Antioxidant, Iron Chelating and Tyrosinase Inhibitory Activities of Extracts from Talinum triangulare Leach Stem
Abstract
:1. Introduction
2. Experimental Section
2.1. Plant
2.2. Determination of Total Phenolics
2.3. Determination of Antioxidant Activity Using the 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging Method
2.4. Iron Binding Ability
2.5. Tyrosinase Enzyme Inhibitory Activity
3. Results and Discussion
3.1. Total Phenolic Contents
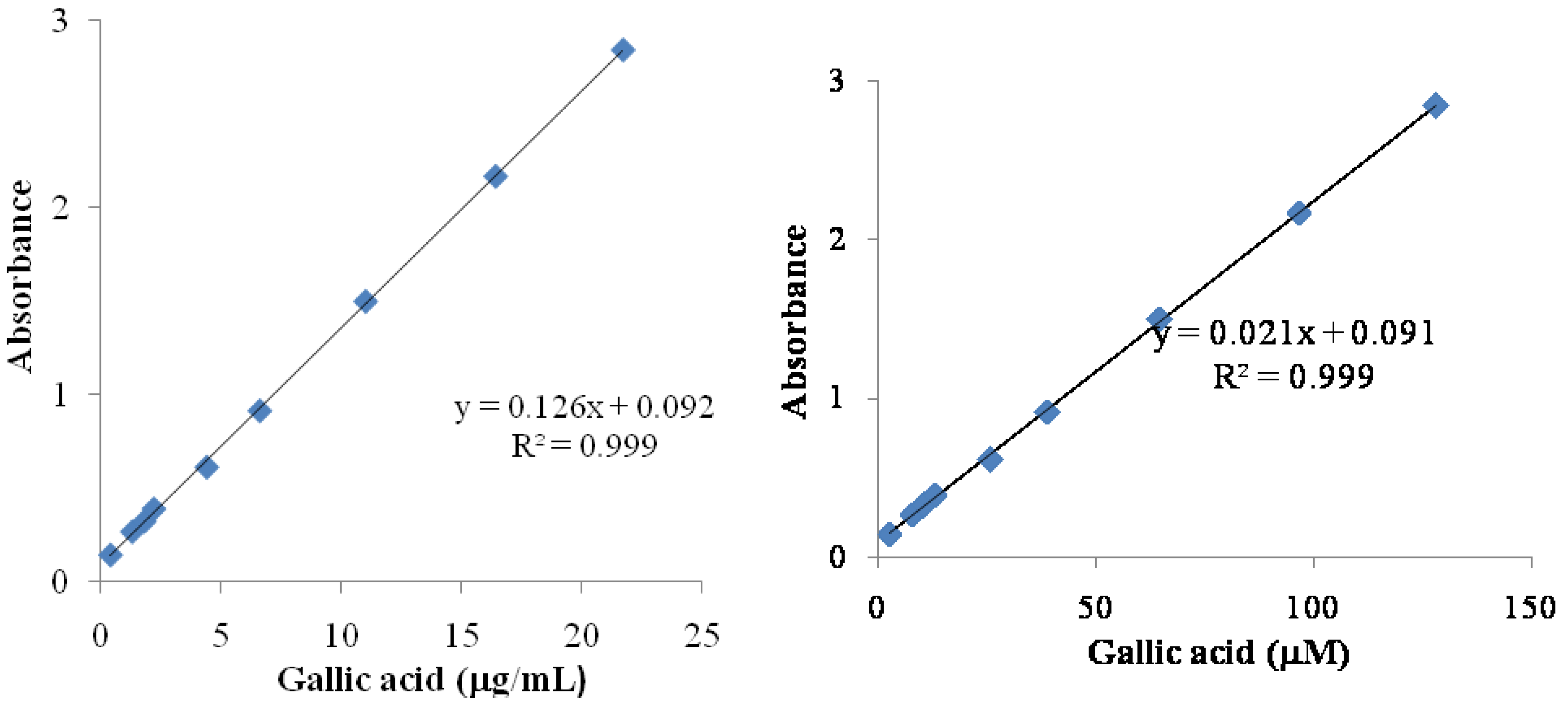
3.2. DPPH Radical Scavenging Activity
| Crude Extract | Fraction 1 | ||||||
|---|---|---|---|---|---|---|---|
| [Sample] a | Absf | ∆Abs b | [DPPHcaptured] c | [Sample] a | Absf | ∆Abs b | [DPPHcaptured] c |
| 0.18 | 0.427 | 0.002 | 0.24 | 0.80 | 0.392 | 0.036 | 4.40 |
| 0.37 | 0.410 | 0.019 | 2.26 | 1.60 | 0.378 | 0.050 | 6.13 |
| 0.74 | 0,395 | 0.034 | 4.08 | 3.20 | 0.351 | 0.077 | 9.3 |
| 1.50 | 0,355 | 0.074 | 8.90 | 6.41 | 0.317 | 0.111 | 13.42 |
| 2.97 | 0.313 | 0.116 | 13.94 | 12.79 | 0.251 | 0.177 | 21.32 |
| 5.95 | 0.191 | 0.238 | 28.60 | 25.59 | 0.165 | 0.264 | 31.70 |


3.3. Iron Binding Ability
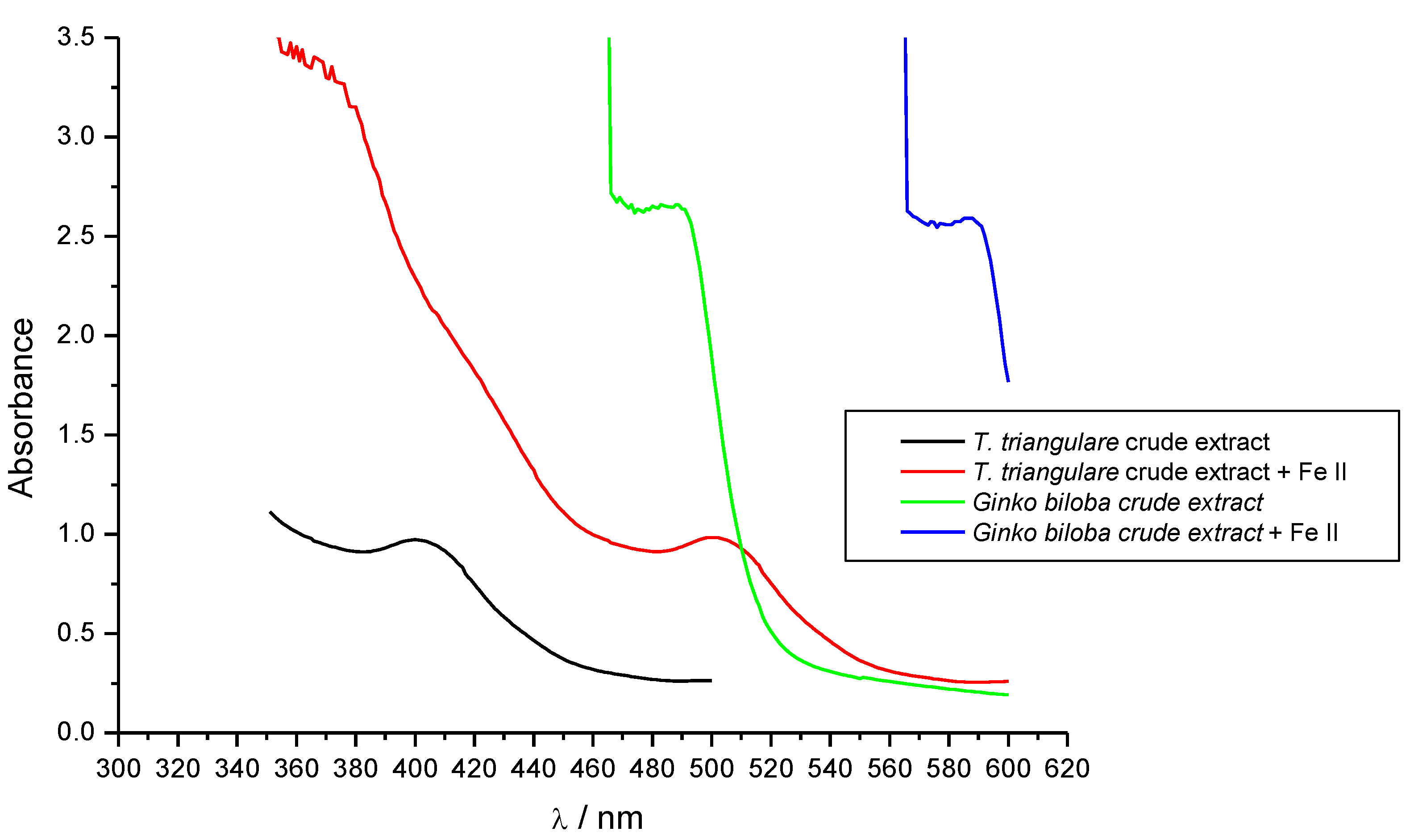
3.4. Tirosinase Inhibitory Activity


4. Conclusions
Acknowledgment
Conflict of Interest
References
- Collinge, S.K.; Prudic, K.L.; Oliver, J.C. Effects of local habitat quality characteristics and landscape context on grassland butterfly diversity. Conserv. Biol. 2003, 17, 178–187. [Google Scholar] [CrossRef]
- Holmes, P.M.; Cowling, R.M. The effects of invasion by Acacia saligna on the guild structure and regeneration capabilities of South Africa fynbos shrublands. J. Appl. Ecol. 1997, 34, 317–332. [Google Scholar] [CrossRef]
- Lindsay, E.A.; French, K. Chrysanthemoides monilifera ssp. rotundata invasion alters decomposition rates in coastal areas of south-eastern Australia. For. Ecol. Manag. 2004, 198, 387–399. [Google Scholar] [CrossRef]
- Manson, T.J.; French, K. Management regimes for a plant invader differentially impact resident communities. Biol. Conserv. 2007, 136, 246–259. [Google Scholar] [CrossRef]
- Souza, V.C.; Lorenzi, H. Botânica Sistemática; Instituto Plantarum de Estudos da Flora Ltda: Nova Odessa, Brazil, 2005. [Google Scholar]
- Ofusori, D.A.; Adelakun, A.E.; Ayoka, A.O.; Oluwayinka, O.P.; Omotoso, E.O.; Odukoya, S.A.; Adeyemi, D.O. Water leaf (Talinum triangulare) enhances cerebral functions in swiss albino mice. J. Neurol. Sci. 2008, 25, 239–246. [Google Scholar] [CrossRef]
- Ekpo, A.; Eseyin, O.; Kalu, N.; Jackson, O.; Edoho, E.J. Studies on the biochemical effects of Talinum triangulare in rat. J. Pharmacol. Toxicol. 2007, 2, 300–303. [Google Scholar] [CrossRef]
- Amorim, A.P.O.; Oliveira, M.C.C.; Echevarria, A. Novo ácido graxo acetiloxi substituído e espergulagenato isolados do caule de Talinum triangulare (Portulacaceae). Available online: http://sec.sbq.org.br/cdrom/31ra/resumos/T0064-1.pdf (accessed on 2 May 2013).
- Cruz, E.M.; Amorim, A.P.O.; Oliveira, M.C.S.; Carvalho, M.G.; Santos, M.A.V. Avaliação da atividade de extratos de Talinum triangulare e Sphagneticola trilobata sobre Trypanosoma cruzi. Available online: http://www.alice.cnptia.embrapa.br/handle/doc/574897 (accessed on 2 May 2013).
- Andarwulan, N.; Batari, R.; Sandrasari, D.A.; Bolling, B.; Agustini, D.; Wijaya, H. Flavonoid content and antioxidant activity of vegetables from Indonesia. Food Chem. 2010, 121, 1231–1235. [Google Scholar] [CrossRef]
- Afonso, V.; Champy, R.; Mitrovic, D.; Collin, P.; Lomri, A. Reactive oxygen species and superoxide dismutases: Role in joint diseases. Joint Bone Spine 2007, 74, 324–329. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Bruck, R.; Aeed, H.; Avni, Y. Melatonin inhibits nuclear factor kappa Bactivation and oxidative stress and protects against thioacetamide induced liver damage in rats. J. Hepatol. 2004, 40, 86–93. [Google Scholar] [CrossRef]
- Fu, W.; Chen, J.; Cai, Y.; Lei, Y.; Chen, L.; Pei, L.; Zhou, D.; Liang, X.; Ruan, J. Antioxidant, free radical scavenging, anti-inflammatory and hepatoprotective potential of the extract from Parathelypteris nipponica (Franch. et Sav.) Ching. J. Ethnopharmacol. 2010, 130, 521–528. [Google Scholar] [CrossRef]
- Chen, J.R.; Hu, T.J.; Zheng, R.L. Antioxidant activities of Sophora subprosrate polysaccharide in immunosuppressed mice. Int. Immunopharmacol. 2007, 7, 547–553. [Google Scholar] [CrossRef]
- Dong Liang, D.; Zhou, Q.; Gong, W.; Wang, Y.; Nie, Z.; He, H.; Li, J.; Wu, J.; Wu, C.; Zhang, J. Studies on the antioxidant and hepatoprotective activities of polyssaccharides from Talinum triangulare. J. Ethnopharmacol. 2011, 136, 316–321. [Google Scholar] [CrossRef]
- Matos, F.J.A. Introdução à Fitoquímica Experimental, 3th ed.; Edições UFC: Fortaleza, Ceará, Brazil, 2009. [Google Scholar]
- Silva, S.; Gomes, L.; Leitão, F.; Coelho, A.V.; Vilas Boas, L. Phenolic compounds and antioxidant activity of Olea europaea L fruits and leaves. Food Sci. Technol. Int. 2006, 12, 385–395. [Google Scholar] [CrossRef]
- Mensor, L.L.; Menezes, F.S.; Leitão, G.G.; Reis, A.S.; Dos Santos, T.C.; Coube, C.S.; Leitão, S.G. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytoter. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef]
- Ikawa, M.; Schaper, T.D.; Dollard, C.A.; Sasner, J.J. Utilization of Folin-Ciocalteu phenol reagent for the detection of certain nitrogen compounds. J. Agric. Food Chem. 2003, 51, 1811–1815. [Google Scholar] [CrossRef]
- Ordoñez, A.A.L.; Gomez, J.D.; Vattuone, M.A.; Isla, M.I. Antioxidant activities of Sechium edule (Jacq.) Swartz extracts. Food Chem. 2006, 97, 452–458. [Google Scholar] [CrossRef]
- Zecca, L.; Youdim, M.B.H.; Riederer, P.; Connor, J.R.; Crichton, R.R. Iron, brain ageing and neurodegenerative disorders. Nat. Rev. Neurosci. 2004, 5, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Moreno, S.R.F.; Freitas, R.S.; Rocha, E.K.; Lima-Filho, G.L.; Bernardo-Filho, M. Protection of plasmid DNA by a Ginkgo biloba extract from the effects of stannous chloride and the action on the labeling of blood elements with technetium-99m. Braz. J. Med. Biol. Res. 2004, 37, 267–271. [Google Scholar] [CrossRef]
- Patil, S.S.; Zucker, M. Potato phenolases: Purification and Properties. J. Biol. Chem. 1965, 240, 3938–3943. [Google Scholar] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Oliveira Amorim, A.P.; Campos de Oliveira, M.C.; De Azevedo Amorim, T.; Echevarria, A. Antioxidant, Iron Chelating and Tyrosinase Inhibitory Activities of Extracts from Talinum triangulare Leach Stem. Antioxidants 2013, 2, 90-99. https://doi.org/10.3390/antiox2030090
Oliveira Amorim AP, Campos de Oliveira MC, De Azevedo Amorim T, Echevarria A. Antioxidant, Iron Chelating and Tyrosinase Inhibitory Activities of Extracts from Talinum triangulare Leach Stem. Antioxidants. 2013; 2(3):90-99. https://doi.org/10.3390/antiox2030090
Chicago/Turabian StyleOliveira Amorim, Ana Paula, Márcia Cristina Campos de Oliveira, Thiago De Azevedo Amorim, and Aurea Echevarria. 2013. "Antioxidant, Iron Chelating and Tyrosinase Inhibitory Activities of Extracts from Talinum triangulare Leach Stem" Antioxidants 2, no. 3: 90-99. https://doi.org/10.3390/antiox2030090
APA StyleOliveira Amorim, A. P., Campos de Oliveira, M. C., De Azevedo Amorim, T., & Echevarria, A. (2013). Antioxidant, Iron Chelating and Tyrosinase Inhibitory Activities of Extracts from Talinum triangulare Leach Stem. Antioxidants, 2(3), 90-99. https://doi.org/10.3390/antiox2030090





