Nanofiltration of Mine Water: Impact of Feed pH and Membrane Charge on Resource Recovery and Water Discharge
Abstract
:1. Introduction
| Authors | Membrane | pH range | Solution | IEP |
|---|---|---|---|---|
| Childress and Elimelech [20] | NF 70 | 2–9 | 0.01 M NaCl | 4 |
| 0.01 M NaCl + 0.001 M CaCl2 | 3–3.5 | |||
| 0.01 M NaCl + 0.001 M Na2SO4 | 4 | |||
| 0.01 M NaCl + 0.001 M MgSO4 | – | |||
| TFCS | 2–9 | 0.01 M NaCl | 3 | |
| 0.01 M NaCl + 0.001 M CaCl2 | 3.5 | |||
| 0.01 M NaCl + 0.001 M Na2SO4 | 3 | |||
| 0.01 M NaCl + 0.001 M MgSO4 | 3 | |||
| Hagmeyer and Gimbel [21] | Desal 5 DK | 3–11 | 0.002 M KCl | 4 |
| NTR-729 | 3–11 | 0.002 M KCl | 4 | |
| Childress and Elimelech [16] | NF 55 | 3–9 | 0.01 M NaCl | 3.2 |
| 0.01 M NaCl + 2 mg L−1 humic acids | no IEP | |||
| 0.01 M NaCl + 1 mM surfactants | no IEP | |||
| Tanninen et al. [22] | NF 270 | – | 0.001 M KCl | 3.3 |
| Desal 5 DK | – | 0.001 M KCl | 4.1 | |
| Desal KH | – | 0.001 M KCl | 4.9 | |
| BTP-NF-1 | – | 0.001 M KCl | 6 | |
| BTP-NF-2 | – | 0.001 M KCl | 5.4 | |
| Artug [15] | NF 270 | 2.5–7 | 0.001 M NaCl | 2.8 |
| 0.001 M CaCl2 | 3.5 | |||
| NF 90 | 2.5–7 | 0.001 M NaCl | 4.3 | |
| 0.001 M CaCl2 | 4.3 | |||
| NF PES 10 | 2.5–7 | 0.001 M NaCl | 3.4 | |
| 0.001 M CaCl2 | 3.5 | |||
| NF 2 | 2.5–7 | 0.001 M NaCl | 3.2 | |
| 0.001 M CaCl2 | 2.9 |
2. Experimental Section
2.1. Membranes and Mine Water Samples
2.2. Methods
2.2.1. IEP Tests
| Parameter | Unit | MW A | MW B | MW C | MW D |
|---|---|---|---|---|---|
| pH | – | 4.56 | 5.50 | 4.10 | 2.60 |
| Aluminium, Al3+ | mg L−1 | 14 | 0.4 | NM | NM |
| Calcium, Ca2+ | mg L−1 | 480 | 260 | 280 | 270 |
| Copper, Cu2+ | mg L−1 | 410 | 270 | 610 | 590 |
| Iron, Fe3+ | mg L−1 | 0.14 | 0.02 | NM | NM |
| Potassium, K+ | mg L−1 | 310 | 340 | NM | NM |
| Magnesium, Mg2+ | mg L−1 | 770 | 870 | 900 | 900 |
| Manganese, Mn3+ | mg L−1 | 440 | 420 | 530 | 500 |
| Sodium, Na+ | mg L−1 | 2000 | 3000 | 3800 | 3600 |
| Sulphate, SO42− | mg L−1 | 6900 | 8700 | 10,500 | 10,200 |
| Chloride, Cl− | mg L−1 | 2300 | NM | 3000 | 2900 |
| Type of Test | Feed Sample | NF Membrane | Feed Flow (L h−1) | Feed Temperature (°C) | Feed Pressure (bar) | Permeate Flux Rate (L m−2 h−1) |
|---|---|---|---|---|---|---|
| IEP Test | NaCl-Na2SO4 | NF 270 | 200 | 37 ± 4.1 | 20 ± 0.0 | 130 ± 0.0 |
| IEP Test | NaCl-Na2SO4 | TS 80 | 225 | 25 ± 0.0 | 10 ± 0.5 | 33 ± 5.8 |
| Feed pH Test | MW A | NF 270 | 200 | 25 ± 0.6 | 7 ± 1.2 | 32 ± 2.5 |
| Feed pH Test | MW B | TS 80 | 225 | 25 ± 0.5 | 19 ± 2.8 | 35 ± 4.6 |
| Recovery Test | MW C | TS 80 | 225 | 25 ± 1.2 | 23 ± 5.0 | 32 ± 2.0 |
| Recovery Test | MW C | NF 270 | 225 | 25 ± 0.5 | 10 ± 2.7 | 34 ± 0.9 |
| Recovery Test | MW D | TS 80 | 225 | 25 ± 0.5 | 22 ± 6.2 | 33 ± 1.6 |
| Recovery Test | MW D | NF 270 | 225 | 25 ± 0.8 | 10 ± 1.9 | 34 ± 1.5 |
2.2.2. Feed pH Tests
2.2.3. Recovery Tests
2.3. Experimental Set-Up
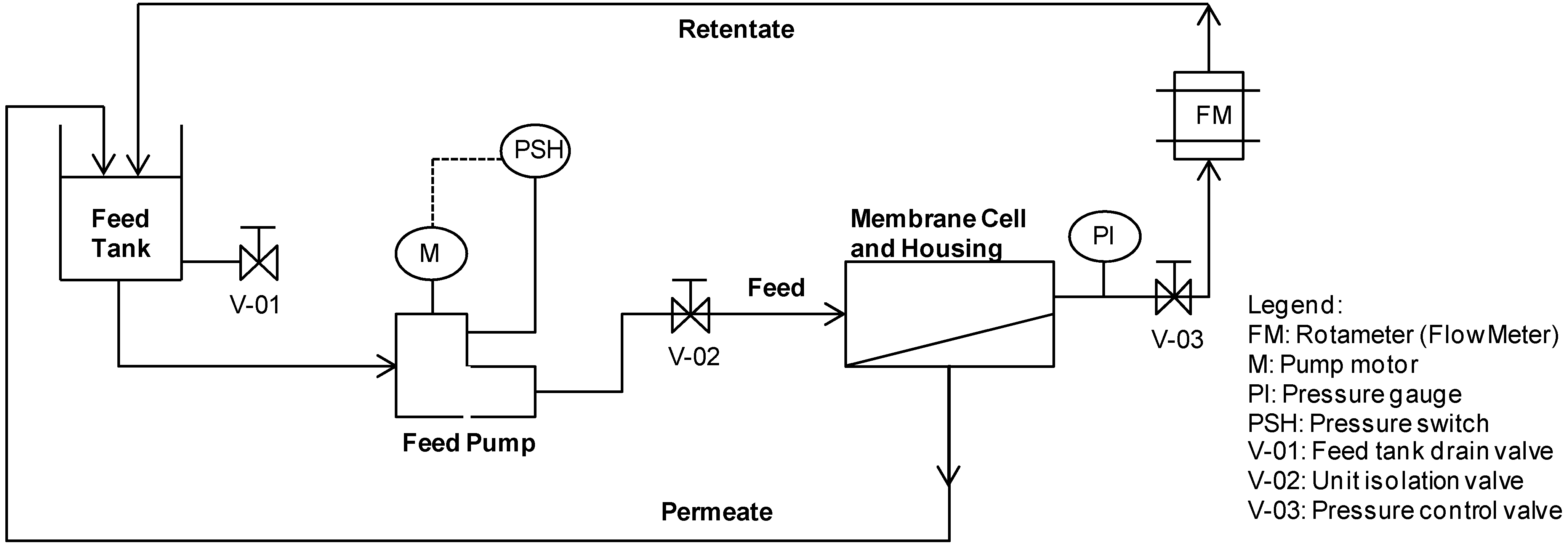
3. Results and Discussion
3.1. IEP Tests

3.2. Feed pH Tests
3.2.1. Feed pH Tests Using NF 270

3.2.2. Feed pH Tests Using TS 80
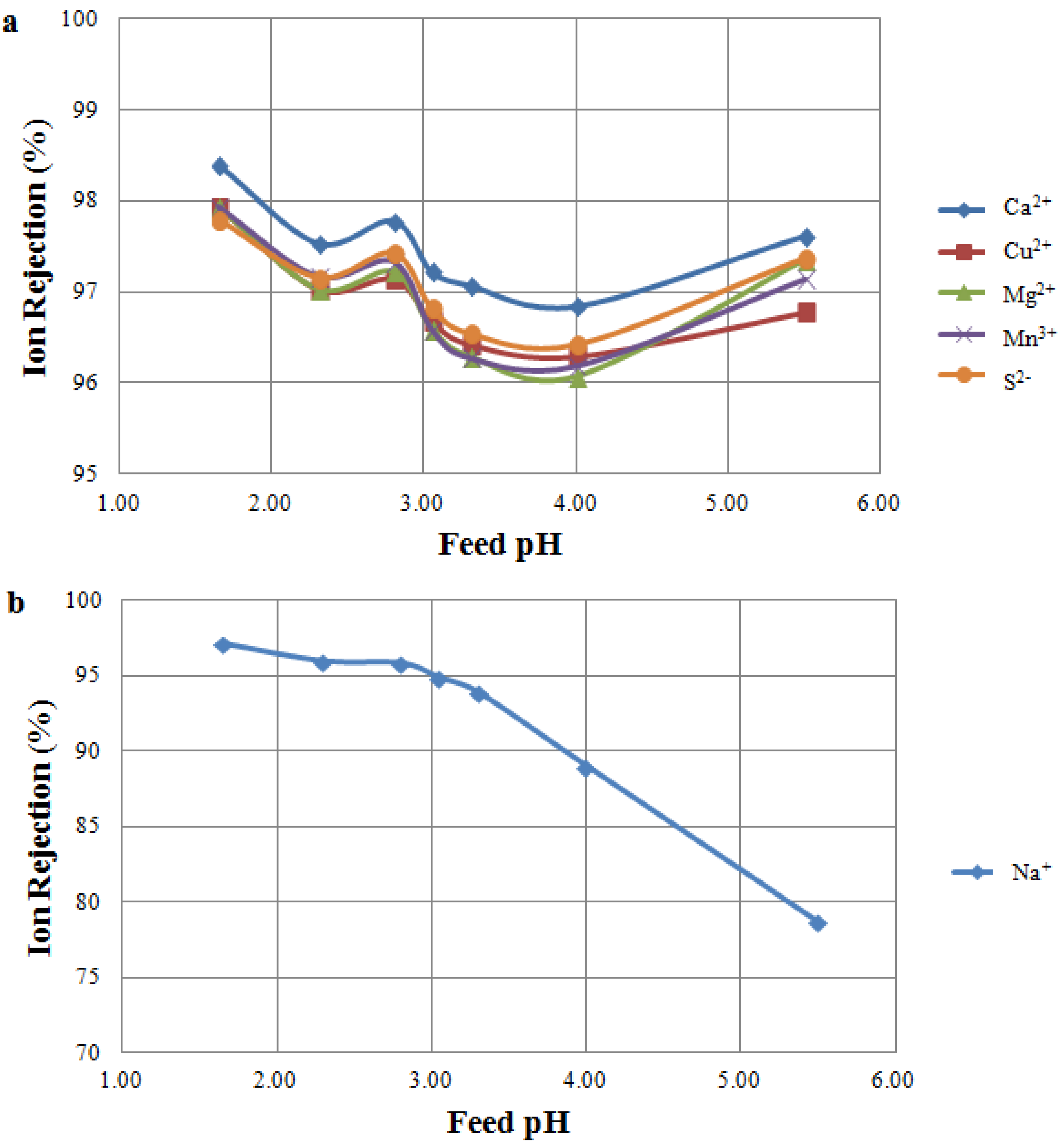
3.2.3. Comparison between NF Membranes
3.3. Recovery Tests
| Ion | Recovery Test on MW D (feed pH = 2.60; recovery = 70%) | Recovery Test on MW C (feed pH = 4.10; recovery = 70%) | Discharge Criteria (mg L−1) | Estimated permeate concentration second pass (mg L−1) | ||||
|---|---|---|---|---|---|---|---|---|
| Rejection (%) | Permeate Concentration (mg L−1) | Rejection (%) | Permeate Concentration (mg L−1) | Feed pH = 2.60 | Feed pH = 4.10 | |||
| TS 80 | ||||||||
| Ca2+ | 98 | 5.7 | 95 | 13 | 50 | 0.1 | 0.6 | |
| Cu2+ | 97 | 15 | 94 | 34 | 1–50 | 0.4 | 1.9 | |
| Mg2+ | 97 | 28 | 94 | 53 | 50 | 0.9 | 3.1 | |
| Mn3+ | 97 | 13 | 95 | 29 | 0.005–0.5 | 0.3 | 1.6 | |
| Na+ | 94 | 200 | 87 | 490 | – | 11 | 63 | |
| SO42− | 98 | 246 | 95 | 510 | 250–1000 | 6 | 25 | |
| Cl− | 84 | 470 | 78 | 650 | – | 76 | 141 | |
| NF 270 | ||||||||
| Ca2+ | 94 | 12 | 93 | 19 | 50 | 0.7 | 1.3 | |
| Cu2+ | 94 | 27 | 91 | 47 | 1–50 | 1.7 | 4.3 | |
| Mg2+ | 95 | 38 | 95 | 49 | 50 | 1.9 | 2.6 | |
| Mn3+ | 95 | 20 | 94 | 27 | 0.005–0.5 | 1.0 | 1.6 | |
| Na+ | 52 | 1300 | 50 | 1600 | – | 626 | 800 | |
| SO42− | 94 | 480 | 95 | 450 | 250–1000 | 31 | 22 | |
| Cl− | 4 | 2200 | −8 | 2800 | – | 2104 | 3015 | |
3.3.1. Metal Rejections at 70% Water Recovery
3.3.2. Nanofiltration of MIW for Environmental Discharge
3.3.3. Nanofiltration of MIW for Metal Recovery

4. Conclusions
Conflicts of Interest
Author Contributions
Acknowledgments
References
- Global Water Intelligence. Water for Mining, Opportunities in scarcity and environmental regulations. Available online: http://www.globalwaterintel.com/market-intelligence-reports/water-mining-opportunities-scarcity-and-environmental-regulation/ (accessed on 30 June 2011).
- Evangelou, V.P.; Zhang, Y.L. A review: Pyrite oxidation mechanisms and acid mine drainage prevention. Crit. Rev. Environ. Sci. Technol. 1995, 25, 141–199. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metals ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Zinck, J.; Griffith, W. Review of Mine Drainage Treatment and Sludge Management Operations. MEND Report 3.43.1. Available online: http://mend-nedem.org/wp-content/uploads/MEND3.43.1_ReviewofMineDrainageTreatmentSludgeManagementOperations.pdf (accessed on 21 March 2014).
- Al-Zoubi, H.; Rieger, A.; Steinberger, P.; Pelz, W.; Haseneder, R.; Hartel, G. Optimization study for treatment of acid mine drainage using membrane technology. Sep. Sci. Technol. 2010, 45, 2004–2016. [Google Scholar] [CrossRef]
- Al-Zoubi, H.; Rieger, A.; Steinberger, P.; Pelz, W.; Haseneder, R.; Hartel, G. Nanofiltration of acid mine drainage. Desalin. Water Treat. 2010, 21, 148–161. [Google Scholar] [CrossRef]
- Nodwell, M.; Kratochvil, D. Sulphide precipitation and ion exchange technologies to treat acid mine water. In Proceedings of the 9th International Conference on Acid Rock Drainage, Ottawa, Ontario, Canada, 20–26 May 2012; Price, W.A., Hogan, C., Tremblay, G., Eds.; Golder Associates Ltd.: Kanata, Ontario, Canada, 2012. [Google Scholar]
- Zhong, C.M.; Xu, Z.L.; Fang, X.H.; Cheng, L. Treatment of acid mine drainage (AMD) by ultra-low-pressure reverse osmosis and nanofiltration. Environ. Eng. Sci. 2007, 24, 1297–1306. [Google Scholar] [CrossRef]
- Rieger, A.; Steinberger, P.; Pelz, W.; Haseneder, R.; Hartel, G. Mine water treatment by membrane filtration processes—Experimental investigations on applicability. Desalin. Water Treat. 2009, 6, 54–60. [Google Scholar] [CrossRef]
- Mortazavi, S.; Chaulk, J. Treatment of acid mine drainage streams using membrane separation. In Proceedings of the 9th International Conference on Acid Rock Drainage, Ottawa, Ontario, Canada, 20–26 May 2012; Price, W.A., Hogan, C., Tremblay, G., Eds.; Golder Associates Ltd.: Kanata, Ontario, Canada, 2012. [Google Scholar]
- Carvalho, A.L.; Maugeri, F.; Pradanos, P.; Silva, V.; Hernandez, A. Separation of potassium clavulanate and potassium chloride by nanofiltration: Transport and evaluation of membranes. Sep. Purif. Technol. 2011, 83, 23–30. [Google Scholar] [CrossRef]
- Tanninen, J.; Manttari, M.; Nystrom, M. Nanofiltration of concentrated acidic copper sulphate solutions. Desalination 2006, 189, 92–96. [Google Scholar] [CrossRef]
- Fornarelli, F.; Mullett, M.; Ralph, D. Factors influencing nanofiltration of acid mine drainage. In Reliable Mine Water Technology; Brown, A., Figueroa, L., Wolkersdorfer, Ch., Eds.; Publication Printers: Denver, CO, USA, 2013; Volume I. [Google Scholar]
- Artug, G. Modelling and Simulation of Nanofiltration Membranes. Ph.D. Thesis, Hamburg-Harburg University, Izmir, Turkey, 2007. [Google Scholar]
- Childress, A.E.; Elimelech, M. Relating Nanofiltration Membrane Performance to Membrane Charge (Electrokinetic) Characteristics. Environ. Sci. Technol. 2000, 34, 3710–3716. [Google Scholar] [CrossRef]
- Qin, J.J.; Oo, M.H.; Lee, H.; Coniglio, B. Effect of feed pH on permeate pH and ion rejection under acidic conditions in NF process. J. Membr. Sci. 2004, 232, 153–159. [Google Scholar] [CrossRef]
- Tay, J.H.; Liu, J.; Sun, D.D. Effect of solution physico-chemistry on the charge property of nanofiltration membranes. Water Res. 2002, 36, 585–598. [Google Scholar] [CrossRef]
- Artug, G.; Hapke, J. Characterization of nanofiltration membranes by their morphology, charge and filtration performance parameters. Desalination 2006, 200, 178–180. [Google Scholar] [CrossRef]
- Childress, A.E.; Elimelech, M. Effect of solution chemistry on the surface charge of polymeric reverse osmosis and nanofiltration membranes. J. Membr. Sci. 1996, 119, 253–268. [Google Scholar] [CrossRef]
- Hagmeyer, G.; Gimbel, R. Modelling the rejection of nanofiltration membranes using zeta potential measurements. Sep. Purif. Technol. 1999, 15, 19–30. [Google Scholar] [CrossRef]
- Tanninen, J.; Platt, S.; Weis, A.; Nystrom, M. Long-term acid resistance and selectivity of NF membranes in very acidic conditions. J. Membr. Sci. 2004, 240, 11–18. [Google Scholar] [CrossRef]
- Ferreira-Esmi, C.; Schrive, L.; Barre, Y.; Palmeri, J.; Deratani, A. Using nanofiltration in a “zero-rejection” process: the removal of Ni2+ and Co2+ from salty wastewater. Desalin. Water Treat. 2013, 51, 476–484. [Google Scholar] [CrossRef]
- Al-Rashdi, B.A.M.; Johnson, D.J.; Hilal, N. Removal of heavy metal ions by nanofiltration. Desalination 2012, 315, 2–17. [Google Scholar] [CrossRef]
- Nghiem, L.D. Removal of Emerging Trace Organic Contaminants by Nanofiltration and Reverese Osmosis. Ph.D. Thesis, School of Civil, Mining and Environmental Engineering, University of Wollongong, Wollongong, Australia, 2005. [Google Scholar]
- Verliefde, A.R.D.; Heijman, S.G.J.; Cornelissen, E.R.; Amy, G.L.; van der Bruggen, B.; van Dijk, J.C. Rejection of trace organic pollutant with high pressure membranes (NF/RO). Environ. Prog. 2008, 27, 180–188. [Google Scholar] [CrossRef]
- Szoke, S.; Patzay, G.; Weiser, L. Characteristics of thin-film nanofiltration membranes at various pH-values. Desalination 2002, 151, 123–129. [Google Scholar]
- Artug, G.; Roosmasari, K.; Richau, K.; Hapke, A. A comprehensive characterization of commercial nanofiltration membranes. Sep. Sci. Technol. 2007, 42, 2947–2986. [Google Scholar] [CrossRef]
- Soldenhoff, K.; McCulloch, J.; Manis, A.; Macintosh, P. Nanofiltration in metal and acid recovery. In Nanofiltration—Principles and Application; Schafer, A.I., Fane, A.G., Waite, T.D., Eds.; Elsevier Advanced Technology: Oxford, UK, 2005; Chapter 19; pp. 459–477. [Google Scholar]
- MacNaughton, S.J.; McCulloch, J.K.; Marshall, K.; Ring, R.J. Application of nanofiltration to the treatment of uranium mill effluents. In Technologies for the Treatment of Effluents from Uranium Mines, Mills and Tailings, Proceedings of a Technical Committee Meeting, Vienna, Austria, 1–4 November 1999; IAEA: Vienna, Austria, 2002; pp. 55–65. [Google Scholar]
- Mortazavi, S. Application of Membrane Separation Technology to Mitigation of Mine Effluente and Acidic Drainage. MEND Report 3.15.1. Available online: http://mend-nedem.org/wp-content/uploads/2013/01/3.15.1.pdf (accessed on 21 March 2014).
- Van der Merwe, I.W. Application of nanofiltration in metal recovery. J. S. Afr. Inst. Min. Metall. 1998, 98, 339–342. [Google Scholar]
- Cameron, R.; Edwards, C. Membrane technology applications in mineral processing. In Proceedings of the 44th Annual Canadian Mineral Processors Operators Conference, Ottawa, Ontario, Canada, 17–19 January 2012.
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mullett, M.; Fornarelli, R.; Ralph, D. Nanofiltration of Mine Water: Impact of Feed pH and Membrane Charge on Resource Recovery and Water Discharge. Membranes 2014, 4, 163-180. https://doi.org/10.3390/membranes4020163
Mullett M, Fornarelli R, Ralph D. Nanofiltration of Mine Water: Impact of Feed pH and Membrane Charge on Resource Recovery and Water Discharge. Membranes. 2014; 4(2):163-180. https://doi.org/10.3390/membranes4020163
Chicago/Turabian StyleMullett, Mark, Roberta Fornarelli, and David Ralph. 2014. "Nanofiltration of Mine Water: Impact of Feed pH and Membrane Charge on Resource Recovery and Water Discharge" Membranes 4, no. 2: 163-180. https://doi.org/10.3390/membranes4020163




