Face Recognition Characteristics in Patients with Age-Related Macular Degeneration Determined Using a Virtual Reality Headset with Eye Tracking
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.2. Face Recognition
2.3. Visual Acuity and Imaging
2.4. Dementia Screening
2.5. Statistical Analysis
3. Results
3.1. Visual Acuity
3.2. AMD Staging
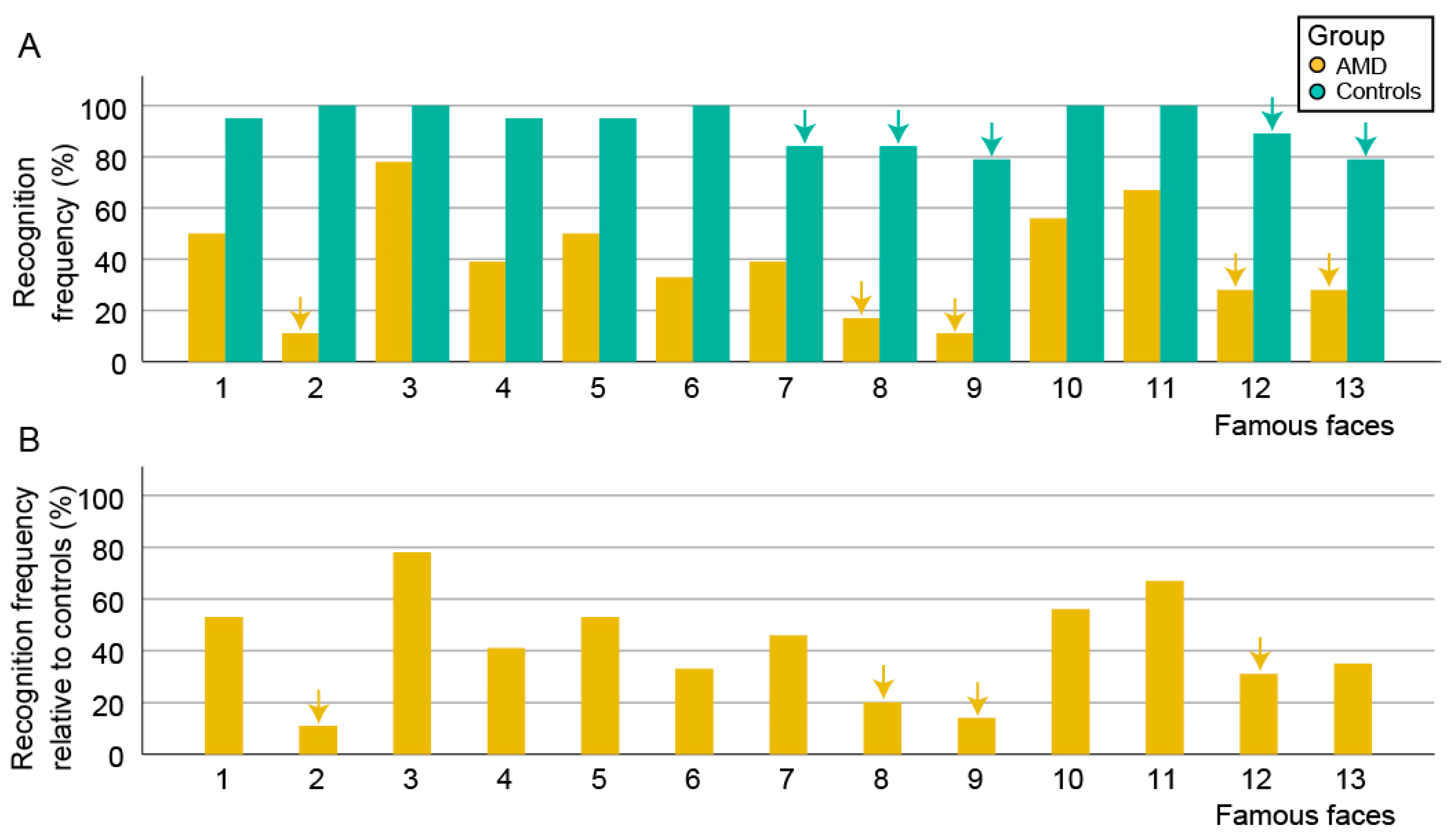
3.3. Face Recognition Accuracy and Time
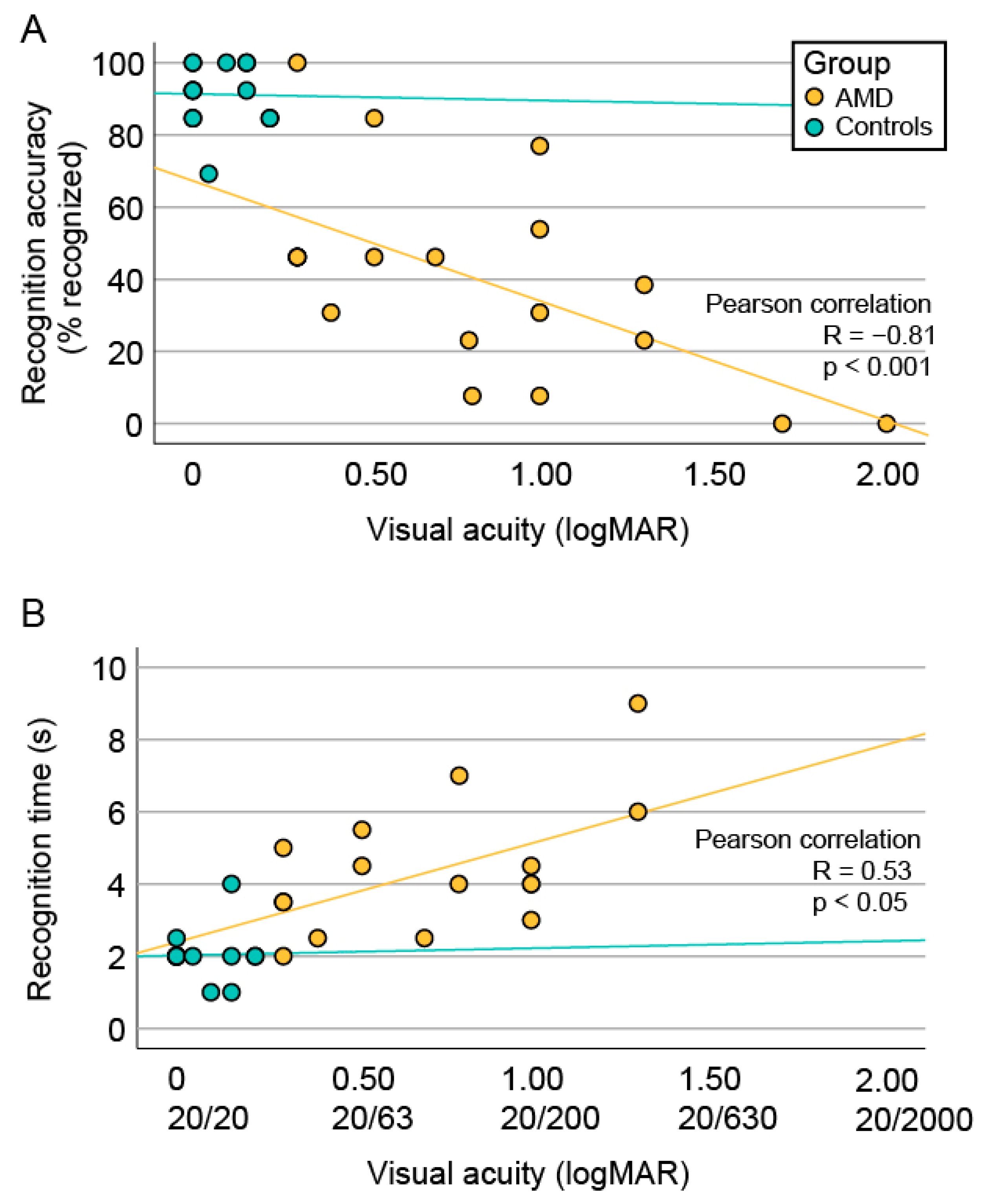
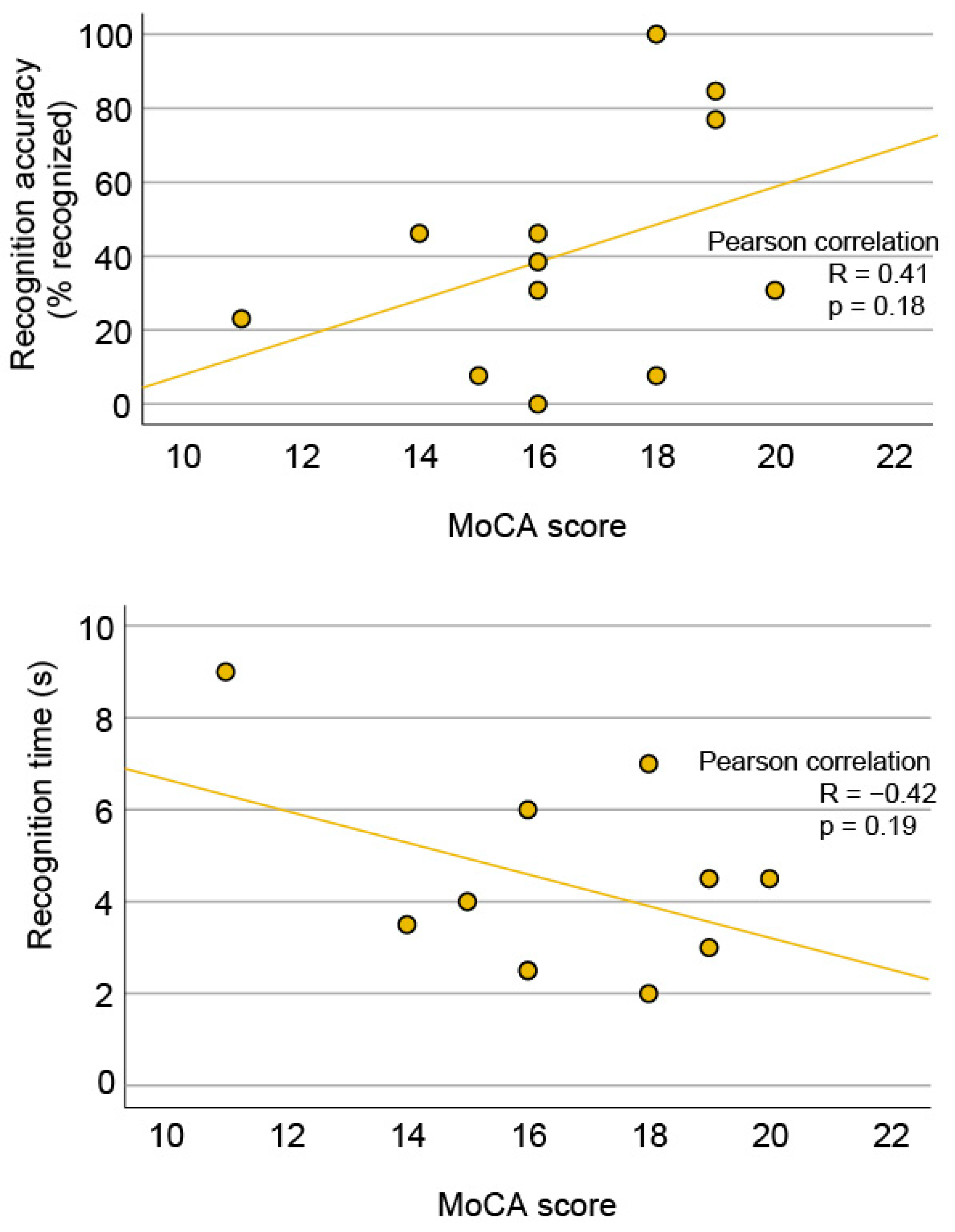
3.4. Correlation between Face Recognition and Visual Acuity
3.5. Correlation between Face Recognition and Structural Biomarkers
3.6. Eye Tracking Data
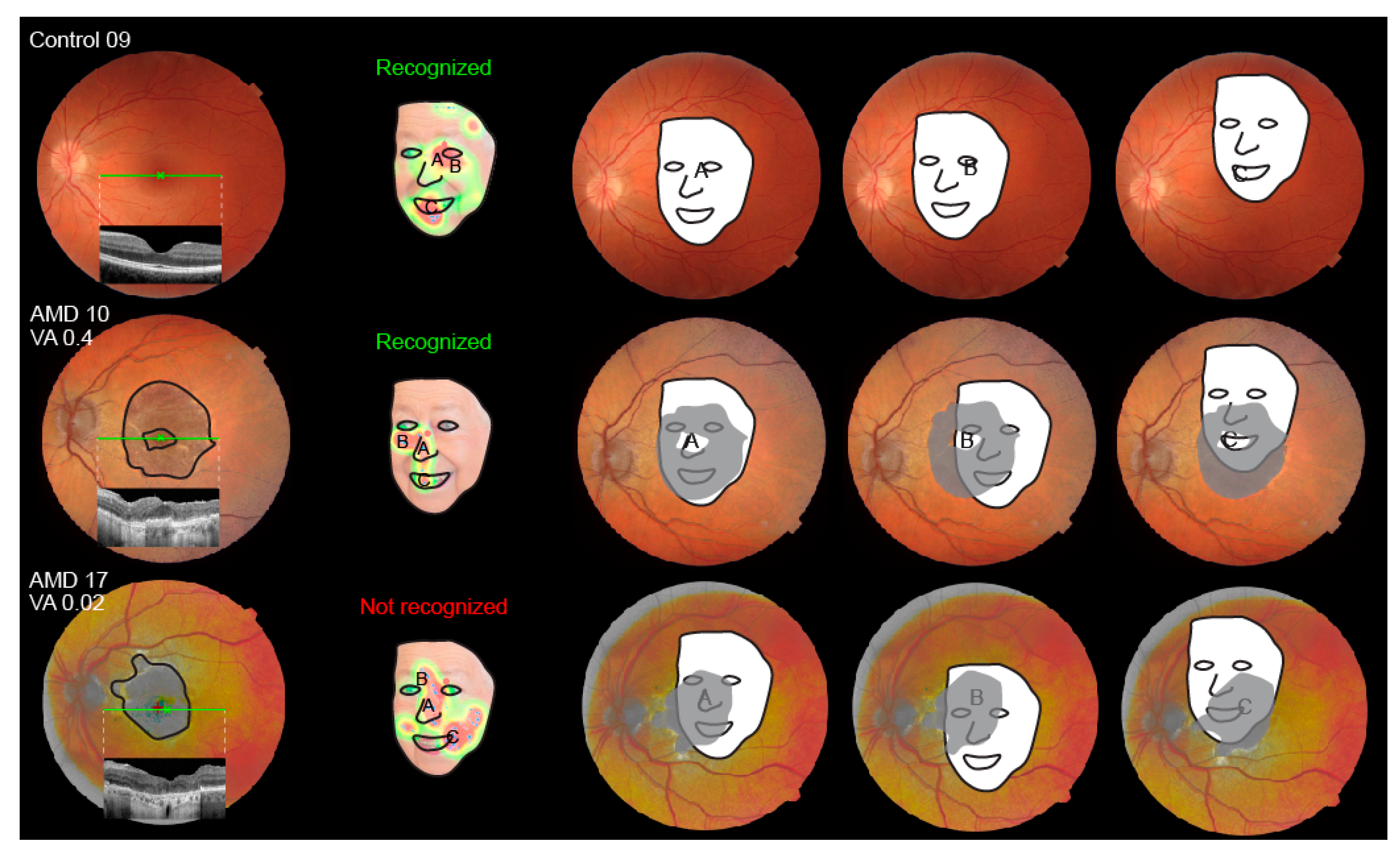
3.6.1. Fixation Heatmaps
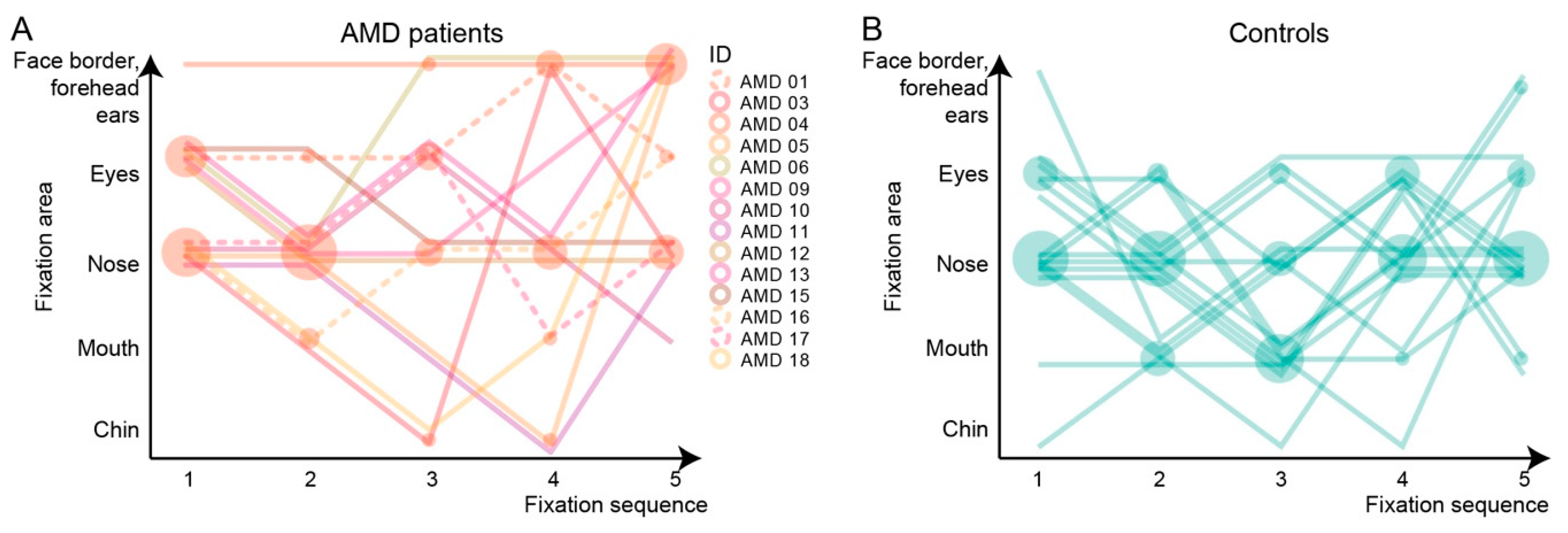
3.6.2. Initial Five-Fixation Sequence
3.6.3. Fixations on the Internal vs. External Facial Features
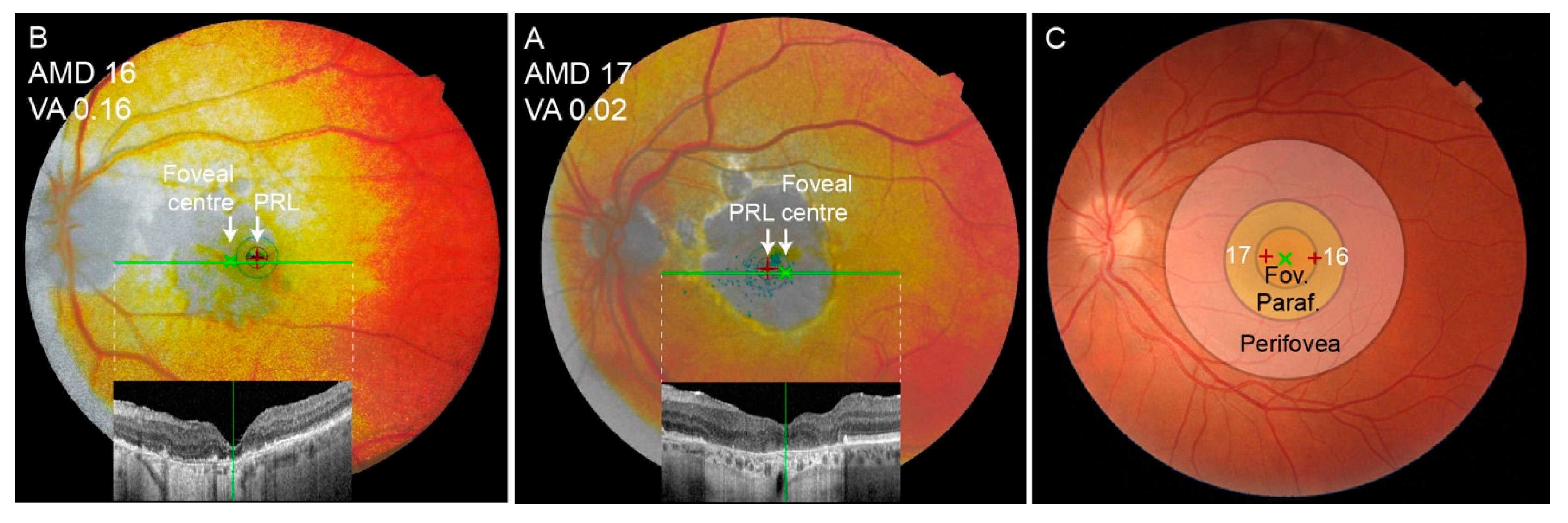
3.7. Shift of the Preferential Retinal Locus
3.8. Dementia
4. Discussion
4.1. Degree of Impaired Face Recognition in AMD Patients
4.2. Insights into Face Recognition Process from the Eye-Tracking Data
4.3. The Impact of Visual Acuity and Geographic Atrophy Diameter on Face Recognition
4.4. Dementia and AMD
4.5. Study Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Al-Zamil, W.M.; Yassin, S.A. Recent developments in age-related macular degeneration: A review. Clin. Interv. Aging 2017, 12, 1313–1330. [Google Scholar] [CrossRef]
- Deng, Y.; Qiao, L.; Du, M.; Qu, C.; Wan, L.; Li, J.; Huang, L. Age-related macular degeneration: Epidemiology, genetics, pathophysiology, diagnosis, and targeted therapy. Genes Dis. 2022, 9, 62–79. [Google Scholar] [CrossRef]
- Taylor, D.J.; Smith, N.D.; Binns, A.M.; Crabb, D.P. The effect of non-neovascular age-related macular degeneration on face recognition performance. Graefes Arch. Clin. Exp. Ophthalmol. 2018, 256, 815–821. [Google Scholar] [CrossRef]
- Garcia Layana, A. Age-related macular degeneration. Rev. Med. Univ. Navarra 1998, 42, 42–48. [Google Scholar]
- Frith, C. Role of facial expressions in social interactions. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 3453–3458. [Google Scholar] [CrossRef]
- Davis, J.M.; McKone, E.; Dennett, H.; O’Connor, K.B.; O’Kearney, R.; Palermo, R. Individual differences in the ability to recognise facial identity are associated with social anxiety. PLoS ONE 2011, 6, e28800. [Google Scholar] [CrossRef]
- Yardley, L.; McDermott, L.; Pisarski, S.; Duchaine, B.; Nakayama, K. Psychosocial consequences of developmental prosopagnosia: A problem of recognition. J. Psychosom. Res. 2008, 65, 445–451. [Google Scholar] [CrossRef]
- Seiple, W.; Rosen, R.B.; Garcia, P.M. Abnormal fixation in individuals with age-related macular degeneration when viewing an image of a face. Optom. Vis. Sci. 2013, 90, 45–56. [Google Scholar] [CrossRef]
- Hsiao, J.H.; Cottrell, G. Two fixations suffice in face recognition. Psychol. Sci. 2008, 19, 998–1006. [Google Scholar] [CrossRef]
- Arizpe, J.; Kravitz, D.J.; Yovel, G.; Baker, C.I. Start position strongly influences fixation patterns during face processing: Difficulties with eye movements as a measure of information use. PLoS ONE 2012, 7, e31106. [Google Scholar] [CrossRef]
- Blais, C.; Jack, R.E.; Scheepers, C.; Fiset, D.; Caldara, R. Culture shapes how we look at faces. PLoS ONE 2008, 3, e3022. [Google Scholar] [CrossRef]
- Mehoudar, E.; Arizpe, J.; Baker, C.I.; Yovel, G. Faces in the eye of the beholder: Unique and stable eye scanning patterns of individual observers. J. Vis. 2014, 14, 6. [Google Scholar] [CrossRef]
- Wegner-Clemens, K.; Rennig, J.; Magnotti, J.F.; Beauchamp, M.S. Using principal component analysis to characterize eye movement fixation patterns during face viewing. J. Vis. 2019, 19, 2. [Google Scholar] [CrossRef]
- Tang, J.; Falkmer, M.; Horlin, C.; Tan, T.; Vaz, S.; Falkmer, T. Face Recognition and Visual Search Strategies in Autism Spectrum Disorders: Amending and Extending a Recent Review by Weigelt et al. PLoS ONE 2015, 10, e0134439. [Google Scholar] [CrossRef]
- Chandra, S.R.; Patwardhan, K.; Pai, A.R. Problems of Face Recognition in Patients with Behavioral Variant Frontotemporal Dementia. Indian J. Psychol. Med. 2017, 39, 653–658. [Google Scholar] [CrossRef]
- Glen, F.C.; Crabb, D.P.; Smith, N.D.; Burton, R.; Garway-Heath, D.F. Do patients with glaucoma have difficulty recognizing faces? Investig. Ophthalmol. Vis. Sci. 2012, 53, 3629–3637. [Google Scholar] [CrossRef]
- Tejeria, L.; Harper, R.A.; Artes, P.H.; Dickinson, C.M. Face recognition in age related macular degeneration: Perceived disability, measured disability, and performance with a bioptic device. Br. J. Ophthalmol. 2002, 86, 1019–1026. [Google Scholar] [CrossRef]
- Bullimore, M.A.; Bailey, I.L.; Wacker, R.T. Face recognition in age-related maculopathy. Investig. Ophthalmol. Vis. Sci. 1991, 32, 2020–2029. [Google Scholar]
- Rong, S.S.; Lee, B.Y.; Kuk, A.K.; Yu, X.T.; Li, S.S.; Li, J.; Guo, Y.; Yin, Y.; Osterbur, D.L.; Yam, J.C.S.; et al. Comorbidity of dementia and age-related macular degeneration calls for clinical awareness: A meta-analysis. Br. J. Ophthalmol. 2019, 103, 1777–1783. [Google Scholar] [CrossRef]
- Wilson, R.S.; Kaszniak, A.W.; Bacon, L.D.; Fox, J.H.; Kelly, M.P. Facial recognition memory in dementia. Cortex 1982, 18, 329–336. [Google Scholar] [CrossRef]
- Barnes, C.S.; De L’Aune, W.; Schuchard, R.A. A test of face discrimination ability in aging and vision loss. Optom. Vis. Sci. 2011, 88, 188–199. [Google Scholar] [CrossRef]
- Bernard, J.B.; Chung, S.T. The Role of External Features in Face Recognition with Central Vision Loss. Optom. Vis. Sci. 2016, 93, 510–520. [Google Scholar] [CrossRef][Green Version]
- Ferris, S.H.; Crook, T.; Clark, E.; McCarthy, M.; Rae, D. Facial recognition memory deficits in normal aging and senile dementia. J. Gerontol. 1980, 35, 707–714. [Google Scholar] [CrossRef]
- Eng, Z.H.D.; Yick, Y.Y.; Guo, Y.; Xu, H.; Reiner, M.; Cham, T.J.; Chen, S.H.A. 3D faces are recognized more accurately and faster than 2D faces, but with similar inversion effects. Vis. Res. 2017, 138, 78–85. [Google Scholar] [CrossRef]
- Burt, A.L.; Crewther, D.P. The 4D Space-Time Dimensions of Facial Perception. Front. Psychol. 2020, 11, 1842. [Google Scholar] [CrossRef]


| ID | Age (Years) | Sex | Recognition Accuracy (%) | Recognition Time (s) | Freq. of External Fixations (%) | VA (logMAR) | AMD Stage | GA Diameter (Degrees) | Spared Foveal Photoreceptors | MoCA Score | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RE | LE | RE | LE | RE | LE | RE | LE | |||||||
| 1 | 91 | F | 0 | N/A | 61.5 | 2.0 | 2.0 * | 4a | 4a | 13.3 | 10.9 | N | N | 16 |
| 2 | 89 | M | 23.1 | 9.0 | N/A | 1.3 | 1.3 * | 4a | 4b | 12.8 | N/A | N | N | 11 |
| 3 | 79 | F | 7.7 | 4.0 | 100.0 | 1.0 * | 1.0 | 4a | 4a | N/A | N/A | N | N | 15 |
| 4 | 87 | F | 30.8 | 4.5 | 100.0 | 1.0 * | 1.7 | 4b | 4b | N/A | N/A | N | N | 20 |
| 5 | 68 | F | 53.9 | 4.0 | N/A | 1.0 * | 2.0 | 4b | 4b | N/A | N/A | N | N | N/A |
| 6 | 86 | F | 46.2 | 2.5 | 100.0 | 0.7 * | 2.0 | 4a | 4a | 14.2 | 11.9 | N | N | 16 |
| 7 | 76 | F | 46.2 | 5.5 | N/A | 1.8 | 0.5 * | 4b | 4a | N/A | 10.3 | N | Y | N/A |
| 8 | 70 | M | 46.2 | 3.5 | N/A | 0.3 * | 0.8 | 4b | 4a | N/A | 14.9 | N | N | N/A |
| 9 | 90 | F | 84.6 | 4,5 | 100.0 | 0.5 * | 1.7 | 4b | 4a | N/A | 9.0 | Y | N | 19 |
| 10 | 83 | F | 30.8 | 2.5 | 30.8 | 1.3 | 0.4 * | 4b | 4b | N/A | N/A | N | Y | 16 |
| 11 | 82 | M | 46.2 | 3.5 | 84.6 | 1.7 | 0.3 * | 4b | 4b | N/A | N/A | N | Y | 14 |
| 12 | 76 | M | 23.1 | 4.0 | 23.1 | 0.8 * | 1.7 | 4a | 4b | 7.9 | N/A | N | N | N/A |
| 13 | 75 | F | 100 | 2.0 | 46.2 | 1.3 | 0.3 * | 3 | 3 | N/A | N/A | Y | Y | 18 |
| 14 | 79 | M | 46.2 | 5.0 | N/A | 0.4 | 0.3 * | 4b | 2 | N/A | N/A | Y | Y | N/A |
| 15 | 75 | F | 76.9 | 3.0 | 46.2 | 1.0 * | 1.7 | 4a | 4a | 7.3 | 8.6 | N | N | 19 |
| 16 | 86 | F | 7.7 | 7.0 | 15.4 | 1.7 | 0.8 * | 4b | 4a | N/A | 10.7 | N | N | 18 |
| 17 | 89 | M | 0.0 | N/A | 76.9 | 1.7 | 1.7 * | 4a | 4a | 17.0 | 12.7 | N | N | N/A |
| 18 | 85 | M | 38.5 | 6.0 | 92.3 | 2.3 | 1.3 * | 4b | 4b | N/A | N/A | N | N | 16 |
| Proportion of Patients That Fixated First on the Area (%) | Proportion of Patients That Included the Area in Their Initial 5-Fixation Sequence (%) | |||
|---|---|---|---|---|
| Fixation Area | AMD (N = 14) | Controls (N = 16) | AMD (N = 14) | Controls (N = 16) |
| Face border, forehead, ears | 7% | 6% | 57% * | 13% * |
| Eyes and near the eyes | 43% | 31% | 64% | 75% |
| Nose or near the nose | 50% | 50% | 86% | 100% |
| Mouth or near the mouth | 0% | 6% | 50% | 88% |
| Chin | 0% | 6% | 29% | 19% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Žugelj, N.; Peterlin, L.; Muznik, U.; Klobučar, P.; Jaki Mekjavić, P.; Vidović Valentinčić, N.; Fakin, A. Face Recognition Characteristics in Patients with Age-Related Macular Degeneration Determined Using a Virtual Reality Headset with Eye Tracking. J. Clin. Med. 2024, 13, 636. https://doi.org/10.3390/jcm13020636
Žugelj N, Peterlin L, Muznik U, Klobučar P, Jaki Mekjavić P, Vidović Valentinčić N, Fakin A. Face Recognition Characteristics in Patients with Age-Related Macular Degeneration Determined Using a Virtual Reality Headset with Eye Tracking. Journal of Clinical Medicine. 2024; 13(2):636. https://doi.org/10.3390/jcm13020636
Chicago/Turabian StyleŽugelj, Nina, Lara Peterlin, Urša Muznik, Pia Klobučar, Polona Jaki Mekjavić, Nataša Vidović Valentinčić, and Ana Fakin. 2024. "Face Recognition Characteristics in Patients with Age-Related Macular Degeneration Determined Using a Virtual Reality Headset with Eye Tracking" Journal of Clinical Medicine 13, no. 2: 636. https://doi.org/10.3390/jcm13020636
APA StyleŽugelj, N., Peterlin, L., Muznik, U., Klobučar, P., Jaki Mekjavić, P., Vidović Valentinčić, N., & Fakin, A. (2024). Face Recognition Characteristics in Patients with Age-Related Macular Degeneration Determined Using a Virtual Reality Headset with Eye Tracking. Journal of Clinical Medicine, 13(2), 636. https://doi.org/10.3390/jcm13020636






