Study of Separation and Fouling of Reverse Osmosis Membranes during Model Hydrolysate Solution Filtration
Abstract
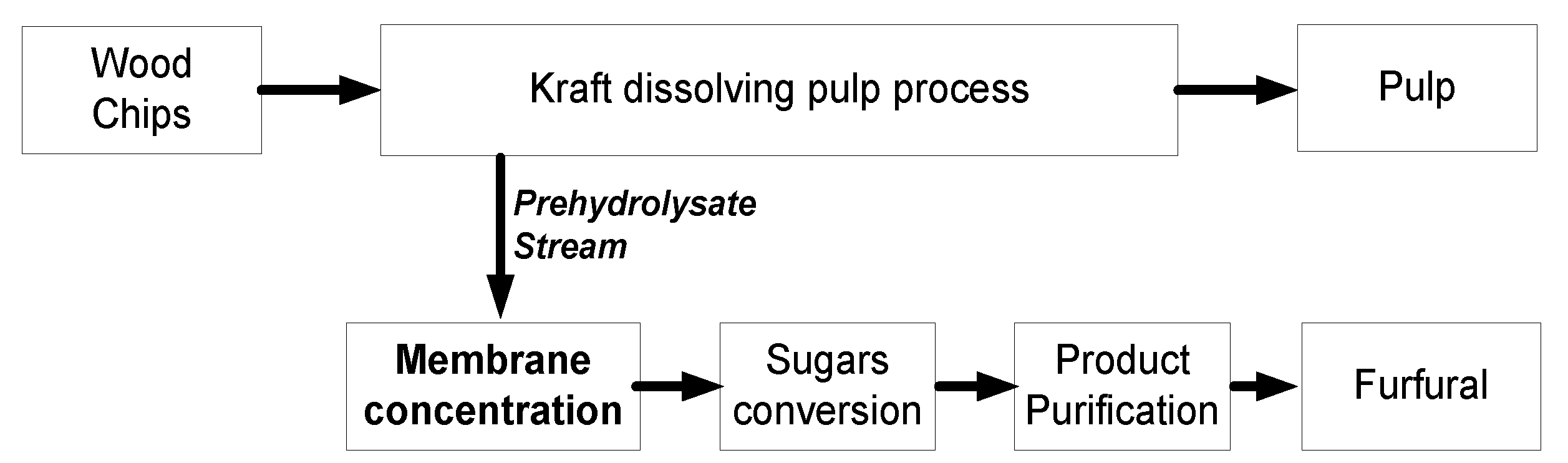
:1. Introduction
2. Materials and Methods
2.1. Membranes
2.2. Experimental Setup
2.3. Model Solution Preparation
2.4. Prehydrolysate Solution
2.5. Filtration Procedure
2.6. Membrane Cleaning Procedure
2.7. Analytical Methods
3. Theory and Computation Method
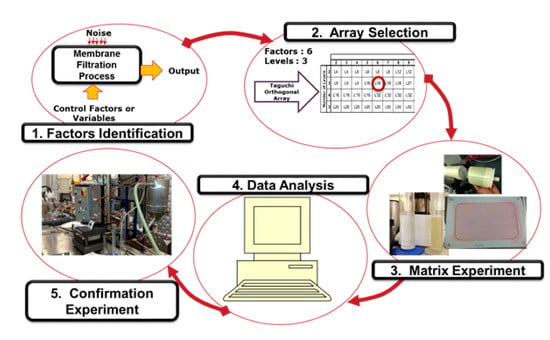
3.1. Design of Experiments (DOE) by the Taguchi Method
- Smaller is better (S/N)S, to minimize the measured response;
- Nominal is the best (S/N)N, to attain a set value for the measured response;
- Larger is better (S/N)L, to maximize the measured response.
3.2. Measures of Membrane Perfomance
- organic fouling from organic compounds
- particulate fouling due to the presence of suspended and colloidal matter
- biofouling due to the growth of bacteria after their adhesion to the membrane surface
- scaling due to the precipitation of compounds that are sparingly soluble.
4. Results and Discussion
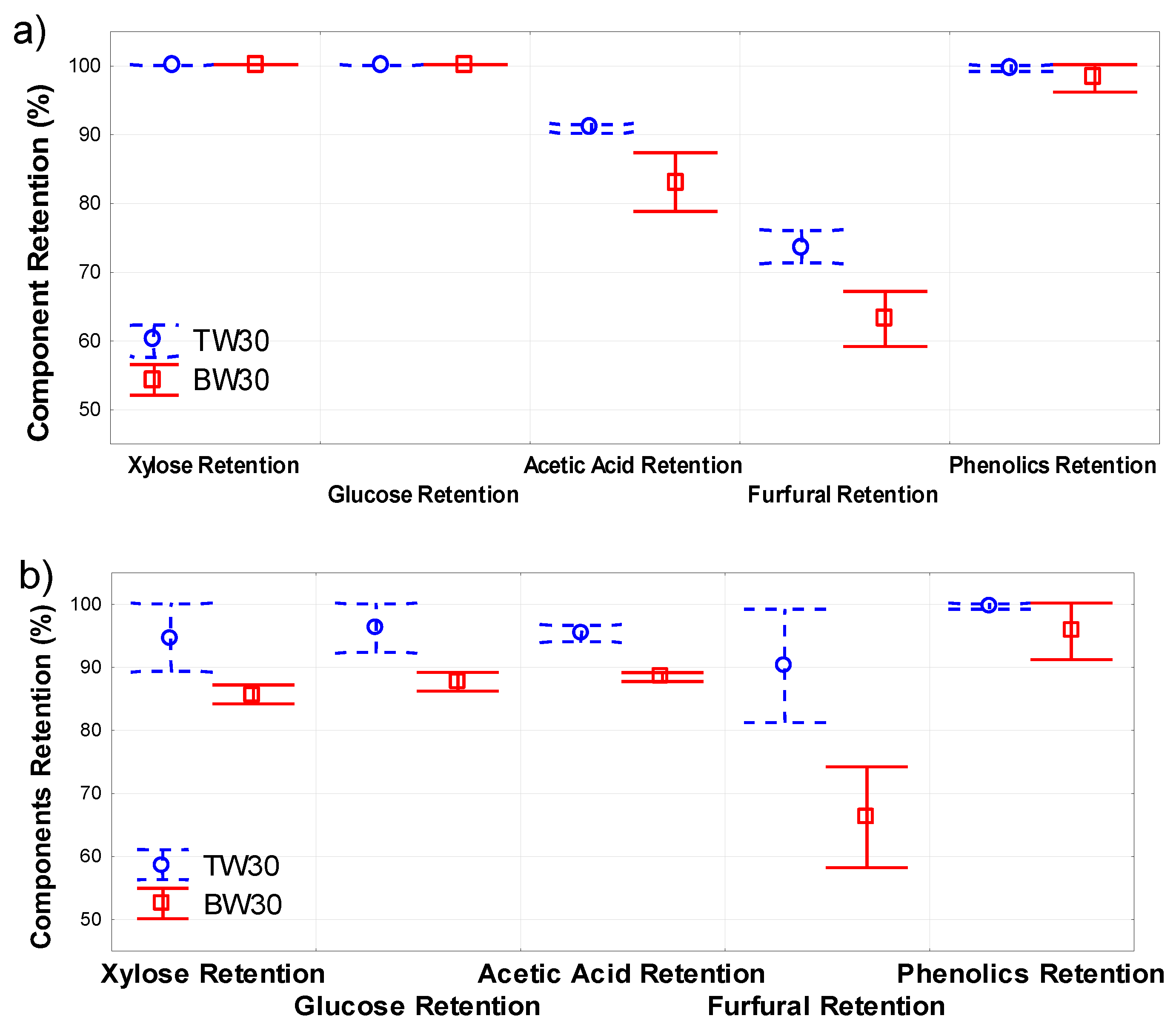
4.1. Membrane Screening
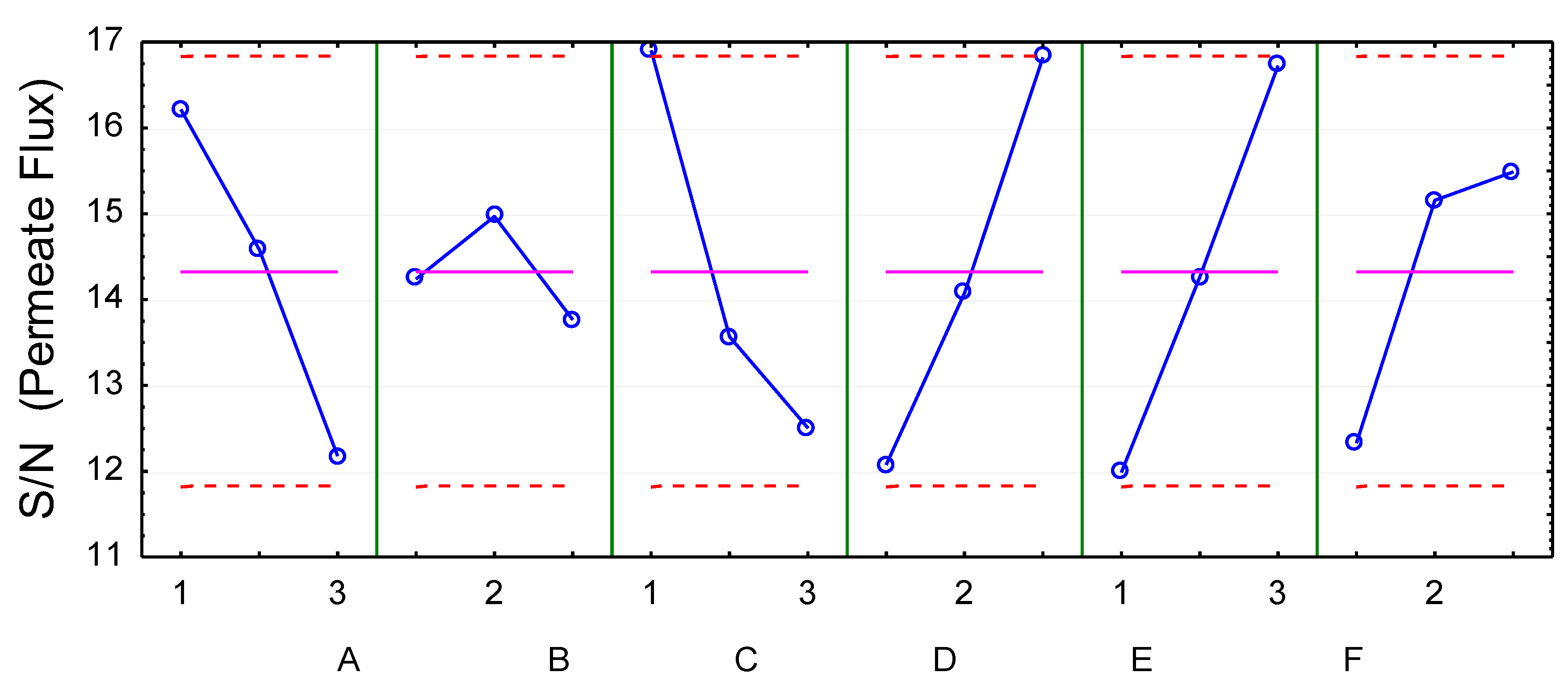
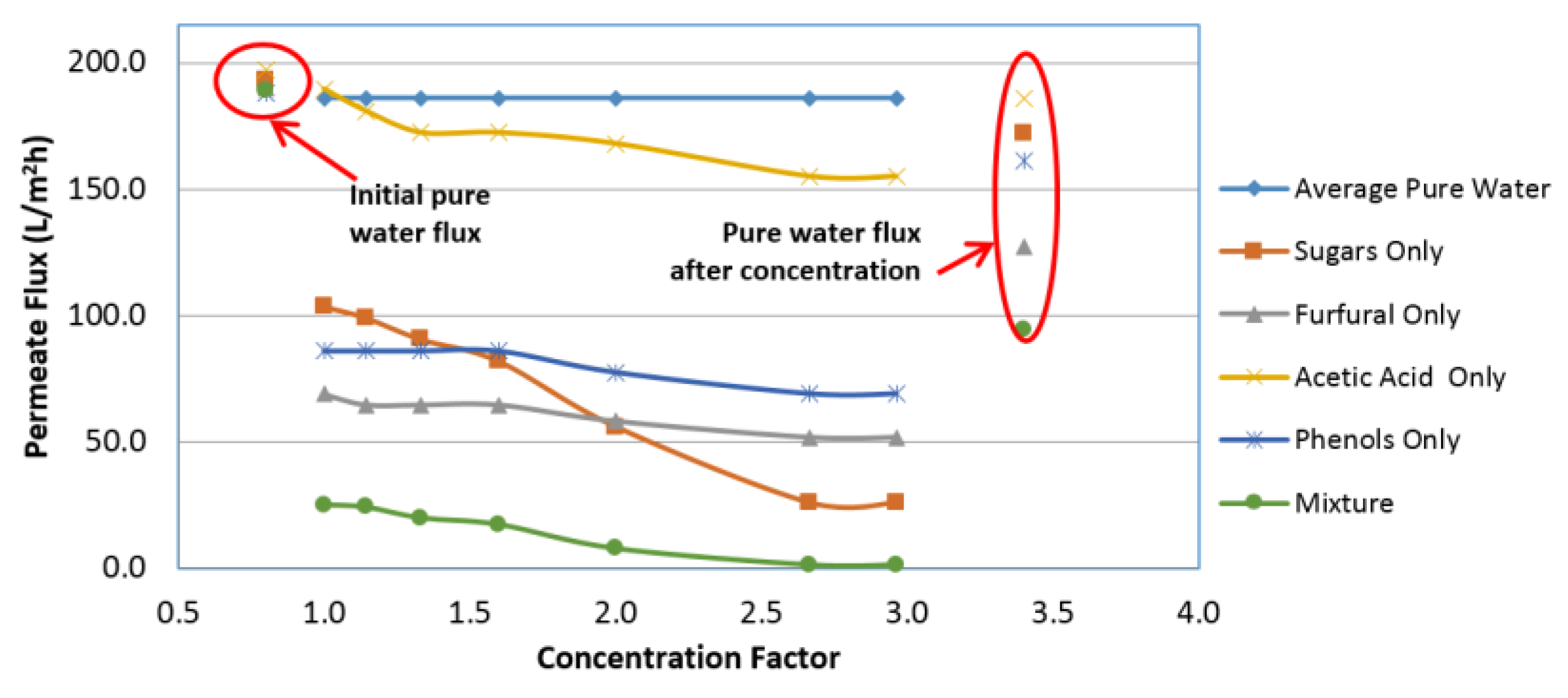
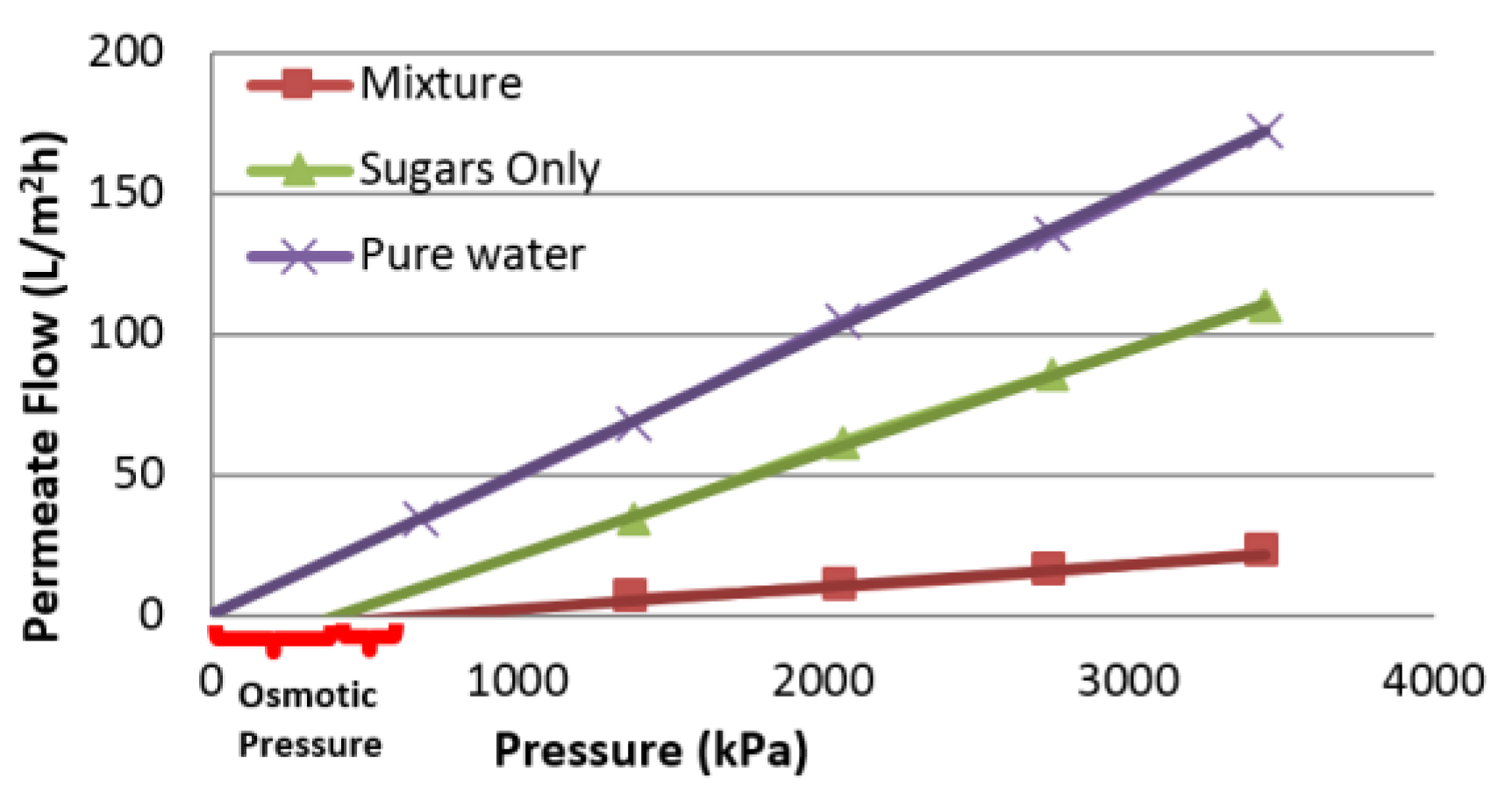
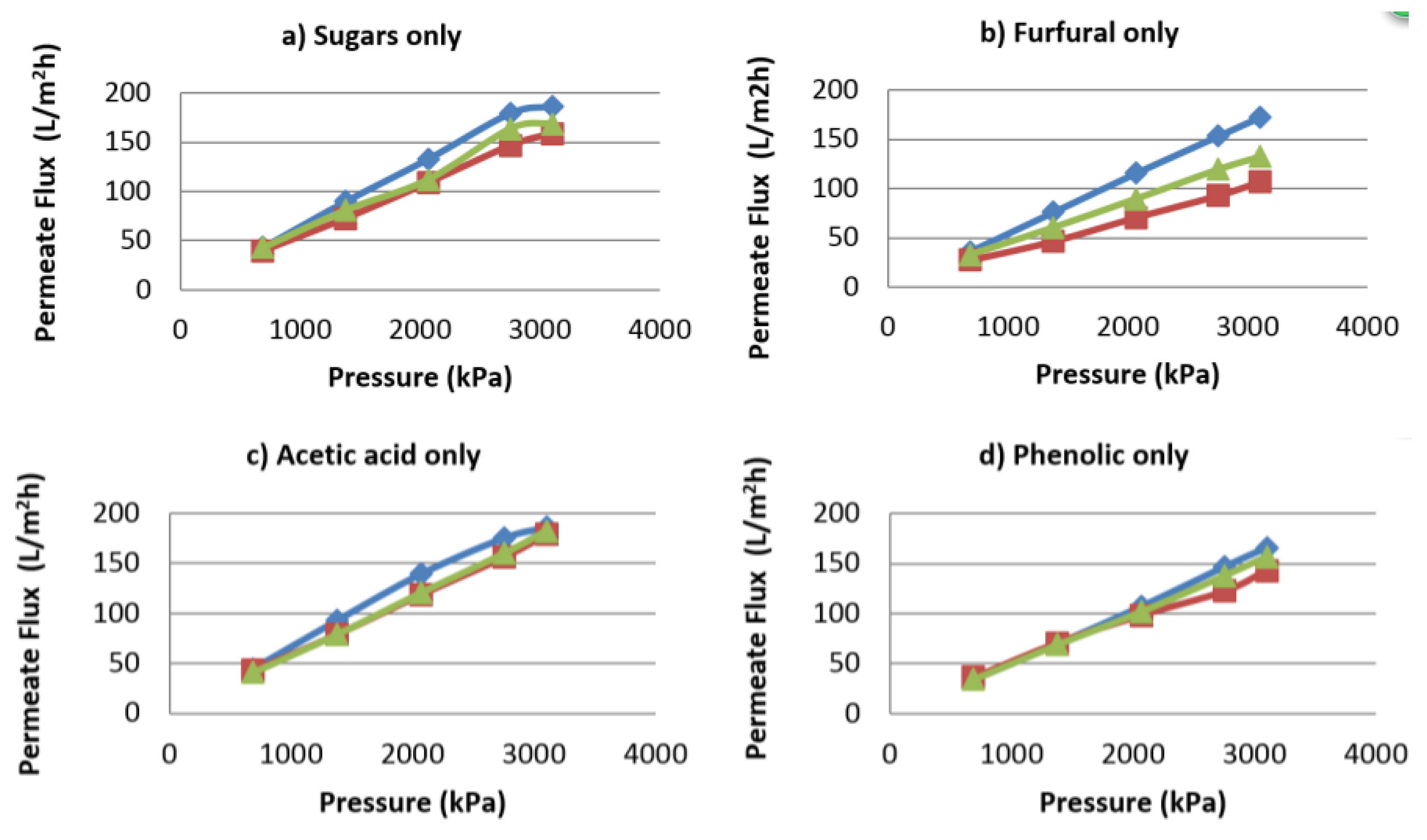
4.2. Influence of Variables on Permeate Flux
4.3. Influence of Variables on Sugar Retention
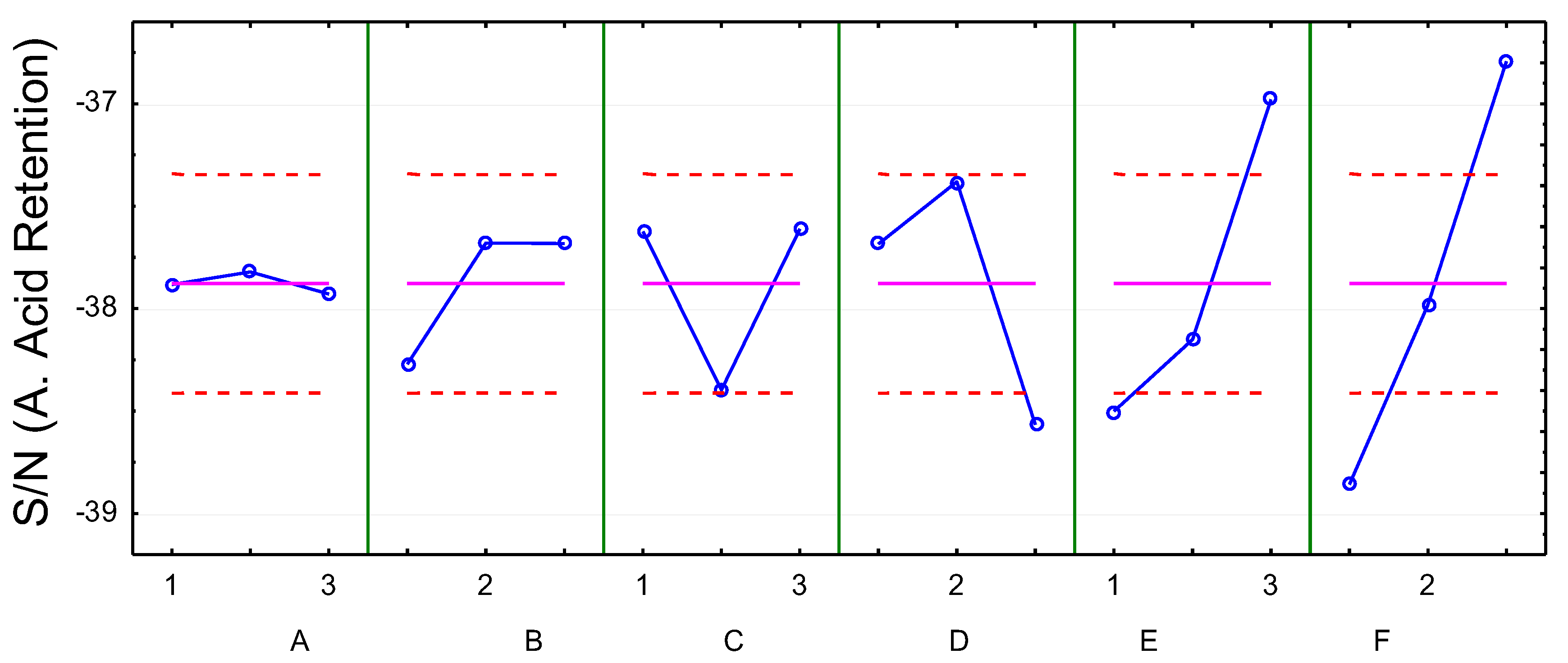
4.4. Influence of Variables on Acetic Acid Retention
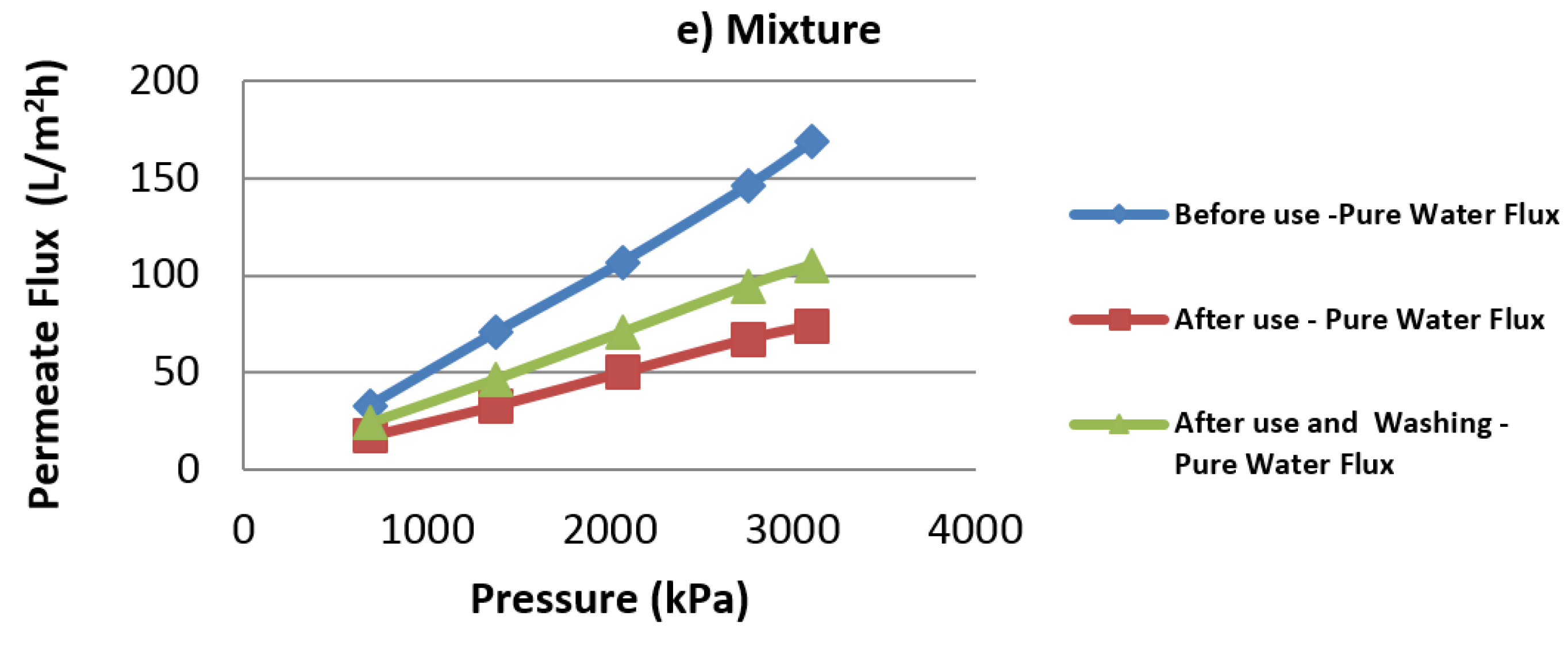
4.5. Influence of Variables on Flux Decline
4.6. Comparison with Filtration of Real Prehydrolysate Solution
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| SS | DF | MS | F | p | I (%) | ||
|---|---|---|---|---|---|---|---|
| 1 | A (Furfural concentration) | 35.7 | 2 | 17.9 | 1.4 | 0.3 | 10.8 |
| 2 | B (Acetic acid concentration) | 5.2 | 2 | 2.6 | 0.2 | 0.8 | 1.6 |
| 3 | C (Phenolics concentration) | 51.8 | 2 | 25.9 | 2.1 | 0.2 | 15.6 |
| 4 | D (Temperature) | 67.7 | 2 | 33.9 | 2.7 | 0.2 | 20.4 |
| 5 | E (Pressure) | 85.6 | 2 | 42.8 | 3.5 | 0.1 | 25.8 |
| 6 | F (Cross-flow velocity) | 23.5 | 2 | 11.7 | 0.9 | 0.4 | 7.1 |
| Residue | 61.9 | 5 | 12.4 | 18.7 |
| SS | DF | MS | F | p | I (%) | ||
|---|---|---|---|---|---|---|---|
| 1 | A (Furfural concentration) | 0.3 | 2 | 0.2 | 1.7 | 0.3 | 15 |
| 2 | B (Acetic acid concentration) | 0.3 | 2 | 0.1 | 1.5 | 0.3 | 13 |
| 3 | C (Phenolics concentration) | 0.3 | 2 | 0.2 | 1.7 | 0.3 | 14.5 |
| 4 | D (Temperature) | 0.2 | 2 | 0.1 | 1.1 | 0.4 | 9.8 |
| 5 | E (Pressure) | 0.1 | 2 | 0 | 0.3 | 0.7 | 3 |
| 6 | F (Cross-flow velocity) | 0.5 | 2 | 0.3 | 2.7 | 0.2 | 23.1 |
| Residue | 0.5 | 5 | 0.1 | 21.7 |
| SS | DF | MS | F | P | I (%) | ||
|---|---|---|---|---|---|---|---|
| 1 | A (Furfural concentration) | 0.03 | 2 | 0.02 | 0.04 | 0.96 | 0.1 |
| 2 | B (Acetic acid concentration) | 1.40 | 2 | 0.70 | 1.61 | 0.29 | 4.7 |
| 3 | C (Phenolics concentration) | 2.4 | 2 | 1.22 | 2.82 | 0.15 | 8.3 |
| 4 | D (Temperature) | 4.6 | 2 | 2.27 | 5.28 | 0.06 | 15.5 |
| 5 | E (Pressure) | 7.6 | 2 | 3.81 | 8.84 | 0.02 | 25.9 |
| 6 | F (Cross-flow velocity) | 12.9 | 2 | 06.45 | 14.94 | 0.01 | 43.8 |
| Residue | 2 | 5 | 0.43 | 1.7 |
| SS | DF | MS | F | P | I (%) | ||
|---|---|---|---|---|---|---|---|
| 1 | A (Furfural concentration) | 0.04 | 2 | 0.02 | 0.04 | 0.96 | 0.1 |
| 2 | B (Acetic acid concentration) | 1.39 | 2 | 0.70 | 1.62 | 0.29 | 4.5 |
| 3 | C (Phenolics concentration) | 2.43 | 2 | 1.22 | 2.82 | 0.15 | 7.8 |
| 4 | D (Temperature) | 4.55 | 2 | 2.28 | 5.28 | 0.06 | 14.6 |
| 5 | E (Pressure) | 7.62 | 2 | 3.81 | 8.84 | 0.02 | 24.5 |
| 6 | F (Cross-flow velocity) | 12.89 | 2 | 6.45 | 14.95 | 0.01 | 41.5 |
| Residue | 2.16 | 5 | 0.43 | 6.9 |

References
- Benali, M.; Périn-Levasseur, Z.; Savulescu, L.; Kouisni, L.; Jemaa, N.; Kudra, T.; Paleologou, M. Implementation of Lignin-Based Biorefinery into a Canadian Softwood Kraft Pulp Mill: Optimal Resources Integration and Economic Viability Assessment. Biomass Bioenergy 2013, 67, 473–482. [Google Scholar] [CrossRef]
- Mateos-Espejel, E.; Radiotis, T.; Jemaa, N. Implications of Converting a Kraft Pulp Mill to a Dissolving Pulp Operation with a Hemicellulose Extraction Stage. Tappi J. 2013, 12, 29–38. [Google Scholar]
- Kang, L.; Lee, Y.Y.; Yoon, S.; Smith, A.J.; Krishnagopalan, G.A. Ethanol Production from the Mixture of Hemicellulose Prehydrolysate and Paper Sludge. BioResources 2012, 7, 3607–3626. [Google Scholar]
- Van Heiningen, A. Lignocellulosic Biorefinery Research; A Personal View. In Proceedings of the 63rd Canadian Chemical Engineering Conference, Fredericton, NB, Canada, 20–23 October 2013. [Google Scholar]
- Ajao, O.; Marinova, M.; Savadogo, O.; Paris, J. Process for Cost and Energy Efficient Production of Furfural from Kraft Hardwood Pre-Hydrolysate. J. Bioprocess Eng. Biorefin. 2014, 3, 296–307. [Google Scholar] [CrossRef]
- Marcotullio, G. The Chemistry and Technology of Furfural Production in Modern Lignocellulose-Feedstock Biorefineries. Ph.D. Thesis, TU Delft, Delft, The Netherlands, 2011. [Google Scholar]
- Lamminpää, K.; Ahola, J.; Tanskanen, J. Kinetics of Xylose Dehydration into Furfural in Formic Acid. Ind. Eng. Chem. Res. 2012, 51, 6297–6303. [Google Scholar] [CrossRef]
- Nakari, O.; Pihlajamäki, A.; Mänttäri, M. Permeability of Dilute Ionic Liquid Solutions through a Nanofiltration Membrane—Effect of Ionic Liquid Concentration, Filtration Pressure and Temperature. Sep. Purif. Technol. 2016, 163, 267–274. [Google Scholar] [CrossRef]
- Al Manasrah, M.; Kallioinen, M.; Ilvesniemi, H.; Mänttäri, M. Recovery of Galactoglucomannan from Wood Hydrolysate Using Regenerated Cellulose Ultrafiltration Membranes. Bioresour. Technol. 2012, 114, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Mänttäri, M.; Pihlajamäki, A.; Kaipainen, E.; Nyström, M. Effect of Temperature and Membrane Pre-Treatment by Pressure on the Filtration Properties of Nanofiltration Membranes. Desalination 2002, 145, 81–86. [Google Scholar] [CrossRef]
- Mänttäri, M.; Puro, L.; Nuortila-Jokinen, J.; Nyström, M. Fouling Effects of Polysaccharides and Humic Acid in Nanofiltration. J. Membr. Sci. 2000, 165, 1–17. [Google Scholar] [CrossRef]
- Jönsson, A.S. Membranes for Lignin and Hemicellulose Recovery in Pulp Mills. Membr. Technol. Biorefin. 2016, 105–133. [Google Scholar] [CrossRef]
- Persson, T.; Jönsson, A.-S. Characterization of Hemicelluloses in Process Streams in Thermomechanical and Chemi-Thermomechanical Pulp Mills. J. Wood Chem. Technol. 2017, 37, 184–190. [Google Scholar] [CrossRef]
- Krawczyk, H.; Oinonen, P.; Jönsson, A.S. Combined Membrane Filtration and Enzymatic Treatment for Recovery of High Molecular Mass Hemicelluloses from Chemithermomechanical Pulp Process Water. Chem. Eng. J. 2013, 225, 292–299. [Google Scholar] [CrossRef]
- Qi, B.; Luo, J.; Chen, X.; Hang, X.; Wan, Y. Separation of Furfural from Monosaccharides by Nanofiltration. Bioresour. Technol. 2011, 102, 7111–7118. [Google Scholar] [CrossRef] [PubMed]
- Egüés, I.; Sanchez, C.; Mondragon, I.; Labidi, J. Separation and Purification of Hemicellulose by Ultrafiltration. Ind. Eng. Chem. Res. 2011, 51, 523–530. [Google Scholar] [CrossRef]
- Sun, Z.; Liu, S. Production of N-Butanol from Concentrated Sugar Maple Hemicellulosic Hydrolysate by Clostridia Acetobutylicum Atcc824. Biomass Bioenergy 2012, 39, 39–47. [Google Scholar] [CrossRef]
- Brás, T.; Guerra, V.; Torrado, I.; Lourenço, P.; Carvalheiro, F.; Duarte, L.C.; Neves, L.A. Detoxification of Hemicellulosic Hydrolysates from Extracted Olive Pomace by Diananofiltration. Process Biochem. 2014, 49, 173–180. [Google Scholar] [CrossRef]
- Nguyen, N.; Fargues, C.; Lewandowski, R.; Guiga, W.; Lameloise, M.L. Assessing Nanofiltration and Reverse Osmosis for the Detoxification of Fermentable Solutions. Procedia Eng. 2012, 44, 1476–1478. [Google Scholar] [CrossRef]
- Teella, A.; Huber, G.W.; Ford, D.M. Separation of Acetic Acid from the Aqueous Fraction of Fast Pyrolysis Bio-Oils Using Nanofiltration and Reverse Osmosis Membranes. J. Membr. Sci. 2011, 378, 495–502. [Google Scholar] [CrossRef]
- Maiti, S.K.; Thuyavan, Y.L.; Singh, S.; Oberoi, H.S.; Agarwal, G.P. Modeling of the Separation of Inhibitory Components from Pretreated Rice Straw Hydrolysate by Nanofiltration Membranes. Bioresour. Technol. 2012, 114, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Sasaki, D.; Sakihama, Y.; Teramura, H.; Yamada, R.; Hasunuma, T.; Ogino, C.; Kondo, A. Ethanol Fermentation by Xylose-Assimilating Saccharomyces Cerevisiae Using Sugars in a Rice Straw Liquid Hydrolysate Concentrated by Nanofiltration. Bioresour. Technol. 2013, 147, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Lyu, H.; Fang, Y.; Ren, S.; Chen, K.; Luo, G.; Zhang, S.; Chen, J. Monophenols Separation from Monosaccharides and Acids by Two-Stage Nanofiltration and Reverse Osmosis in Hydrothermal Liquefaction Hydrolysates. J. Membr. Sci. 2016, 504, 141–152. [Google Scholar] [CrossRef]
- Nguyen, N.; Fargues, C.; Guiga, W.; Lameloise, M.L. Assessing Nanofiltration and Reverse Osmosis for the Detoxification of Lignocellulosic Hydrolysates. J. Membr. Sci. 2015, 487, 40–50. [Google Scholar] [CrossRef]
- Ajao, O.; le Hir, M.; Rahni, M.; Marinova, M.; Chadjaa, H.; Savadogo, O. Concentration and Detoxification of Kraft Prehydrolysate by Combining Nanofiltration with Flocculation. Ind. Eng. Chem. Res. 2015, 54, 1113–1122. [Google Scholar] [CrossRef]
- Mechmech, F.; Chadjaa, H.; Rahni, M.; Marinova, M.; Akacha, N.B.; Gargouri, M. Improvement of Butanol Production from a Hardwood Hemicelluloses Hydrolysate by Combined Sugar Concentration and Phenols Removal. Bioresour. Technol. 2015, 192, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Malmali, M.; Stickel, J.J.; Wickramasinghe, S.R. Sugar Concentration and Detoxification of Clarified Biomass Hydrolysate by Nanofiltration. Sep. Purif. Technol. 2014, 132, 655–665. [Google Scholar] [CrossRef]
- Ajao, O.; Rahni, M.; Marinova, M.; Chadjaa, H.; Savadogo, O. Retention and Flux Characteristics of Nanofiltration Membranes during Hemicellulose Prehydrolysate Concentration. Chem. Eng. J. 2015, 260, 605–615. [Google Scholar] [CrossRef]
- Luo, J.; Zeuner, B.; Morthensen, S.T.; Meyer, A.S.; Pinelo, M. Separation of Phenolic Acids from Monosaccharides by Low-Pressure Nanofiltration Integrated with Laccase Pre-Treatments. J. Membr. Sci. 2015, 482, 83–91. [Google Scholar] [CrossRef]
- Gautam, A.; Menkhaus, T.J. Performance Evaluation and Fouling Analysis for Reverse Osmosis and Nanofiltration Membranes during Processing of Lignocellulosic Biomass Hydrolysate. J. Membr. Sci. 2014, 451, 252–265. [Google Scholar] [CrossRef]
- Hwang, K.-J.; Chen, K.-S.; Chen, S.-T.; Chang, K.-L. Sugar Purification from Enzymatic Rice Straw Hydrolysis Products Using Cross-Flow Diafiltration. Sep. Sci. Technol. 2012, 47, 52–61. [Google Scholar] [CrossRef]
- Sueb, M.S.M.; Zdarta, J.; Jesionowski, T.; Jonsson, G.; Meyer, A.S.; Jørgensen, H.; Pinelo, M. High-Performance Removal of Acids and Furans from Wheat Straw Pretreatment Liquid by Diananofiltration. Sep. Sci. Technol. 2017, 52, 1901–1912. [Google Scholar] [CrossRef]
- Almanasrah, M. Hot Water Extraction and Membrane Filtration Processes in Fractionation and Recovery of Value-Added Compounds from Wood and Plant Residues. Ph.D. Thesis, Lappeenranta University of Technology, Lappeenranta, Finland, 2017. [Google Scholar]
- Krawczyk, H.; Arkell, A.; Jönsson, A.S. Impact of Prefiltration on Membrane Performance during Isolation of Hemicelluloses Extracted from Wheat Bran. Sep. Purif. Technol. 2013, 116, 192–198. [Google Scholar] [CrossRef]
- González-Muñoz, M.J.; Rivas, S.; Santos, V.; Parajó, J.C. Fractionation of Extracted Hemicellulosic Saccharides from Pinus Pinaster Wood by Multistep Membrane Processing. J. Membr. Sci. 2013, 428, 281–289. [Google Scholar] [CrossRef]
- Koivula, E.; Kallioinen, M.; Preis, S.; Testova, L.; Sixta, H.; Mänttäri, M. Evaluation of Various Pretreatment Methods to Manage Fouling in Ultrafiltration of Wood Hydrolysates. Sep. Purif. Technol. 2011, 83, 50–56. [Google Scholar] [CrossRef]
- Saadatmand, S.; Edlund, U.; Albertsson, A.; Danielsson, S.; Dahlman, O. Prehydrolysis in Softwood Pulping Produces a Valuable Biorefinery Fraction for Material Utilization. Environ. Sci. Technol. 2012, 46, 8389–8396. [Google Scholar] [CrossRef] [PubMed]
- Edlund, U.; Ryberg, Y.Z.; Albertsson, A. Barrier Films from Renewable Forestry Waste. Biomacromolecules 2010, 11, 2532–2538. [Google Scholar] [CrossRef] [PubMed]
- Khayati, G.; Gilani, H.G.; Keyvani, Z.S. Extraction of Cu(Ii) Ions from Aqueous Media Using Peg/Sulphate Salt Aqueous Two-Phase System. Sep. Sci. Technol. 2016, 51, 601–608. [Google Scholar] [CrossRef]
- Mogi, N.; Sugai, E.; Fuse, Y.; Funazukuri, T. Infinite Dilution Binary Diffusion Coefficients for Six Sugars at 0.1 Mpa and Temperatures from (273.2 to 353.2) K. J. Chem. Eng. Data 2006, 52, 40–43. [Google Scholar] [CrossRef]
- Haynes, W.M. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Yui, K.; Yamazaki, N.; Funazukuri, T. Infinite Dilution Binary Diffusion Coefficients for Compounds Derived from Biomass in Water at 0.1 Mpa and Temperatures from (298.2 to 353.2) K. J. Chem. Eng. Data 2012, 58, 183–186. [Google Scholar] [CrossRef]
- Ragnar, M.; Lindgren, C.T.; Nilvebrant, N. Pka-Values of Guaiacyl and Syringyl Phenols Related to Lignin. J. Wood Chem. Technol. 2000, 20, 277–305. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Wang, K.-S.; Chen, J.-H.; Huang, Y.-H.; Huang, S.-L. Integrated Taguchi Method and Response Surface Methodology to Confirm Hydrogen Production by Anaerobic Fermentation of Cow Manure. Int. J. Hydrog. Energy 2013, 38, 45–53. [Google Scholar] [CrossRef]
- Chowdhury, A.; Chakraborty, R.; Mitra, D.; Biswas, D. Optimization of the Production Parameters of Octyl Ester Biolubricant Using Taguchi’s Design Method and Physico-Chemical Characterization of the Product. Ind. Crops Prod. 2014, 52, 783–789. [Google Scholar] [CrossRef]
- Mohagheghi, E.; Alemzadeh, I.; Vossoughi, M. Study and Optimization of Amino Acid Extraction by Emulsion Liquid Membrane. Sep. Sci. Technol. 2008, 43, 3075–3096. [Google Scholar] [CrossRef]
- Roy, R.K. Design of Experiments Using the Taguchi Approach: 16 Steps to Product and Process Improvement; Wiley: Chichester, NY, USA, 2001. [Google Scholar]
- Sundaramoorthy, S.; Srinivasan, G.; Murthy, D.V.R. An Analytical Model for Spiral Wound Reverse Osmosis Membrane Modules: Part II—Experimental Validation. Desalination 2011, 277, 257–264. [Google Scholar] [CrossRef]
- Stade, S.; Kallioinen, M.; Mikkola, A.; Tuuva, T.; Mänttäri, M. Reversible and Irreversible Compaction of Ultrafiltration Membranes. Sep. Purif. Technol. 2013, 118, 127–134. [Google Scholar] [CrossRef]
- Mareth, B. A Reverse Osmosis Treatment Process for Produced Water: Optimization, Process Control, and Renewable Energy Application. Master’s Thesis, Texas A&M University, College Station, TX, USA, 2006. [Google Scholar]
- Alhadidi, A.; Blankert, B.; Kemperman, A.J.B.; Schippers, J.C.; Wessling, M.; van der Meer, W.G.J. Effect of Testing Conditions and Filtration Mechanisms on SDI. J. Membr. Sci. 2011, 381, 142–151. [Google Scholar] [CrossRef]
- Marriott, J.I. Detailed Modelling and Optimal Design of Membrane Separation Systems. Ph.D. Thesis, University of London, London, UK, 2001. [Google Scholar]
- Verliefde, A.R.D.; Cornelissen, E.R.; Heijman, S.G.J.; Verberk, J.Q.J.C.; Amy, G.L.; van der Bruggen, B.; van Dijk, J.C. The Role of Electrostatic Interactions on the Rejection of Organic Solutes in Aqueous Solutions with Nanofiltration. J. Membr. Sci. 2008, 322, 52–66. [Google Scholar] [CrossRef]


| Memb. Type | Prehydrolysate Type (Compounds Present) | Objectives | Key Results/Observations | Ref. |
|---|---|---|---|---|
| NF | Synthetic solution: (xylose, glucose, furfural) | Sugar purification for ethanol | Concentration and purification can be accomplished | [15] |
| UF | Corn waste hydrolysis liquor: (glucose, xylose, arabinose, and acetic acid) | Hydrolysate purification bioproducts | Ultrafiltration can be used for hemicellulose fractionation and purification | [16] |
| NF | Hemicellulosic hydrolysate | Inhibitor removal for butanol | Removal of nearly all small molecular organic acids, furfural, and HMF is possible | [17] |
| NF | Rice straw hydrolysate (glucose, xylose, arabinose, cellobiose, fructose, sucrose, acetic acid, HMF, rurfural, ferulic acid, vanilic acid) | Inhibitor removal | Simultaneous concentration of sugars and separation of inhibitors achievable | [21] |
| NF | Three sugar solution (glucose solution, diluted sugar beet molasses, and liquid hydrolysate of dilute acid-pretreated rice straw, glucose, xylose, acetate formate, furfural, and HMF) | Sugar concentration and inhibitor removal | Sugars can be concentrated and fermentation inhibitors removed at low pressures prior to successful fermentation | [22] |
| RO & NF | Hydrothermal iquefaction (HTL) hydrolysates (glucose, xylose, acetic acid, lactic acid, levulinic acid, phenol, 2-methoxyphenol, and 2,6-dimethoxyphenol) | Hydrolysate fractionation | Two-stage membrane process is feasible for fractionating model HTL hydrolysates | [23] |
| RO & NF | Lignocellulosic hydrolysate model solution (C5 and C6 sugars from acetic acid, furfural, 5-hydroxymethyl furfural, and vanillin in a model solution) | Inhibitor removal | RO had the highest sugar retention but inhibitor removal was lower than for NF | [24] |
| RO & NF | Hemicelluloses prehydrolysate (glucose, mannose, galactose, xylose, arabinose, acetic acid, furfural) | Inhibitor removal | Membrane filtration not efficient for phenolic inhibitors removal except in combination with flocculation | [25,26] |
| RO & NF | Corn stover hydrolysate (glucose, xylose, acetic acid, furfural, and HMF) Model solution (glucose, xylose, acetic acid, furfural, and HMF) | Inhibitor removal | Hydrolysis degradation products can be removed, but membrane surface characteristics play a role | [27] |
| RO & NF | Hemicellulose prehydrolysate (glucose, mannose, galactose, xylose, arabinose, acetic acid, furfural) | Sugar and inhibitor concentration | Retention and flux characteristics determined, but no indication of the impact of components | [28] |
| NF | Model solution (vanillic acid, p-coumaric acid, ferulic acid, xylose, arabinose, and glucose) | Phenolics removal | Enzymes can be used to polymerize phenolic compounds and facilitate their separation from sugars | [29] |
| RO & NF | Lignocellulosic hydrolysate mix (glucose, xylose, mannose, galactose, and arabinose, furfural, HMF, acetic, and other unidentified organic acids) Synthetic pure sugar mix (glucose, xylose, mannose, galactose, and arabinose) | Sugar concentration and inhibitor removal | Higher inhibitor separation comes with sugar losses, and reversible fouling was mainly responsible for flux reduction | [30] |
| UF & MF | Rice straw hydrolysate (sugar mix indicated by reducing sugars) | Sugar recovery and inhibitor removal | The effects of membrane type, pore size, cross-flow velocity, and transmembrane pressure on the filtration flux, and sugar rejection elucidated | [31] |
| NF | Wheat straw pretreatment liquor (mono and oligosaccharides, acids and furans) Model solutions (sodium chloride, potassium chloride, acetic acid, formic acid, 5-hydroxymethylfurfural (HMF), d-xylose, l-arabinose, d-glucose, and xylan) | Acid and furan removal | Diananofiltration strategy shown to be promising for the recovery of high-purity streams of monosaccharides | [32] |
| RO, NF & UF | Spruce wood autohydrolysate | Recovery of hemicelluloses | Diafiltration and pulsed corona discharge (PCD) improves recovery | [33] |
| UF | Wheat bran hemicelluloses (araboxylan) solution | Concentration and purification | Product purity and ultrafiltration performance can be improved by dead-end prefiltration | [34] |
| NF & UF | Pine wood autohydrolysis liquor from containing poly- and oligosaccharides (POHS), and monosaccharides | Concentration, purification, and fractionation | The purified POHS/monosaccharides ratio can be altered by different membrane combinations | [35] |
| UF | Birch chips and spruce saw-dust hydrolysate | Fouling reduction while removing inhibitors | Pulsed corona discharge (PCD) and activated carbon treatments reduces fouling | [36] |
| Chemical | d-Glucose | d-Xylose | Acetic Acid | Syringaldehyde | Furfural |
|---|---|---|---|---|---|
| Formula | C6H12O6 | C5H10O5 | C2H4O2 | C9H10O4 | C5H4O2 |
| Molecular Structure |  |  |  |  |  |
| MW (g/mol) | 180.16 | 150.13 | 60.05 | 182.17 | 96.08 |
| D (×10−5 cm2/s) | 0.67 [40] | 0.75 [40] | 1.29 [41] | n/a | 1.01 [42] |
| pKa | 12.46 [41] | 12.14 [41] | 4.76 [41] | 7.34 [43] | n/a |
| Controlling Factors | Levels | Units | ||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| A (Furfural concentration) | 0.6 | 1.8 | 3.5 | g/L |
| B (Acetic acid concentration) | 0.5 | 3.5 | 10 | g/L |
| C (Phenolics concentration) | 0.3 | 2.8 | 6 | g/L |
| D (Temperature) | 20 | 30 | 40 | °C |
| E (Pressure) | 3100 | 3800 | 4500 | kPa |
| F (Cross-flow velocity) | 0.3 | 0.4 | 0.5 | m/s |
| Exp Nrs. | Levels of parameters | RS | RA | JP-i | JP-i/JP-f | pH | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | (%) | (%) | (L/m2h) | |||
| 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0.95 | 0.74 | 14 | 32 | 3.65 |
| 2 | 1 | 2 | 2 | 2 | 2 | 2 | 0.92 | 0.70 | 26 | 5 | 3.25 |
| 3 | 1 | 3 | 3 | 3 | 3 | 3 | 1.00 | 0.78 | 38 | 3 | 2.68 |
| 4 | 2 | 1 | 1 | 2 | 2 | 3 | 1.00 | 0.83 | 28 | 3 | 3.25 |
| 5 | 2 | 2 | 2 | 3 | 3 | 1 | 0.86 | 0.88 | 24 | 18 | 2.67 |
| 6 | 2 | 3 | 3 | 1 | 1 | 2 | 0.98 | 0.81 | 9 | 4 | 2.69 |
| 7 | 3 | 1 | 2 | 1 | 3 | 2 | 0.99 | 0.81 | 16 | 4 | 3.23 |
| 8 | 3 | 2 | 3 | 2 | 1 | 3 | 0.98 | 0.79 | 10 | 3 | 2.94 |
| 09 | 3 | 3 | 1 | 3 | 2 | 1 | 0.97 | 0.61 | 21 | 16 | 2.70 |
| 10 | 1 | 1 | 3 | 3 | 2 | 2 | 0.90 | 0.62 | 35 | 21 | 3.28 |
| 11 | 1 | 2 | 1 | 1 | 3 | 3 | 0.99 | 0.79 | 57 | 2 | 3.36 |
| 12 | 1 | 3 | 2 | 2 | 1 | 1 | 0.95 | 0.66 | 17 | 10 | 2.66 |
| 13 | 2 | 1 | 2 | 3 | 1 | 3 | 0.97 | 0.71 | 31 | 7 | 3.06 |
| 14 | 2 | 2 | 3 | 1 | 2 | 1 | 0.96 | 0.91 | 16 | 5 | 2.79 |
| 15 | 2 | 3 | 1 | 2 | 3 | 2 | 1.00 | 0.79 | 47 | 3 | 2.61 |
| 16 | 3 | 1 | 3 | 2 | 3 | 1 | 0.98 | 0.84 | 17 | 4 | 3.20 |
| 17 | 3 | 2 | 1 | 3 | 1 | 2 | 0.99 | 0.70 | 34 | 6 | 2.85 |
| 18 | 3 | 3 | 2 | 1 | 2 | 3 | 1.00 | 0.91 | 14 | 4 | 2.66 |
| Expt Nr. | Description | A (g/L) | B (g/L) | C (g/L) | Sugars | |
|---|---|---|---|---|---|---|
| X (g/L) | G (g/L) | |||||
| 1 | Sugars only | 0 | 0 | 0 | 35 | 10 |
| 2 | Acetic acid only | 3.5 | 0 | 0 | 0 | 0 |
| 3 | Furfural only | 0 | 3.5 | 0 | 0 | 0 |
| 4 | Phenols only | 0 | 0 | 2.8 | 0 | 0 |
| 5 | Mixture | 3.5 | 3.5 | 2.8 | 35 | 10 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajao, O.; Rahni, M.; Marinova, M.; Chadjaa, H.; Savadogo, O. Study of Separation and Fouling of Reverse Osmosis Membranes during Model Hydrolysate Solution Filtration. Membranes 2017, 7, 68. https://doi.org/10.3390/membranes7040068
Ajao O, Rahni M, Marinova M, Chadjaa H, Savadogo O. Study of Separation and Fouling of Reverse Osmosis Membranes during Model Hydrolysate Solution Filtration. Membranes. 2017; 7(4):68. https://doi.org/10.3390/membranes7040068
Chicago/Turabian StyleAjao, Olumoye, Mohamed Rahni, Mariya Marinova, Hassan Chadjaa, and Oumarou Savadogo. 2017. "Study of Separation and Fouling of Reverse Osmosis Membranes during Model Hydrolysate Solution Filtration" Membranes 7, no. 4: 68. https://doi.org/10.3390/membranes7040068