3.1. Characterization of Gold ENMs
The gold ENMs were selected to provide a range of characteristics such as shape, morphology and surface charges. Various types of shaped and charged gold ENMs were used to determine the effect on toxicity towards
D. magna. A summary of the gold ENM characteristics can be seen in
Table 1.
Gold ENMs were characterized using multiple methods to ensure adequate characterization, including DLS, DCS, UV-Vis and TEM.
DLS results provided hydrodynamic diameter of a spherical ENM which is a slightly higher value compared to the core size as ENMs dispersed in medium have a thin layer of solvent that surrounds the surface and hydrodynamic diameter is a measure of ENM diameter and the absorbed surrounding layer. DCS is a useful technique to measure rod shaped ENMs: sedimentation of ENMs follows Stokes’ law, where the dimensions of a rod shaped ENM can be converted to an equivalent spherical diameter following Equation (1):
where
dst is the equivalent spherical diameter (Stokes’ diamter),
dc is the diameter of the cylinder and β is the aspect ratio (length vs. diameter) of the ENM. According to this equation, equivalent spherical diameter depends directly on the diameter of the rod though only marginally on the length [
18], which is why both short (length 60 nm) and long (length 146 nm) rod ENMs appear to have a size value of 33 nm as both rods have the same diameter of 25 nm. Gold ENMs have localized surface plasmon resonance (LSPR) resulting in a strong absorbance band between 500–600 nm which can be seen for spherical, short rod and long rod gold ENMs. Gold ENMs that have a second dimension (rods) have a second peak which is red-shifted to the right with longer diameters resulting in a further red-shift. Characterization of ENMs were completed in fresh HH Combo medium except for TEM images which were prepared in DI water to avoid drying effects from salts present in the media. TEM reflects better the physical dimensions of ENMs providing their dry characteristics and core size, although does not explain the ENMs behavior in dispersion and, depending on sample preparation, may overexpress aggregation, or cause particle dehydration. UV-Vis spectra and TEM images for spherical and rod gold ENMs are provided in
Figure S1.
3.2. Effect of Gold ENMs on D. magna Survival
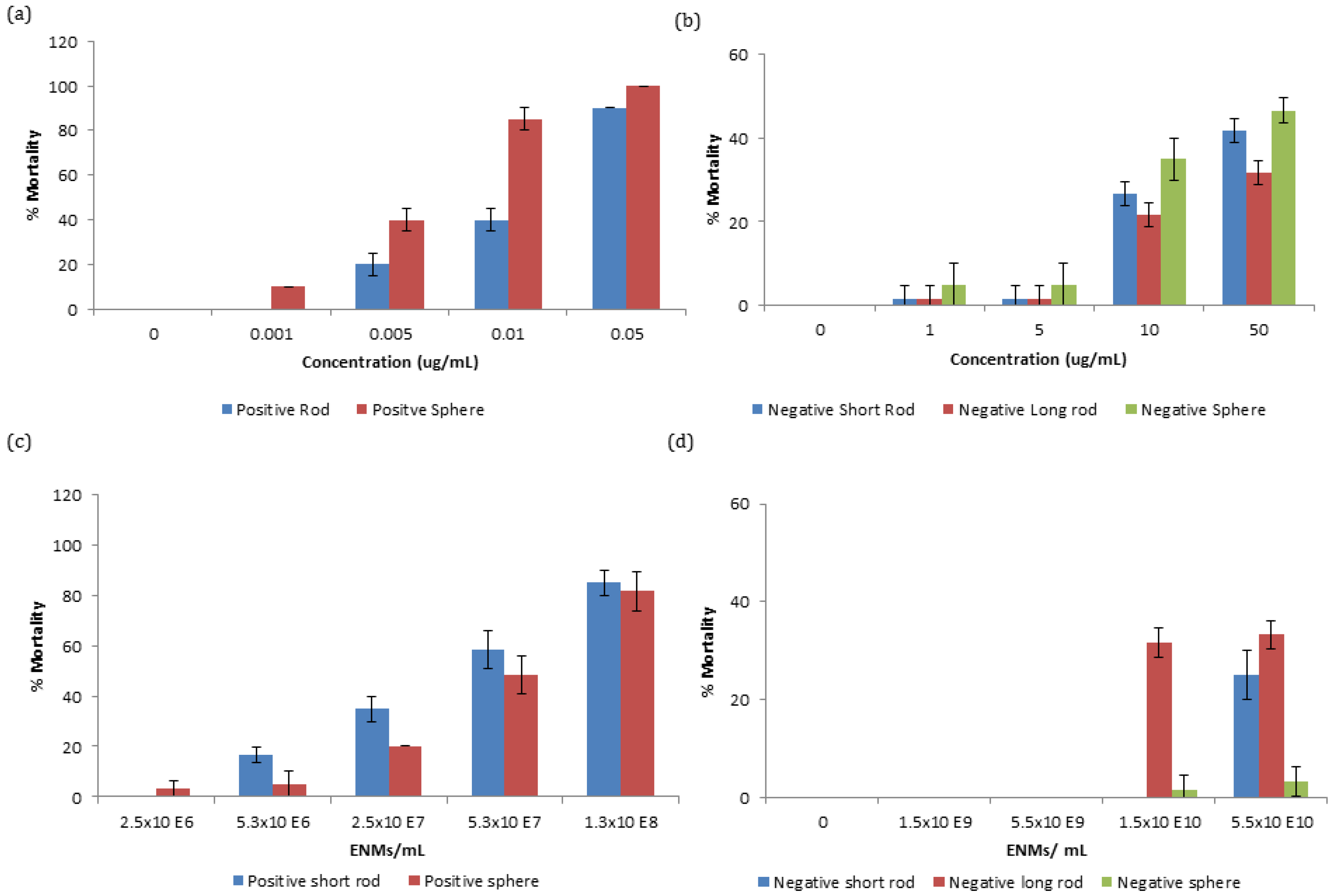
Zooplankton were exposed to a range of concentrations of gold ENMs in order to assess survivorship. It was observed that positively charged ENMs, regardless of shape, were orders of magnitude more toxic compared to negatively charged ENMs with respect to mass concentration. The EC
50 for positively charged spheres and short rods were 6.11 and 18 µg/L respectively. Differences in mortality between positively charged spheres and short rods were statistically significant at concentrations between 0.005 and 0.01 µg/mL at
p < 0.01 and 0.001, respectively, as determined by students-test. The increased toxicity of positively charged ENMs can be attributed to the positive charge on the NM being attracted to the negative charge on the phospholipid membrane of organisms increasing their interaction [
12]. A similar toxicity trend can be observed with negatively charged gold ENMs (
Figure 1b) where spheres appear to be most toxic, followed by short rods and then by longer rods although there is no EC
50 value even at concentrations as high as 50 µg/mL indicating low toxicity of these ENMs. Negatively charged spheres were not significantly different in toxicity compared to short rods, though were statistically significant compared to long rods at 10 and 50 µg/mL at
p < 0.05 and 0.01, respectively.
Particle number concentration is an important comparison basis to consider for dose-metric issues for ENMs, where mass concentration is not an optimal comparison, especially when considering ENMs of different sizes/shapes. Toxicity of ENMs is not always size dependent and ENM number or surface area exposure have been demonstrated as more optimal choices for NM comparison [
19]. Gold ENMs of various number concentrations were exposed to
D. magna neonates and as expected, regardless of number or mass concentration exposure, positively charged ENMs were still orders of magnitude more toxic than negatively charged ENMs. Interestingly, with respect to number concentration, long rods were found to be most toxic, followed by short rods and finally spheres as seen in
Figure 1c,d for both positively and negatively charged ENMs, respectively. Spherical and short rod shaped NMs that were exposed to neonates at the same number concentration have a different amount of surface area exposed to
D. magna depending on the type of ENM. This can explain why positively charged short rods appear to be most toxic as they have a larger surface area available for interaction (5694 nm
2) compared to spheres (1963 nm
2) as can be identified in
Table 2. The EC
50 values for the positively charged ENMs for spheres and short rods with respect to number concentration are 5.69 × 10
7 ENMs/mL and 4.30 × 10
7 ENMs/mL respectively. Toxicity of ENMs may also depend on their ability to agglomerate, as
D. magna preferentially take-up larger entities. It was determined that ENMs remained stable in HH Combo medium for 24 h (
Table S2) with no statistical significance in zeta-potential values over time.
Though an increased surface area could explain an increase in toxicity, this would only hold true if the charge per surface area on each of the ENMs types were the same and therefore charge per surface area must also be taken into account to verify the potency of the NM toxicity.
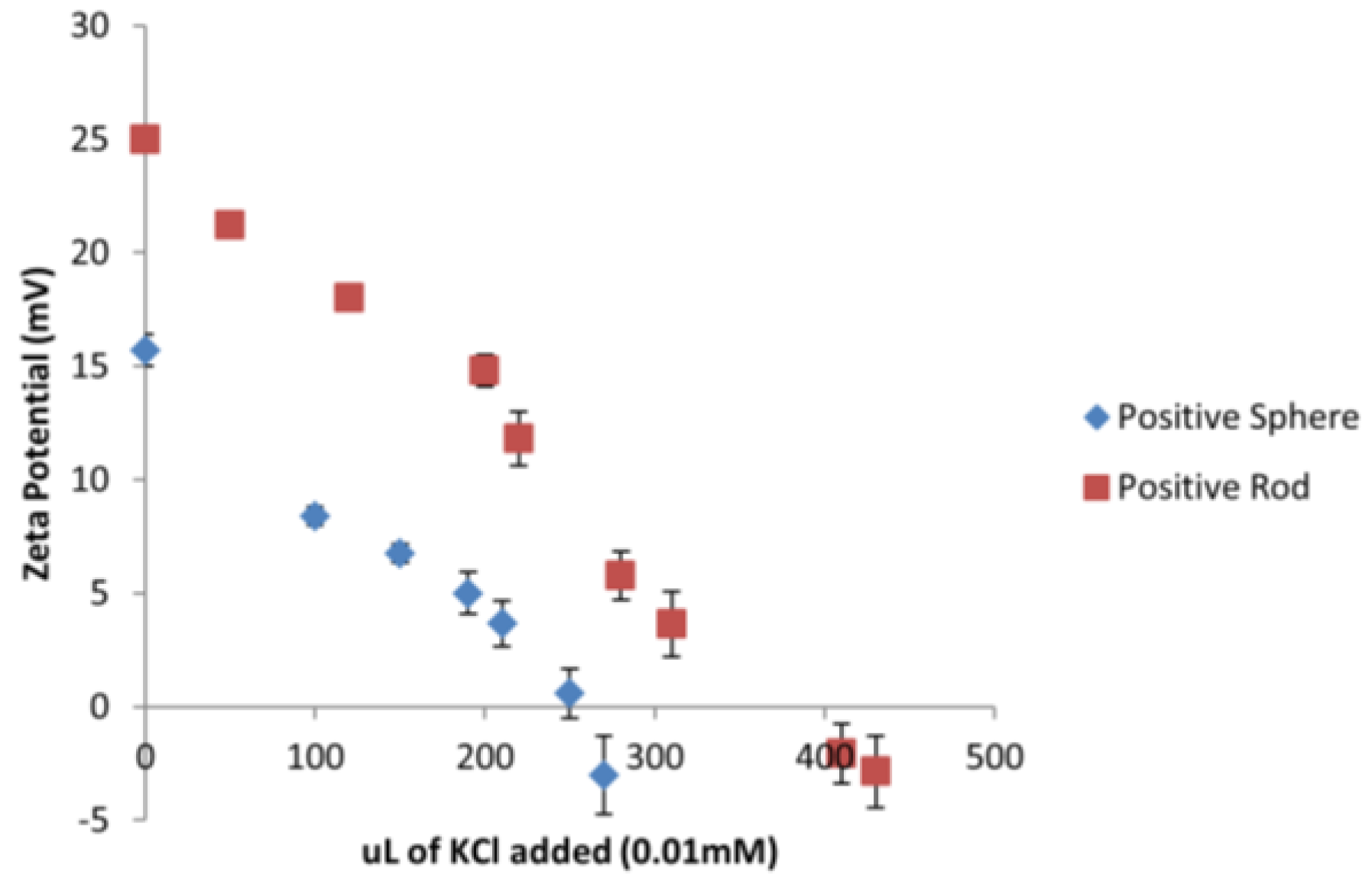
Considering that all the tested ENMs are of different sizes and shapes, a different amount of surface area is being exposed to
D. magna depending on the type of ENM. A series of titrations were conducted using 0.01 mM KCl to neutralize the charge on the gold ENMs in order to determine how many
n of KCl was needed to neutralize the same amount of
n of gold ENMs. It was determined that 270 µL and 430 µL of 0.01 mM KCl was needed to neutralize the charges on positively charged spheres and positively charged short rods respectively as can be seen in
Figure 2 which corresponds to 2 positive charges/nm
2 on spheres and 3 positive charges per nm
2 on short rods (see
Supplementary Materials for sample calculation).
Analysis of the charge per surface area can begin to explain the toxicity effects of positively charged ENMs on
D. magna as short rods have a much higher surface area compared to spheres. Results also indicate that short rods hold a higher number of positive charges per nm
2 compared to spheres which in turn provides a substantially higher amount of overall positive charge to be available to interact with
D. magna. Therefore short rods appear to have a dual toxic effect of a larger surface area and an increased charge to surface area ratio compared to the spheres. It was determined for negatively charged long rods, short rods and spheres, that the charge per surface area corresponded to 2 and 3 charges/nm
2 as seen in
Figure S3.
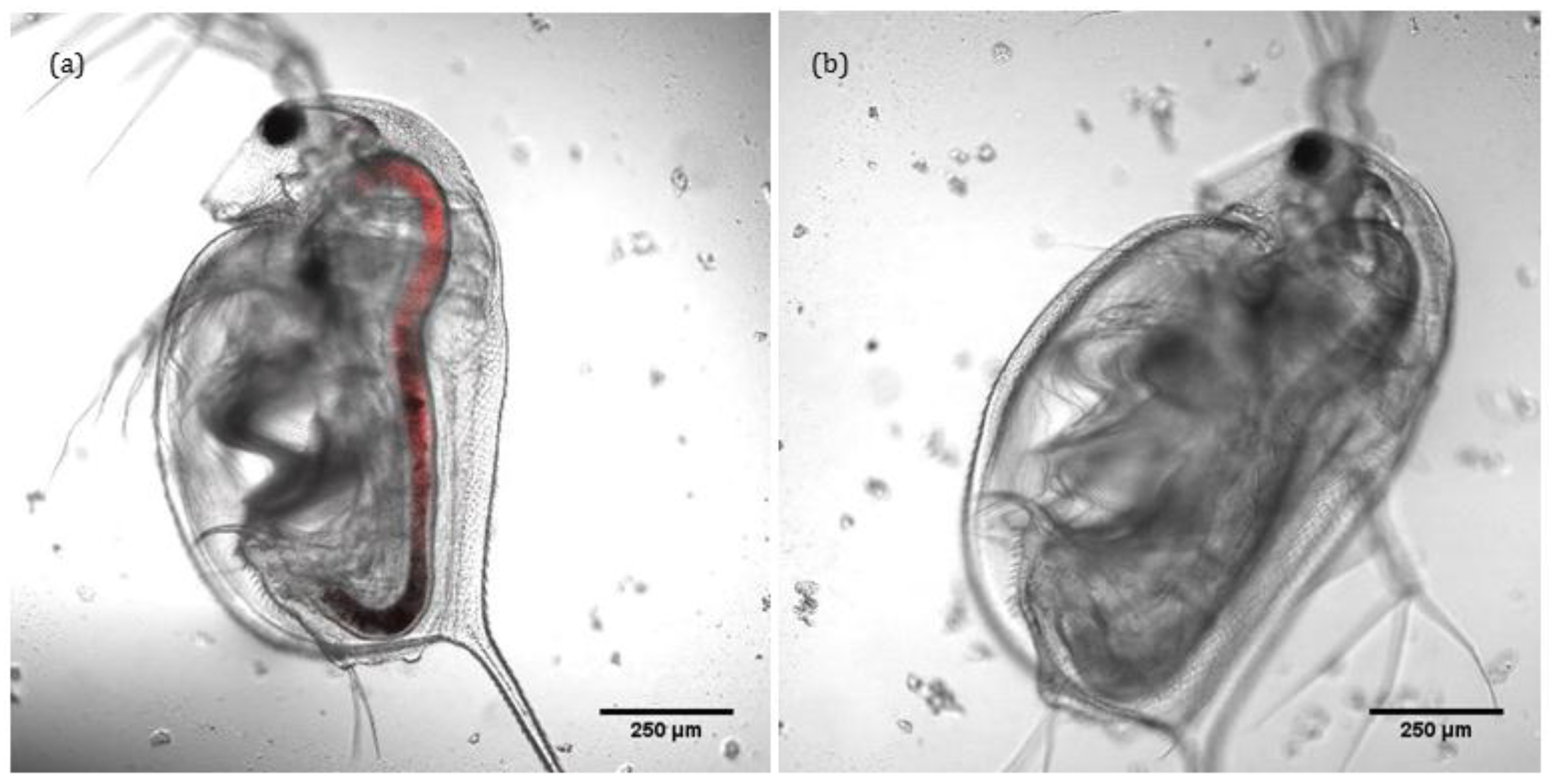
Confocal and transmitted light imaging was used to confirm that the intaken ENMs remain localised solely within the gut of
D. magna and do not translocate into other tissues, as shown in
Figure 3a,b. Thus, lumen cells and microvilli along the bush boarder are exposed to ENMs, and observed toxicity could be due to selective interaction of ENMs with intracellular organelles (mitochondria or DNA) of lumen cells and microvilli. Charge density is also a moderator of the degree of toxicity (see
Table S3). ENMs do not appear to translocate any further than the gut, illustrating its effectiveness as a barrier. Note that the fluorescence was added via conjugation to ~50% of the –NH
2 groups on the surface of the positively charged ENMs. Transmitted light images of the same can be found in
Figure S4. As gold ENM are inherently reflectant, use of reflectance confocal microscopy as a means to quantify uptake and localisation was also attempted. However, the carapace itself is highly reflectant and as
D. magna tend to consume the carapace (in the absence of food sources) and the keratin protein that constitutes the carapace is itself highly reflectant, such that control organisms (with no ENMs) were actually more reflectant that the gold ENM-exposed
D. magna.
3.3. Effect of Gold ENMs on ROS Production in D. magna
It was determined that gold ENMs regardless of their shape or charge are able to influence
D. magna survival and therefore an analysis of the toxicity mechanism was performed. It is widely recorded that
D. magna produce ROS as an indicator of stress [
20]. The production of ROS within organisms is an excellent indicator of stress at a biochemical level and can take the form of superoxide anion radical, hydrogen peroxide and hydroxyl radical, each of which are known to be produced by
D. magna [
20]. As seen in
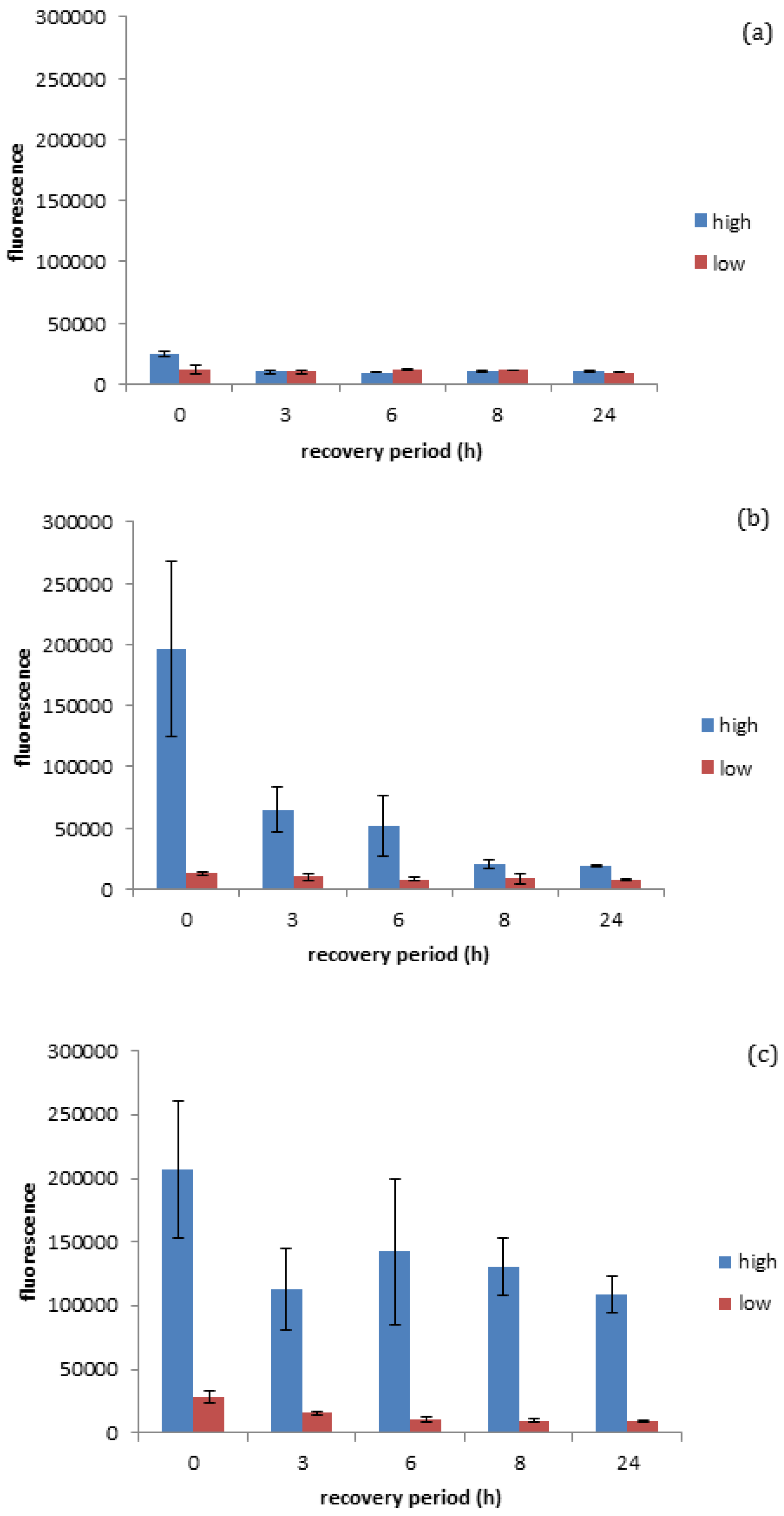
Figure 4a, it was quantified that negatively charged gold spheres prompt minimal ROS production in
D. magna exposed to either high (EC
10) or low (EC
3) number concentrations of negatively charged gold ENMs (for EC
10 of negatively charged spheres see
Figure S2), while positively charged spherical gold ENMs prompted significant ROS production in
D. magna exposed to either high (EC
40) or low (EC
5) number concentrations of positively charged gold ENMs. EC values were chosen as appropriate ‘high’ and ‘low’ values based on survivorship results where ENMs of the same charge are comparable. Further explanation of the selection of the ENM number particle concentrations is given in the
supplementary information alongside
Table S1. This is complementary to the survivorship results in
Figure 1 and
Figure 2, as negatively charged gold ENMs were shown to be orders of magnitude less toxic compared to positively charged gold ENMs.
As seen in
Figure 4b, positively charged spheres prompt a higher degree of ROS generation (determined immediately after the exposure period, so at 0 h recovery) compared to negatively charged spheres at the high exposure dose seen in
Figure 4a, although the accumulated ROS is reduced by approximately 65% within the three hours of the recovery period and reaches steady state levels (approximately 10%) after 8 h of recovery (insignificant
t-test
p < 0.05). This is in line with survivorship results as high concentrations of positive spherical ENMs can cause a high generation of ROS where coping mechanisms are not able to fully overcome the ROS produced, resulting in increased
D. magna mortality. ROS generated by positive and negatively charged spheres when exposed to high and low concentrations of ENMs are significantly different at 0 h recovery (significant
t-test
p < 0.05 and
p < 0.001 respectively) and statistically significant at low concentrations at 24 h recovery (
p < 0.05). Positively charged rods also appear to prompt a high degree of ROS generation, although
D. manga coping mechanisms appear to be unable to fully recover from the stress in this case, unlike with positively charged spheres. This inability to recover could be attributed to rods having an increased surface area as well as a higher surface area to charge ratio, thus causing them to induce a higher degree of toxicity/ROS activation as seen in
Figure 4c. It was observed that Daphnia were unable to successfully remove ROS accumulation even after 24 h recovery, which can be due to decreasing levels of anti-oxidant enzymes and reduced anti-buffering capacity where there is insufficient enzyme available to handle ROS production and therefore a ROS spike occurs at 8 h into recovery. This prompts the de novo synthesis of anti-oxidant enzymes to handle the excess ROS and can be seen as ROS levels start to decrease again. It is interesting as this may be a factor to explain why rods are more toxic compared to spheres at the same number concentration, as
D. magna are able to effectively remove ROS when exposed to high levels of spheres though are unable to when exposed to rods, which could potentially be due to the ingestion of the ENMs or increased adsorption of ENMs to the carapace leading to a decrease in moulting. Of course, the production or ROS is not the only factor influencing ENM toxicity, whereby both ROS and high toxicity could correlate to cellular stress, whereby damage to the mitochondria could cause a decrease in cellular respiration and ATP depletion. Cellular stress caused by damage to DNA can result in the cells undergoing unregulated necrotic cell death. Upon exposure to cellular stress, homeostasis between net growth and death is altered, whereupon cells may employ a protective cellular response. However, if cellular stress is too high this will result in the activation of cell death pathways [
21] with ROS functioning at all stages of this process, either as an initiating event or as a symptom of cellular stress.
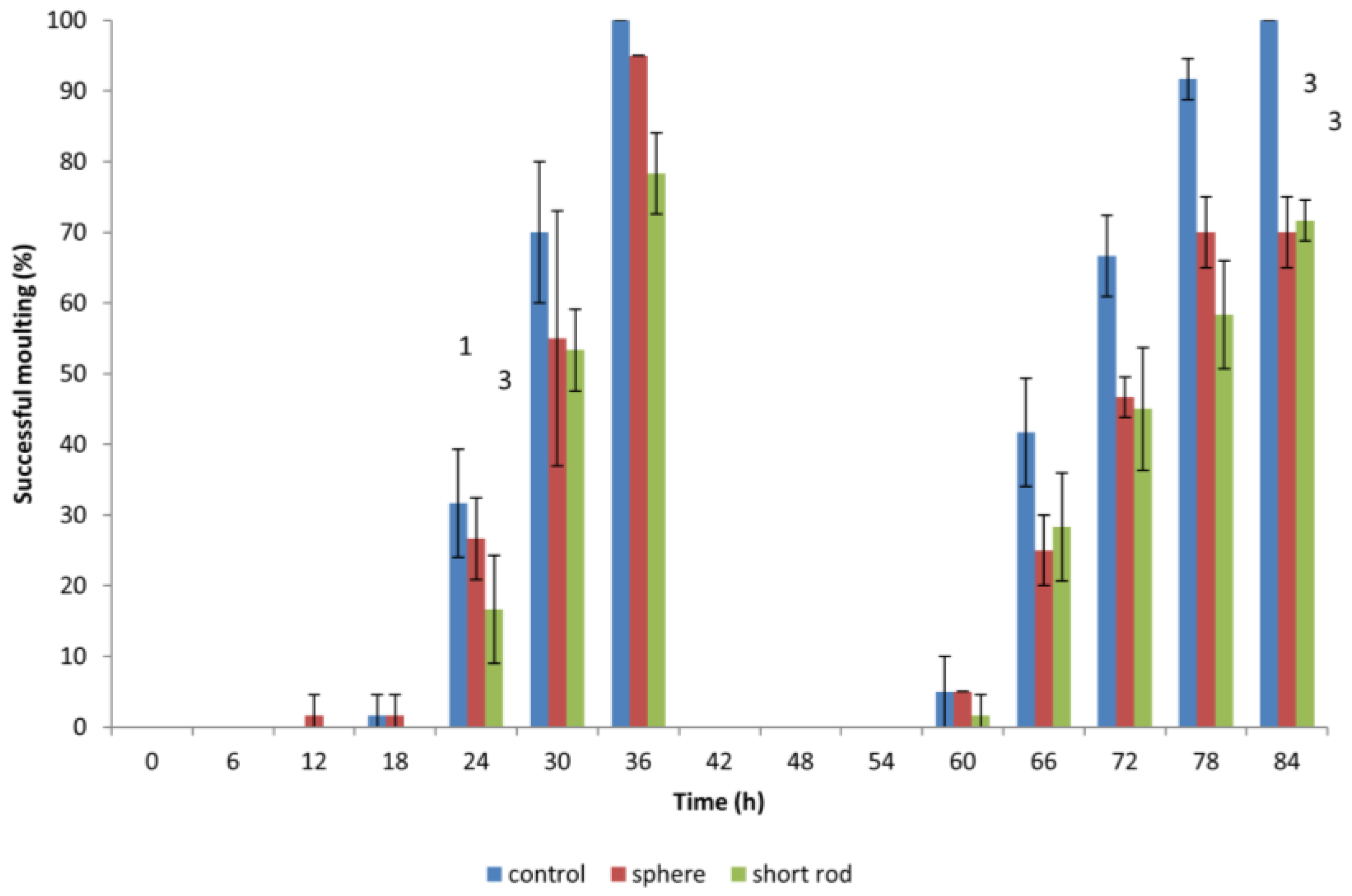
3.4. Effect of Gold ENMs on Moulting of D. magna
When
D. magna neonates were previously exposed to gold ENMs, neonates would appear to halt in mid-swim and then return to a regular swimming pattern which prompted the idea if ENMs were adhering to the surface of
D. magna and if this would have an effect on shedding of the carapace, hindering further development. During the first moulting, which occurs approximately 24 h after birth,
D. magna shed their exoskeleton, and this is known to be a mechanism for removing metal pollutants also, so we postulated that any adhered gold ENMs would also be shed. Our results indicate that all living
D. magna neonates that are exposed to gold ENMs at a concentration of 5.3 × 10
6 ENMs/mL (regardless of shape or charge) successfully complete the first round of moulting. The concept of adsorption of ENMs to organisms has been well established that the presence of environmental stressors induces difficulties in moulting [
22]. Biological surface coating leads to ENMs adhering to the filtering apparatus of
D. magna [
10] resulting in an increased chance of uptake by filter feeding. Our results indicate that exposure to gold ENMs plays a major role in the decrease of moulting ability of
D. magna. The coating of the exoskeleton by gold ENMs increases both the weight along with physical stamina required by each neonate, which consequently increases the energy demand of each neonate. For neonates also struggling with high ROS levels, we postulate that the required energy for moulting is unavailable due to the high demand for energy used in an attempt to remove accumulating ROS for neonates exposed to positively charged spheres or short rods. Our results clearly indicate that an 84 h exposure to 5.3 × 10
6 ENMs/mL of spherical and rod shaped gold ENMs significantly reduces the success of a second moulting compared to 100% moulting success in the control group (**,
p < 0.01 for spheres) and (**,
p < 0.01 for short rods).
D. magna exposed to positively charged gold rods, moult less, though not significantly, compared to those exposed to spheres at the end of the exposure period (84 h), as seen in
Figure 5. The inability of
D. magna to moult combined with accumulated ROS generation result in
D. magna immobility and mortality. This is consistent with findings from Dabrunz et al. for nanoscale titanium dioxide ENMs (nTiO
2), who hypothesized a mechanistic chain of events for nTiO
2 toxicity in
D. magna that involves the coating of the organism surface with nTiO
2 combined with a moulting disruption [
10].