Hydrothermal Synthesis of Nanoclusters of ZnS Comprised on Nanowires
Abstract
:1. Introduction
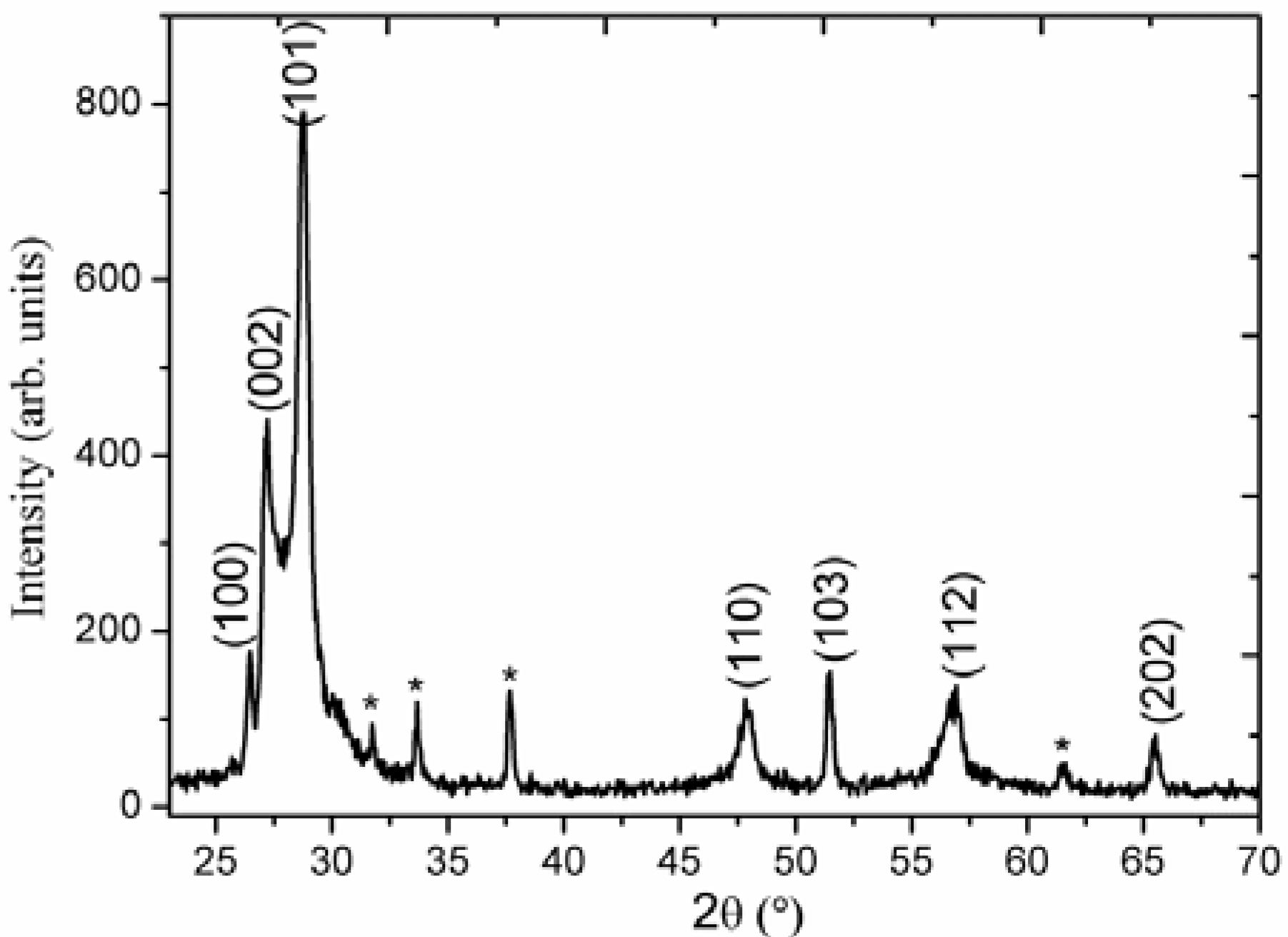
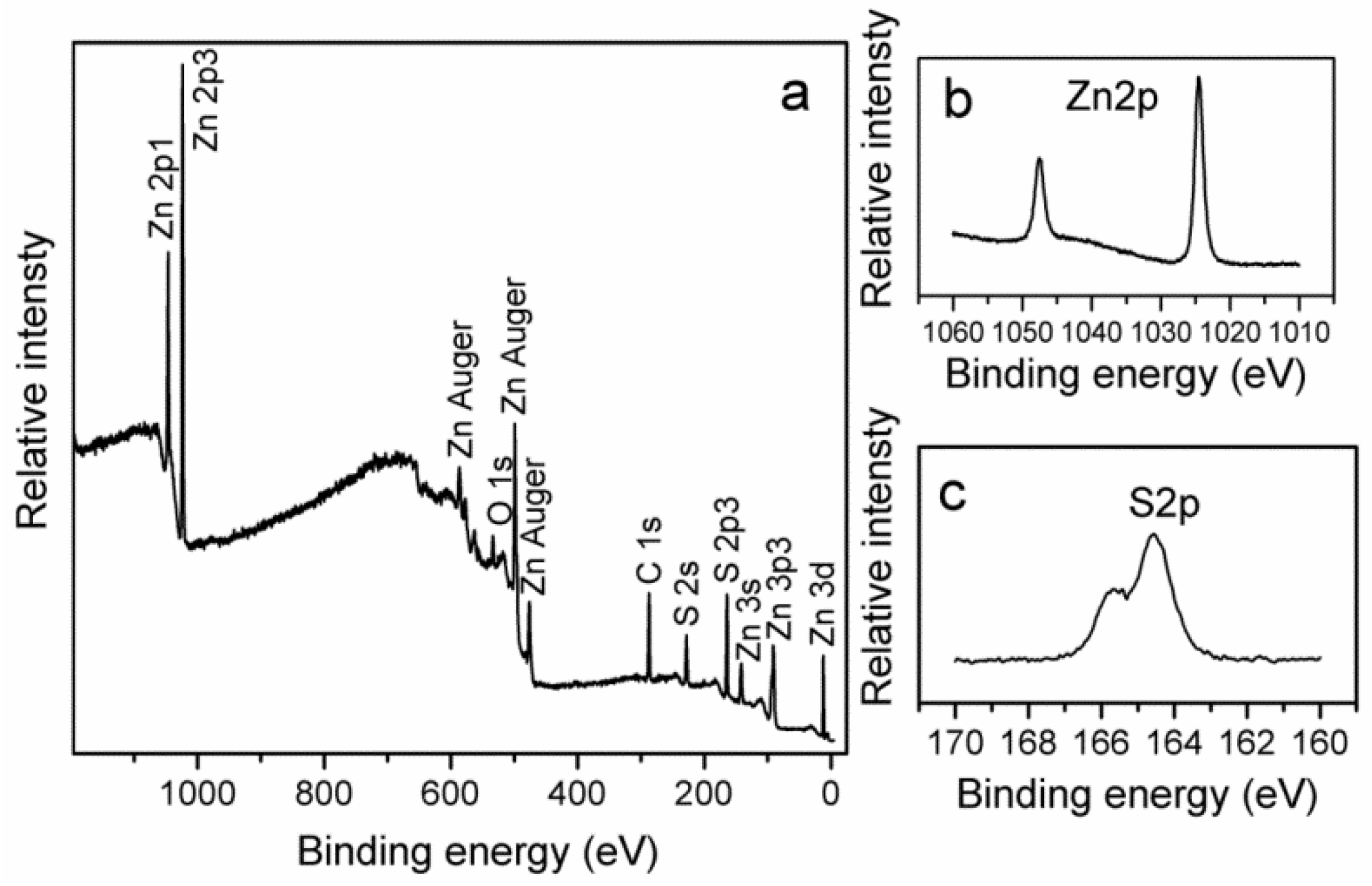
2. Results and Discussion


3. Experimental Section
4. Conclusions
Conflicts of Interest
References
- Wang, D.W.; Zhao, S.L.; Xu, Z.; Kong, C.; Gong, W. The improvement of near-ultraviolet electroluminescence of ZnO nanorods/MEH-PPV heterostructure by using a ZnS buffer layer. Org. Electro. 2011, 12, 92–97. [Google Scholar] [CrossRef]
- Boutaud, G.; Cranton, W.M.; Koutsogeorgis, D.C.; Ranson, R.M.; Tsakonas, C.; Thomas, C.B. Growth optimisation of ZnS:Mn thin film phosphors for high intensity miniature electroluminescent displays. Mater. Sci. Eng. B 2009, 165, 202–206. [Google Scholar] [CrossRef]
- Liang, Y.; Xu, H.Y.; Hark, S.K. Orientation and structure controllable epitaxial growth of ZnS nanowire arrays on GaAs substrates. J. Phys. Chem. C 2010, 114, 8343–8347. [Google Scholar]
- Kao, C.C.; Liu, Y.C. Intense green emission of ZnS:Cu, Al phosphor obtained by using diode structure of carbon nano-tubes field emission display. Mater. Chem. Phys. 2009, 115, 463–466. [Google Scholar] [CrossRef]
- Hu, J.S.; Ren, L.L.; Guo, Y.G.; Liang, H.P.; Cao, A.M.; Wan, L.J.; Bai, C.L. Mass production and high photocatalytic activity of ZnS nanoporous nanoparticles. Angew. Chem. Int. Ed. 2005, 44, 1269–1273. [Google Scholar]
- Yu, J.H.; Joo, J.; Park, H.M.; Baik, S.I.; Kim, Y.W.; Kim, S.C.; Hyeon, T.J. Synthesis of quantum-sized cubic ZnS nanorods by the oriented attachment mechanism. J. Am. Chem. Soc. 2005, 127, 5662–5670. [Google Scholar]
- Ma, C.; Moore, D.; Li, J.; Wang, Z.L. Nanobelts, nanocombs and nanowindmills of wurtzite ZnS. Adv. Mater. 2003, 15, 228–231. [Google Scholar] [CrossRef]
- Yao, W.T.; Yu, S.H.; Pan, L.; Li, J.; Wu, Q.S.; Zhang, L.; Jiang, J. Flexible wurtzite-type ZnS nanobelts with quantum-size effects: A diethylenetriamine-assisted solvothermal approach. Small 2005, 1, 320–325. [Google Scholar] [CrossRef]
- Yin, L.W.; Bando, Y.; Zhan, J.H.; Li, M.S.; Golberg, D. Self-assembled highly faceted wurtzite-type ZnS single-crystalline nanotubes with hexagonal cross-sections. Adv. Mater. 2005, 17, 1972–1977. [Google Scholar] [CrossRef]
- Yu, S.H.; Yoshimura, M. Shape and phase control of ZnS nanocrystals: Template fabrication of wurtzite ZnS single-crystal nanosheets and ZnO flake-like dendrites from a lamellar molecular precursor ZnS·(NH2CH2CH2NH2)0.5. Adv. Mater. 2002, 14, 296–300. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Bando, Y.; Xue, D.F.; Golberg, D. Nanocable-aligned ZnS tetrapod nanocrystals. J. Am. Chem. Soc. 2003, 125, 16196–16197. [Google Scholar] [CrossRef]
- Moore, D.F.; Ding, Y.; Wang, Z.L. Crystal orientation-ordered ZnS nanowire bundles. J. Am. Chem. Soc. 2004, 126, 14372–14373. [Google Scholar] [CrossRef]
- Zhu, Y.C.; Bando, Y.; Xue, D.F.; Golberg, D. Oriented assemblies of ZnS one-dimensional nanostructures. Adv. Mater. 2004, 16, 831–834. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, S.; Pan, S.; Li, G.; Hou, J. A simple solution route to ZnS nanotubes and hollow nanospheres and their optical properties. Nanotechnology 2004, 15, 945–948. [Google Scholar] [CrossRef]
- Liu, H.; Ni, Y.; Han, M.; Liu, Q.; Xu, Z.; Hong, J.; Ma, X. A facile template-free route for synthesis of hollow hexagonal ZnS nano- and submicro-spheres. Nanotechnology 2005, 16. [Google Scholar] [CrossRef]
- Wolosiuk, A.; Armagan, O.; Braun, P.V. Double direct templating of periodically nanostructured ZnS hollow microspheres. J. Am. Chem. Soc. 2005, 127, 16356–16367. [Google Scholar] [CrossRef]
- Peng, Q.; Xu, S.; Zhuang, Z.; Wang, X.; Li, Y. A small extension to “costs and rewards of children: The effects of becoming a parent on adults’ lives”. Small 2005, 1, 216–221. [Google Scholar] [CrossRef]
- Jiang, Y.; Meng, X.M.; Liu, J.; Hong, Z.R.; Lee, C.S.; Lee, S.T. ZnS nanowires with wurtzite polytype modulated structure. Adv. Mater. 2003, 15, 1195–1198. [Google Scholar] [CrossRef]
- Meng, X.M.; Liu, J.; Jiang, Y.; Chen, W.W.; Lee, C.S.; Bello, I.; Lee, S.T. Structure- and size-controlled ultrafine ZnS nanowires. Chem. Phys. Lett. 2003, 382, 434–438. [Google Scholar]
- Lin, M.; Sudhiranjan, T.; Boothroyd, C.; Loh, K.P. Influence of Au catalyst on the growth of ZnS nanowires. Chem. Phys. Lett. 2004, 400, 175–178. [Google Scholar] [CrossRef]
- Kar, S.; Biswas, S.; Chaudhuri, S. Catalytic growth and photoluminescence properties of ZnS nanowires. Nanotechnology 2005, 16, 737–740. [Google Scholar] [CrossRef]
- Moore, D.; Morber, J.R.; Snyder, R.L.; Wang, Z.L. Growth of ultralong ZnS/SiO2 core-shell nanowires by volume and surface diffusion VLS process. J. Phys. Chem. C 2008, 112, 2895–2903. [Google Scholar] [CrossRef]
- Kim, D.; Shimpi, P.; Gao, P.X. Zigzag zinc blende ZnS nanowires: Large scale synthesis and their structure evolution induced by electron irradiation. Nano Res. 2009, 2, 966–974. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, W. Preparation and photoluminescence of zinc sulfide nanowires. Mater. Lett. 2004, 58, 3836–3838. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, S.; Zuo, M.; Li, G.; Hou, J. Synthesis of ZnS nanowires and assemblies by carbothermal chemical vapor deposition and their photoluminescence. Eur. J. Inorg. Chem. 2005, 2005, 47–50. [Google Scholar] [CrossRef]
- Xu, X.J.; Fei, G.T.; Yu, W.H.; Wang, X.W.; Chen, L.; Zhang, L.D. Preparation and formation mechanism of ZnS semiconductor nanowires made by the electrochemical deposition method. Nanotechnology 2006, 17, 426–429. [Google Scholar] [CrossRef]
- Sun, H.Y.; Li, X.H.; Li, W.; Li, F.; Liu, B.T.; Zhang, X.Y. Low-temperature synthesis of wurtzite ZnS single-crystal nanowire arrays. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Chang, M.; Cao, X.L.; Xu, X.J.; Zhang, L. Fabrication and photoluminescence properties of highly ordered ZnS nanowire arrays embedded in anodic alumina membrane. Phys. Lett. A 2008, 372, 273–276. [Google Scholar] [CrossRef]
- Chai, L.; Du, J.; Xiong, S.; Li, H.; Zhu, Y.; Qian, Y.J. Synthesis of wurtzite ZnS nanowire bundles using a solvothermal technique. Phys. Chem. C 2007, 111, 12658–12662. [Google Scholar] [CrossRef]
- Shi, L.; Xu, Y.; Li, Q. Shape-selective synthesis and optical properties of highly ordered one-dimensional ZnS nanostructures. Cryst. Growth. Des. 2009, 9, 2214–2219. [Google Scholar] [CrossRef]
- Qian, G.; Huo, K.; Chu, P.K. Statistical theory of protein sequence design by random mutation. J. Phys. Chem. C 2009, 113, 5520–5527. [Google Scholar] [CrossRef]
- Rabenau, A. The role of hydrothermal synthesis in preparative chemistry. Angew. Chem. Int. Ed. 1985, 24, 1026–1040. [Google Scholar] [CrossRef]
- Walton, R.I. Subcritical solvothermal synthesis of condensed inorganic materials. Chem. Soc. Rev. 2002, 31, 230–238. [Google Scholar] [CrossRef]
- Murakoshi, K.; Hosokawa, H.; Tanaka, N.; Saito, M.; Wada, Y.; Sakata, T.; Mori, H.; Yanagida, S. Phase transition of ZnS nanocrystallites induced by surface modification at ambient temperature and pressure confirmed by electron diffraction. Chem. Commun. 1998, 3, 321–322. [Google Scholar]
- Wang, X.; Zhuang, J.; Peng, Q.; Li, Y.D. A simple chemical technique can be used to create a large family of high-quality nanocrystals. Nature 2005, 437, 121–124. [Google Scholar] [CrossRef]
- Zhao, J.G.; Zhang, H.H. Hydrothermal synthesis and characterization of ZnS hierarchical microspheres. Superlatt. Microstruct. 2012, 51, 663–667. [Google Scholar] [CrossRef]
- Joint Committee on Powder Diffraction Standards (JCPDS). Card no. 80-0007. Available online: http://jcpds.crystalstar.org/ (accessed on 6 September 2013).
- Maiti, U.N.; Nandy, S.; Karan, S.; Mallik, B.; Chattopadhyay, K.K. Effect of Ni doping on the dielectric constant of ZnO and its frequency dependent exchange interaction. Appl. Surf. Sci. 2008, 25, 7266–7271. [Google Scholar]
- Ramimoghadam, D.; Bin Hussein, M.Z.; Taufiq-Yap, Y.H. The effect of sodium dodecyl sulfate (SDS) and cetyltrimethylammonium bromide (CTAB) on the properties of ZnO synthesized by hydrothermal method. Int. J. Mol. Sci. 2012, 13, 13275–13293. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, J.C.; Ngo, C.; Prikhodko, S.; Kodambaka, S.; Li, J.L.; Richards, R. Gram-scale wet chemical synthesis of wurtzite-8H nanoporous ZnS spheres with high photocatalytic activity. Appl. Catal. B 2011, 106, 212–219. [Google Scholar]
- Ullah, M.H.; Kim, I.; Ha, C.S. pH selective synthesis of ZnS nanocrystals and their growth and photoluminescence. Mater. Lett. 2007, 61, 4267–4271. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ibupoto, Z.H.; Khun, K.; Liu, X.; Willander, M. Hydrothermal Synthesis of Nanoclusters of ZnS Comprised on Nanowires. Nanomaterials 2013, 3, 564-571. https://doi.org/10.3390/nano3030564
Ibupoto ZH, Khun K, Liu X, Willander M. Hydrothermal Synthesis of Nanoclusters of ZnS Comprised on Nanowires. Nanomaterials. 2013; 3(3):564-571. https://doi.org/10.3390/nano3030564
Chicago/Turabian StyleIbupoto, Zafar Hussain, Kimleang Khun, Xianjie Liu, and Magnus Willander. 2013. "Hydrothermal Synthesis of Nanoclusters of ZnS Comprised on Nanowires" Nanomaterials 3, no. 3: 564-571. https://doi.org/10.3390/nano3030564
APA StyleIbupoto, Z. H., Khun, K., Liu, X., & Willander, M. (2013). Hydrothermal Synthesis of Nanoclusters of ZnS Comprised on Nanowires. Nanomaterials, 3(3), 564-571. https://doi.org/10.3390/nano3030564




