
- Lv, R.; Li, Q.; Botello-Méndez, A.R.; Hayashi, T.; Wang, B.; Berkdemir, A.; Hao, Q.; Elías, A.L.; Cruz-Silva, R.; Gutiérrez, H.R.; et al. Nitrogen-doped graphene: Beyond single substitution and enhanced molecular sensing. Sci. Rep. 2012, 2, 586. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Wu, B.; Jiang, L.; Guo, Y.; Huang, L.; Chen, J.; Tan, J.; Geng, D.; Luo, B.; Hu, W.; et al. Low temperature growth of highly nitrogen-doped single crystal graphene arrays by chemical vapor deposition. J. Am. Chem. Soc. 2012, 134, 11060–11063. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Lim, S.; Tian, Z.; Shang, J.; Lai, L.; MacDonald, B.; Fu, C.; Shen, Z.; Yu, T.; Lin, J. Pyridinic N-doped graphene: Synthesis, electronic structure, and electrocatalytic property. J. Mater. Chem. 2011, 21, 8038–8044. [Google Scholar] [CrossRef]
- Zhang, L.; Xia, J.; Zhao, Q.; Liu, L.; Zhang, Z. Functional graphene oxide as a nanocarrier for controlled loading and targeted delivery of mixed anticancer drugs. Small 2010, 6, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Raj, S.; Kolanthai, E.; Sood, A.K.; Sampath, S.; Chatterjee, K. Chemical functionalization of graphene to augment stem cell osteogenesis and inhibit biofilm formation on polymer composites for orthopedic applications. ACS Appl. Mater. Interfaces 2015, 7, 3237–3252. [Google Scholar] [CrossRef] [PubMed]
- Long, D.; Li, W.; Ling, L.; Miyawaki, J.; Mochida, I.; Yoon, S.-H. Preparation of nitrogen-doped graphene sheets by a combined chemical and hydrothermal reduction of graphene oxide. Langmuir 2010, 26, 16096–16102. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, L.; Tian, C.; Tan, T.; Xie, Y.; Shi, K.; Li, M.; Fu, H. Nitrogen-doped graphene with high nitrogen level via a one-step hydrothermal reaction of graphene oxide with urea for superior capacitive energy storage. RSC Adv. 2012, 2, 4498–4506. [Google Scholar] [CrossRef]
- Zhang, Y.; Fugane, K.; Mori, T.; Niu, L.; Ye, J. Wet chemical synthesis of nitrogen-doped graphene towards oxygen reduction electrocatalysts without high-temperature pyrolysis. J. Mater. Chem. 2012, 22, 6575–6580. [Google Scholar] [CrossRef]
- Sun, H.; Wang, Y.; Liu, S.; Ge, L.; Wang, L.; Zhu, Z.; Wang, S. Facile synthesis of nitroge N-doped reduced graphene oxide as a superior metal-free catalyst for oxidation. Chem. Commun. 2013, 49, 9914–9916. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.-L.; Zhang, H.-B.; Hu, Q.-H.; Li, W.-J.; Jiang, Z.-G.; Yu, Z.-Z.; Dasari, A. Functionalization and reduction of graphene oxide with p-phenylene diamine for electrically conductive and thermally stable polystyrene composites. ACS Appl. Mater. Interfaces 2012, 4, 1948–1953. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Shen, J.; Li, N.; Shi, M.; Ma, H.; Yan, B.; Wang, W.; Huang, W.; Ye, M. Amino-functionalization of graphene sheets and the fabrication of their nanocomposites. Polym. Compos. 2010, 31, 1987–1994. [Google Scholar] [CrossRef]
- Ai, W.; Zhou, W.; Du, Z.; Du, Y.; Zhang, H.; Jia, X.; Xie, L.; Yi, M.; Yu, T.; Huang, W. Benzoxazole and benzimidazole heterocycle-grafted graphene for high-performance supercapacitor electrodes. J. Mater. Chem. 2012, 22, 23439–23446. [Google Scholar] [CrossRef]
- Ryu, S.H.; Shanmugharaj, A.M. Influence of long-chain alkylamine-modified graphene oxide on the crystallization, mechanical and electrical properties of isotactic polypropylene nanocomposites. Chem. Eng. J. 2014, 244, 552–560. [Google Scholar] [CrossRef]
- Han, Z.; Tang, Z.; Li, P.; Yang, G.; Zheng, Q.; Yang, J. Ammonia solution strengthened three-dimensional macro-porous graphene aerogel. Nanoscale 2013, 5, 5462–5467. [Google Scholar] [CrossRef] [PubMed]
- Hung, W.-S.; Tsou, C.-H.; De Guzman, M.; An, Q.-F.; Liu, Y.-L.; Zhang, Y.-M.; Hu, C.-C.; Lee, K.-R.; Lai, J.-Y. Cross-linking with diamine monomers to prepare composite graphene oxide-framework membranes with varying d-spacing. Chem. Mater. 2014, 26, 2983–2990. [Google Scholar] [CrossRef]
- Kumar, A.; Khandelwal, M. Amino acid mediated functionalization and reduction of graphene oxide-synthesis and the formation mechanism of nitrogen-doped graphene. New J. Chem. 2014, 38, 3457–3467. [Google Scholar] [CrossRef]
- Shanmugharaj, A.M.; Yoon, J.H.; Yang, W.J.; Ryu, S.H. Synthesis, characterization, and surface wettability properties of amine functionalized graphene oxide films with varying amine chain lengths. J. Colloid Interface Sci. 2013, 401, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Jang, J.; Nagase, S. Hydrazine and thermal reduction of graphene oxide: Reaction mechanisms, product structures, and reaction design. J. Phys. Chem. C 2010, 114, 832–842. [Google Scholar] [CrossRef]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, T.; Sun, H.; Peng, T.; Liu, B.; Zhao, X. Structural Regulation and Electroconductivity Change of Nitrogen-Doping Reduced Graphene Oxide Prepared Using p-Phenylene Diamine as Modifier. Nanomaterials 2017, 7, 292. https://doi.org/10.3390/nano7100292
Peng T, Sun H, Peng T, Liu B, Zhao X. Structural Regulation and Electroconductivity Change of Nitrogen-Doping Reduced Graphene Oxide Prepared Using p-Phenylene Diamine as Modifier. Nanomaterials. 2017; 7(10):292. https://doi.org/10.3390/nano7100292
Chicago/Turabian StylePeng, Tiefeng, Hongjuan Sun, Tongjiang Peng, Bo Liu, and Xiaolong Zhao. 2017. "Structural Regulation and Electroconductivity Change of Nitrogen-Doping Reduced Graphene Oxide Prepared Using p-Phenylene Diamine as Modifier" Nanomaterials 7, no. 10: 292. https://doi.org/10.3390/nano7100292






