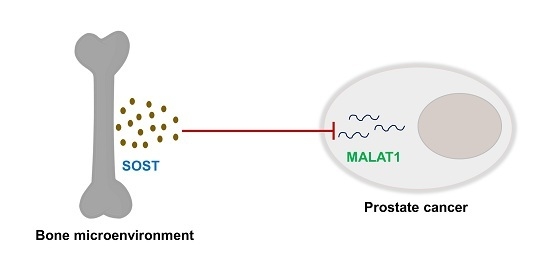
Cancer–Osteoblast Interaction Reduces Sost Expression in Osteoblasts and Up-Regulates lncRNA MALAT1 in Prostate Cancer
Abstract
:1. Introduction
2. Experimental Section
2.1. Animals
2.2. Cell Culture
2.3. Microarrays
2.4. Functional Annotation
2.5. Immunocytochemistry
2.6. Quantitative PCR
3. Results
3.1. Molecular Changes in Osteoblasts Co-Cultured with Prostate Cancer Cells

| Gene | Log2 Fold Change | Adjusted p-Value |
|---|---|---|
| Mmp13 | 7.379 | 0.00268 |
| Lbp | 4.111 | 0.00578 |
| Wisp2 | 3.857 | 0.01510 |
| Slpi | 3.636 | 0.00614 |
| A2m | 3.472 | 0.01160 |
| Cp | 3.436 | 0.01553 |
| Il1rl1 | 3.408 | 0.01955 |
| Igfbp5 | 3.005 | 0.01046 |
| Il6 | 2.808 | 0.01311 |
| Cxcl1 | 2.551 | 0.01750 |
| Mgp | 2.518 | 0.03807 |
| Fam20c | 2.289 | 0.01510 |
| Sfrp4 | 2.284 | 0.01831 |
| Fst | 2.161 | 0.03814 |
| Adamts4 | 2.044 | 0.03251 |
| Sostdc1 | 1.968 | 0.01953 |
| Lum | 1.745 | 0.01986 |
| Enpp3 | 1.745 | 0.03105 |
| Sned1 | 1.733 | 0.03146 |
| Adamts1 | 1.64 | 0.03251 |
| Gpx3 | 1.587 | 0.02450 |
| Adamts5 | 1.515 | 0.03793 |
| Fndc1 | 1.454 | 0.03494 |
| Igfbp4 | 1.452 | 0.03072 |
| Col3a1 | 1.435 | 0.03793 |
| Col14a1 | 1.427 | 0.04376 |
| Clec11a | 1.425 | 0.03089 |
| Ecm1 | 1.352 | 0.04301 |
| Enpp1 | 1.328 | 0.03564 |
| C1qtnf1 | 1.308 | 0.04556 |
| Ctgf | 1.30 | 0.03807 |
| Tgfb2 | 1.194 | 0.04545 |
| Cgref1 | −1.174 | 0.04376 |
| Pcsk6 | −1.485 | 0.03793 |
| Apln | −1.563 | 0.02821 |
| Bmp3 | −1.722 | 0.02335 |
| Cd55 | −1.738 | 0.02369 |
| Metrnl | −1.849 | 0.01974 |
| Bmper | −2.128 | 0.01553 |
| Sost | −2.83 | 0.01510 |
| Mamdc2 | −2.859 | 0.01953 |
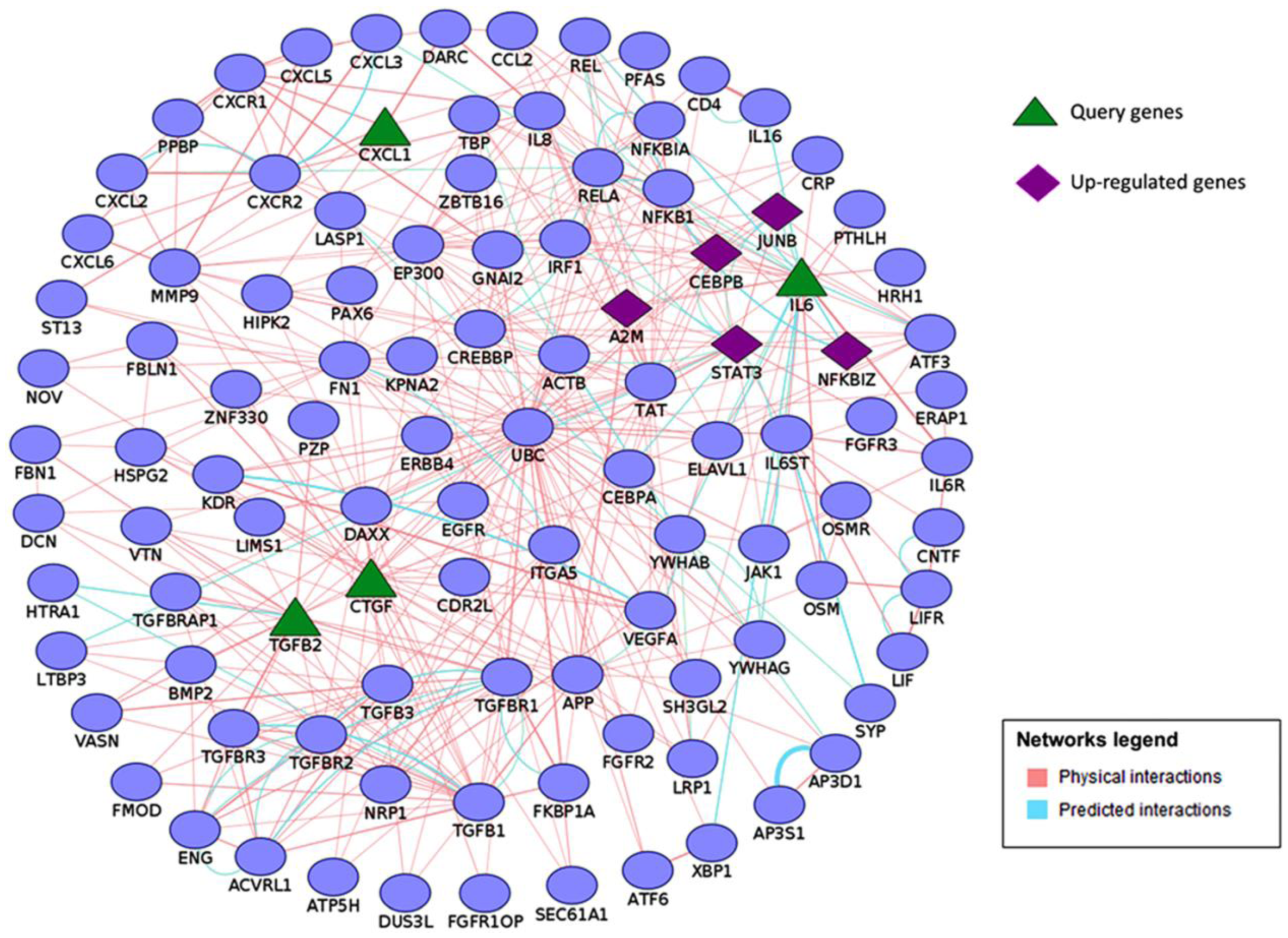
3.2. Functional Analysis of Differentially Regulated Genes
| GO (Gene Ontology) ID | GO Term | Genes |
|---|---|---|
| GO:0001503 | Ossification | Sost, Col13a1, Cebpb, Cebpd ,Dhrs3, Enpp1, Ifitm1, Ecm1, Sbno2, Tgfb2, Phex, Bmp3, Junb, Fam20c, Igfbp5, Mgp, Phospho1, Egr2, Jag1, Ctgf, Gpnmb, Mmp13, Fhl2, Csgalnact1, Il6 |
| GO:0009725 | Response to hormone | Hhex, Sost, Sfrp4, Rarb, Enpp1, Fos, Dusp1, Gatm, Cd55, Irf1, Cav1, Nr4a1, Stat3, Tgfb2, Cryab, Junb, Btg2, Egr1, Egr2, Ass1, Kcnma1, Prkar1b, Ctgf, Fhl2, Gng5, Il6 |
| GO:0030198 | Extracellular matrix organization | Col3a1, A2m, Col13a1, Adamts4, Pdgfra, Pecam1, Adamts5, Itgb3, Tgfb2, Col14a1, Ctgf, Lum, Mmp13, Csgalnact1 |
| GO:0016477 | Cell migration | Col3a1, Pde4b, Ddx58, Pdgfra, Pecam1, Ifitm1, Cav1, Lbp, Nr4a1, Ecm1, Cxcl1, Adarb1, Itgb3, Stat3, Ednrb, Tgfb2, Thbd, Igfbp5, Jag1, Ctgf, Bmper, Nexn, Il6 |
| GO:0045595 | Regulation of cell differentiation | Col3a1, Sox6, Sfrp4, Rarb, Cebpb, Cebpd, Pdgfra, Metrnl, Enpp1, Fos, Ntrk1 ,Dusp6, Tmem176a, Ifitm1, Sostdc1, Irf1 , Irx3 ,Cav1, Itgb3, Zfp36, Stat3, Ednrb, Tgfb2, Junb, Fam20c, Bcl11b, Igfbp5, Jag1, Errfi1, Col14a1, Mafb, Ctgf, Maff, Tesc, Fst, Il6, Maf |
| GO:0008283 | Cell proliferation | Hhex, Sfrp4, Rarb, Cebpb, Pdgfra, Adamts1, Ntrk1, Cd55, Etv4, Ifitm1, Irf1, Cav1, Nr4a1, Ecm1, Cxcl1, Adarb1, Itgb3, Stat3, Ednrb, Tgfb2, Wisp2, Tbx18, Slfn3, Junb, Bcl11b, Btg2, Apln, Igfbp4, Igfbp5, Egr1, Ifitm3, Cgref1, Clec11a, Jag1, Prkar1b, Ctgf, Il1rl1, Tesc, Gpnmb, Bmper, Fst, Csgalnact1, Il6 |
| GO:0001944 | Vasculature development | Col3a1, Hhex, Sfrp4, Ddah1, Pdgfra, Adamts1, Ntrk1, Pecam1, Cav1, Nr4a1, Ecm1, Itgb3, Tgfb2, Junb, Zfp36l1, Egr1, Jag1, Errfi1, Ctgf, Bmper, Il6 |
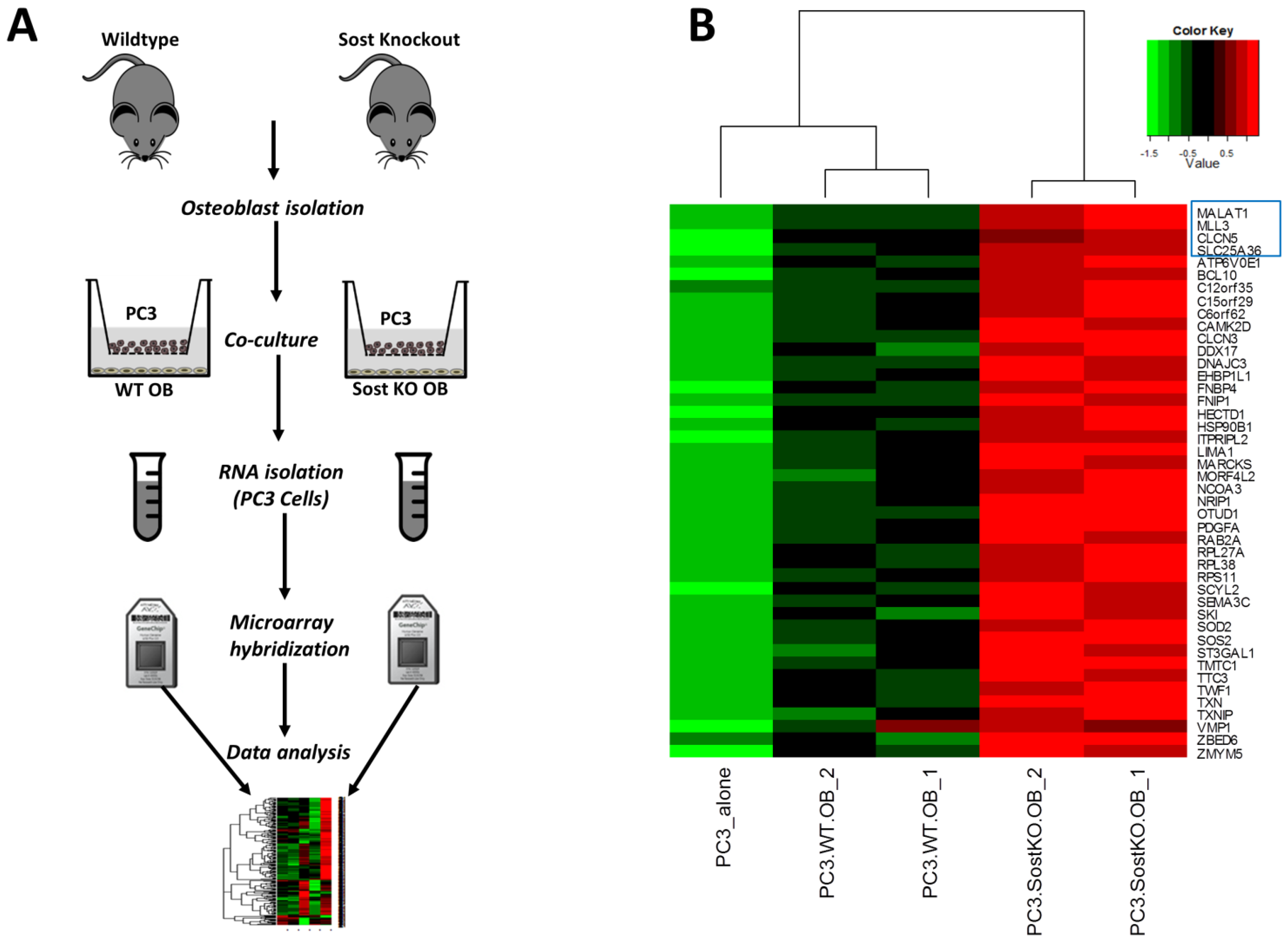
3.3. Effect of Bone Microenvironment Derived Sost on PC3 Gene Expression
| GO ID | GO Term | Genes |
|---|---|---|
| GO:0043624 | Cellular protein complex disassembly | RPL27A, LIMA1, RPL37A, RPL38, MAP1B, EML4, RPS11, TWF1 |
| GO:0006468 | Protein phosphorylation | SCYL2, JAK1, MET, HSP90B1, BMPR2, PTPRA, PDGFA, SQSTM1, MAP4K5, CAMK2D, CLK4, FER, FNIP1, ST3GAL1, BCL10, GNAS, DNAJC3, TWF1 |
| GO:1902680 | Positive regulation of RNA biosynthetic process | NCOA2, MET, NCOA3, NRIP1, DDX17, ATRX, MORF4L2, SQSTM1, KAT6A, ELF1, E2F3, BCL10, MLLT10, SKI, NR1D2 |
| GO:0006935 | Chemotaxis | EZR, MET, WASL, SEMA3C, BMPR2, PTPRA, PDGFA, FER, NRP2, SOS2 |
| GO:0045595 | Regulation of cell differentiation | BHLHE41, MET, NCOA3, BMPR2, DDX17, PTPRA, MAP1B, MORF4L2, KAT6A, FNIP1, GNAS, SKI, TTC3, NR1D2, SOD2 |
| GO:0007015 | Actin filament organization | EZR, HSP90B1, WASL, LIMA1, FER, TWF1 |
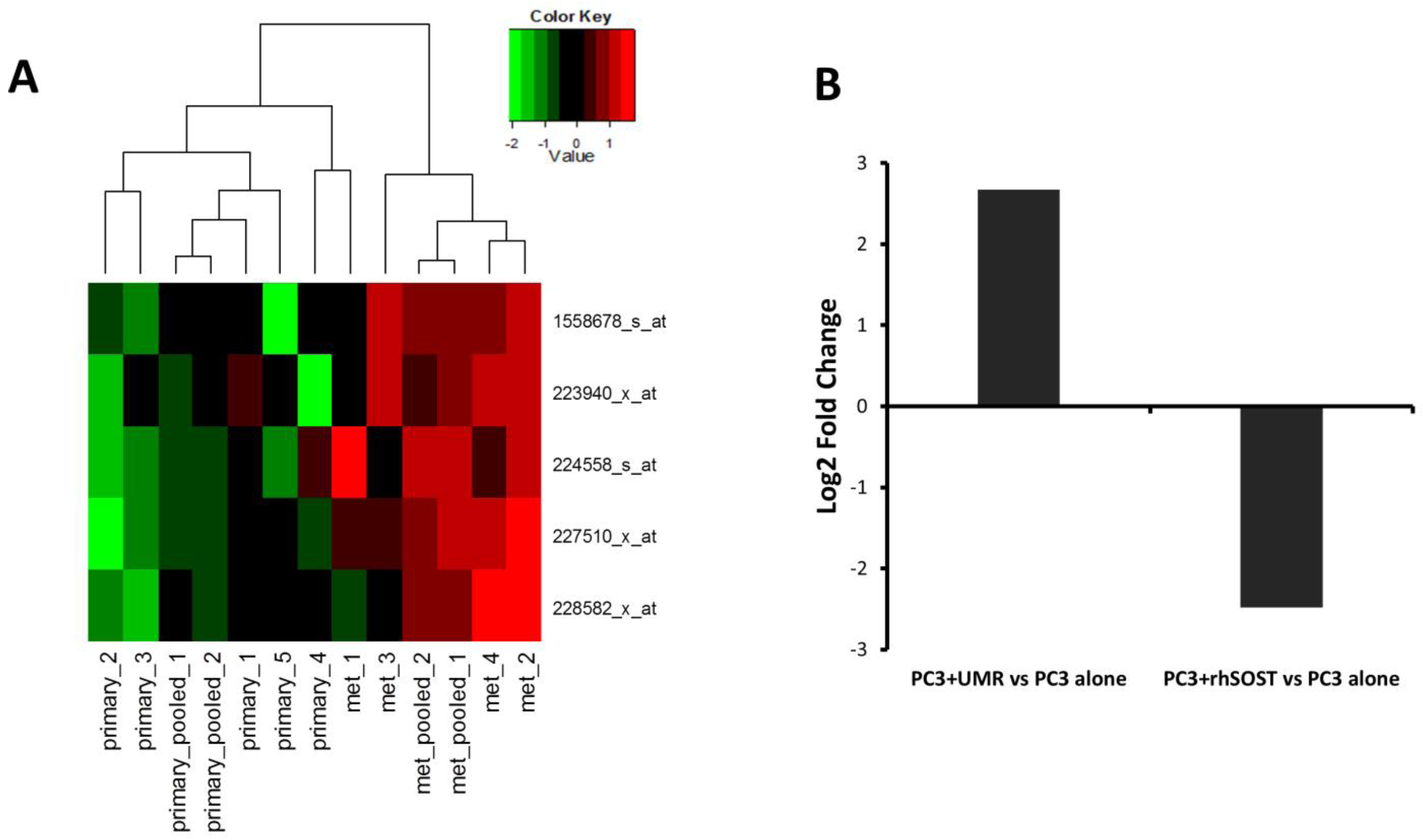
3.4. Sost Is a Regulator of MALAT1 in Prostate Cancer Cells
4. Discussion
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2013. CA Cancer J. Clin. 2013, 63, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Tantivejkul, K.; Kalikin, L.M.; Pienta, K.J. Dynamic process of prostate cancer metastasis to bone. J. Cell Biochem. 2004, 91, 706–717. [Google Scholar] [CrossRef] [PubMed]
- Krzeszinski, J.Y.; Wan, Y. New therapeutic targets for cancer bone metastasis. Trends Pharmacol. Sci. 2015, 36, 360–373. [Google Scholar] [CrossRef] [PubMed]
- Logothetis, C.J.; Lin, S.H. Osteoblasts in prostate cancer metastasis to bone. Nat. Rev. Cancer 2005, 5, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Morrison, C.; Mancini, S.; Cipollone, J.; Kappelhoff, R.; Roskelley, C.; Overall, C. Microarray and proteomic analysis of breast cancer cell and osteoblast co-cultures: Role of osteoblast matrix metalloproteinase (MMP)-13 in bone metastasis. J. Biol. Chem. 2011, 286, 34271–34285. [Google Scholar] [CrossRef] [PubMed]
- Florencio-Silva, R.; Sasso, G.R.; Sasso-Cerri, E.; Simoes, M.J.; Cerri, P.S. Biology of bone tissue: Structure, function, and factors that influence bone cells. BioMed Res. Int. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Giunciuglio, D.; Cai, T.; Filanti, C.; Manduca, P.; Albini, A. Effect of osteoblast supernatants on cancer cell migration and invasion. Cancer Lett. 1995, 97, 69–74. [Google Scholar] [CrossRef]
- Krishnan, V.; Shuman, L.A.; Sosnoski, D.M.; Dhurjati, R.; Vogler, E.A.; Mastro, A.M. Dynamic interaction between breast cancer cells and osteoblastic tissue: Comparison of two- and three-dimensional cultures. J. Cell Physiol. 2011, 226, 2150–2158. [Google Scholar] [CrossRef] [PubMed]
- Rajski, M.; Vogel, B.; Baty, F.; Rochlitz, C.; Buess, M. Global gene expression analysis of the interaction between cancer cells and osteoblasts to predict bone metastasis in breast cancer. PLoS ONE 2012, 7, e29743. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Fizazi, K.; Peleg, S.; Sikes, C.R.; Raymond, A.K.; Jamal, N.; Hu, M.; Olive, M.; Martinez, L.A.; Wood, C.G.; et al. Prostate cancer cells induce osteoblast differentiation through a Cbfa1-dependent pathway. Cancer Res. 2001, 61, 5652–5659. [Google Scholar] [PubMed]
- Collette, N.M.; Genetos, D.C.; Economides, A.N.; Xie, L.; Shahnazari, M.; Yao, W.; Lane, N.E.; Harland, R.M.; Loots, G.G. Targeted deletion of Sost distal enhancer increases bone formation and bone mass. Proc. Natl. Acad. Sci. USA 2012, 109, 14092–14097. [Google Scholar] [CrossRef] [PubMed]
- Bellows, C.G.; Sodek, J.; Yao, K.L.; Aubin, J.E. Phenotypic differences in subclones and long-term cultures of clonally derived rat bone cell lines. J. Cell Biochem. 1986, 31, 153–169. [Google Scholar] [CrossRef] [PubMed]
- Gentleman, R.C.; Carey, V.J.; Bates, D.M.; Bolstad, B.; Dettling, M.; Dudoit, S.; Ellis, B.; Gautier, L.; Ge, Y.; Gentry, J.; et al. Bioconductor: Open software development for computational biology and bioinformatics. Genome Biol. 2004, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Irizarry, R.A.; Hobbs, B.; Collin, F.; Beazer-Barclay, Y.D.; Antonellis, K.J.; Scherf, U.; Speed, T.P. Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 2003, 4, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Smyth, G.K. Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet. Mol. Biol. 2004, 3, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Bardes, E.E.; Aronow, B.J.; Jegga, A.G. Toppgene suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res. 2009, 37, W305–W311. [Google Scholar] [CrossRef] [PubMed]
- Montojo, J.; Zuberi, K.; Rodriguez, H.; Bader, G.D.; Morris, Q. Genemania: Fast gene network construction and function prediction for cytoscape. F1000Research 2014, 3. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−∆∆Ct method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Frara, N.; Abdelmagid, S.M.; Sondag, G.R.; Moussa, F.M.; Yingling, V.R.; Owen, T.A.; Popoff, S.N.; Barbe, M.F.; Safadi, F.F. Transgenic expression of osteoactivin/gpnmb enhances bone formation in vivo and osteoprogenitor differentiation ex vivo. J. Cell Physiol. 2015, 231, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Brun, J.; Fromigue, O.; Dieudonne, F.X.; Marty, C.; Chen, J.; Dahan, J.; Wei, Y.; Marie, P.J. The LIM-only protein FHL2 controls mesenchymal cell osteogenic differentiation and bone formation through Wnt5a and Wnt10b. Bone 2013, 53, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Julien, M.; Khoshniat, S.; Lacreusette, A.; Gatius, M.; Bozec, A.; Wagner, E.F.; Wittrant, Y.; Masson, M.; Weiss, P.; Beck, L.; et al. Phosphate-dependent regulation of MGP in osteoblasts: Role of ERK1/2 and Fra-1. J. Bone Miner. Res. 2009, 24, 1856–1868. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, N.C.; Huesa, C.; Rutsch, F.; MacRae, V.E. New insights into NPP1 function: Lessons from clinical and animal studies. Bone 2012, 51, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Rowe, P.S. A unified model for bone-renal mineral and energy metabolism. Curr. Opin. Pharmacol. 2015, 22, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Casimiro, S.; Guise, T.A.; Chirgwin, J. The critical role of the bone microenvironment in cancer metastases. Mol. Cell Endocrinol. 2009, 310, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Bonfil, R.D.; Chinni, S.; Fridman, R.; Kim, H.R.; Cher, M.L. Proteases, growth factors, chemokines, and the microenvironment in prostate cancer bone metastasis. Urol. Oncol. 2007, 25, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Honasoge, M.; Rao, A.D.; Rao, S.D. Sclerostin: Recent advances and clinical implications. Curr. Opin. Endocrinol. Diabetes Obes. 2014, 21, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Ellies, D.L.; Economou, A.; Viviano, B.; Rey, J.P.; Paine-Saunders, S.; Krumlauf, R.; Saunders, S. Wise regulates bone deposition through genetic interactions with Lrp5. PLoS ONE 2014, 9, e96257. [Google Scholar] [CrossRef] [PubMed]
- Tawara, K.; Oxford, J.T.; Jorcyk, C.L. Clinical significance of interleukin (IL)-6 in cancer metastasis to bone: Potential of anti-IL-6 therapies. Cancer Manag. Res. 2011, 3, 177–189. [Google Scholar] [PubMed]
- Vindrieux, D.; Escobar, P.; Lazennec, G. Emerging roles of chemokines in prostate cancer. Endocr. Relat. Cancer 2009, 16, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Hardaway, A.L.; Herroon, M.K.; Rajagurubandara, E.; Podgorski, I. Marrow adipocyte-derived CXCL1 and CXCL2 contribute to osteolysis in metastatic prostate cancer. Clin. Exp. Metastasis 2015, 32, 353–368. [Google Scholar] [CrossRef] [PubMed]
- Buijs, J.T.; Stayrook, K.R.; Guise, T.A. TGF-β in the bone microenvironment: Role in breast cancer metastases. Cancer Microenviron. 2011, 4, 261–281. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Tuxhorn, J.A.; Ressler, S.J.; McAlhany, S.J.; Dang, T.D.; Rowley, D.R. Stromal expression of connective tissue growth factor promotes angiogenesis and prostate cancer tumorigenesis. Cancer Res. 2005, 65, 8887–8895. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Tian, Y.; Wang, J.S.; Jiang, Y.G.; Luo, Y.; Chen, Y.T. Pituitary tumor-transforming gene 1 regulates invasion of prostate cancer cells through MMP13. Tumour Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Mendonsa, A.M.; VanSaun, M.N.; Ustione, A.; Piston, D.W.; Fingleton, B.M.; Gorden, D.L. Host and tumor derived MMP13 regulate extravasation and establishment of colorectal metastases in the liver. Mol. Cancer 2015, 14. [Google Scholar] [CrossRef] [PubMed]
- Mochizuki, S.; Okada, Y. Adams in cancer cell proliferation and progression. Cancer Sci. 2007, 98, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Ishimi, Y.; Miyaura, C.; Jin, C.H.; Akatsu, T.; Abe, E.; Nakamura, Y.; Yamaguchi, A.; Yoshiki, S.; Matsuda, T.; Hirano, T.; et al. IL-6 is produced by osteoblasts and induces bone resorption. J. Immunol. 1990, 145, 3297–3303. [Google Scholar] [PubMed]
- Onan, D.; Allan, E.H.; Quinn, J.M.; Gooi, J.H.; Pompolo, S.; Sims, N.A.; Gillespie, M.T.; Martin, T.J. The chemokine CXCL1 is a novel target gene of parathyroid hormone (PTH)/PTH-related protein in committed osteoblasts. Endocrinology 2009, 150, 2244–2253. [Google Scholar] [CrossRef] [PubMed]
- Mundy, G.R.; Boyce, B.; Hughes, D.; Wright, K.; Bonewald, L.; Dallas, S.; Harris, S.; Ghosh-Choudhury, N.; Chen, D.; Dunstan, C.; et al. The effects of cytokines and growth factors on osteoblastic cells. Bone 1995, 17, 71S–75S. [Google Scholar] [CrossRef]
- Arnott, J.A.; Lambi, A.G.; Mundy, C.; Hendesi, H.; Pixley, R.A.; Owen, T.A.; Safadi, F.F.; Popoff, S.N. The role of connective tissue growth factor (CTGF/CCN2) in skeletogenesis. Crit. Rev. Eukaryot. Gene Expr. 2011, 21, 43–69. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.Y.; Herber, R.P.; Ho, S.P.; Alliston, T. Matrix metalloproteinase-13 is required for osteocytic perilacunar remodeling and maintains bone fracture resistance. J. Bone Miner. Res. 2012, 27, 1936–1950. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Huang, J.; Guo, R.; Wang, Y.; Chen, D.; Xing, L. Smurf1 inhibits mesenchymal stem cell proliferation and differentiation into osteoblasts through JunB degradation. J. Bone Miner. Res. 2010, 25, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Kenner, L.; Hoebertz, A.; Beil, T.; Keon, N.; Karreth, F.; Eferl, R.; Scheuch, H.; Szremska, A.; Amling, M.; Schorpp-Kistner, M.; et al. Mice lacking JunB are osteopenic due to cell-autonomous osteoblast and osteoclast defects. J. Cell Biol. 2004, 164, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Henriquez, B.; Hepp, M.; Merino, P.; Sepulveda, H.; van Wijnen, A.J.; Lian, J.B.; Stein, G.S.; Stein, J.L.; Montecino, M. C/EBPβ binds the P1 promoter of the Runx2 gene and up-regulates Runx2 transcription in osteoblastic cells. J. Cell Physiol. 2011, 226, 3043–3052. [Google Scholar] [CrossRef] [PubMed]
- Itoh, S.; Udagawa, N.; Takahashi, N.; Yoshitake, F.; Narita, H.; Ebisu, S.; Ishihara, K. A critical role for interleukin-6 family-mediated Stat3 activation in osteoblast differentiation and bone formation. Bone 2006, 39, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Tang, W.; Li, Y. Matrix metalloproteinase 13 (MMP13) is a direct target of osteoblast-specific transcription factor osterix (Osx) in osteoblasts. PLoS ONE 2012, 7, e50525. [Google Scholar] [CrossRef] [PubMed]
- Nannuru, K.C.; Futakuchi, M.; Varney, M.L.; Vincent, T.M.; Marcusson, E.G.; Singh, R.K. Matrix metalloproteinase (MMP)-13 regulates mammary tumor-induced osteolysis by activating MMP9 and transforming growth factor-β signaling at the tumor-bone interface. Cancer Res. 2010, 70, 3494–3504. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Huang, D.; Blick, T.; Connor, A.; Reiter, L.A.; Hardink, J.R.; Lynch, C.C.; Waltham, M.; Thompson, E.W. An MMP13-selective inhibitor delays primary tumor growth and the onset of tumor-associated osteolytic lesions in experimental models of breast cancer. PLoS ONE 2012, 7, e29615. [Google Scholar] [CrossRef] [PubMed]
- Akech, J.; Wixted, J.J.; Bedard, K.; van der Deen, M.; Hussain, S.; Guise, T.A.; van Wijnen, A.J.; Stein, J.L.; Languino, L.R.; Altieri, D.C.; et al. Runx2 association with progression of prostate cancer in patients: Mechanisms mediating bone osteolysis and osteoblastic metastatic lesions. Oncogene 2010, 29, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Sottnik, J.L.; Hall, C.L.; Zhang, J.; Keller, E.T. Wnt and Wnt inhibitors in bone metastasis. BoneKEy Rep. 2012, 1. [Google Scholar] [CrossRef] [PubMed]
- Rabbani, S.A.; Arakelian, A.; Farookhi, R. LRP5 knockdown: Effect on prostate cancer invasion growth and skeletal metastasis in vitro and in vivo. Cancer Med. 2013, 2, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Tai, H.C.; Chang, A.C.; Yu, H.J.; Huang, C.Y.; Tsai, Y.C.; Lai, Y.W.; Sun, H.L.; Tang, C.H.; Wang, S.W. Osteoblast-derived Wnt-induced secreted protein 1 increases VCAM-1 expression and enhances prostate cancer metastasis by down-regulating miR-126. Oncotarget 2014, 5, 7589–7598. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Li, L.; Huang, Q.; Xu, W.; Cai, X.; Zhang, J.; Yan, W.; Song, D.; Liu, T.; Zhou, W.; et al. Wnt signaling through Snail1 and Zeb1 regulates bone metastasis in lung cancer. Am. J. Cancer Res. 2015, 5, 748–755. [Google Scholar] [PubMed]
- Varambally, S.; Yu, J.; Laxman, B.; Rhodes, D.R.; Mehra, R.; Tomlins, S.A.; Shah, R.B.; Chandran, U.; Monzon, F.A.; Becich, M.J.; et al. Integrative genomic and proteomic analysis of prostate cancer reveals signatures of metastatic progression. Cancer Cell 2005, 8, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Liu, Y.; Xu, W.; Sun, Y.; Lu, J.; Wang, F.; Wei, M.; Shen, J.; Hou, J.; Gao, X.; et al. Long noncoding RNA MALAT-1 is a new potential therapeutic target for castration resistant prostate cancer. J. Urol. 2013, 190, 2278–2287. [Google Scholar] [CrossRef] [PubMed]
- Gutschner, T.; Hammerle, M.; Diederichs, S. MALAT1—A paradigm for long noncoding RNA function in cancer. J. Mol. Med. 2013, 91, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Chen, C.; Liu, Y.; Wu, C. 17β-Estradiol treatment inhibits breast cell proliferation, migration and invasion by decreasing MALAT-1 RNA level. Biochem. Biophys. Res. Commun. 2014, 445, 388–393. [Google Scholar] [CrossRef] [PubMed]
- Tano, K.; Mizuno, R.; Okada, T.; Rakwal, R.; Shibato, J.; Masuo, Y.; Ijiri, K.; Akimitsu, N. MALAT-1 enhances cell motility of lung adenocarcinoma cells by influencing the expression of motility-related genes. FEBS Lett. 2010, 584, 4575–4580. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.C.; Yang, Z.; Zhou, L.; Zhu, Q.Q.; Xie, H.Y.; Zhang, F.; Wu, L.M.; Chen, L.M.; Zheng, S.S. Long non-coding RNA MALAT-1 overexpression predicts tumor recurrence of hepatocellular carcinoma after liver transplantation. Med. Oncol. 2012, 29, 1810–1816. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Wu, Y.; Tan, D.; Meng, H.; Wang, K.; Bai, Y.; Yang, K. Up-regulation of long noncoding RNA MALAT1 contributes to proliferation and metastasis in esophageal squamous cell carcinoma. J. Exp. Clin. Cancer Res. CR 2015, 34. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, M.; Wang, Z.; Han, S.; Tang, X.; Ge, Y.; Zhou, L.; Zhou, C.; Yuan, Q.; Yang, M. Silencing of long noncoding RNA MALAT1 by miR-101 and miR-217 inhibits proliferation, migration, and invasion of esophageal squamous cell carcinoma cells. J. Biol. Chem. 2015, 290, 3925–3935. [Google Scholar] [CrossRef] [PubMed]
- Jiao, F.; Hu, H.; Yuan, C.; Wang, L.; Jiang, W.; Jin, Z.; Guo, Z. Elevated expression level of long noncoding RNA MALAT-1 facilitates cell growth, migration and invasion in pancreatic cancer. Oncol. Rep. 2014, 32, 2485–2492. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Yang, M.; Tian, J.; Wang, X.; Li, Z. MALAT-1: A long non-coding RNA and its important 3′ end functional motif in colorectal cancer metastasis. Int. J. Oncol. 2011, 39, 169–175. [Google Scholar] [PubMed]
- Ying, L.; Chen, Q.; Wang, Y.; Zhou, Z.; Huang, Y.; Qiu, F. Upregulated MALAT-1 contributes to bladder cancer cell migration by inducing epithelial-to-mesenchymal transition. Mol. BioSyst. 2012, 8, 2289–2294. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Li, Y.; Liu, Y.; Wang, J.; Li, G. Inhibition of metastasis-associated lung adenocarcinoma transcript 1 in CaSki human cervical cancer cells suppresses cell proliferation and invasion. Acta Biochim. Biophys. Sin. 2010, 42, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Liang, G.; Yuan, B.; Yang, C.; Gao, R.; Zhou, X. MALAT1 promotes the proliferation and metastasis of osteosarcoma cells by activating the PI3K/Akt pathway. Tumour Biol. 2015, 36, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Kan, J.Y.; Wu, D.C.; Yu, F.J.; Wu, C.Y.; Ho, Y.W.; Chiu, Y.J.; Jian, S.F.; Hung, J.Y.; Wang, J.Y.; Kuo, P.L. Chemokine (C–C motif) ligand 5 is involved in tumor-associated dendritic cell-mediated colon cancer progression through non-coding RNA MALAT-1. J. Cell Physiol. 2015, 230, 1883–1894. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.S.; Wang, X.A.; Wu, W.G.; Hu, Y.P.; Li, M.L.; Ding, Q.; Weng, H.; Shu, Y.J.; Liu, T.Y.; Jiang, L.; et al. MALAT1 promotes the proliferation and metastasis of gallbladder cancer cells by activating the ERK/MAPK pathway. Cancer Biol. Ther. 2014, 15, 806–814. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Ellis, J.D.; Shen, Z.; Song, D.Y.; Pan, Q.; Watt, A.T.; Freier, S.M.; Bennett, C.F.; Sharma, A.; Bubulya, P.A.; et al. The nuclear-retained noncoding RNA MALAT1 regulates alternative splicing by modulating SR splicing factor phosphorylation. Mol. Cell 2010, 39, 925–938. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Shen, Z.; Chakraborty, A.; Giri, S.; Freier, S.M.; Wu, X.; Zhang, Y.; Gorospe, M.; Prasanth, S.G.; Lal, A.; et al. Long noncoding RNA MALAT1 controls cell cycle progression by regulating the expression of oncogenic transcription factor B-MYB. PLoS Genet. 2013, 9, e1003368. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sebastian, A.; Hum, N.R.; Hudson, B.D.; Loots, G.G. Cancer–Osteoblast Interaction Reduces Sost Expression in Osteoblasts and Up-Regulates lncRNA MALAT1 in Prostate Cancer. Microarrays 2015, 4, 503-519. https://doi.org/10.3390/microarrays4040503
Sebastian A, Hum NR, Hudson BD, Loots GG. Cancer–Osteoblast Interaction Reduces Sost Expression in Osteoblasts and Up-Regulates lncRNA MALAT1 in Prostate Cancer. Microarrays. 2015; 4(4):503-519. https://doi.org/10.3390/microarrays4040503
Chicago/Turabian StyleSebastian, Aimy, Nicholas R. Hum, Bryan D. Hudson, and Gabriela G. Loots. 2015. "Cancer–Osteoblast Interaction Reduces Sost Expression in Osteoblasts and Up-Regulates lncRNA MALAT1 in Prostate Cancer" Microarrays 4, no. 4: 503-519. https://doi.org/10.3390/microarrays4040503