Electrospun Nanofibers: from Food to Energy by Engineered Electrodes in Microbial Fuel Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Nanofibers Synthesis
2.2. MFC Architecture and Configuration
3. Results and Discussion
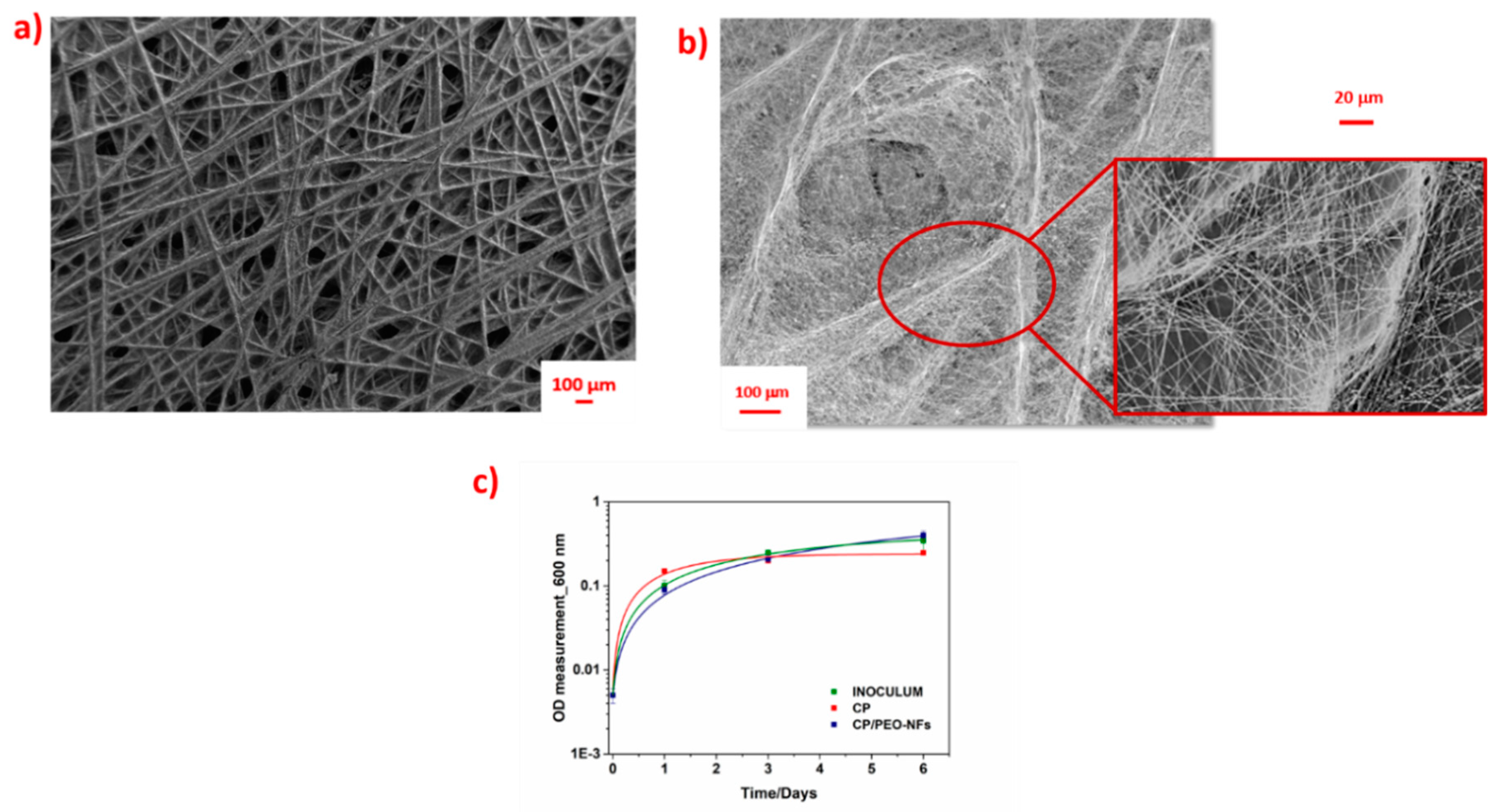
3.1. Electrospun Nanofibers Onto Carbon-Based Materials and Their Role as a Biomass Carrier
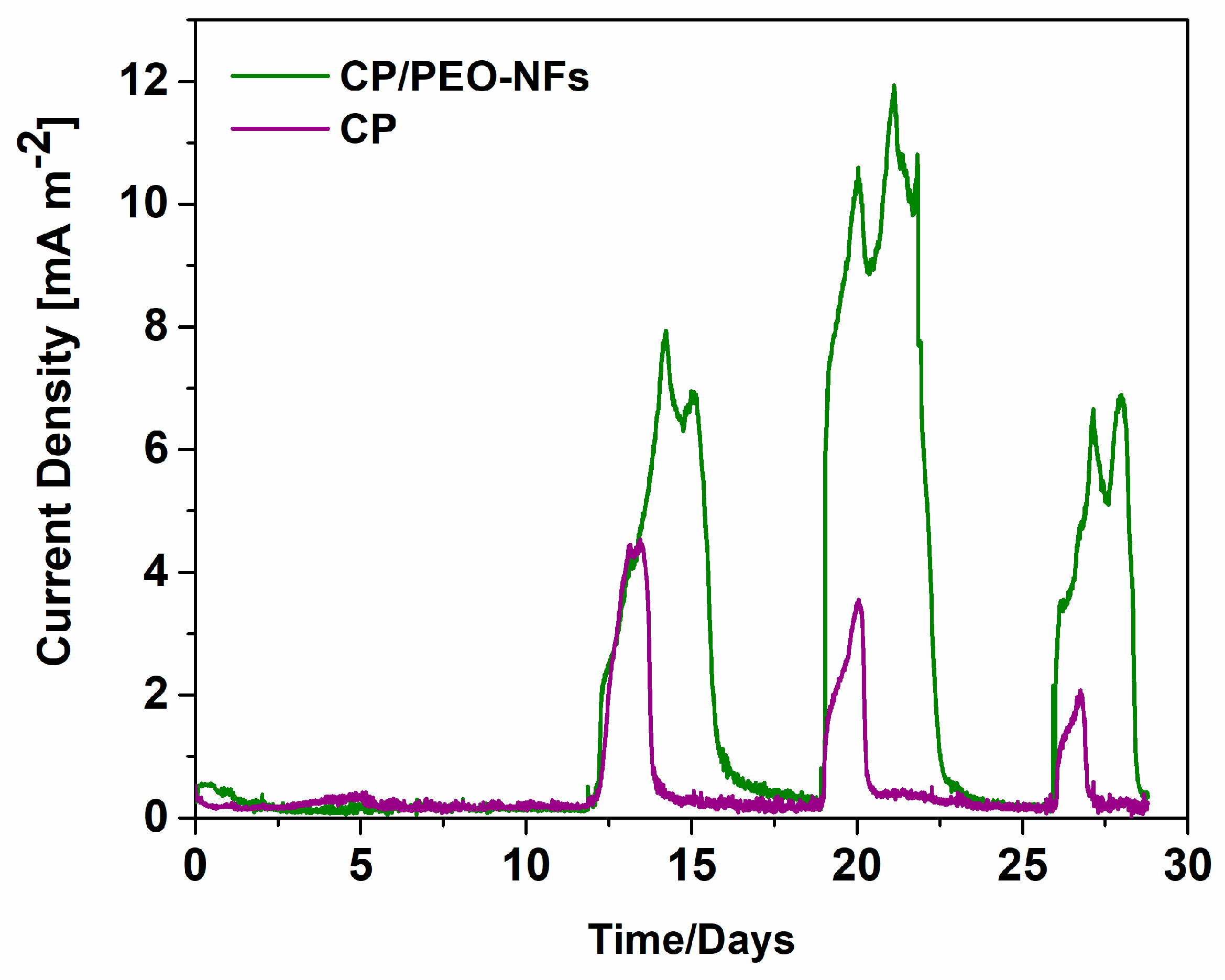
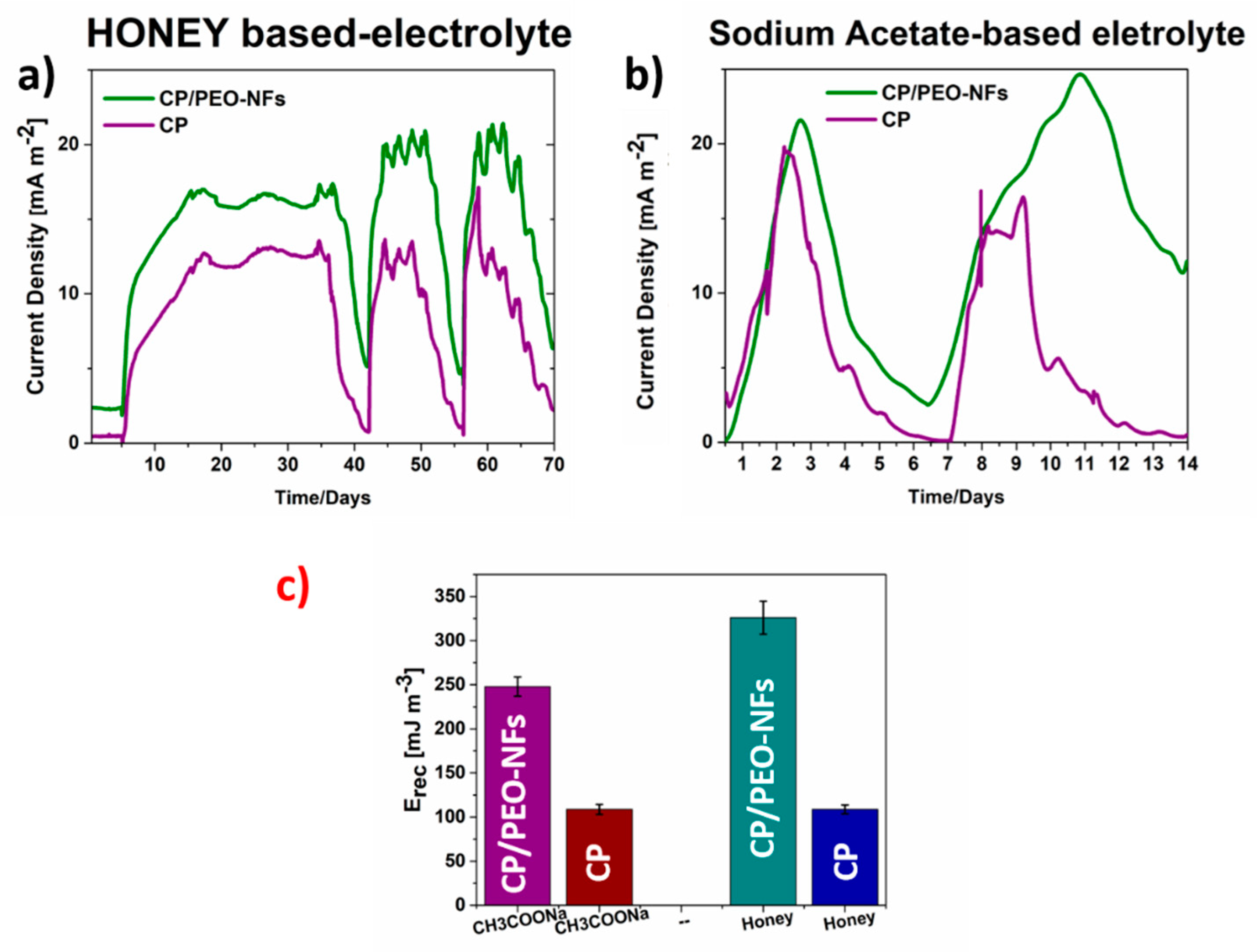
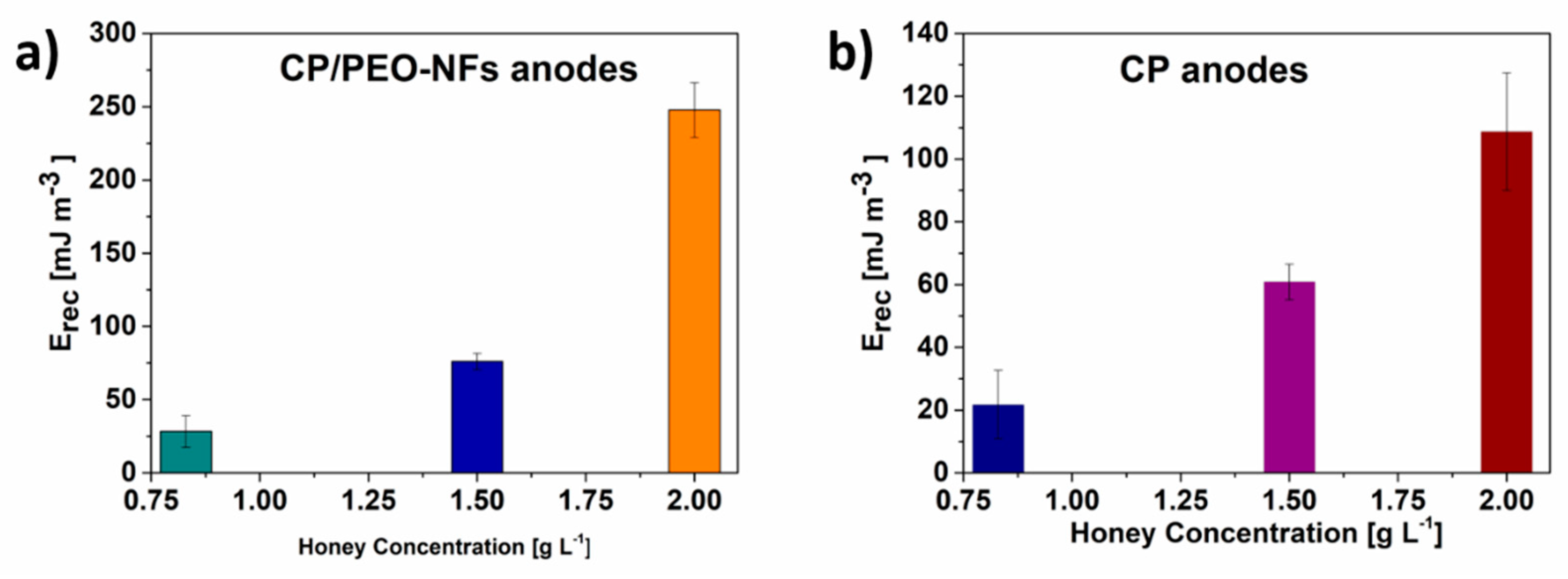
3.2. SCMFCs Performance
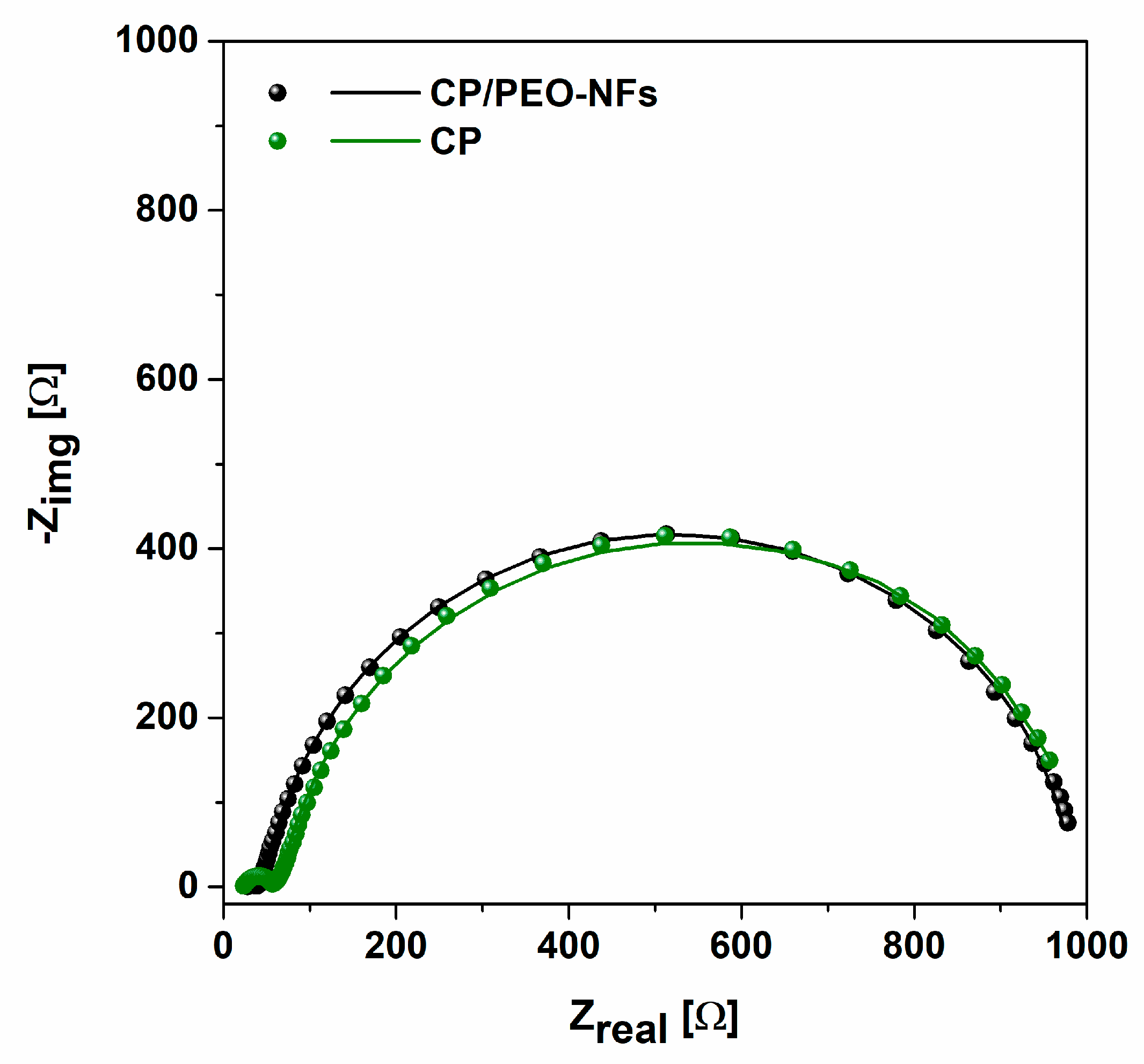
3.3. Electrochemical Impedance Spectroscopy Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Slate, A.J.; Whiteland, K.A.; Brownson, D.A.C.; Banks, C.E. Microbial fuel cells: An overview of current technology. Renew. Sustain. Energy Rev. 2019, 101, 60–81. [Google Scholar] [CrossRef]
- Logan, B.E. Microbial Fuel Cells; John Wiley & Sons Inc.: New York, NY, USA, 2008. [Google Scholar]
- Santoro, C.; Arbizzani, C.; Erable, B.; Ieropoulos, I. Microbial fuel cells: From fundamentals to applications. A review. J. Power Sources 2017, 356, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Harnisch, F.; Aulenta, F.; Schroeder, U. Microbial Fuel Cells and Bioelectrochemical Systems: Industrial and Environmental Biotechnologies Based on Extracellular Electron Transfer. In Comprehensive Biotechnology, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 644–659. [Google Scholar]
- Melhuish, C.; Ieropoulos, I.; Greenman, J.; Horsfield, I. Energetically autonomous robots: Food for thought. Auton. Robots 2006, 21, 187–198. [Google Scholar] [CrossRef]
- Sanket, G. From waste to watts in micro-devices: Review on development of Membraned and Membraneless Microfluidic Microbial Fuel Cell. Appl. Mater. Today 2018, 11, 270–279. [Google Scholar]
- Li, W.-W.; Yu, H.-Q.; He, Z. Towards sustainable wastewater treatment by using microbial fuel cells-centered technologies. Energy Environ. Sci. 2014, 7, 911–924. [Google Scholar] [CrossRef] [Green Version]
- Chang, I.S.; Jang, J.K.; Gil, G.C.; Kim, M.; Kim, H.J.; Cho, B.W.; Kim, B.H. Continuous determination of biochemical oxygen demand using microbial fuel cell type biosensor. Biosens. Bioelectron. 2004, 19, 607–613. [Google Scholar] [CrossRef]
- Shantaram, A.; Beyenal, H.; Raajan, R.; Veluchamy, A.; Lewandowski, Z. Wireless sensors powered by microbial fuel cells. Environ. Sci. Technol. 2005, 39, 5037–5042. [Google Scholar] [CrossRef]
- Donovan, C.; Dewan, A.; Heo, D.; Beyenal, H. Batteryless, wireless sensor powered by a sediment microbial fuel cell. Environ. Sci. Technol. 2008, 42, 8591–8596. [Google Scholar] [CrossRef]
- Tender, L.M.; Gray, S.A.; Groveman, E.; Lowy, D.A.; Kauffman, P.; Melhado, J.; Tyce, R.C.; Flynn, D.; Petrecca, R.; Dobarro, J. The first demonstration of a microbial fuel cell as a viable power supply: powering a meteorological buoy. J. Power Sources 2008, 179, 571–575. [Google Scholar] [CrossRef]
- Venkata Mohan, S.; Raghavulu, V.S.; Sarma, P.N. Biochemical evaluation of bioelectricity production process from anaerobic wastewater treatment in a single chambered microbial fuel cell (MFC) employing glass wool membrane. Biosens. Bioelectron. 2008, 23, 1326–1332. [Google Scholar] [CrossRef]
- Cecconet, D.; Molognoni, D.; Callegari, A.; Capodaglio, A.G. Agro-food industry wastewater treatment with microbial fuel cells: Energetic recovery issues. Int. J. Hydrogen Energy 2018, 43, 500–511. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, X.; Logan, B.E.; Lee, H. Brewery wastewater treatment using air-cathode microbial fuel cells. Appl. Microbiol. Biotechnol. 2008, 78, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Feng, Y.J.; Lee, H. Electricity production from beer brewery wastewater using single chamber microbial fuel cell. Water Sci. Technol. 2008, 57, 1117–1121. [Google Scholar] [CrossRef] [PubMed]
- Sciarra, T.P.; Tenca, A.; D’Epifanio, A.; Mecheri, B.; Merlino, G.; Barbato, M.; Borin, S.; Licoccia, S.; Garaviglia, V.; Adani, F. Using olive mill wastewater to improve performance in producing electricity from domestic wastewater by using single-chamber microbial fuel cell. Bioresour. Technol. 2013, 147, 246–253. [Google Scholar] [CrossRef] [Green Version]
- Sciarra, T.P.; Merlino, G.; Scaglia, B.; D’Epifanio, A.; Mecheri, B.; Borin, S.; Licoccia, S.; Adani, F. Electricity generation using white and red wine lees in air cathode microbial fuel cells. J. Power Sources 2015, 274, 393–399. [Google Scholar] [CrossRef]
- Penteado, E.D.; Fernandez-Marchante, C.M.; Zaiat, M.; Canizares, P.; Gonzales, E.R.; Rodrigo, M.A.R. Energy recovery from winery wastewater using a dual chamber microbial fuel cell. J Chem. Technol. Biotechnol. 2016, 91, 1802–1808. [Google Scholar] [CrossRef]
- Mardanpour, M.M.; Esfahany, M.N.; Behzad, T.; Sedaqatvand, R. Single chamber microbial fuel cell with spiral anode for dairy wastewater treatment. Biosens. Bioelectron. 2012, 38, 264–269. [Google Scholar] [CrossRef]
- Oh, S.T.; Kim, J.R.; Premier, G.C.; Lee, T.H.; Kim, C.; Sloan, W.T. Sustainable wastewater treatment: How might microbial fuel cells contribute. Biotechnology Advances. 2010, 28, 871–881. [Google Scholar] [CrossRef]
- Yang, G.; Wang, J.; Zhang, H.; Jia, H.; Zhang, Y.; Cui, Z.; Gao, F. Maximizing energy recovery from homeostasis in microbial fuel cell by synergistic conversion of short-chain volatile fatty acid. Bioresour. Technol. Rep. 2019, 7, 100200. [Google Scholar] [CrossRef]
- Lee, H.S.; Parameswaran, P.; Kato-Marcus, A.; Torres, C.I.; Rittmann, B.E. Evaluation of energy-conversion efficiencies in microbial fuel cells (MFCs) utilizing fermentable and non-fermentable substrates. Water Res. 2008, 24, 1501–1510. [Google Scholar] [CrossRef]
- Ge, Z.; Ping, Q.; Xiao, L.; He, Z. Reducing effluent discharge and recovering bioenergy in an osmotic microbial fuel cell treating domestic wastewater. Desalination. 2013, 312, 52–59. [Google Scholar] [CrossRef]
- Fornero, J.J.; Rosenbaum, M.; Angenent, L.T. Electric Power Generation from Municipal, Food, and Animal Wastewaters Using Microbial Fuel Cells. Electroanalysis. 2010, 22, 832–843. [Google Scholar] [CrossRef]
- Zhang, L.S.; Wu, W.; Wang, J. Immobilization of activated sludge using improved polyvinyl alcohol (PVA) gel. J. Environ. Sci. 2007, 19, 1293–1297. [Google Scholar] [CrossRef]
- Jiang, D.; Li, B. Novel electrode materials to enhance the bacterial adhesion and increase the power generation in microbial fuel cells (MFCs). Water Sci. Technol. 2009, 59, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Ye, Z.F.; Li, Y.F.; Zhou, L.C.; Yang, L.Q. Preparation of crosslinked macroporous PVA foam carrier for immobilization of microorganisms. Process Biochem. 2010, 45, 60–66. [Google Scholar] [CrossRef]
- Bruce, P.G. Structure and electrochemistry of polymer electrolytes. Electrochim. Acta 1995, 40, 2077–2086. [Google Scholar] [CrossRef]
- Xie, J.; Duan, R.G.; Han, Y.; Kerr, J.B. Morphological, rheological and electrochemical studies of Poly(ethylene oxide) electrolytes containing fumed silica nanoparticles. Solid State Ionics 2004, 175, 755–758. [Google Scholar] [CrossRef]
- Quaglio, M.; Chiodoni, A.; Massaglia, G.; Delmondo, L.; Sacco, A.; Garino, N.; Castellino, M.; Bianco, S.; Margaria, V.; Salvador, G.P.; et al. Electrospinning-on-Electrode Assembly for Air-Cathodes in Microbial Fuel Cells. Adv. Mater. Interfaces 2018, 5, 1801107. [Google Scholar] [CrossRef]
- Massaglia, G.; Margaria, V.; Sacco, A.; Tommasi, T.; Pentassugli, S.; Ahmed, D.; Mo, R.; Pirri, C.F.; Quaglio, M. In situ continuous current production from marine floating microbial fuel cells. Appl. Energy 2018, 230, 78–85. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.M.; Tulipani, S.; Romandini, S.; Bertoli, E.; Battino, M. Contribution of honey in nutrition and human health: A review. Mediterr. J. Nutr. Metab. 2010, 3, 15–23. [Google Scholar] [CrossRef]
- Burlando, B.; Cornara, L. Honey in dermatology and skin care: A review. J. Cosmet. Dermatol. 2013, 12, 306–313. [Google Scholar] [CrossRef] [PubMed]
- De-Melo, A.A.M.; Almeida-Muradina, L.B.; Sancho, M.T.; Pascual-Mate, A. Composition and properties of Apis mellifera honey: A review. J. Apicult. Res. 2018, 57, 5–37. [Google Scholar] [CrossRef]
- Doner, L.W. Honey. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Finglas, P.M., Trugo, L.C., Eds.; Academic Press: London, UK, 2003; pp. 3125–3130. [Google Scholar]
- Sabatini, A.G. Il miele: Origine, composizione e proprieta. In Conscere il Miele; Sabatini, A.G., Botolotti, L., Marcazzan, G.L., Eds.; Avenue Media: Bologna/Milano, Italy, 2007; pp. 3–37. [Google Scholar]
- Capodaglio, A.G.; Molognoni, D.; Dallago, E.; Liberale, A.; Cella, R.; Longoni, P.; Pantaleoni, L. Microbial Fuel Cells for Direct Electrical Energy Recovery from Urban Wastewaters. Sci. World J. 2013, 2013, 634738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massaglia, G.; Gerosa, M.; Agostino, V.; Cingolani, A.; Sacco, A.; Saracco, G.; Margaria, V.; Quaglio, M. Fluid Dynamic Modeling for Microbial Fuel Cell Based Biosensor Optimization. Fuel Cells 2017, 17, 627–634. [Google Scholar] [CrossRef]
- Hidalgo, D.; Sacco, A.; Hernàndez, S.; Tommasi, T. Electrochemical and impedance characterization of Microbial Fuel Cells based on 2D and 3D anodic electrodes working with seawater microorganisms under continuous operation. Bioresour. Technol. 2015, 195, 139–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massaglia, G.; Frascella, F.; Chiadò, A.; Sacco, A.; Marasso, S.L.; Cocuzza, M.; Pirri, C.F.; Quaglio, M. Electrospun Nanofibers: from Food to Energy by Engineered Electrodes in Microbial Fuel Cells. Nanomaterials 2020, 10, 523. https://doi.org/10.3390/nano10030523
Massaglia G, Frascella F, Chiadò A, Sacco A, Marasso SL, Cocuzza M, Pirri CF, Quaglio M. Electrospun Nanofibers: from Food to Energy by Engineered Electrodes in Microbial Fuel Cells. Nanomaterials. 2020; 10(3):523. https://doi.org/10.3390/nano10030523
Chicago/Turabian StyleMassaglia, Giulia, Francesca Frascella, Alessandro Chiadò, Adriano Sacco, Simone Luigi Marasso, Matteo Cocuzza, Candido F. Pirri, and Marzia Quaglio. 2020. "Electrospun Nanofibers: from Food to Energy by Engineered Electrodes in Microbial Fuel Cells" Nanomaterials 10, no. 3: 523. https://doi.org/10.3390/nano10030523
APA StyleMassaglia, G., Frascella, F., Chiadò, A., Sacco, A., Marasso, S. L., Cocuzza, M., Pirri, C. F., & Quaglio, M. (2020). Electrospun Nanofibers: from Food to Energy by Engineered Electrodes in Microbial Fuel Cells. Nanomaterials, 10(3), 523. https://doi.org/10.3390/nano10030523








