Nanotreatment and Nanodiagnosis of Prostate Cancer: Recent Updates
Abstract
:1. Introduction
2. Nanomaterials: Applications in Treatment of Prostate Cancer
2.1. Mesoporous Silica Nanoparticles
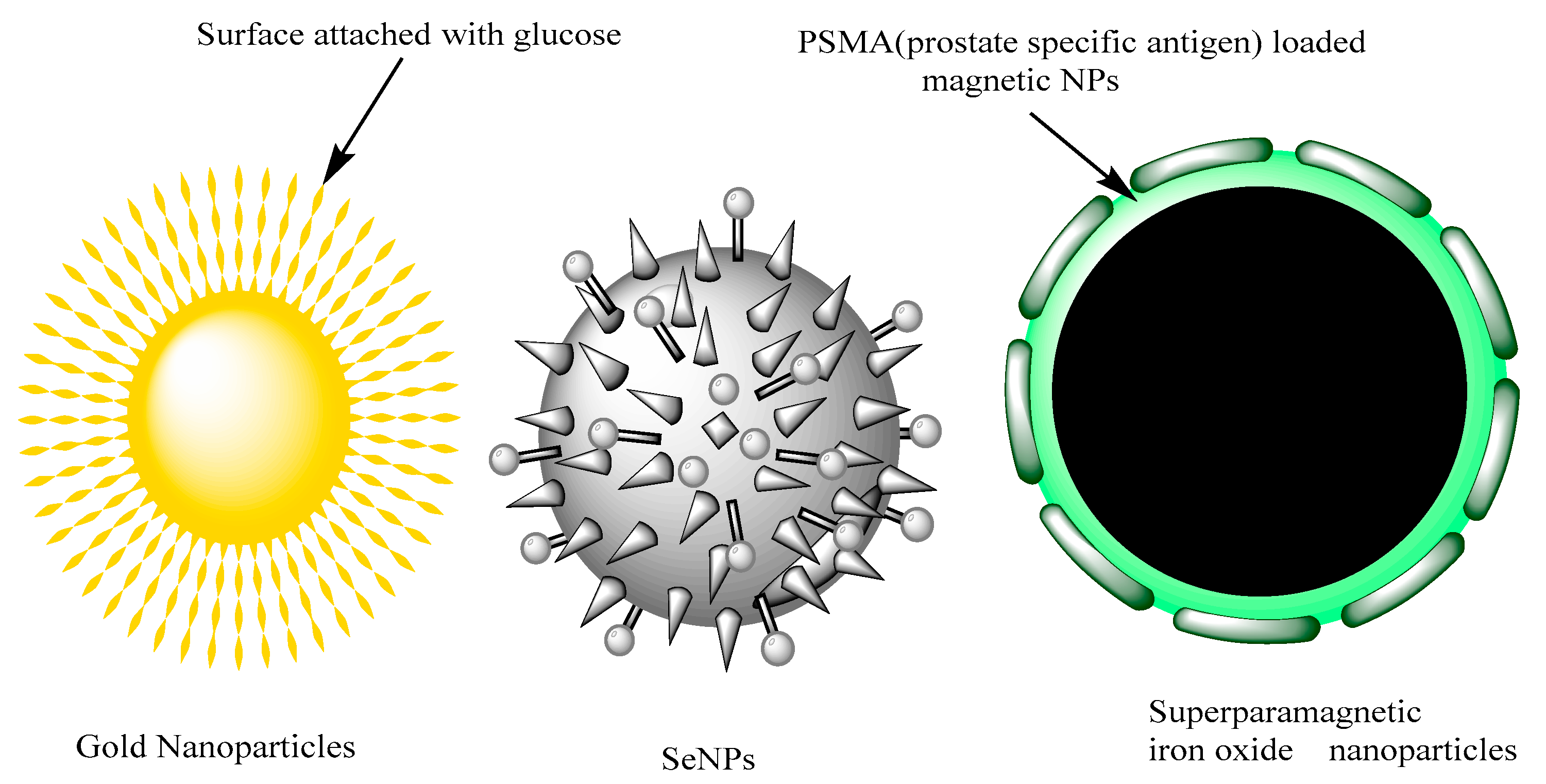
2.2. Selenium, Magnetic and Gold Nanoparticles
2.3. Quantum Dots
2.4. Carbon Nanotube
2.5. Polymeric Nanoparticles with Block Copolymers
2.6. Liposomes
2.7. Nanoemulsion
2.8. Niosomes
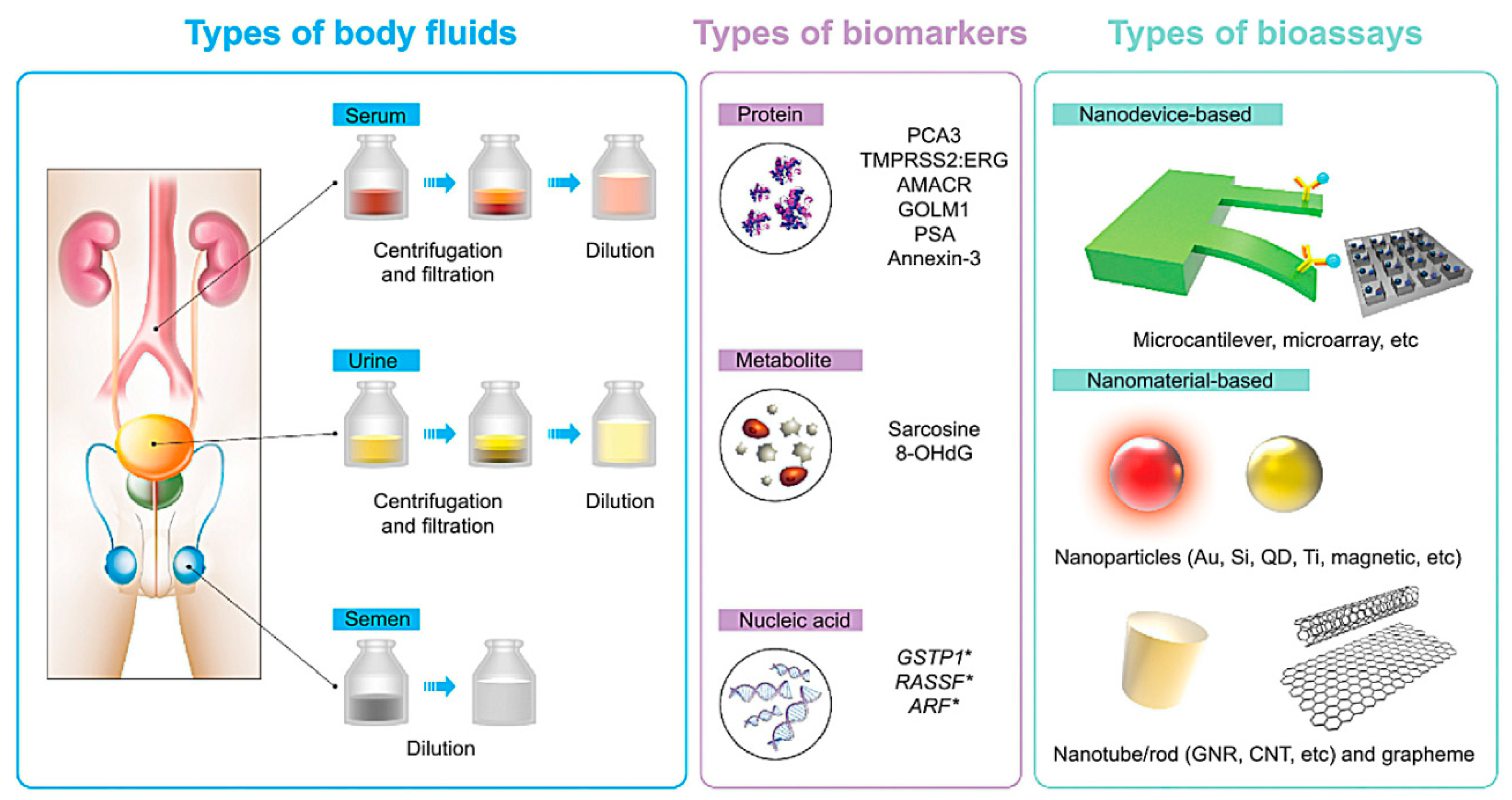
3. Nanomaterials towards Diagnosis and Biosensing of Prostate Cancer
3.1. Nanomaterials for Prostate Cancer Diagnosis and Biosensing
3.2. Magnetic Nanoparticles
3.3. Gold Nanoparticles
3.4. Quantum Dots
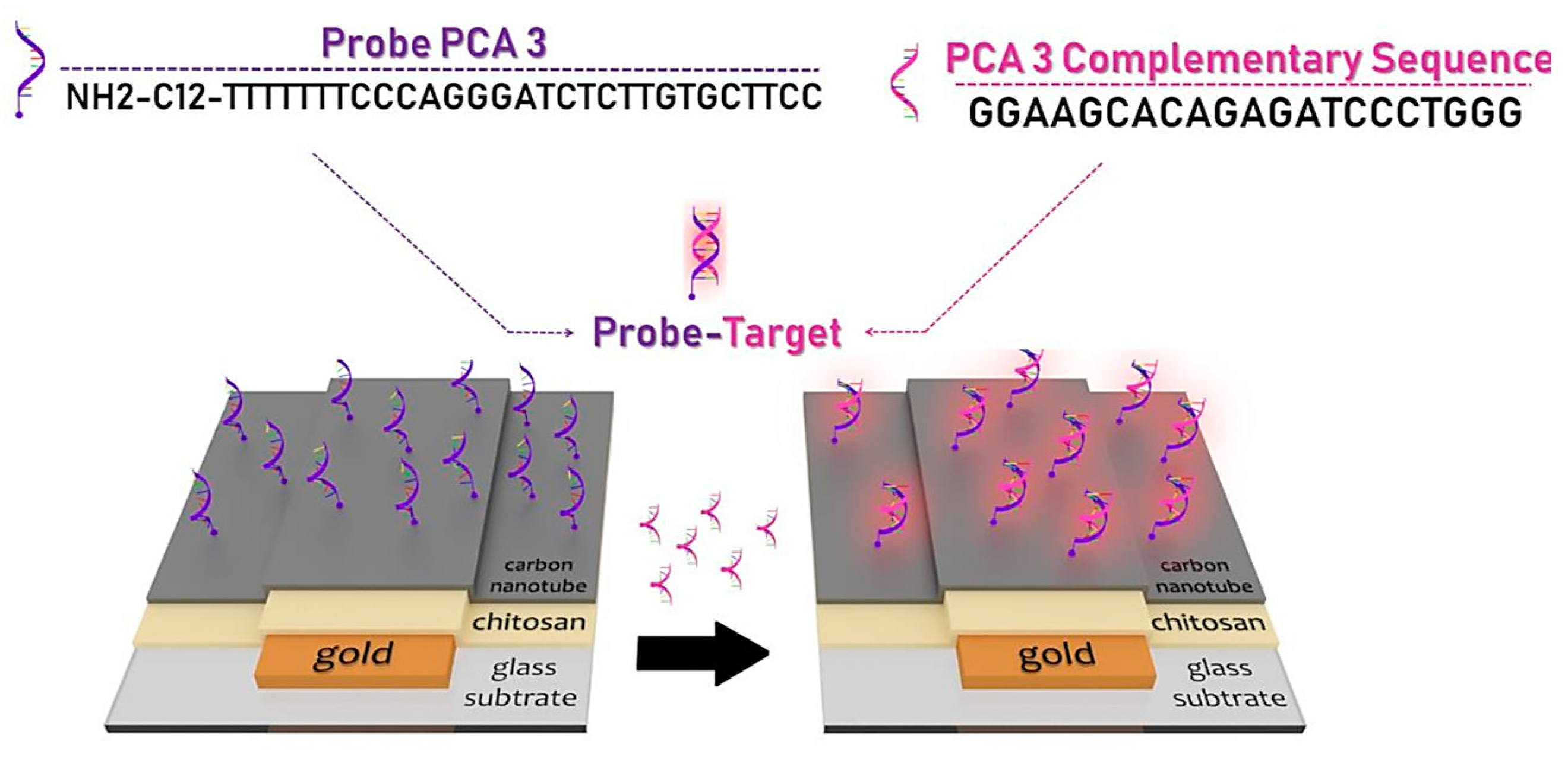
3.5. Carbon Nanotubes
3.6. Graphene
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Taitt, H.E. Global Trends and Prostate Cancer: A Review of Incidence, Detection, and Mortality as Influenced by Race, Ethnicity, and Geographic Location. Am. J. Men’s Health 2018, 12, 1807–1823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rawla, P. Epidemiology of prostate cancer. World J. Oncol. 2019, 10, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.; Huang, V.; Xu, X.; Livingstone, J.; Soares, F.; Jeon, J.; Zeng, Y.; Hua, J.T.; Petricca, J.; Guo, H.; et al. Widespread and Functional RNA Circularization in Localized Prostate Cancer. Cell 2019, 176, 831–843.e22. [Google Scholar] [CrossRef] [Green Version]
- Mei, W.; Lin, X.; Kapoor, A.; Gu, Y.; Zhao, K.; Tang, D. The Contributions of Prostate Cancer Stem Cells in Prostate Cancer Initiation and Metastasis. Cancers 2019, 11, 434. [Google Scholar] [CrossRef] [Green Version]
- Fujita, K.; Hayashi, T.; Matsushita, M.; Uemura, M.; Nonomura, N. Obesity, Inflammation, and Prostate Cancer. J. Clin. Med. 2019, 8, 201. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, P.P.; Banerjee, S.; Brown, T.R.; Zirkin, B.R. Androgen action in prostate function and disease. Am. J. Clin. Exp. Urol. 2018, 6, 62–77. [Google Scholar]
- Ejike, C.E.; Ezeanyika, L.U.S. Metabolic syndrome in sub-Saharan Africa: “smaller twin” of a region’s prostatic diseases? Int. Urol. Nephrol. 2008, 40, 909–920. [Google Scholar] [CrossRef]
- Chaurasia, V.; Pal, S.; Tiwari, B. Prediction of benign and malignant breast cancer using data mining techniques. J. Algorithms Comput. Technol. 2018, 12, 119–126. [Google Scholar] [CrossRef] [Green Version]
- Moradi, H.; Tang, S.; Salcudean, T. Toward Intra-Operative Prostate Photoacoustic Imaging: Configuration Evaluation and Implementation Using the da Vinci Research Kit. IEEE Trans. Med. Imaging 2018, 38, 57–68. [Google Scholar] [CrossRef]
- Calais, J.; Czernin, J.; Fendler, W.P.; Elashoff, D.; Nickols, N.G. Randomized prospective phase III trial of 68 Ga-PSMA-11 PET/CT molecular imaging for prostate cancer salvage radiotherapy planning [PSMA-SRT]. BMC Cancer 2019, 19, 1–11. [Google Scholar]
- Kishan, A.U.; Dang, A.; Katz, A.J.; Mantz, C.A.; Collins, S.P.; Aghdam, N.; Chu, F.-I.; Kaplan, I.D.; Appelbaum, L.; Fuller, D.B.; et al. Long-term Outcomes of Stereotactic Body Radiotherapy for Low-Risk and Intermediate-Risk Prostate Cancer. JAMA Netw. Open 2019, 2, e188006. [Google Scholar] [CrossRef] [Green Version]
- Stish, B.J.; Davis, B.J.; Mynderse, L.A.; McLaren, R.H.; Deufel, C.L.; Choo, R. Low dose rate prostate brachytherapy. Transl. Androl. Urol. 2018, 7, 341–356. [Google Scholar] [CrossRef]
- Zhang, J.; Agrawal, S.; Dangi, A.; Frings, N.; Kothapalli, S.-R. Photons Plus Ultrasound: Imaging and Sensing 2019. In Computer Assisted Photoacoustic Imaging Guided Device for Safer Percutaneous Needle Operations; International Society for Optics and Photonics: Bellingham, WA, USA, 2019; p. 1087866. [Google Scholar]
- Sabharwal, N.; Sharifi, N. HSD3B1 Genotypes Conferring Adrenal-Restrictive and Adrenal-Permissive Phenotypes in Prostate Cancer and Beyond. Endocrinology 2019, 160, 2180–2188. [Google Scholar] [CrossRef]
- Karpuz, M.; Gunay, M.S.; Ozer, A.Y. Liposomes and phytosomes for phytoconstituents. In Advances and Avenues in the Development of Novel Carriers for Bioactives and Biological Agents; Elsevier: Amsterdam, The Netherlands, 2020; pp. 525–553. [Google Scholar]
- Porter, C.M.; Shrestha, E.; Peiffer, L.B.; Sfanos, K.S. The microbiome in prostate inflammation and prostate cancer. Prostate Cancer Prostatic Dis. 2018, 21, 345–354. [Google Scholar] [CrossRef]
- Ozgur, E.; Gezer, U. Enzalutamide restores the testosterone effect on H19 expression in prostate cancer cells but not in exosomes. Ann. Med. Res. 2019, 26, 1056–1059. [Google Scholar] [CrossRef]
- Dobbs, R.W.; Malhotra, N.R.; Greenwald, D.T.; Wang, A.Y.; Prins, G.S.; Abern, M.R. Estrogens and prostate cancer. Prostate Cancer Prostatic Dis. 2018, 22, 185–194. [Google Scholar] [CrossRef]
- Selvi, I.; Basar, H. Subcapsular orchiectomy versus total orchiectomy and LHRH analogue in the treatment of hormone-sensitive metastatic prostate cancer: A different perspective in evaluation of the psychosocial effects. Support. Care Cancer 2020, 1–14. [Google Scholar] [CrossRef]
- Puca, L.; Bareja, R.; Prandi, D.; Shaw, R.; Benelli, M.; Karthaus, W.R.; Hess, J.; Sigouros, M.; Donoghue, A.; Kossai, M.; et al. Patient derived organoids to model rare prostate cancer phenotypes. Nat. Commun. 2018, 9, 2404. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.-H.; Kantoff, P. Treatment of Metastatic Prostate Cancer in 2018. JAMA Oncol. 2019, 5, 263–264. [Google Scholar] [CrossRef]
- Oun, R.; Moussa, Y.; Wheate, N.J. The side effects of platinum-based chemotherapy drugs: A review for chemists. Dalton Trans. 2018, 47, 6645–6653. [Google Scholar] [CrossRef]
- Bax, C.; Taverna, G.; Eusebio, L.; Sironi, S.; Grizzi, F.; Guazzoni, G.; Capelli, L. Innovative Diagnostic Methods for Early Prostate Cancer Detection through Urine Analysis: A Review. Cancers 2018, 10, 123. [Google Scholar] [CrossRef] [Green Version]
- Kasivisvanathan, V.; Rannikko, A.; Borghi, M.; Panebianco, V.; Mynderse, L.A.; Vaarala, M.H.; Briganti, A.; Budäus, L.; Hellawell, G.; Hindley, R.G.; et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis. N. Engl. J. Med. 2018, 378, 1767–1777. [Google Scholar] [CrossRef]
- Espinoza, S.M.; Patil, H.I.; Martinez, E.S.M.; Pimentel, R.C.; Ige, P.P. Poly-ε-caprolactone (PCL), a promising polymer for pharmaceutical and biomedical applications: Focus on nanomedicine in cancer. Int. J. Polym. Mater. 2019, 69, 85–126. [Google Scholar] [CrossRef]
- Wakaskar, R.R. Promising effects of nanomedicine in cancer drug delivery. J. Drug Target. 2017, 26, 319–324. [Google Scholar] [CrossRef]
- Wu, L.-P.; Wang, D.; Li, Z.; Lin-Ping, W. Grand challenges in nanomedicine. Mater. Sci. Eng. C 2020, 106, 110302. [Google Scholar] [CrossRef]
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Adeli-Sardou, M. Evaluation of Carum-loaded Niosomes on Breast Cancer Cells: Physicochemical Properties, In Vitro Cytotoxicity, Flow Cytometric, DNA Fragmentation and Cell Migration Assay. Sci. Rep. 2019, 9, 7139. [Google Scholar] [CrossRef] [Green Version]
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Lohrasbi-Nejad, A.; Nematollahi, M.H. A new formulation of hydrophobin-coated niosome as a drug carrier to cancer cells. Mater. Sci. Eng. C 2020, 113, 110975. [Google Scholar] [CrossRef]
- Öztürk-Atar, K.; Eroğlu, H.; Çalış, S. Novel advances in targeted drug delivery. J. Drug Target. 2017, 26, 633–642. [Google Scholar] [CrossRef]
- Alavi, M.; Hamidi, M. Passive and active targeting in cancer therapy by liposomes and lipid nanoparticles. Drug Metab. Pers. Ther. 2019, 34, 20180032. [Google Scholar] [CrossRef]
- Marquardt, S.; Solanki, M.; Spitschak, A.; Vera, J.; Puetzer, B.M. Seminars in cancer biology. In Emerging Functional Markers for Cancer Stem Cell-Based Therapies: Understanding Signaling Networks for Targeting Metastasis; Elsevier: Amsterdam, The Netherlands, 2018; pp. 90–109. [Google Scholar]
- Bennett, C.H.; Brassard, G. Quantum cryptography: Public key distribution and coin tossing. In Proceedings of the International Conference on Computers, Systems and Signal Processing, Bangalore, India, 10–12 December 1984. [Google Scholar]
- Barani, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Nematollahi, M.H. Lawsone-loaded Niosome and its antitumor activity in MCF-7 breast Cancer cell line: A Nano-herbal treatment for Cancer. DARU J. Pharm. Sci. 2018, 26, 11–17. [Google Scholar] [CrossRef]
- Barani, M.; Nematollahi, M.H.; Zaboli, M.; Mirzaei, M.; Torkzadeh-Mahani, M.; Pardakhty, A.; Karam, G.A. In silico and in vitro study of magnetic niosomes for gene delivery: The effect of ergosterol and cholesterol. Mater. Sci. Eng. C 2019, 94, 234–246. [Google Scholar] [CrossRef]
- Hajizadeh, M.R.; Parvaz, N.; Barani, M.; Khoshdel, A.; Fahmidehkar, M.A.; Mahmoodi, M.; Torkzadeh-Mahani, M. Diosgenin-loaded niosome as an effective phytochemical nanocarrier: Physicochemical characterization, loading efficiency, and cytotoxicity assay. DARU J. Pharm. Sci. 2019, 27, 329–339. [Google Scholar] [CrossRef]
- Cui, J.; Björnmalm, M.; Ju, Y.; Caruso, F. Nanoengineering of Poly(ethylene glycol) Particles for Stealth and Targeting. Langmuir 2018, 34, 10817–10827. [Google Scholar] [CrossRef]
- Rosenblum, D.; Joshi, N.; Tao, W.; Karp, J.M.; Peer, D. Progress and challenges towards targeted delivery of cancer therapeutics. Nat. Commun. 2018, 9, 1410. [Google Scholar] [CrossRef] [Green Version]
- Peng, L.; Liu, D.; Cheng, H.; Zhou, S.; Zu, M. A Multilayer Film Based Selective Thermal Emitter for Infrared Stealth Technology. Adv. Opt. Mater. 2018, 6, 1801006. [Google Scholar] [CrossRef]
- Huckaby, J.T.; Lai, S.K. PEGylation for enhancing nanoparticle diffusion in mucus. Adv. Drug Deliv. Rev. 2018, 124, 125–139. [Google Scholar] [CrossRef]
- Ghimici, L.; Nichifor, M. Dextran derivatives application as flocculants. Carbohydr. Polym. 2018, 190, 162–174. [Google Scholar] [CrossRef]
- Adjei, I.M.; Temples, M.N.; Brown, S.B.; Sharma, B. Targeted Nanomedicine to Treat Bone Metastasis. Pharmaceutics 2018, 10, 205. [Google Scholar] [CrossRef] [Green Version]
- Cifuentes-Rius, A.; Butler, L.M.; Voelcker, N.H. Precision nanomedicines for prostate cancer. Nanomedicine 2018, 13, 803–807. [Google Scholar] [CrossRef] [Green Version]
- Kyzas, G.Z.; Matis, K.A. Electroflotation process: A review. J. Mol. Liq. 2016, 220, 657–664. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Bomis, G.; Kosheleva, R.I.; Efthimiadou, E.K.; Favvas, E.P.; Kostoglou, M.; Mitropoulos, A.C. Nanobubbles effect on heavy metal ions adsorption by activated carbon. Chem. Eng. J. 2019, 356, 91–97. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Kyzas, G.Z. Composts as Biosorbents for Decontamination of Various Pollutants: A Review. Water Air Soil Pollut. 2015, 226, 61. [Google Scholar] [CrossRef]
- He, Z.; Cheng, X.; Kyzas, G.Z.; Fu, J. Pharmaceuticals pollution of aquaculture and its management in China. J. Mol. Liq. 2016, 223, 781–789. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Lazaridis, N.K.; Kostoglou, M. Adsorption/desorption of a dye by a chitosan derivative: Experiments and phenomenological modeling. Chem. Eng. J. 2014, 248, 327–336. [Google Scholar] [CrossRef]
- Deliyanni, E.A.; Kyzas, G.Z.; Matis, K.A. Various flotation techniques for metal ions removal. J. Mol. Liq. 2017, 225, 260–264. [Google Scholar] [CrossRef]
- Ganipineni, L.P.; Danhier, F.; Préat, V. Drug delivery challenges and future of chemotherapeutic nanomedicine for glioblastoma treatment. J. Control. Release 2018, 281, 42–57. [Google Scholar] [CrossRef]
- Contreras-Cáceres, R.; Cabeza, L.; Perazzoli, G.; Díaz, A.; Lopez-Romero, J.M.; Melguizo, C.; Prados, J. Electrospun Nanofibers: Recent Applications in Drug Delivery and Cancer Therapy. Nanomaterials 2019, 9, 656. [Google Scholar] [CrossRef] [Green Version]
- Dziawer, Ł.; Majkowska-Pilip, A.; Gawel, D.; Godlewska, M.; Pruszynski, M.; Jastrzębski, J.; Wąs, B.; Bilewicz, A. Trastuzumab-Modified Gold Nanoparticles Labeled with 211At as a Prospective Tool for Local Treatment of HER2-Positive Breast Cancer. Nanomaterials 2019, 9, 632. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Pinel, B.; Porras-Alcalá, C.; Rodríguez, A.O.; Sarabia, F.; Prados, J.; Melguizo, C.; Lopez-Romero, J.M. Lipid-Based Nanoparticles: Application and Recent Advances in Cancer Treatment. Nanomaterials 2019, 9, 638. [Google Scholar] [CrossRef] [Green Version]
- Gu, H.; Tang, H.; Xiong, P.; Zhou, Z. Biomarkers-based Biosensing and Bioimaging with Graphene for Cancer Diagnosis. Nanomaterials 2019, 9, 130. [Google Scholar] [CrossRef] [Green Version]
- Gurunathan, S.; Qasim, M.; Park, C.H.; Iqbal, M.A.; Yoo, H.; Hwang, J.H.; Uhm, S.J.; Song, H.; Seo, H.G.; Choi, Y.S.; et al. Cytotoxicity and Transcriptomic Analyses of Biogenic Palladium Nanoparticles in Human Ovarian Cancer Cells (SKOV3). Nanomaterials 2019, 9, 787. [Google Scholar] [CrossRef] [Green Version]
- Iannazzo, D.; Pistone, A.; Celesti, C.; Triolo, C.; Patanè, S.; Giofrè, S.V.; Romeo, R.; Ziccarelli, I.; Mancuso, R.; Gabriele, B.; et al. A Smart Nanovector for Cancer Targeted Drug Delivery Based on Graphene Quantum Dots. Nanomaterials 2019, 9, 282. [Google Scholar] [CrossRef] [Green Version]
- Ranathunge, T.A.; Karunaratne, D.; Rajapakse, R.G.; Watkins, D.L. Doxorubicin Loaded Magnesium Oxide Nanoflakes as pH Dependent Carriers for Simultaneous Treatment of Cancer and Hypomagnesemia. Nanomaterials 2019, 9, 208. [Google Scholar] [CrossRef] [Green Version]
- Wu, P.-T.; Lin, C.-L.; Lin, C.-W.; Chang, N.-C.; Tsai, W.-B.; Yu, J. Methylene-Blue-Encapsulated Liposomes as Photodynamic Therapy Nano Agents for Breast Cancer Cells. Nanomaterials 2018, 9, 14. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Wang, Y.; Zhao, Y.; Liu, H.; Zhao, Y.; Li, X.; Lin, Q. Biodegradable Micelles for NIR/GSH-Triggered Chemophototherapy of Cancer. Nanomaterials 2019, 9, 91. [Google Scholar] [CrossRef] [Green Version]
- Astolfi, M.; Rispoli, G.; Anania, G.; Nevoso, V.; Artioli, E.; Landini, N.; Benedusi, M.; Melloni, E.; Secchiero, P.; Tisato, V.; et al. Colorectal Cancer Study with Nanostructured Sensors: Tumor Marker Screening of Patient Biopsies. Nanomaterials 2020, 10, 606. [Google Scholar] [CrossRef] [Green Version]
- Borg, H.; Zámbó, D.; Elmansi, H.; Hashem, H.M.; Nasr, J.J.M.; Walash, M.I.; Bigall, N.C.; Belal, F. Preconcentration and Detection of Gefitinib Anti-Cancer Drug Traces from Water and Human Plasma Samples by Means of Magnetic Nanoparticles. Nanomaterials 2020, 10, 1196. [Google Scholar] [CrossRef]
- Chantada-Vázquez, M.D.P.; García-Vence, M.; Vázquez-Estévez, S.; Bravo, S.B.; Núñez, C. Identification of a Profile of Neutrophil-Derived Granule Proteins in the Surface of Gold Nanoparticles after Their Interaction with Human Breast Cancer Sera. Nanomaterials 2020, 10, 1223. [Google Scholar] [CrossRef]
- De Matteis, V.; Rizzello, L.; Cascione, M.; Liatsi-Douvitsa, E.; Apriceno, A.; Rinaldi, R. Green Plasmonic Nanoparticles and Bio-Inspired Stimuli-Responsive Vesicles in Cancer Therapy Application. Nanomaterials 2020, 10, 1083. [Google Scholar] [CrossRef]
- Haque, S.T.; Karim, E.; Abidin, S.A.Z.; Othman, I.; Holl, M.M.B.; Chowdhury, E.H. Fe/Mg-Modified Carbonate Apatite with Uniform Particle Size and Unique Transport Protein-Related Protein Corona Efficiently Delivers Doxorubicin into Breast Cancer Cells. Nanomaterials 2020, 10, 834. [Google Scholar] [CrossRef]
- Knights, O.; Freear, S.; McLaughlan, J.R. Improving Plasmonic Photothermal Therapy of Lung Cancer Cells with Anti-EGFR Targeted Gold Nanorods. Nanomaterials 2020, 10, 1307. [Google Scholar] [CrossRef]
- Miao, T.; Little, A.C.; Aronshtam, A.; Marquis, T.; Fenn, S.L.; Hristova, M.; Krementsov, D.N.; van der Vliet, A.; Spees, J.L.; Oldinski, R.A. Internalized FGF-2-loaded nanoparticles increase nuclear ERK1/2 content and result in lung cancer cell death. Nanomaterials 2020, 10, 612. [Google Scholar] [CrossRef] [Green Version]
- Pennetta, C.; Floresta, G.; Graziano, A.C.E.; Cardile, V.; Rubino, L.; Galimberti, M.; Rescifina, A.; Barbera, V. Functionalization of Single and Multi-Walled Carbon Nanotubes with Polypropylene Glycol Decorated Pyrrole for the Development of Doxorubicin Nano-Conveyors for Cancer Drug Delivery. Nanomaterials 2020, 10, 1073. [Google Scholar] [CrossRef]
- Piehler, S.; Dähring, H.; Grandke, J.; Göring, J.; Couleaud, P.; Aires, A.; Cortajarena, A.L.; Courty, J.; Latorre, A.; Somoza, Á.; et al. Iron Oxide Nanoparticles as Carriers for DOX and Magnetic Hyperthermia after Intratumoral Application into Breast Cancer in Mice: Impact and Future Perspectives. Nanomaterials 2020, 10, 1016. [Google Scholar] [CrossRef]
- Reczyńska, K.; Marszałek, M.; Zarzycki, A.; Reczyński, W.; Kornaus, K.; Pamuła, E.; Chrzanowski, W. Superparamagnetic Iron Oxide Nanoparticles Modified with Silica Layers as Potential Agents for Lung Cancer Treatment. Nanomaterials 2020, 10, 1076. [Google Scholar] [CrossRef]
- Nicolosi, P.; Ledet, E.; Yang, S.; Michalski, S.; Freschi, B.; O’Leary, E.; Esplin, E.D.; Nussbaum, R.L.; Sartor, O. Prevalence of Germline Variants in Prostate Cancer and Implications for Current Genetic Testing Guidelines. JAMA Oncol. 2019, 5, 523–528. [Google Scholar] [CrossRef] [Green Version]
- Koo, K.; Mainwaring, P.N.; Tomlins, S.A.; Trau, M. Merging new-age biomarkers and nanodiagnostics for precision prostate cancer management. Nat. Rev. Urol. 2019, 16, 302–317. [Google Scholar] [CrossRef]
- Junqueira, H.; Quinn, T.A.; Biringer, R.; Hussein, M.; Smeriglio, C.; Barrueto, L.; Finizio, J.; Huang, X.Y. “Michelle” Accuracy of Canine Scent Detection of Non-Small Cell Lung Cancer in Blood Serum. J. Am. Osteopat. Assoc. 2019, 119, 413. [Google Scholar] [CrossRef] [Green Version]
- Bhana, S.; Wang, Y.; Huang, X. Nanotechnology for enrichment and detection of circulating tumor cells. Nanomedicine 2015, 10, 1973–1990. [Google Scholar] [CrossRef]
- Fung, Y. Identification and Sensing of Biomarkers for Early Diagnosis of Prostate Cancer. Ph.D. Thesis, Hong Kong Baptist University, Hong Kong, China, 30 August 2019. [Google Scholar]
- He, M.-H.; Chen, L.; Zheng, T.; Tu, Y.; He, Q.; Fu, H.-L.; Lin, J.-C.; Zhang, W.; Shu, G.; He, L.; et al. Potential Applications of Nanotechnology in Urological Cancer. Front. Pharmacol. 2018, 9, 745. [Google Scholar] [CrossRef] [Green Version]
- Yallapu, M.M.; Khan, S.; Maher, D.M.; Ebeling, M.C.; Sundram, V.; Chauhan, N.; Ganju, A.; Balakrishna, S.; Gupta, B.K.; Zafar, N.; et al. Anti-cancer activity of curcumin loaded nanoparticles in prostate cancer. Biomaterials 2014, 35, 8635–8648. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.; Gill, A.A.; Nlooto, M.; Karpoormath, R. Prostate cancer biomarkers detection using nanoparticles based electrochemical biosensors. Biosens. Bioelectron. 2019, 137, 213–221. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; You, X.; Xian, T.; Wu, J.; Pang, J. Nanoparticle Therapy for Prostate Cancer: Overview and Perspectives. Curr. Top. Med. Chem. 2019, 19, 57–73. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, L.; Wu, H. Nanomaterials for cancer therapies. Nanotechnol. Rev. 2017, 6, 473–496. [Google Scholar] [CrossRef]
- Chaudhary, Z.; Subramaniam, S.; Khan, G.M.; Abeer, M.M.; Qu, Z.; Janjua, T.; Kumeria, T.; Batra, J.; Popat, A. Encapsulation and Controlled Release of Resveratrol within Functionalized Mesoporous Silica Nanoparticles for Prostate Cancer Therapy. Front. Bioeng. Biotechnol. 2019, 7, 225. [Google Scholar] [CrossRef]
- Gu, C.; Li, C.; Zhang, J.; Li, X.; Wang, L.; Ju, Y.; Liu, Y.; Xu, Y. Ultra-effective near-infrared Photothermal therapy for the prostate cancer Nursing care through novel intended and surface tailored photo-responsive Ga-Au@MPS nanovesicles. J. Photochem. Photobiol. B Biol. 2020, 202, 111685. [Google Scholar] [CrossRef]
- Liu, C.-M.; Chen, G.-B.; Chen, H.-H.; Zhang, J.-B.; Li, H.-Z.; Sheng, M.-X.; Weng, W.-B.; Guo, S.-M. Cancer cell membrane-cloaked mesoporous silica nanoparticles with a pH-sensitive gatekeeper for cancer treatment. Colloids Surfaces B Biointerfaces 2018, 175, 477–486. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, Y.; Yu, H.; Wu, D.; Ma, H.; Li, H.; Du, B.; Wei, Q. Label-free electrochemical immunosensor for prostate-specific antigen based on silver hybridized mesoporous silica nanoparticles. Anal. Biochem. 2013, 434, 123–127. [Google Scholar] [CrossRef]
- Badr, G.; Al-Sadoon, M.K.; Rabah, D.; Sayed, D. Snake (Walterinnesia aegyptia) venom-loaded silica nanoparticles induce apoptosis and growth arrest in human prostate cancer cells. Apoptosis 2012, 18, 300–314. [Google Scholar] [CrossRef]
- Sanna, V.; Sechi, M. Nanoparticle therapeutics for prostate cancer treatment. Maturitas 2012, 73, 27–32. [Google Scholar] [CrossRef]
- Chen, J.; Hu, C.; Niestroj, M.; Yuan, D.; Chang, S. Treating cancer stem cells and cancer metastasis using glucose-coated gold nanoparticles. Int. J. Nanomed. 2015, 10, 2065–2077. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Xing, J.Z.; Chen, J.; Ko, L.; Amanie, J.; Gulavita, S.; Pervez, N.; Yee, D.; Moore, R.B.; Roa, W. Enhanced radiation sensitivity in prostate cancer by gold-nanoparticles. Clin. Investig. Med. 2008, 31, E160–E167. [Google Scholar] [CrossRef] [Green Version]
- Rastinehad, A.; Anastos, H.; Wajswol, E.; Winoker, J.S.; Sfakianos, J.P.; Doppalapudi, S.K.; Carrick, M.R.; Knauer, C.J.; Taouli, B.; Lewis, S.C.; et al. Gold nanoshell-localized photothermal ablation of prostate tumors in a clinical pilot device study. Proc. Natl. Acad. Sci. USA 2019, 116, 18590–18596. [Google Scholar] [CrossRef] [Green Version]
- Shukla, R.; Chanda, N.; Zambre, A.; Upendran, A.; Katti, K.; Kulkarni, R.R.; Nune, S.K.; Casteel, S.W.; Smith, C.J.; Vimal, J.; et al. Laminin receptor specific therapeutic gold nanoparticles (198AuNP-EGCg) show efficacy in treating prostate cancer. Proc. Natl. Acad. Sci. USA 2012, 109, 12426–12431. [Google Scholar] [CrossRef] [Green Version]
- Liao, G.; Tang, J.; Wang, D.; Zuo, H.; Zhang, Q.; Liu, Y.; Xiong, H. Selenium nanoparticles (SeNPs) have potent antitumor activity against prostate cancer cells through the upregulation of miR-16. World J. Surg. Oncol. 2020, 18, 1–11. [Google Scholar] [CrossRef]
- Xia, Y.; Zhong, J.; Zhao, M.; Tang, Y.; Han, N.; Hua, L.; Xu, T.; Wang, C.; Zhu, B. Galactose-modified selenium nanoparticles for targeted delivery of doxorubicin to hepatocellular carcinoma. Drug Deliv. 2019, 26, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Sonkusre, P. Specificity of Biogenic Selenium Nanoparticles for Prostate Cancer Therapy with Reduced Risk of Toxicity: An in vitro and in vivo Study. Front. Oncol. 2020, 9, 1541. [Google Scholar] [CrossRef] [Green Version]
- Khurana, A.; Tekula, S.; Saifi, M.A.; Venkatesh, P.; Godugu, C. Therapeutic applications of selenium nanoparticles. Biomed. Pharmacother. 2019, 111, 802–812. [Google Scholar] [CrossRef]
- Yamkamon, V.; Htoo, K.P.P.; Yainoy, S.; Suksrichavalit, T.; Tangchaikeeree, T.; Eiamphungporn, W. Urinary PCA3 detection in prostate cancer by magnetic nanoparticles coupled with colorimetric enzyme-linked oligonucleotide assay. EXCLI J. 2020, 19, 501–513. [Google Scholar]
- Khramtsov, P.V.; Kropaneva, M.; Bochkova, M.; Timganova, V.; Zamorina, S.; Rayev, M. Solid-phase nuclear magnetic resonance immunoassay for the prostate-specific antigen by using protein-coated magnetic nanoparticles. Microchim. Acta 2019, 186, 768. [Google Scholar] [CrossRef]
- Wang, H.-M.; Huang, X.-Q.; Wang, A.-J.; Luo, X.; Liu, W.-D.; Yuan, P.-X.; Feng, J.-J. Construction of efficient “on-off-on” fluorescence aptasensor for ultrasensitive detection of prostate specific antigen via covalent energy transfer between g-C3N4 quantum dots and palladium triangular plates. Anal. Chim. Acta 2020, 1104, 53–59. [Google Scholar] [CrossRef]
- Garcia-Cortes, M.; Encinar, J.R.; Costa-Fernandez, J.M.; Sanz-Medel, A. Highly sensitive nanoparticle-based immunoassays with elemental detection: Application to Prostate-Specific Antigen quantification. Biosens. Bioelectron. 2016, 85, 128–134. [Google Scholar] [CrossRef]
- Rastogi, V.; Yadav, P.; Bhattacharya, S.S.; Mishra, A.K.; Verma, N.; Verma, A.; Pandit, J.K. Carbon Nanotubes: An Emerging Drug Carrier for Targeting Cancer Cells. J. Drug Deliv. 2014, 2014, 1–23. [Google Scholar] [CrossRef] [Green Version]
- Dai, P.; Liu, C.; Xie, C.; Ke, J.; He, Y.; Wei, L.; Chen, L.; Jin, J. TiO2 nanotubes loaded with CdS nanocrystals as enhanced emitters of electrochemiluminescence: Application to an assay for prostate-specific antigen. Anal. Bioanal. Chem. 2020, 412, 1375–1384. [Google Scholar] [CrossRef]
- Cai, J.; Deng, W.; Tan, Y.; Xie, Q. Electrocatalytic activity of Co3O4 quantum dots supported on aminated carbon nanotubes and their application for sensitive electrochemical immunosensing of prostate-specific antigen. J. Electroanal. Chem. 2020, 862, 114023. [Google Scholar] [CrossRef]
- Sanna, V.; Roggio, A.M.; Posadino, A.M.; Cossu, A.; Marceddu, S.; Mariani, A.; Alzari, V.; Uzzau, S.; Pintus, G.; Sechi, M. Novel docetaxel-loaded nanoparticles based on poly(lactide-co-caprolactone) and poly(lactide-co-glycolide-co-caprolactone) for prostate cancer treatment: Formulation, characterization, and cytotoxicity studies. Nanoscale Res. Lett. 2011, 6, 260. [Google Scholar] [CrossRef] [Green Version]
- Farokhzad, O.C.; Karp, J.M.; Langer, R. Nanoparticle–aptamer bioconjugates for cancer targeting. Expert Opin. Drug Deliv. 2006, 3, 311–324. [Google Scholar] [CrossRef]
- Kamaly, N.; Fredman, G.; Subramanian, M.; Gadde, S.; Pesic, A.; Cheung, L.; Fayad, Z.A.; Langer, R.; Tabas, I.; Farokhzad, O.C. Development and in vivo efficacy of targeted polymeric inflammation-resolving nanoparticles. Proc. Natl. Acad. Sci. USA 2013, 110, 6506–6511. [Google Scholar] [CrossRef] [Green Version]
- Mondal, S.; Adhikari, N.; Banerjee, S.; Amin, S.A.; Jha, T. Matrix metalloproteinase-9 (MMP-9) and its inhibitors in cancer: A minireview. Eur. J. Med. Chem. 2020, 194, 112260. [Google Scholar] [CrossRef]
- Dhar, S.; Gu, F.X.; Langer, R.; Farokhzad, O.C.; Lippard, S.J. Targeted delivery of cisplatin to prostate cancer cells by aptamer functionalized Pt(IV) prodrug-PLGA-PEG nanoparticles. Proc. Natl. Acad. Sci. USA 2008, 105, 17356–17361. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, R.; Tyagi, P.; Li, S.; Huang, L. Anisamide-targeted stealth liposomes: A potent carrier for targeting doxorubicin to human prostate cancer cells. Int. J. Cancer 2004, 112, 693–700. [Google Scholar] [CrossRef]
- Trojan, L.; Bode, C.; Weiss, C.; Kraenzlin, B.; Michaelis, U.; Teifel, M.; Alken, P.; Michel, M.S. Paclitaxel encapsulated in cationic liposomes: A new option for neovascular targeting for the treatment of prostate cancer. Oncol. Rep. 2009, 22, 321–326. [Google Scholar] [CrossRef] [Green Version]
- Narayanan, N.K.; Nargi, D.; Randolph, C.; Narayanan, B.A. Liposome encapsulation of curcumin and resveratrol in combination reduces prostate cancer incidence in PTEN knockout mice. Int. J. Cancer 2009, 125, 1–8. [Google Scholar] [CrossRef]
- Goldstein, D.; Gofrit, O.; Nyska, A.; Benita, S. Anti-HER2 Cationic Immunoemulsion as a Potential Targeted Drug Delivery System for the Treatment of Prostate Cancer. Cancer Res. 2007, 67, 269–275. [Google Scholar] [CrossRef] [Green Version]
- Morris, M.J.; Reuter, V.E.; Kelly, W.K.; Slovin, S.F.; Kenneson, K.; Verbel, D.; Osman, I.; Scher, H.I. HER-2 profiling and targeting in prostate carcinoma. Cancer 2002, 94, 980–986. [Google Scholar] [CrossRef]
- Wang, J.; Sui, M.; Fan, W. Nanoparticles for tumor targeted therapies and their pharmacokinetics. Curr. Drug Metab. 2010, 11, 129–141. [Google Scholar] [CrossRef] [Green Version]
- Chen, B.-H.; Tsai, Y.-J. Preparation of catechin extracts and nanoemulsions from green tea leaf waste and their inhibition effect on prostate cancer cell PC-3. Int. J. Nanomed. 2016, 11, 1907–1926. [Google Scholar] [CrossRef] [Green Version]
- Panda, P.K.; Saraf, S.; Tiwari, A.; Verma, A.; Raikwar, S.; Jain, A.; Jain, S.K. Novel Strategies for Targeting Prostate Cancer. Curr. Drug Deliv. 2019, 16, 712–727. [Google Scholar] [CrossRef]
- Guan, Y.-B.; Zhou, S.-Y.; Zhang, Y.-Q.; Wang, J.-L.; Tian, Y.-D.; Jia, Y.-Y.; Sun, Y.-J. Therapeutic effects of curcumin nanoemulsions on prostate cancer. Acta Acad. Med. Wuhan 2017, 37, 371–378. [Google Scholar] [CrossRef]
- El-Mahdy, M.M.; Hassan, A.S.; El-Badry, M.; El-Gindy, G.E.-D.A. Performance of Curcumin in Nanosized Carriers Niosomes and Ethosomes as Potential Anti-Inflammatory Delivery System for Topical Application. Bull. Pharm. Sci. Assiut 2020, 43, 105–122. [Google Scholar] [CrossRef]
- Akbarzadeh, I.; Yaraki, M.T.; Bourbour, M.; Noorbazargan, H.; Lajevardi, A.; Shilsar, S.M.S.; Heidari, F.; Mousavian, S.M. Optimized doxycycline-loaded niosomal formulation for treatment of infection-associated prostate cancer: An in-vitro investigation. J. Drug Deliv. Sci. Technol. 2020, 57, 101715. [Google Scholar] [CrossRef]
- Fletcher, R.H. Guideline: Experts recommend against prostate cancer screening with prostate-specific antigen test. Ann. Intern. Med. 2019, 170, JC2. [Google Scholar] [CrossRef]
- Pinsky, P.F.; Miller, E.A.; Prorok, P.; Grubb, R.; Crawford, E.; Andriole, G. Extended follow-up for prostate cancer incidence and mortality in the PLCO randomized cancer screening trial. BJU Int. 2019, 123, 854. [Google Scholar] [CrossRef]
- Abida, W.; Cyrta, J.; Heller, G.; Prandi, D.; Armenia, J.; Coleman, I.; Cieslik, M.; Benelli, M.; Robinson, D.; Van Allen, E.M.; et al. Genomic correlates of clinical outcome in advanced prostate cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 11428–11436. [Google Scholar] [CrossRef] [Green Version]
- Gao, R.; Cheng, Z.; Wang, X.; Yu, L.; Guo, Z.; Zhao, G.; Choo, J. Simultaneous immunoassays of dual prostate cancer markers using a SERS-based microdroplet channel. Biosens. Bioelectron. 2018, 119, 126–133. [Google Scholar] [CrossRef]
- Lee, J.R.; Ooi, C.; Wang, S.X. In Vitro Cancer Diagnostics. In Bioanalysis; Springer Science and Business Media LLC: Berlin, Germany, 2018; pp. 109–132. [Google Scholar]
- Cahill, L.C.; Wu, Y.; Yoshitake, T.; Ponchiardi, C.; Giacomelli, M.G.; Wagner, A.A.; Rosen, S.; Fujimoto, J.G. Nonlinear microscopy for detection of prostate cancer: Analysis of sensitivity and specificity in radical prostatectomies. Mod. Pathol. 2019, 33, 916–923. [Google Scholar] [CrossRef]
- Ahmad, M.K.; Srivastava, S.; Mahdi, A.A. Molecular Diagnostic in Prostate Cancer. In Molecular Diagnostics in Cancer Patients; Springer Science and Business Media LLC: Berlin, Germany, 2019; pp. 187–198. [Google Scholar]
- Caracciolo, G.; Vali, H.; Moore, A.; Mahmoudi, M. Challenges in molecular diagnostic research in cancer nanotechnology. Nano Today 2019, 27, 6–10. [Google Scholar] [CrossRef]
- El-Sayed, A.; Kamel, M.S. Advances in nanomedical applications: Diagnostic, therapeutic, immunization, and vaccine production. Environ. Sci. Pollut. Res. 2019, 27, 19200–19213. [Google Scholar] [CrossRef]
- Lydiard, J.; Nemeroff, C.B. Biomarker-Guided Tailored Therapy. In Advances in Experimental Medicine and Biology; Springer Science and Business Media LLC: Berlin, Germany, 2019; Volume 1192, pp. 199–224. [Google Scholar]
- Gao, Y.; Zhang, M.; Li, X.; Zeng, P.; Wang, P.; Zhang, L. Serum PSA levels in patients with prostate cancer and other 33 different types of diseases. Prog. Mol. Biol. Transl. Sci. 2019, 162, 377–390. [Google Scholar] [CrossRef]
- Moradi, A.; Srinivasan, S.; Clements, J.; Batra, J. Beyond the biomarker role: Prostate-specific antigen (PSA) in the prostate cancer microenvironment. Cancer Metastasis Rev. 2019, 38, 333–346. [Google Scholar] [CrossRef]
- Murray, N.P.; Fuentealba, C.; Reyes, E.; Lopez, M.A.; Aníbal, A.; Minzer, S.; Munoz, L.; Orrego, S.; Guzman, E.; Arzeno, L.; et al. Predictive Value of Neutrophil to Lymphocyte Ratio in the Diagnosis of Significant Prostate Cancer at Initial Biopsy: A Comparison with Free Percent Prostate Specific Antigen, Prostate Specific Antigen Density and Primary Circulating Prostate Cells. Asian Pac. J. Cancer Prev. 2019, 20, 3385–3389. [Google Scholar] [CrossRef]
- Wang, A.; Lazo, M.; Carter, H.B.; Groopman, J.; Nelson, W.G.; Platz, E.A. Association Between Liver Fibrosis and Serum Prostate Specific Antigen (PSA) Among US Men: National Health and Nutrition Examination Survey (NHANES) 2001–2010. Cancer Epidemiol. Biomark. Prev. 2019, 28, 1331–1338. [Google Scholar] [CrossRef]
- Wang, K.; Prosperi, M.; Qiu, P.; Cheng, T.-Y.D.; Bird, V.Y.; Chen, X.; Song, M. Circulating testosterone in modifying the association of BMI change rate with serum PSA in prostate cancer-free men with initial-PSA less than 4 ng/mL. In Proceedings of the AACR Annual Meeting 2019, Atlanta, Georgia, 29 March–3 April 2019. [Google Scholar]
- Samad, A.; Fayyaz, N.; Siddiqa, A.; Akhter, N.; Saeed, R.; Rafiq, M.; Bukhari, A.A.; Afzal, A. Association of Serum PSA Levels with Histopathological Pattern of Prostate Lesions. J. Islam. Med. Dent. Coll. 2019, 8, 92–95. [Google Scholar] [CrossRef] [Green Version]
- Preston, M.A.; Gerke, T.A.; Carlsson, S.V.; Signorello, L.; Sjoberg, D.D.; Markt, S.C.; Kibel, A.S.; Trinh, Q.-D.; Steinwandel, M.; Blot, W.; et al. Baseline Prostate-specific Antigen Level in Midlife and Aggressive Prostate Cancer in Black Men. Eur. Urol. 2019, 75, 399–407. [Google Scholar] [CrossRef]
- Lee, K.; Kang, B.J.; Jeun, M.; Jang, G.H.; Song, S.H.; Jeong, I.G.; Kim, C.-S.; Searson, P.C. Diagnosis of prostate cancer via nanotechnological approach. Int. J. Nanomed. 2015, 10, 6555–6569. [Google Scholar] [CrossRef] [Green Version]
- Jazi, M.S. A Mini-Review of Nanotechnology and Prostate Cancer: Approaches in Early Diagnosis. J. Clin. Basic Res. (JCBR) 2020, 4, 21–31. [Google Scholar]
- Basso, L.; Bazzanella, N.; Cazzanelli, M.; Miotello, A. On the route towards a facile fluorescent nanodiamonds laser-synthesis. Carbon 2019, 153, 148–155. [Google Scholar] [CrossRef]
- Khivrich, I.; Ilani, S. Electric and Magnetic Field Nano-Sensing Using a New, Atomic-like Qubit in a Carbon Nanotube. arXiv 2019, arXiv:1908.08249. [Google Scholar]
- Zhao, Y.; Zhang, H.; Hou, Y.; Li, M.; Luo, Z. Progress toward Safe Tumor Diagnosis and Therapy via Degradable Inorganic Nanomaterials Constructed with Metabolically Safe Elements. ACS Appl. Nano Mater. 2019, 3, 1028–1042. [Google Scholar] [CrossRef]
- Farshchi, F.; Hasanzadeh, M.; Mokhtarzadeh, A. A novel electroconductive interface based on Fe3O4 magnetic nanoparticle and cysteamine functionalized AuNPs: Preparation and application as signal amplification element to minoring of antigen-antibody immunocomplex and biosensing of prostate cancer. J. Mol. Recognit. 2019, 33, 2825. [Google Scholar] [CrossRef]
- Xia, N.; Deng, D.; Wang, Y.; Fang, C.; Li, S.-J. Gold nanoparticle-based colorimetric method for the detection of prostate-specific antigen. Int. J. Nanomed. 2018, 13, 2521–2530. [Google Scholar] [CrossRef] [Green Version]
- Ivanov, Y.; Pleshakova, T.; Malsagova, K.; Kurbatov, L.; Popov, V.P.; Glukhov, A.; Smirnov, A.; Enikeev, D.; Potoldykova, N.; Alekseev, B.; et al. Detection of Marker miRNAs, Associated with Prostate Cancer, in Plasma Using SOI-NW Biosensor in Direct and Inversion Modes. Sensors 2019, 19, 5248. [Google Scholar] [CrossRef] [Green Version]
- Rong, Z.; Bai, Z.; Li, J.; Tang, H.; Shen, T.; Wang, Q.; Wang, C.; Xiao, R.; Wang, S. Dual-color magnetic-quantum dot nanobeads as versatile fluorescent probes in test strip for simultaneous point-of-care detection of free and complexed prostate-specific antigen. Biosens. Bioelectron. 2019, 145, 111719. [Google Scholar] [CrossRef]
- Sabahi, A.; Salahandish, R.; Ghaffarinejad, A.; Omidinia, E. Electrochemical nano-genosensor for highly sensitive detection of miR-21 biomarker based on SWCNT-grafted dendritic Au nanostructure for early detection of prostate cancer. Talanta 2020, 209, 120595. [Google Scholar] [CrossRef]
- Jonous, Z.A.; Shayeh, J.S.; Yazdian, F.; Yadegari, A.; Hashemi, M.; Omidi, M. An electrochemical biosensor for prostate cancer biomarker detection using graphene oxide-gold nanostructures. Eng. Life Sci. 2019, 19, 206–216. [Google Scholar] [CrossRef] [Green Version]
- Chen, N.; Rong, M.; Shao, X.; Zhang, H.; Liu, S.; Dong, B.; Xue, W.; Wang, T.; Li, T.; Pan, J. Surface-enhanced Raman spectroscopy of serum accurately detects prostate cancer in patients with prostate-specific antigen levels of 4–10 ng/mL. Int. J. Nanomed. 2017, 12, 5399. [Google Scholar] [CrossRef] [Green Version]
- Wu, G.; Datar, R.H.; Hansen, K.M.; Thundat, T.; Cote, R.J.; Majumdar, A. Bioassay of prostate-specific antigen (PSA) using microcantilevers. Nat. Biotechnol. 2001, 19, 856–860. [Google Scholar] [CrossRef]
- Kadimisetty, K.; Mosa, I.M.; Malla, S.; Satterwhite-Warden, J.E.; Kuhns, T.; Faria, R.C.; Lee, N.H.; Rusling, J.F. 3D-printed supercapacitor-powered electrochemiluminescent protein immunoarray. Biosens. Bioelectron. 2015, 77, 188–193. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Yu, B.; Wang, S.; Shen, Y.; Cong, H. Preparation, surface functionalization and application of Fe3O4 magnetic nanoparticles. Adv. Colloid Interface Sci. 2020, 281, 102165. [Google Scholar] [CrossRef]
- Amiri, M.; Salavati-Niasari, M.; Akbari, A. Magnetic nanocarriers: Evolution of spinel ferrites for medical applications. Adv. Colloid Interface Sci. 2019, 265, 29–44. [Google Scholar] [CrossRef]
- Kiplagat, A.; Martin, D.R.; Onani, M.O.; Meyer, M. Aptamer-conjugated magnetic nanoparticles for the efficient capture of cancer biomarker proteins. J. Magn. Magn. Mater. 2020, 497, 166063. [Google Scholar] [CrossRef]
- Thanh, B.T.; Van Sau, N.; Ju, H.; Bashir, M.J.; Jun, H.K.; Phan, T.B.; Ngo, Q.M.; Tran, N.Q.; Hai, T.H.; Van, P.H.; et al. Immobilization of Protein A on Monodisperse Magnetic Nanoparticles for Biomedical Applications. J. Nanomater. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Ho, S.-L.; Xu, D.; Wong, M.S.; Li, H.-W. Direct and multiplex quantification of protein biomarkers in serum samples using an immuno-magnetic platform. Chem. Sci. 2016, 7, 2695–2700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, L.; Lee, J.-R.; Hao, S.; Ling, X.B.; Brooks, J.D.; Wang, S.X.; Gambhir, S.S. Improved detection of prostate cancer using a magneto-nanosensor assay for serum circulating autoantibodies. PLoS ONE 2019, 14, e0221051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elahi, N.; Kamali, M.; Baghersad, M.H. Recent biomedical applications of gold nanoparticles: A review. Talanta 2018, 184, 537–556. [Google Scholar] [CrossRef]
- Miao, Z.; Gao, Z.; Chen, R.; Yu, X.; Su, Z.; Wei, G. Surface-bioengineered Gold Nanoparticles for Biomedical Applications. Curr. Med. Chem. 2018, 25, 1920–1944. [Google Scholar] [CrossRef]
- Sharifi, M.; Hosseinali, S.H.; Alizadeh, R.H.; Hasan, A.; Attar, F.; Salihi, A.; Shekha, M.S.; Amen, K.M.; Aziz, F.M.; Saboury, A.A.; et al. Plasmonic and chiroplasmonic nanobiosensors based on gold nanoparticles. Talanta 2020, 212, 120782. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, Z.; Zhu, D.; Tao, X.; Gao, Y.; Zhu, H.; Mao, Z.; Ling, J. Gold nanoparticles coated with polysarcosine brushes to enhance their colloidal stability and circulation time in vivo. J. Colloid Interface Sci. 2016, 483, 201–210. [Google Scholar] [CrossRef]
- Luo, D.; Wang, X.; Zeng, S.; Ramamurthy, G.; Burda, C.; Basilion, J. Prostate-specific membrane antigen targeted gold nanoparticles for prostate cancer radiotherapy: Does size matter for targeted particles? Chem. Sci. 2019, 10, 8119–8128. [Google Scholar] [CrossRef] [Green Version]
- Hoang, T.B.; Akselrod, G.M.; Mikkelsen, M.H. Ultrafast Room-Temperature Single Photon Emission from Quantum Dots Coupled to Plasmonic Nanocavities. Nano Lett. 2015, 16, 270–275. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, Y.; Deng, H.; Farooq, U.; Yang, X.; Khan, J.; Tang, J.; Song, H. High Quantum Yield Blue Emission from Lead-Free Inorganic Antimony Halide Perovskite Colloidal Quantum Dots. ACS Nano 2017, 11, 9294–9302. [Google Scholar] [CrossRef]
- Cao, S.; Ji, W.; Zhao, J.; Yang, W.; Li, C.; Zheng, J. Color-tunable photoluminescence of Cu-doped Zn–In–Se quantum dots and their electroluminescence properties. J. Mater. Chem. C 2016, 4, 581–588. [Google Scholar] [CrossRef]
- Pu, C.-D.; Dai, X.; Shu, Y.; Zhu, M.; Deng, Y.; Jin, Y.; Peng, X. Electrochemically-stable ligands bridge the photoluminescence-electroluminescence gap of quantum dots. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karimzadeh, A.; Hasanzadeh, M.; Shadjou, N.; De La Guardia, M. Electrochemical biosensing using N-GQDs: Recent advances in analytical approach. TrAC Trends Anal. Chem. 2018, 105, 484–491. [Google Scholar] [CrossRef]
- Xie, R.; Wang, Z.; Zhou, W.; Liu, Y.; Fan, L.; Li, Y.; Li, X. Graphene quantum dots as smart probes for biosensing. Anal. Methods 2016, 8, 4001–4016. [Google Scholar] [CrossRef]
- Ehzaria, H.; Amirib, M.; Safari, M. Enzyme-free sandwich-type electrochemical immunosensor for highly sensitive prostate specific antigen based on conjugation of quantum dots and antibody on surface of modified glassy carbon electrode with core–shell magnetic metal-organic frameworks. Talanta 2020, 210, 120641. [Google Scholar] [CrossRef]
- Ahmed, W.; Elhissi, A.; Dhanak, V.; Subramani, K. Carbon nanotubes: Applications in cancer therapy and drug delivery research. In Emerging Nanotechnologies in Dentistry; Elsevier: Amsterdam, The Netherlands, 2018; pp. 371–389. [Google Scholar]
- Gu, Z. Preparation of Carbon Nanotube/MnO2 Nanocomposite as an Electrode Modifier for Prostate-Specific Antigen (PSA) Determination. Int. J. Electrochem. Sci. 2017, 12, 10726–10736. [Google Scholar] [CrossRef]
- Quintero-Jaime, A.F.; Berenguer-Murcia, Á.; Cazorla-Amorós, D.; Morallón, E. Carbon Nanotubes Modified with Au for Electrochemical Detection of Prostate Specific Antigen: Effect of Au Nanoparticle Size Distribution. Front. Chem. 2019, 7, 147. [Google Scholar] [CrossRef] [Green Version]
- Soares, J.C.; Soares, A.C.; Rodrigues, V.C.; Melendez, M.E.; Santos, A.C.; Faria, E.F.; Reis, R.M.; Carvalho, A.L.; Oliveira, O.N. Detection of the Prostate Cancer Biomarker PCA3 with Electrochemical and Impedance-Based Biosensors. ACS Appl. Mater. Interfaces 2019, 11, 46645–46650. [Google Scholar] [CrossRef]
- Justino, C.I.; Gomes, A.R.; Freitas, A.C.; Duarte, A.C.; Rocha-Santos, T. Graphene based sensors and biosensors. TrAC Trends Anal. Chem. 2017, 91, 53–66. [Google Scholar] [CrossRef]
- Palys, B. Biosensors and Advanced Sensors. In Handbook of Graphene; John Wiley & Sons: Hoboken, NY, USA, 2019; Volume 6. [Google Scholar]
- Li, Z.; Zhang, W.; Xing, F. Graphene Optical Biosensors. Int. J. Mol. Sci. 2019, 20, 2461. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Hao, Z.; Yu, S.; De Moraes, C.G.; Suh, L.H.; Zhao, X.; Lin, Q. An Ultraflexible and Stretchable Aptameric Graphene Nanosensor for Biomarker Detection and Monitoring. Adv. Funct. Mater. 2019, 29, 1905202. [Google Scholar] [CrossRef]
- Jeong, B.; Kim, Y.J.; Jeong, J.-Y.; Kim, Y.J. Label-free electrochemical quantification of microRNA-375 in prostate cancer cells. J. Electroanal. Chem. 2019, 846, 113127. [Google Scholar] [CrossRef]
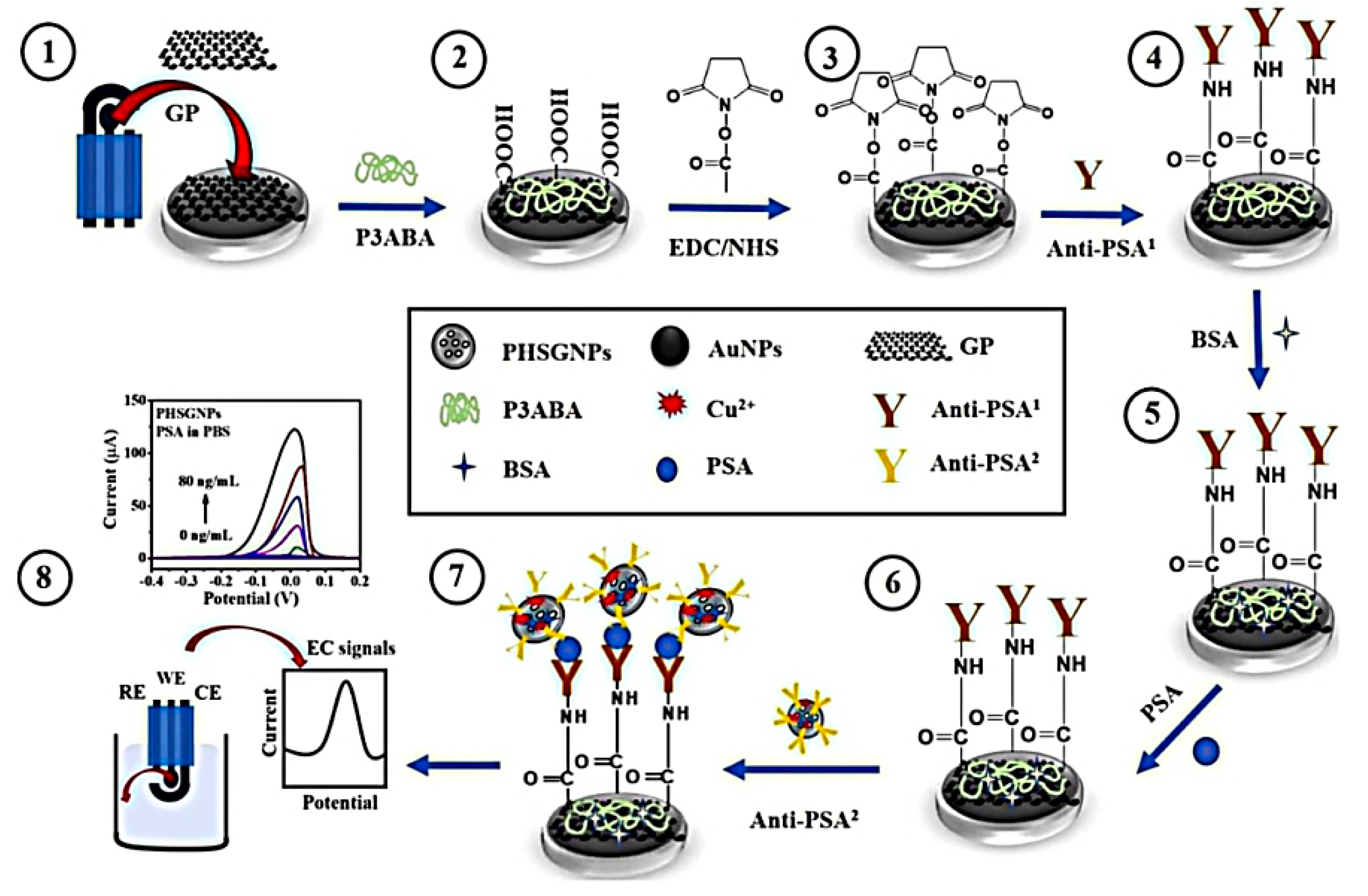
- Pothipor, C.; Wiriyakun, N.; Putnin, T.; Ngamaroonchote, A.; Jakmunee, J.; Ounnunkad, K.S.; Laocharoensuk, R.; Aroonyadet, N. Highly sensitive biosensor based on graphene–poly (3-aminobenzoic acid) modified electrodes and porous-hollowed-silver-gold nanoparticle labelling for prostate cancer detection. Sens. Actuators B Chem. 2019, 296, 126657. [Google Scholar] [CrossRef]
- Khondakar, K.R.; Dey, S.; Wuethrich, A.; Ibn Sina, A.A.; Trau, M. Toward Personalized Cancer Treatment: From Diagnostics to Therapy Monitoring in Miniaturized Electrohydrodynamic Systems. Acc. Chem. Res. 2019, 52, 2113–2123. [Google Scholar] [CrossRef]
- Koo, K.; Wee, E.J.H.; Wang, Y.; Trau, M. Enabling miniaturised personalised diagnostics: From lab-on-a-chip to lab-in-a-drop. Lab Chip 2017, 17, 3200–3220. [Google Scholar] [CrossRef]
- Fernández-La-Villa, A.; Pozo-Ayuso, D.F.; Castaño-Álvarez, M. Microfluidics and electrochemistry: An emerging tandem for next-generation analytical microsystems. Curr. Opin. Electrochem. 2019, 15, 175–185. [Google Scholar] [CrossRef]
- Koo, K.M. Label-Free Surface-Enhanced Raman Scattering Detection System for Clinical Biomarker Targets. In Mafic-Ultramafic Intrusions in Beishan and Eastern Tianshan at Southern CAOB: Petrogenesis, Mineralization and Tectonic Implication; Springer Science and Business Media LLC: Berlin, Germany, 2019; pp. 45–55. [Google Scholar]
- Zhang, J.J.; Lan, T.; Lu, Y. Translating in vitro diagnostics from centralized laboratories to point-of-care locations using commercially-available handheld meters. TrAC Trends Anal. Chem. 2020, 124, 115782. [Google Scholar] [CrossRef]
- Niederberger, E.; Parnham, M.J.; Maas, J.; Geisslinger, G. 4 Ds in health research—Working together toward rapid precision medicine. EMBO Mol. Med. 2019, 11, e10917. [Google Scholar] [CrossRef]
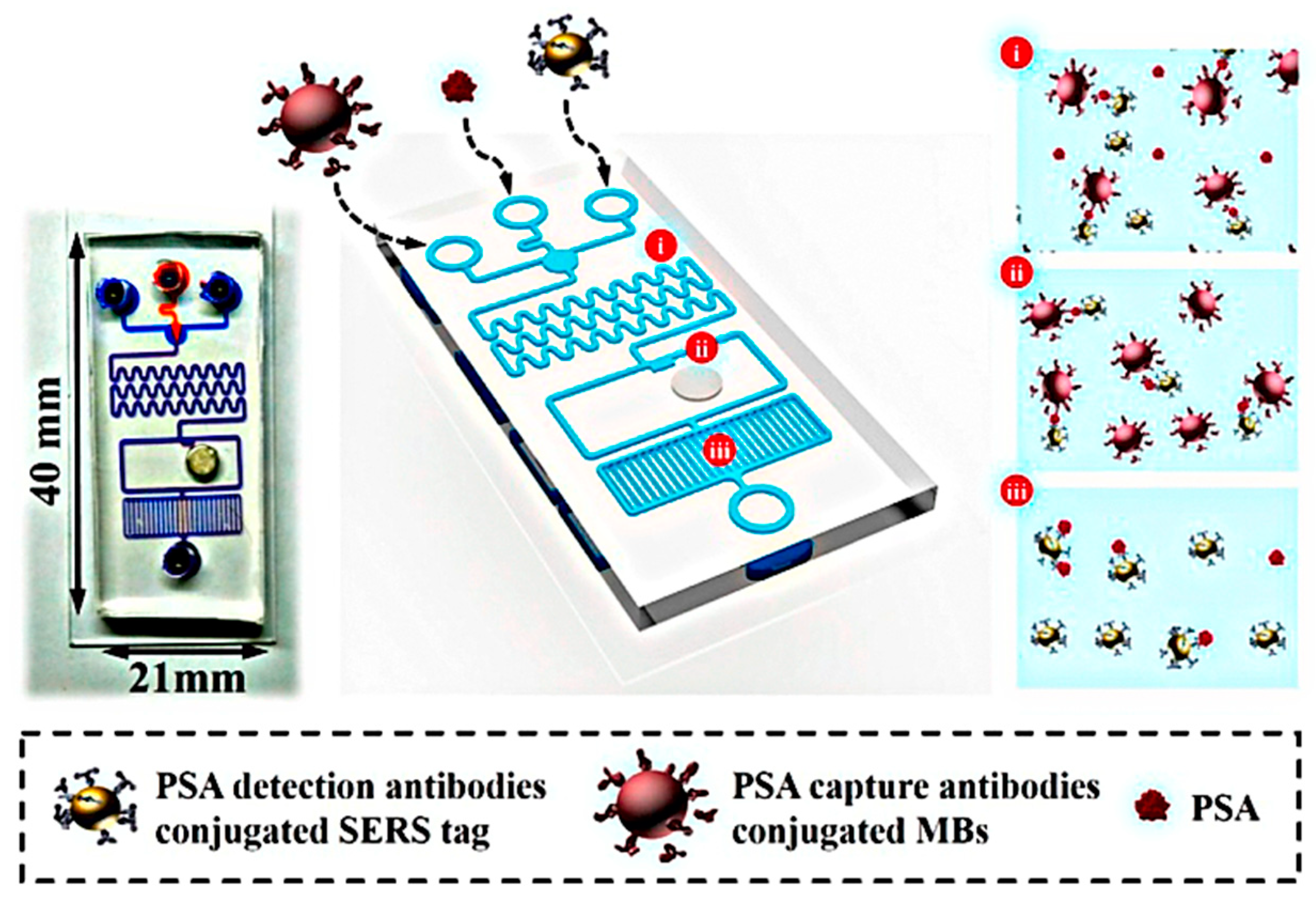
- Gao, R.; Lv, Z.; Mao, Y.; Yu, L.; Bi, X.; Xu, S.; Cui, J.; Wu, Y.-C. SERS-Based Pump-Free Microfluidic Chip for Highly Sensitive Immunoassay of Prostate-Specific Antigen Biomarkers. ACS Sens. 2019, 4, 938–943. [Google Scholar] [CrossRef]
- Feng, Z.; Zhi, S.; Guo, L.; Zhou, Y.; Lei, C. An integrated magnetic microfluidic chip for rapid immunodetection of the prostate specific antigen using immunomagnetic beads. Microchim. Acta 2019, 186, 252. [Google Scholar] [CrossRef]






| Nanomethodology | Biomarker | Detection Medium | Feature | Ref. |
|---|---|---|---|---|
| Magnetic nanoparticle | PSA | Human plasma | Appropriate linear range between 0.001 and 1 μg/L (via SWV method) with a 0.001 μg/L LLOQ. | [139] |
| Gold nanoparticles | PSA | Serums of healthy and prostate patients | Linear range 0~0.8 ng/mL for PSA measurement with a detection maximum of 0.02 ng/mL. | [140] |
| Silicon nanowires | miRNA 183 and 484 | Plasma | Target nucleic acid molecules can be detected with a high sensitivity of 3.3 × 10−16 M. | [141] |
| Quantum dots | f-PSA and cPSA | Two human serum | At the same time, detect f-PSA and c-PSA with detection limits of 0.009 ng/mL, in a quick assay time of 60 min. | [142] |
| Carbon nanotubes | miR-21 | Human serum | Strong linear relation with miR-21 target concentration (0.01 fmol/L to 1 μmol/L) and low experimental detection limit of 0.01 fmol/L. | [143] |
| Graphene | PSA | Blood | The detection limit for total and free PSA antigen was about 0.2 and 0.07 ng/mL, respectively. | [144] |
| Surface-enhanced Raman scattering (SERS) nanoparticles | PSA | Blood serum | Technique can substantially distinguish between low-risk and high-risk PCa with 92.3% accuracy, 89.5% sensitivity and 95% specificity. | [145] |
| Micro-cantilever or Piezoelectric material | PSA | HP and HSA | Offer a strong platform for DNA-protein, protein-protein binding, and DNA hybridization interactions with high-throughput label-free analyzes. | [146] |
| Lab-on-a-chip systems | PSA, PSMA and PF-4 | Serum | Detection limits for the 3 proteins in undiluted calf serum was 300–500 fg/mL. | [147] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barani, M.; Sabir, F.; Rahdar, A.; Arshad, R.; Kyzas, G.Z. Nanotreatment and Nanodiagnosis of Prostate Cancer: Recent Updates. Nanomaterials 2020, 10, 1696. https://doi.org/10.3390/nano10091696
Barani M, Sabir F, Rahdar A, Arshad R, Kyzas GZ. Nanotreatment and Nanodiagnosis of Prostate Cancer: Recent Updates. Nanomaterials. 2020; 10(9):1696. https://doi.org/10.3390/nano10091696
Chicago/Turabian StyleBarani, Mahmood, Fakhara Sabir, Abbas Rahdar, Rabia Arshad, and George Z. Kyzas. 2020. "Nanotreatment and Nanodiagnosis of Prostate Cancer: Recent Updates" Nanomaterials 10, no. 9: 1696. https://doi.org/10.3390/nano10091696









