Serum Levels of Glutamate-Pyruvate Transaminase, Glutamate-Oxaloacetate Transaminase and Gamma-Glutamyl Transferase in 1494 Patients with Various Genotypes for the Alpha-1 Antitrypsin Gene
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Patients
2.3. Genetic Analysis
Determination of AAT and Transaminase Serum Levels
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Relationship between Genotypes and Transaminase Levels
3.3. Relationship between Transaminases and Serum AAT Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Thoracic Society; European Respiratory Society statement. American Thoracic Society/European Respiratory Society statement: Standards for the diagnosis and management of individuals with alpha-1 antitrypsin deficiency. Am. J. Respir. Crit. Care Med. 2003, 168, 818–900. [Google Scholar] [CrossRef] [PubMed]
- Stoller, J.K.; Aboussouan, L.S. Alpha1-antitrypsin deficiency. Lancet 2005, 365, 2225–2236. [Google Scholar] [CrossRef]
- de Serres, F.; Blanco, I. Role of alpha-1 antitrypsin in human health and disease. J. Intern. Med. 2014, 276, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Mela, M.; Smeeton, W.; Davies, S.E.; Miranda, E.; Scarpini, C.; Coleman, N.; Alexander, G.J. The Alpha-1 Antitrypsin Polymer Load Correlates with Hepatocyte Senescence, Fibrosis Stage and Liver-Related Mortality. Chronic Obstr. Pulm. Dis. 2020, 7, 151–162. [Google Scholar] [CrossRef]
- Teckman, J.H.; Blomenkamp, K.S. Pathophysiology of Alpha-1 Antitrypsin Deficiency Liver Disease. Methods Mol. Biol. 2017, 1639, 1–8. [Google Scholar] [CrossRef]
- Mahadeva, R.; Chang, W.S.W.; Dafforn, T.R.; Oakley, D.J.; Foreman, R.C.; Calvin, J.; Wight, D.G.; Lomas, D.A. Heteropolymerization of S, I, and Z α1-antitrypsin and liver cirrhosis. J. Clin. Investig. 1999, 103, 999–1006. [Google Scholar] [CrossRef] [Green Version]
- Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 3rd ed.; Worth Publishers: New York, NY, USA, 2000; pp. 628–631. [Google Scholar]
- Lay, M.J.; Wittwer, C.T. Real-time fluorescence genotyping of factor V Leiden during rapid-cycle PCR. Clin. Chem. 1997, 43, 2262–2267. [Google Scholar] [CrossRef] [Green Version]
- Hernández, P.J.; Ramos, D.R.; Fumero, G.S.; Pérez, P.J. Description of alpha-1-antitrypsin deficiency associated with PI* Q0ourém allele in La Palma Island (Spain) and a genotyping assay for its detection. Arch. Bronconeumol. 2015, 51. [Google Scholar] [CrossRef]
- Tanash, H.A.; Nystedt-Düzakin, M.; Montero, L.C.; Sveger, T.; Piitulainen, E. The Swedish α1-Antitrypsin Screening Study: Health Status and Lung and Liver Function at Age. Ann. Am. Thorac. Soc. 2015, 12, 807–812. [Google Scholar] [CrossRef]
- Alagille, D. α-1-Antitrypsin deficiency. Hepatology 1984, 4 (Suppl. 1), 11–14. [Google Scholar] [CrossRef]
- Silverman, E.K.; Sandhaus, R.A. Alpha1-antitrypsin deficiency. N. Engl. J. Med. 2009, 360, 2749–2757. [Google Scholar] [CrossRef] [PubMed]
- Regev, A.; Guaqueta, C.; Molina, E.G.; Conrad, A.; Mishra, V.; Brantly, M.L.; Torres, M.; De Medina, M.; Tzakis, A.G.; Schiff, E.R. Does the heterozygous state of alpha-1 antitrypsin deficiency have a role in chronic liver diseases? Interim results of a large case-control study. J. Pediatr. Gastroenterol. Nutr. 2006, 43 (Suppl. 1), 30–35. [Google Scholar] [CrossRef] [PubMed]
- Berg, N.O.; Eriksson, S. Liver disease in adults with alpha-1 -antitrypsin deficiency. N. Engl. J. Med. 1972, 287, 1264–1267. [Google Scholar] [CrossRef] [PubMed]
- Mandorfer, M.; Bucsics, T.; Hutya, V.; Schmid-Scherzer, K.; Schaefer, B.; Zoller, H.; Ferlitsch, A.; Peck-Radosavljevic, M.; Trauner, M.; Ferenci, P.; et al. Liver disease in adults with α1-antitrypsin deficiency. United Eur. Gastroenterol. J. 2018, 6, 710–718. [Google Scholar] [CrossRef] [Green Version]
- Larsson, C. Natural history and life expectancy in severe alpha1-antitrypsin deficiency, Pi Z. Acta Med. Scand. 1978, 204, 345–351. [Google Scholar] [CrossRef]
- Eigenbrodt, M.L.; McCashland, T.M.; Dy, R.M.; Clark, J.; Galati, J. Heterozygous α1-antitrypsin phenotypes in patients with end stage liver disease. Am. J. Gastroenterol. 1997, 92, 602–607. [Google Scholar]
- Strnad, P.; Buch, S.; Hamesch, K.; Fischer, J.; Rosendahl, J.; Schmelz, R.; Brueckner, S.; Brosch, M.; Heimes, C.V.; Woditsch, V.; et al. Heterozygous carriage of the alpha1-antitrypsin Pi*Z variant increases the risk to develop liver cirrhosis. Gut 2019, 68, 1099–1107. [Google Scholar] [CrossRef]
- Sveger, T. The natural history of liver disease in α1-antitrypsin deficient children. Acta Paediatr. Scand. 1988, 77, 847–851. [Google Scholar] [CrossRef]
- Blanco, I.; Lara, B. Déficit de Alfa-1 Antitripsina: Fisiopatología, Enfermedades Relacionadas y Tratamiento; Issuu Inc.: Copenhagen, Denmark, 2017. [Google Scholar]
- Tan, L.; Dickens, J.A.; DeMeo, D.L.; Miranda, E.; Perez, J.; Rashid, S.T.; Day, J.; Ordoñez, A.; Marciniak, S.J.; Haq, I.; et al. Circulating polymers in α1-antitrypsin deficiency. Eur. Respir. J. 2014, 43, 1501–1504. [Google Scholar] [CrossRef] [Green Version]
- Mostafavi, B.; Diaz, S.; Tanash, H.A.; Piitulainen, E. Liver function in alpha-1-antitrypsin deficient individuals at 37 to 40 years of age. Medicine 2017, 96. [Google Scholar] [CrossRef]
- Tanash, H.A.; Piitulainen, E. Liver disease in adults with severe alpha-1-antitrypsin deficiency. J. Gastroenterol. 2019, 54, 541–548. [Google Scholar] [CrossRef] [PubMed] [Green Version]

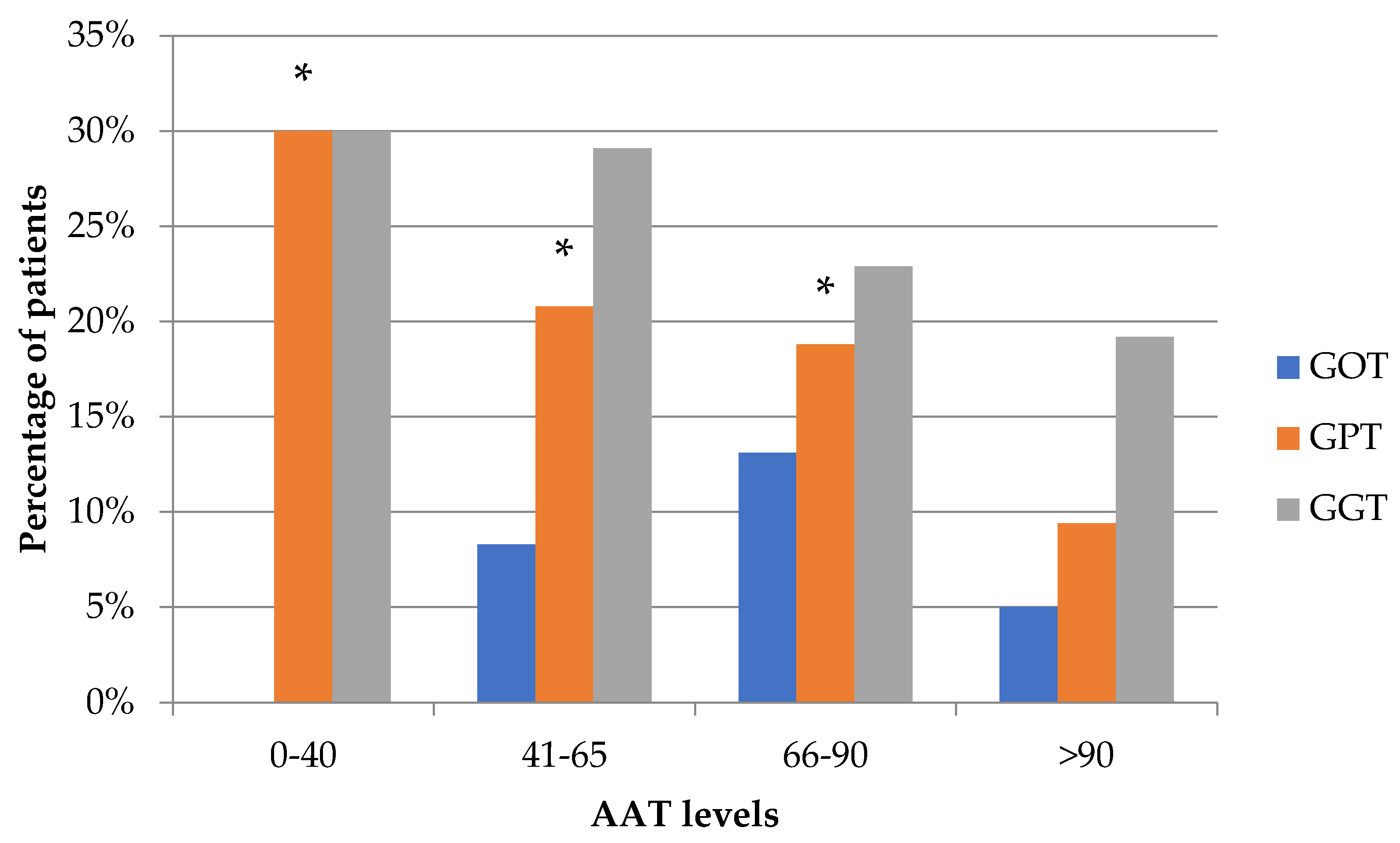
| Pi*MM n = 1018 | Pi*MS n = 287 | Pi*SS n = 23 | Pi*MZ n = 112 | Pi*SZ n = 26 | Pi*ZZ n = 7 | Rare Variants n = 21 | Degree of Significance PiMM-Pi ≠ MM | |
|---|---|---|---|---|---|---|---|---|
| Male, n | 612 (59.18%) | 142 (49.47%) | 10 (43.47%) | 62 (53.35%) | 11 (42.30%) | 4 (57.14%) | 6 (28.57%) | p < 0.0001 |
| Age, years, median (range) | 57 (11–94) | 52.05 (1–86) | 57.13 (16–86) | 48.68 (1–89) | 46.54 (10–81) | 57.86 (45–87) | 40.57 (1–75) | p < 0.004 |
| Weight, kg median (range) | 82.22 (39–152) | 79.94 (3.6–135) | 74.76 (49–116) | 79.67 (4.6–119) | 80.52 (54–115) | 65.67 (47–97) | 70.22 (4.4–92) | N/S |
| BMI, kg/m2 median (range) | 30.16 (14.9–53.6) | 29.06 (15–52.5) | 27.43 (19–40.1) | 28.67 (17.9–42.2) | 29.14 (21.4–44.1) | 23.03 (16.3–31.3) | 26.20 (19.6–37) | N/S |
| Serum AAT levels, mg/dL median (range) | 135.9 (82.8–308.2) | 115.05 (76.2–126.6) | 90.31 (74.6–125) | 85.15 (62.3–137.6) | 60.18 (43.9–82.1) | 18.94 (5–36.3) | 69.51 (7–111) | p < 0.05 |
| Genotype | GOT | GPT | GGT |
|---|---|---|---|
| Pi*MZ | 2.49 (CI: 1.28–4.85) | 4.65 (1.68–12.84) | 0.91 (0.55–1.51) |
| p < 0.005 | p < 0.001 | N/S | |
| Pi*SZ | 2 (CI: 1.17–3.42) | 2.75 (CI: 1.07–7) | 6.87 (CI: 1.52–31.15) |
| p < 0.01 | p < 0.02 | p < 0.003 | |
| Pi*ZZ | Not applicable | 2.72 (1.23–6) | 2.78 (0.61–12.52) |
| N/S | p < 0.01 | N/S |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández Pérez, J.M.; Blanco, I.; Jesús Sánchez Medina, A.; Díaz Hernández, L.; Antonio Pérez Pérez, J. Serum Levels of Glutamate-Pyruvate Transaminase, Glutamate-Oxaloacetate Transaminase and Gamma-Glutamyl Transferase in 1494 Patients with Various Genotypes for the Alpha-1 Antitrypsin Gene. J. Clin. Med. 2020, 9, 3923. https://doi.org/10.3390/jcm9123923
Hernández Pérez JM, Blanco I, Jesús Sánchez Medina A, Díaz Hernández L, Antonio Pérez Pérez J. Serum Levels of Glutamate-Pyruvate Transaminase, Glutamate-Oxaloacetate Transaminase and Gamma-Glutamyl Transferase in 1494 Patients with Various Genotypes for the Alpha-1 Antitrypsin Gene. Journal of Clinical Medicine. 2020; 9(12):3923. https://doi.org/10.3390/jcm9123923
Chicago/Turabian StyleHernández Pérez, José María, Ignacio Blanco, Agustín Jesús Sánchez Medina, Laura Díaz Hernández, and José Antonio Pérez Pérez. 2020. "Serum Levels of Glutamate-Pyruvate Transaminase, Glutamate-Oxaloacetate Transaminase and Gamma-Glutamyl Transferase in 1494 Patients with Various Genotypes for the Alpha-1 Antitrypsin Gene" Journal of Clinical Medicine 9, no. 12: 3923. https://doi.org/10.3390/jcm9123923
APA StyleHernández Pérez, J. M., Blanco, I., Jesús Sánchez Medina, A., Díaz Hernández, L., & Antonio Pérez Pérez, J. (2020). Serum Levels of Glutamate-Pyruvate Transaminase, Glutamate-Oxaloacetate Transaminase and Gamma-Glutamyl Transferase in 1494 Patients with Various Genotypes for the Alpha-1 Antitrypsin Gene. Journal of Clinical Medicine, 9(12), 3923. https://doi.org/10.3390/jcm9123923





