Functionalization of Cotton Fabrics with Polycaprolactone Nanoparticles for Transdermal Release of Melatonin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
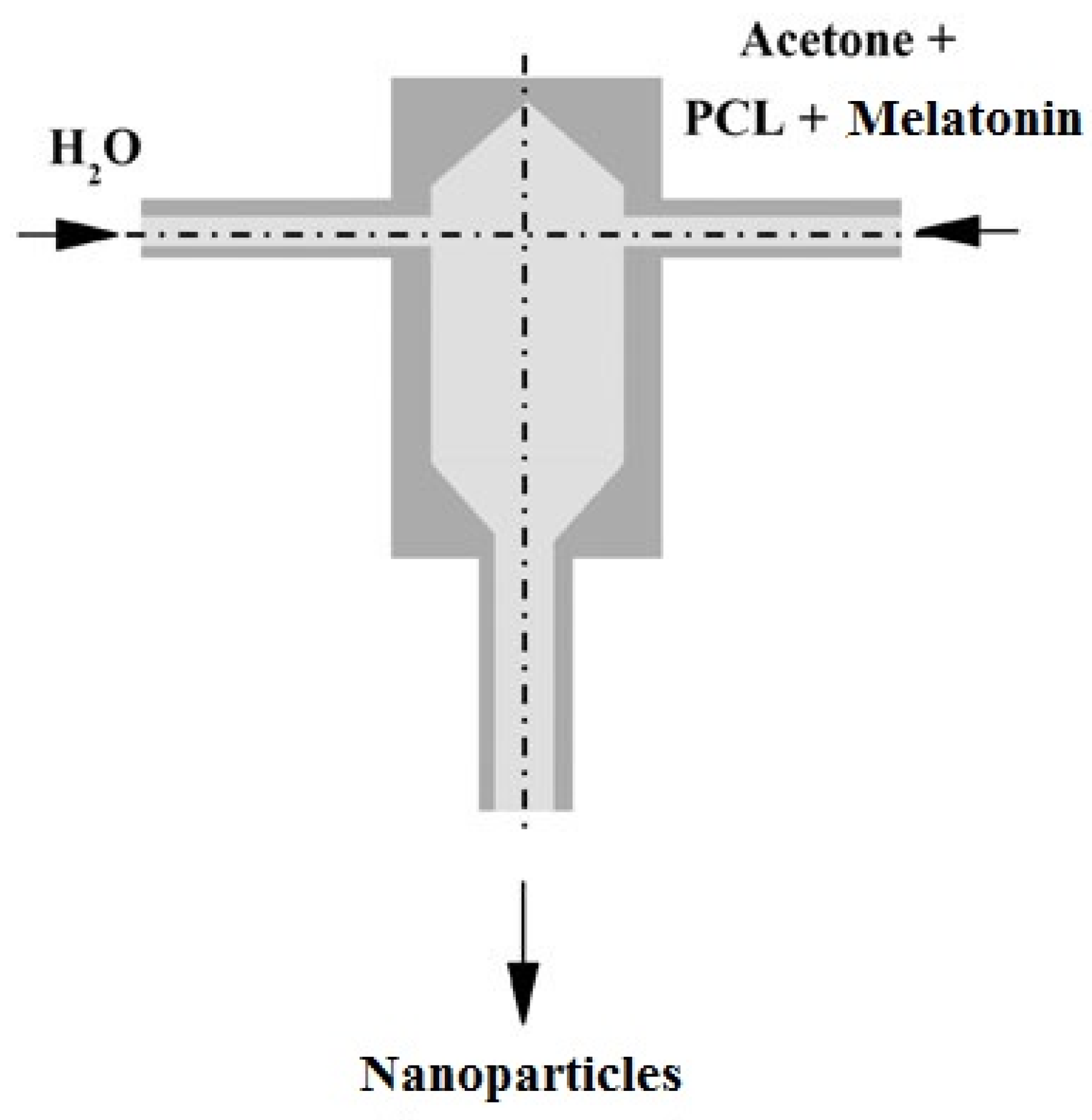
2.2. Nanoparticles Preparation
2.3. Nanoparticle Characterization
2.3.1. Dynamic Light Scattering
2.3.2. Determination of Loading Capacity (LC) and Encapsulation Efficiency (EE)
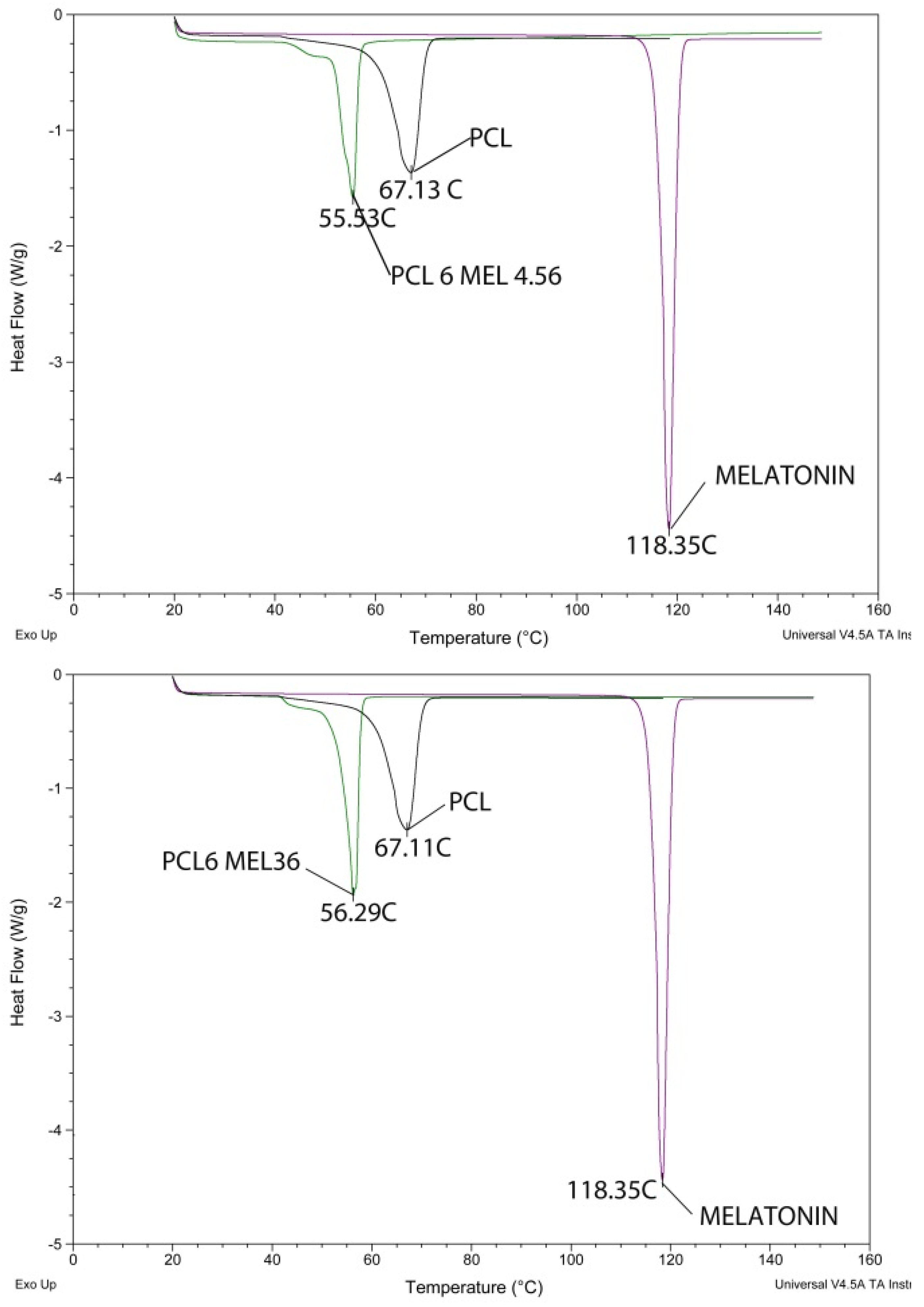
2.3.3. Differential Scanning Calorimetry
2.4. Fabric Functionalization
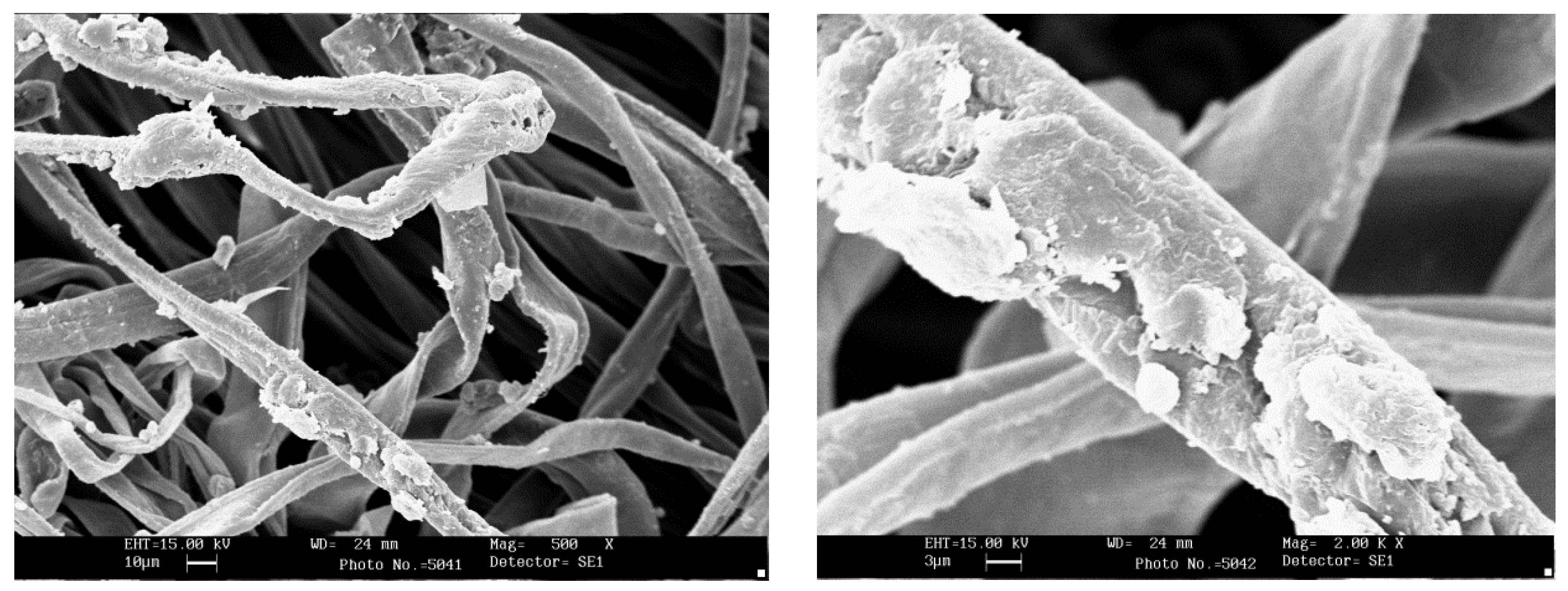
2.5. Scanning Electron Microscopy
2.6. In Vitro Release Test
2.7. Release Kinetics Modeling
3. Results and Discussion
3.1. Nanoparticles Size
3.1.1. Effect of Flow Rate
3.1.2. Effect of Melatonin and Polymer Initial Concentration
3.2. Zeta Potential
3.3. Loading Capacity and Encapsulation Efficiency
3.4. Differential Scanning Calorimetry
3.5. Scanning Electron Microscopy
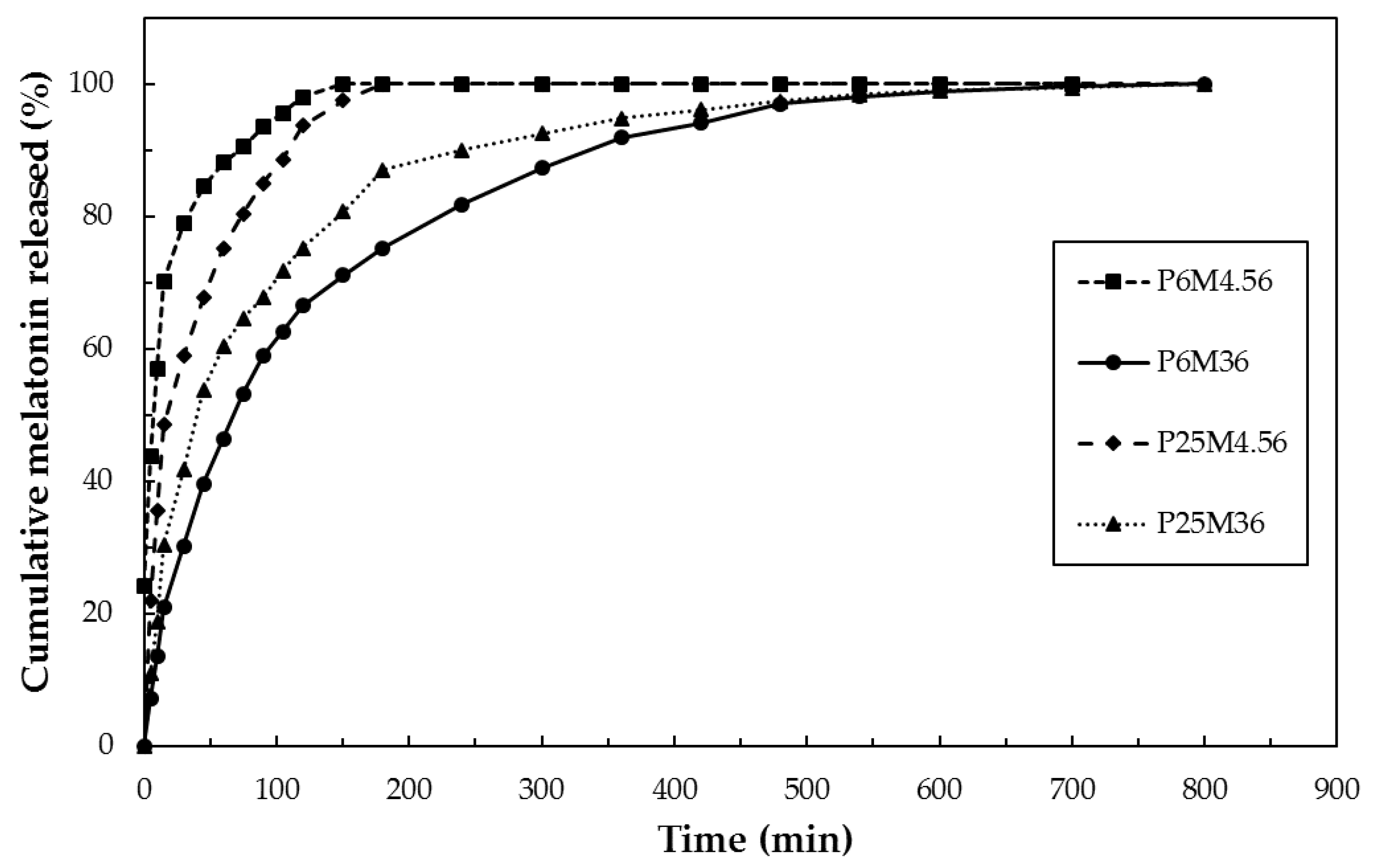
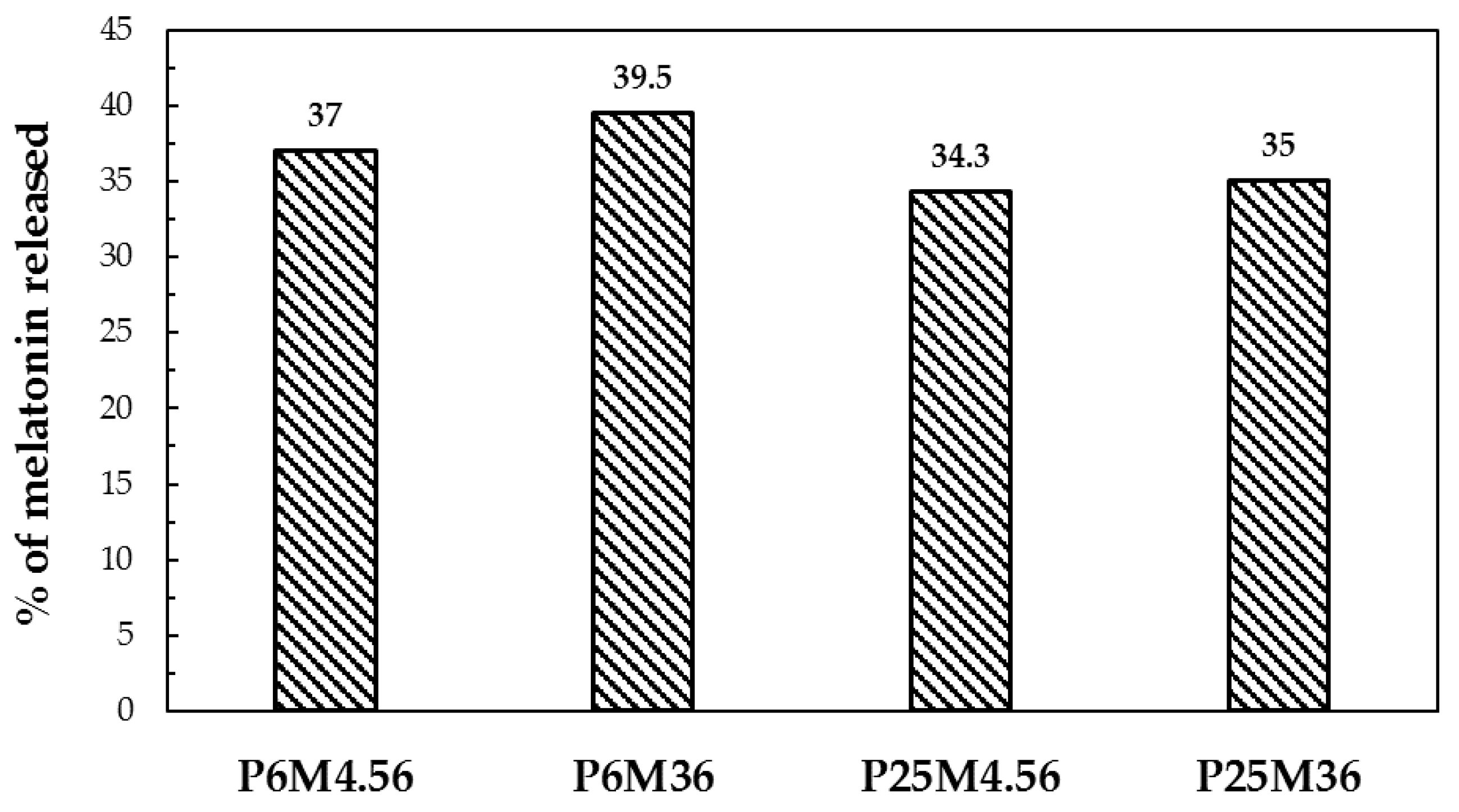
3.6. In Vitro Release Test
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Goyal, R.; Macri, L.K.; Kaplan, H.M.; Kohn, J. Nanoparticles and nanofibers for topical drug delivery. J. Control. Release 2016, 240, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Prow, T.W.; Grice, J.E.; Lin, L.L.; Faye, R.; Butler, M.; Becker, W.; Wurm, E.M.T.; Yoong, C.; Robertson, T.A.; Soyer, H.P.; et al. Nanoparticles and microparticles for skin drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 470–491. [Google Scholar] [CrossRef] [PubMed]
- DeLouise, L.A. Applications of nanotechnology in dermatology. J. Investig. Dermatol. 2012, 132, 964–975. [Google Scholar] [CrossRef] [PubMed]
- Hipler, U.-C.; Elsner, P. (Eds.) Current Problems in Dermatology. In Biofunctional Textiles and the Skin; S. Karger AG: Basel, Switzerland, 2006; Volume 33, ISBN 978-3-8055-8121-9. [Google Scholar]
- Mihailiasa, M.; Caldera, F.; Li, J.; Peila, R.; Ferri, A.; Trotta, F. Preparation of functionalized cotton fabrics by means of melatonin loaded β-cyclodextrin nanosponges. Carbohydr. Polym. 2016, 142, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Ruela, A.L.M.; Perissinato, A.G.; de Lino Sousa, M.E.; Mudrik, P.S.; Pereira, G.R. Evaluation of skin absorption of drugs from topical and transdermal formulations. Braz. J. Pharm. Sci. 2016, 52, 527–544. [Google Scholar] [CrossRef]
- Roberts, M.; Mohammed, Y.; Pastore, M.; Namjoshi, S.; Yousef, S.; Alinaghi, A.; Haridass, I.; Abd, E.; Leite-Silva, V.; Benson, H.; et al. Topical and cutaneous delivery using nanosystems. J. Control. Release 2017, 247, 86–105. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.A.; Jalalpure, S.S.; Kempwade, A.A.; Peram, M.R. Fabrication and in-vivo evaluation of lipid nanocarriers based transdermal patch of colchicine. J. Drug Deliv. Sci. Technol. 2017, 41, 444–453. [Google Scholar] [CrossRef]
- Rubio, L.; Alonso, C.; Coderch, L.; Parra, J.L.; Martí, M.; Cebrián, J.; Navarro, J.A.; Lis, M.; Valldeperas, J. Skin delivery of caffeine contained in biofunctional textiles. Text. Res. J. 2010, 80, 1214–1221. [Google Scholar] [CrossRef]
- Pinho, E.; Henriques, M.; Oliveira, R.; Dias, A.; Soares, G. Development of biofunctional textiles by the application of resveratrol to cotton, bamboo, and silk. Fibers Polym. 2010, 11, 271–276. [Google Scholar] [CrossRef] [Green Version]
- Martí, M.; Martínez, V.; Rubio, L.; Coderch, L.; Parra, J.L. Biofunctional textiles prepared with liposomes: In vivo and in vitro assessment. J. Microencapsul. 2011, 28, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Palmer, B.; DeLouise, L. Nanoparticle-enabled transdermal drug delivery systems for enhanced dose control and tissue targeting. Molecules 2016, 21, 1719. [Google Scholar] [CrossRef] [PubMed]
- Cevc, G.; Vierl, U. Nanotechnology and the transdermal route. A state of the art review and critical appraisal. J. Control. Release 2010, 141, 277–299. [Google Scholar] [CrossRef] [PubMed]
- Soppimath, K.S.; Aminabhavi, T.M.; Kulkarni, A.R.; Rudzinski, W.E. Biodegradable polymeric nanoparticles as drug delivery devices. J. Control. Release 2001, 70, 1–20. [Google Scholar] [CrossRef]
- Hans, M.L.; Lowman, A.M. Biodegradable nanoparticles for drug delivery and targeting. Curr. Opin. Solid State Mater. Sci. 2002, 6, 319–327. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K.; Yadav, S.C. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B Biointerfaces 2010, 75, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Dash, T.K.; Konkimalla, V.B. Poly-ε-caprolactone based formulations for drug delivery and tissue engineering: A review. J. Control. Release 2012, 158, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Kevadiya, B.D.; Thumbar, R.P.; Rajput, M.M.; Rajkumar, S.; Brambhatt, H.; Joshi, G.V.; Dangi, G.P.; Mody, H.M.; Gadhia, P.K.; Bajaj, H.C. Montmorillonite/poly-(ε-caprolactone) composites as versatile layered material: Reservoirs for anticancer drug and controlled release property. Eur. J. Pharm. Sci. 2012, 47, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Sinha, V.R.; Bansal, K.; Kaushik, R.; Kumria, R.; Trehan, A. Poly-ϵ-caprolactone microspheres and nanospheres: An overview. Int. J. Pharm. 2004, 278, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Madhaiyan, K.; Sridhar, R.; Sundarrajan, S.; Venugopal, J.R.; Ramakrishna, S. Vitamin B12 loaded polycaprolactone nanofibers: A novel transdermal route for the water soluble energy supplement delivery. Int. J. Pharm. 2013, 444, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Allen, C. Polycaprolactone—b-poly(ethylene oxide) copolymer micelles as a delivery vehicle for dihydrotestosterone. J. Control. Release 2000, 63, 275–286. [Google Scholar] [CrossRef]
- Szczęch, M.; Szczepanowicz, K.; Jantas, D.; Piotrowski, M.; Kida, A.; Lasoń, W.; Warszyński, P. Neuroprotective action of undecylenic acid (UDA) encapsulated into PCL nanocarriers. Colloids Surf. A Physicochem. Eng. Asp. 2017, 532, 41–47. [Google Scholar] [CrossRef]
- Baek, J.-S.; Choo, C.C.; Qian, C.; Tan, N.S.; Shen, Z.; Loo, S.C.J. Multi-drug-loaded microcapsules with controlled release for management of Parkinson’s disease. Small 2016, 12, 3712–3722. [Google Scholar] [CrossRef] [PubMed]
- Uto, K.; Ebara, M. Magnetic-responsive microparticles that switch shape at 37 °C. Appl. Sci. 2017, 7, 1203. [Google Scholar] [CrossRef]
- Tshweu, L.; Katata, L.; Kalombo, L.; Chiappetta, D.A.; Hocht, C.; Sosnik, A.; Swai, H. Enhanced oral bioavailability of the antiretroviral efavirenz encapsulated in poly(epsilon-caprolactone) nanoparticles by a spray-drying method. Nanomedicine 2014, 9, 1821–1833. [Google Scholar] [CrossRef] [PubMed]
- Fessi, H.; Puisieux, F.; Devissaguet, J.P.; Ammoury, N.; Benita, S. Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int. J. Pharm. 1989, 55, R1–R4. [Google Scholar] [CrossRef]
- Johnson, B.K.; Prud’homme, R.K. Flash nanoprecipitation of organic actives and block copolymers using a confined impinging jets mixer. Aust. J. Chem. 2003, 56, 1021–1024. [Google Scholar] [CrossRef]
- Valente, I.; Celasco, E.; Marchisio, D.L.; Barresi, A.A. Nanoprecipitation in confined impinging jets mixers: Production, characterization and scale-up of pegylated nanospheres and nanocapsules for pharmaceutical use. Chem. Eng. Sci. 2012, 77, 217–227. [Google Scholar] [CrossRef]
- Lince, F.; Marchisio, D.L.; Barresi, A.A. Strategies to control the particle size distribution of poly-ε-caprolactone nanoparticles for pharmaceutical applications. J. Colloid Interface Sci. 2008, 322, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.F.; Sun, C.C.; Chow, A.H.L. Assessment of the relative performance of a confined impinging jets mixer and a multi-inlet vortex mixer for curcumin nanoparticle production. Eur. J. Pharm. Biopharm. 2014, 88, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Zhu, Z.; Qian, H.; Wohl, A.R.; Beaman, C.J.; Hoye, T.R.; Macosko, C.W. A simple confined impingement jets mixer for flash nanoprecipitation. J. Pharm. Sci. 2012, 101, 4018–4023. [Google Scholar] [CrossRef] [PubMed]
- Zelenková, T.; Fissore, D.; Marchisio, D.L.; Barresi, A.A. Size control in production and freeze-drying of poly-ε-caprolactone nanoparticles. J. Pharm. Sci. 2014, 103, 1839–1850. [Google Scholar] [CrossRef] [PubMed]
- Ferri, A.; Kumari, N.; Peila, R.; Barresi, A.A. Production of menthol-loaded nanoparticles by solvent displacement. Can. J. Chem. Eng. 2017, 95. [Google Scholar] [CrossRef]
- Mossotti, R.; Ferri, A.; Innocenti, R.; Zelenková, T.; Dotti, F.; Marchisio, D.L.; Barresi, A.A. Cotton fabric functionalisation with menthol/PCL micro- and nano-capsules for comfort improvement. J. Microencapsul. 2015, 32, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.C.; Vigil, R.D.; Fox, R.O. A competitive aggregation model for flash nanoprecipitation. J. Colloid Interface Sci. 2010, 351, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Lavino, A.D.; Di Pasquale, N.; Carbone, P.; Marchisio, D.L. A novel multiscale model for the simulation of polymer flash nano-precipitation. Chem. Eng. Sci. 2017, 171, 485–494. [Google Scholar] [CrossRef]
- Di Pasquale, N.; Marchisio, D.L.; Barresi, A.A. Model validation for precipitation in solvent-displacement processes. Chem. Eng. Sci. 2012, 84, 671–683. [Google Scholar] [CrossRef]
- Margulis, K.; Magdassi, S.; Lee, H.S.; Macosko, C.W. Formation of curcumin nanoparticles by flash nanoprecipitation from emulsions. J. Colloid Interface Sci. 2014, 434, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Ansell, S.M.; Johnstone, S.A.; Tardi, P.G.; Lo, L.; Xie, S.; Shu, Y.; Harasym, T.O.; Harasym, N.L.; Williams, L.; Bermudes, D.; et al. Modulating the therapeutic activity of nanoparticle delivered Paclitaxel by manipulating the hydrophobicity of prodrug conjugates. J. Med. Chem. 2008, 51, 3288–3296. [Google Scholar] [CrossRef] [PubMed]
- Akbulut, M.; Ginart, P.; Gindy, M.E.; Theriault, C.; Chin, K.H.; Soboyejo, W.; Prud’homme, R.K. Generic method of preparing multifunctional fluorescent nanoparticles using flash nanoprecipitation. Adv. Funct. Mater. 2009, 19, 718–725. [Google Scholar] [CrossRef]
- Martínez Rivas, C.J.; Tarhini, M.; Badri, W.; Miladi, K.; Greige-Gerges, H.; Nazari, Q.A.; Galindo Rodríguez, S.A.; Román, R.Á.; Fessi, H.; Elaissari, A. Nanoprecipitation process: From encapsulation to drug delivery. Int. J. Pharm. 2017, 532, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Tam, Y.T.; To, K.K.W.; Chow, A.H.L. Fabrication of doxorubicin nanoparticles by controlled antisolvent precipitation for enhanced intracellular delivery. Colloids Surf. B Biointerfaces 2016, 139, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Massella, D.; Ferri, A.; Barresi, A.A. Production of PCL nanoparticles by flash nanoprecipitation for controlled release of caffeine. In Proceedings of the Merck Young Chemists Symposium (MYCS 2017), Milano Marittima, Italy, 13–15 November 2017; p. 80, ISBN 978-88-86208-89-5. [Google Scholar]
- Allen, S.; Osorio, O.; Liu, Y.-G.; Scott, E. Facile assembly and loading of theranostic polymersomes via multi-impingement flash nanoprecipitation. J. Control. Release 2017, 262, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Zetner, D.; Andersen, L.; Rosenberg, J. Pharmacokinetics of alternative administration routes of melatonin: A systematic review. Drug Res. 2015, 66, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Aeschbach, D.; Lockyer, B.J.; Dijk, D.-J.; Lockley, S.W.; Nuwayser, E.S.; Nichols, L.D.; Czeisler, C.A. Use of transdermal melatonin delivery to improve sleep maintenance during daytime. Clin. Pharmacol. Ther. 2009, 86, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Flo, A.; Calpena, A.C.; Halbaut, L.; Araya, E.I.; Fernández, F.; Clares, B. Melatonin delivery: Transdermal and transbuccal evaluation in different vehicles. Pharm. Res. 2016, 33, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- Kikwai, L.; Kanikkannan, N.; Babu, R.J.; Singh, M. Effect of vehicles on the transdermal delivery of melatonin across porcine skin in vitro. J. Control. Release 2002, 83, 307–311. [Google Scholar] [CrossRef]
- Rodrigues, M.R.; Lanzarini, C.M.; Ricci-Junior, E. Preparation, in vitro characterization and in vivo release of naproxen loaded in poly-caprolactone nanoparticles. Pharm. Dev. Technol. 2011, 16, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.; Sousa Lobo, J.M. Modeling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 2001, 13, 123–133. [Google Scholar] [CrossRef]
- Barresi, A.A.; Vanni, M.; Fissore, D.; Zelenková, T. Synthesis and preservation of polymer nanoparticles for pharmaceutical applications. In Handbook of Polymers for Pharmaceutical Technologies; Thakur, V.K., Thakur, M.K., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 229–280. ISBN 978-1-119-04141-2. [Google Scholar]
- Mihailiasa, M.; Izzo, M.; Ferri, A.; Peila, R.; Barresi, A.A. Study on trandermal delivery of melatonin from functionalized cotton fabrics. Presented at the 15th AUTEX World Textile Conference 2015, Bucharest, Romania, 10–12 June 2015; p. 13. [Google Scholar]
- Puglia, C.; Offerta, A.; Tirendi, G.G.; Tarico, M.S.; Curreri, S.; Bonina, F.; Perrotta, R.E. Design of solid lipid nanoparticles for caffeine topical administration. Drug Deliv. 2016, 23, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Souguir, H.; Salaün, F.; Douillet, P.; Vroman, I.; Chatterjee, S. Nanoencapsulation of curcumin in polyurethane and polyurea shells by an emulsion diffusion method. Chem. Eng. J. 2013, 221, 133–145. [Google Scholar] [CrossRef]



| Sample Name | Melatonin Concentration (mg/mL) | Polycaprolactone (PCL) Concentration (mg/mL) | Mass Ratio (MR) |
|---|---|---|---|
| P6M4.56 | 4.56 | 6 | 0.76 |
| P6M12 | 12 | 6 | 2 |
| P6M18 | 18 | 6 | 3 |
| P6M24 | 24 | 6 | 4 |
| P6M36 | 36 | 6 | 6 |
| P10M4.56 | 4.56 | 10 | 0.456 |
| P10M12 | 12 | 10 | 1.2 |
| P10M18 | 18 | 10 | 1.8 |
| P10M24 | 24 | 10 | 2.4 |
| P10M36 | 36 | 10 | 3.6 |
| P25M4.56 | 4.56 | 25 | 0.1824 |
| P25M12 | 12 | 25 | 0.48 |
| P25M18 | 18 | 25 | 0.72 |
| P25M24 | 24 | 25 | 0.96 |
| P25M36 | 36 | 25 | 1.44 |
| Mathematical Model | Equation |
|---|---|
| Zeroth order | F = kt |
| First order | Ln (1 − F) = −kt |
| Higuchi | F = kt1/2 |
| Baker–Lonsdale | 3/2[1 − (1 − F)2/3] − F = kt |
| Hixon–Crowell | 1 − (1 − F)1/3 = kt |
| Square root of mass | 1 − (1 − F)1/2 = kt |
| Three seconds root of mass | 1 − (1 − F)2/3 = kt |
| Formulation | MR | Mean Diameter (nm) | LC% | EE% |
|---|---|---|---|---|
| P25M4.56 | 0.18 | 852.8 | 13.0 | 81.6 |
| P6M4.56 | 0.76 | 259.9 | 39.6 | 86.2 |
| P25M36 | 1.44 | 2316.3 | 56.4 | 89.9 |
| P6M36 | 6 | 378.1 | 84.4 | 90.4 |
| Sample | Zeroth Order | First Order | Higuchi | Hixon–Crowell | Baker–Lonsdale | Square Root of Mass | Three Second Root of Mass |
|---|---|---|---|---|---|---|---|
| P6M4.56 | 0.33 | 0.94 | 0.88 | 0.79 | 0.97 | 0.69 | 0.56 |
| P6M36 | 0.63 | 0.90 | 0.97 | 0.82 | 0.99 | 0.77 | 0.71 |
| P25M4.56 | −0.46 | 0.71 | 0.65 | 0.45 | 0.94 | 0.28 | 0.07 |
| P25M36 | 0.84 | 0.95 | 0.98 | 0.92 | 0.99 | 0.91 | 0.88 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massella, D.; Leone, F.; Peila, R.; Barresi, A.A.; Ferri, A. Functionalization of Cotton Fabrics with Polycaprolactone Nanoparticles for Transdermal Release of Melatonin. J. Funct. Biomater. 2018, 9, 1. https://doi.org/10.3390/jfb9010001
Massella D, Leone F, Peila R, Barresi AA, Ferri A. Functionalization of Cotton Fabrics with Polycaprolactone Nanoparticles for Transdermal Release of Melatonin. Journal of Functional Biomaterials. 2018; 9(1):1. https://doi.org/10.3390/jfb9010001
Chicago/Turabian StyleMassella, Daniele, Federica Leone, Roberta Peila, Antonello A. Barresi, and Ada Ferri. 2018. "Functionalization of Cotton Fabrics with Polycaprolactone Nanoparticles for Transdermal Release of Melatonin" Journal of Functional Biomaterials 9, no. 1: 1. https://doi.org/10.3390/jfb9010001








