Magnetoencephalography: Clinical and Research Practices
Abstract
:1. Introduction
1.1. What Is MEG?
1.2. Clinical Uses for MEG
2. MEG at Wake Forest Baptist Health
General MEG Scanning Protocols
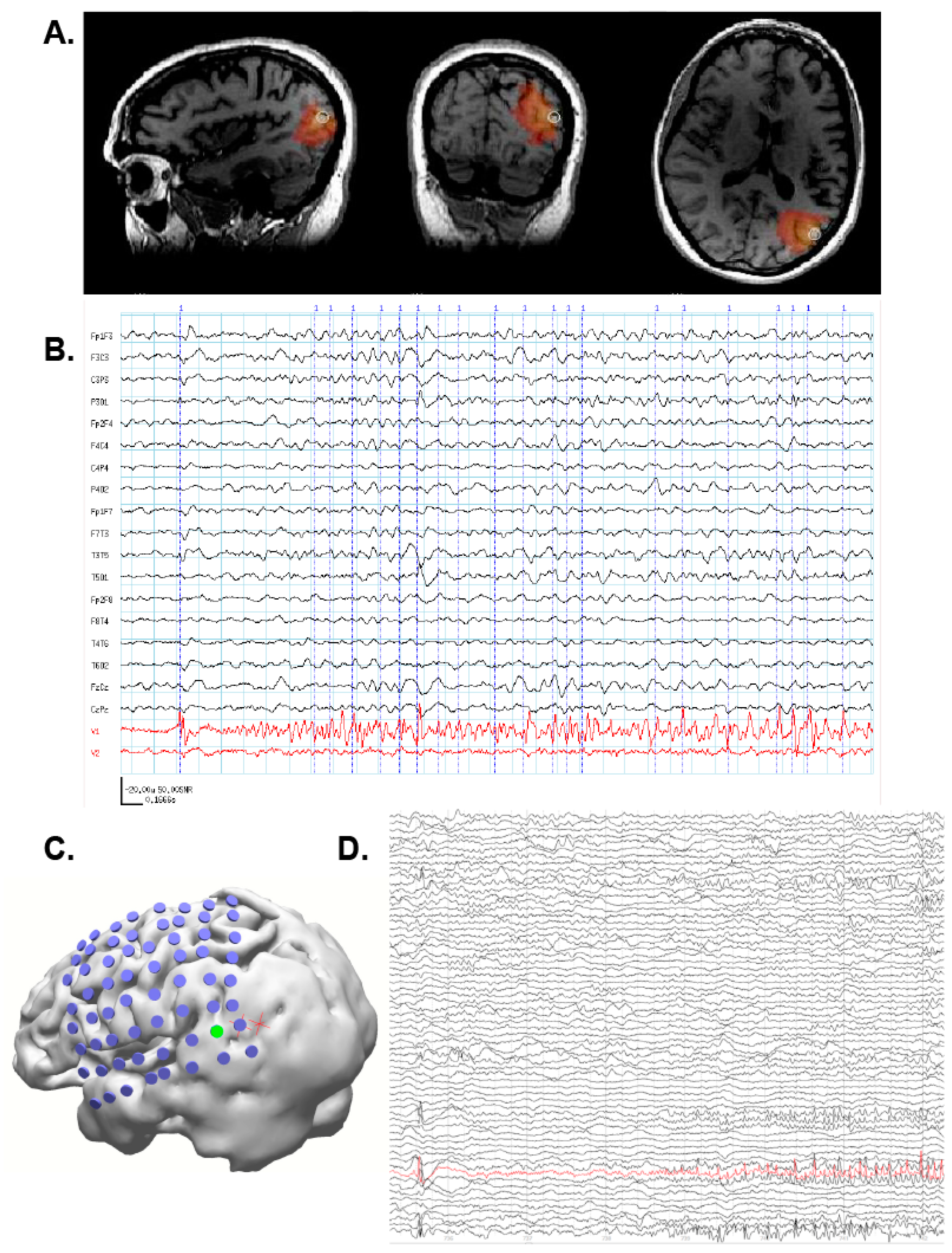
3. Illustrative Clinical Cases
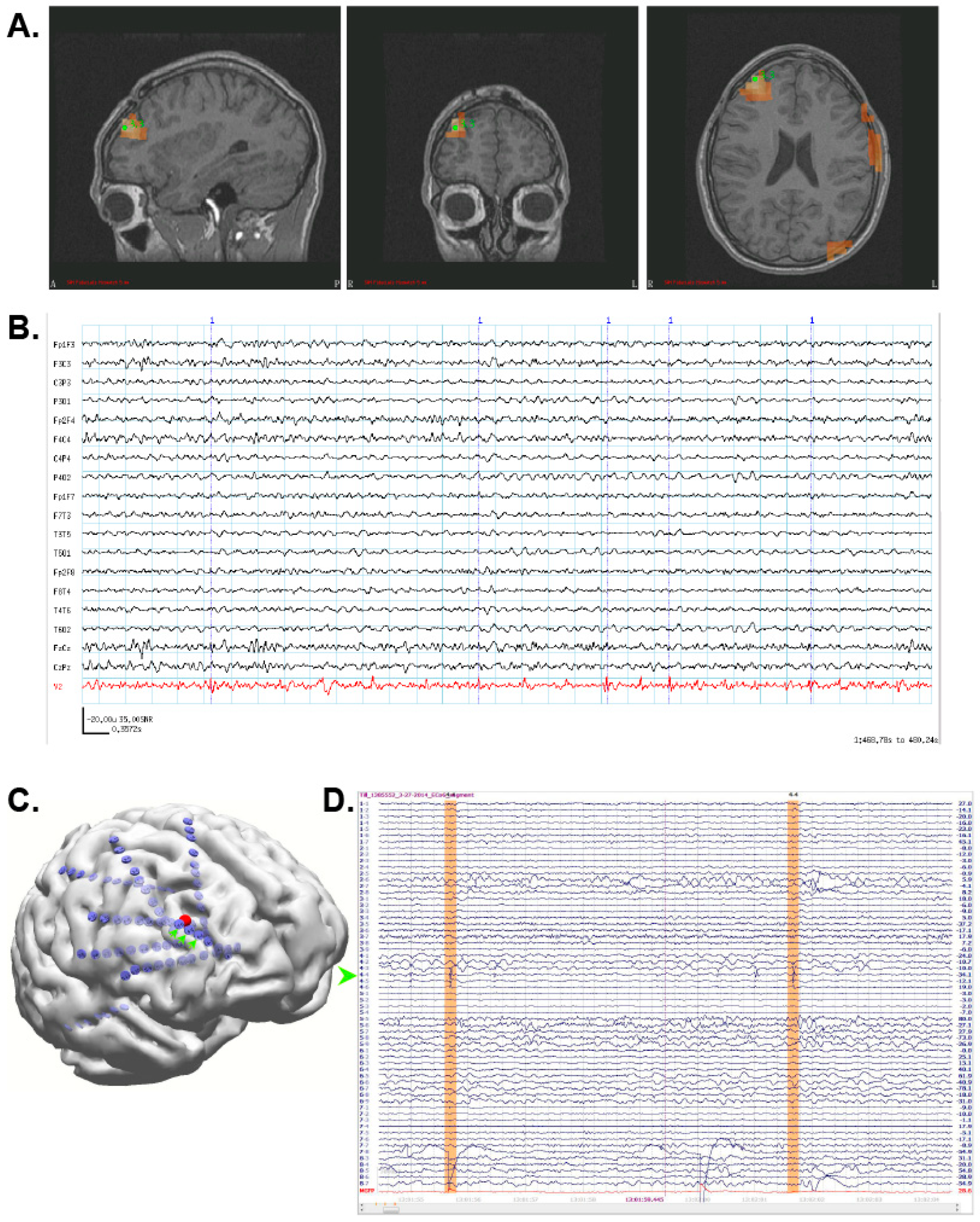
3.1. Case 1: MEG Localization Is Concordant with Multiple Other Modalities
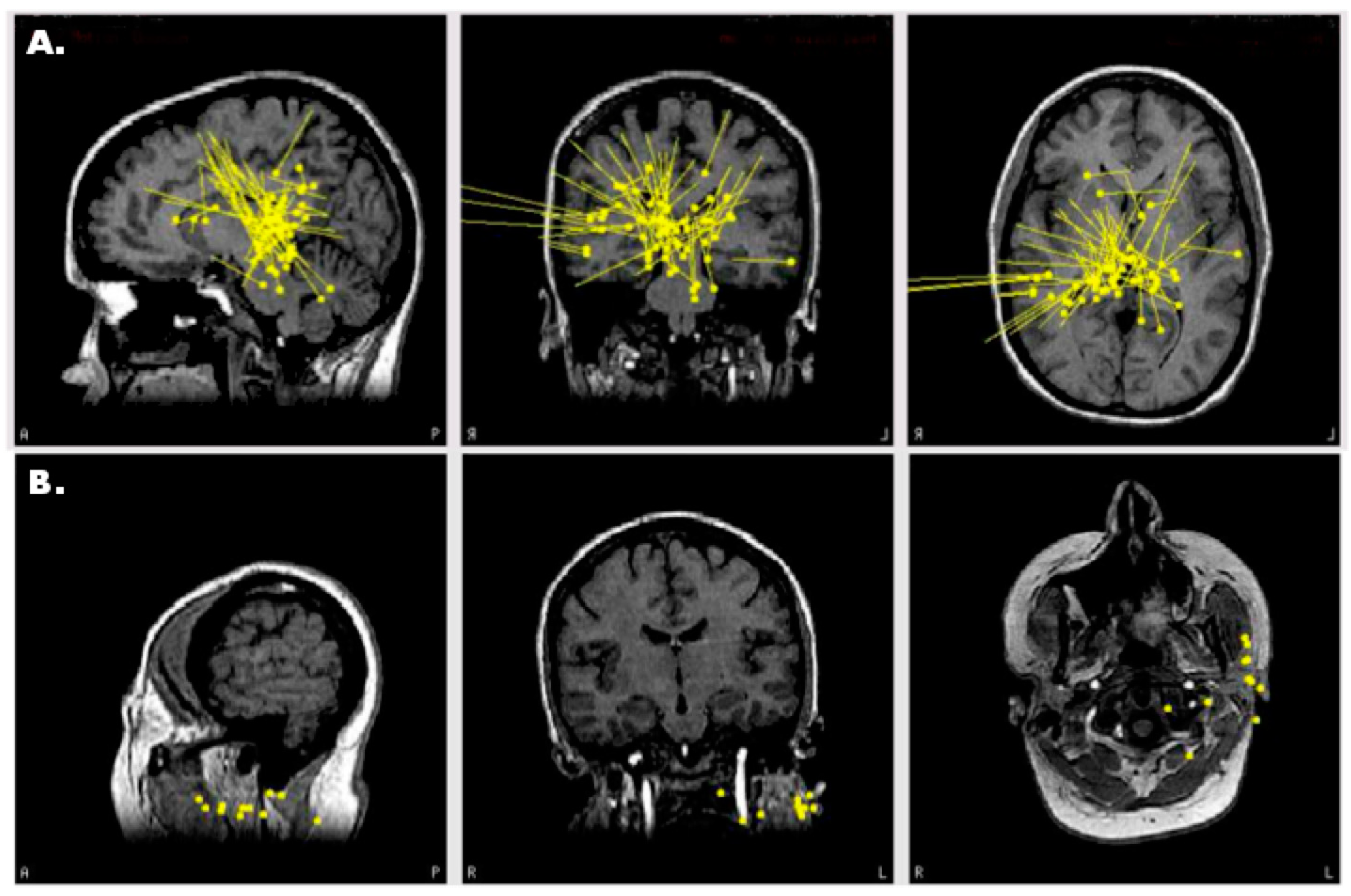
3.2. Case 2: MEG Discrimates among Multiple Seizure Foci
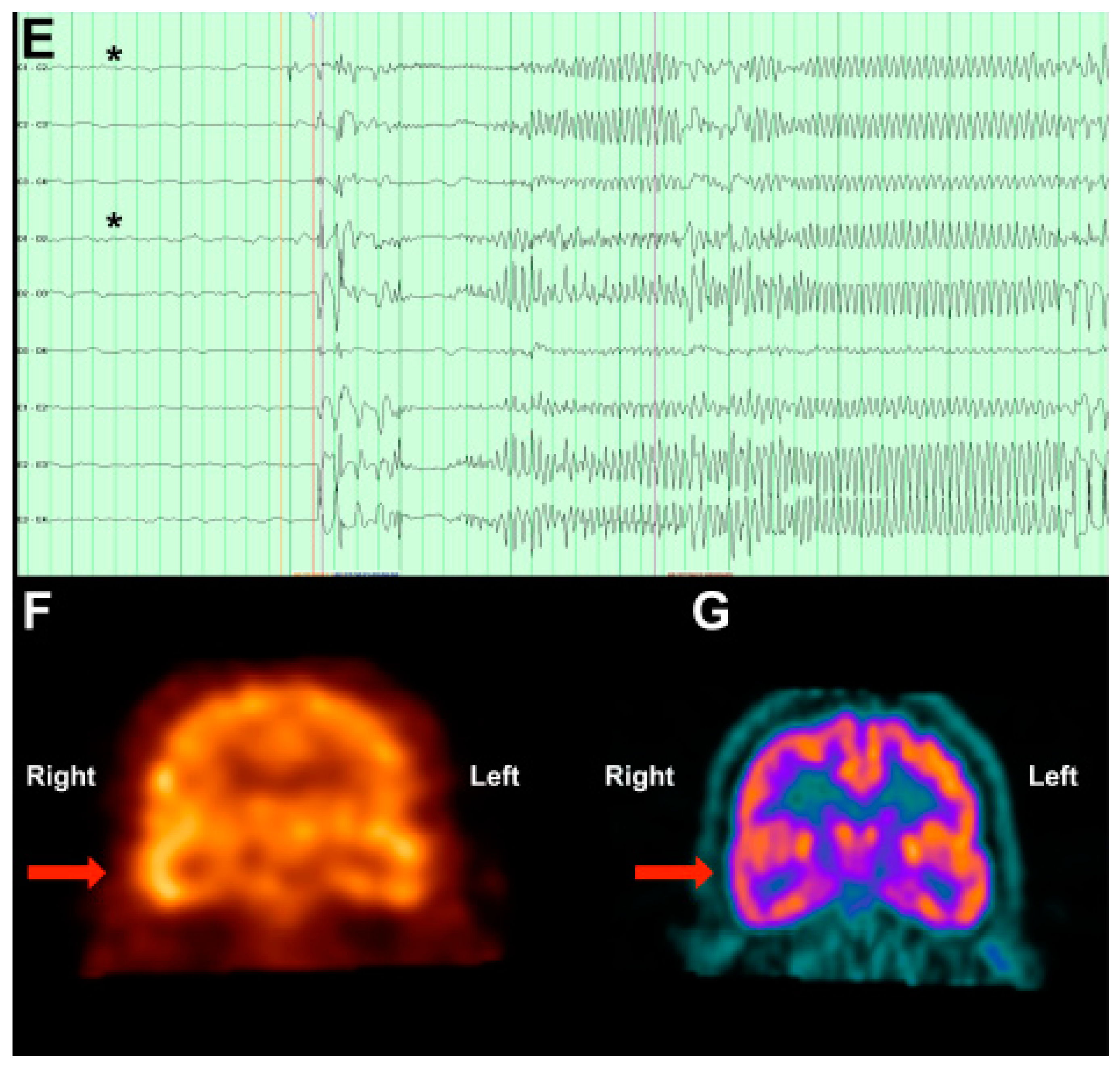
3.3. Case 3: MEG Identifies an Unexpected Seizure Focus
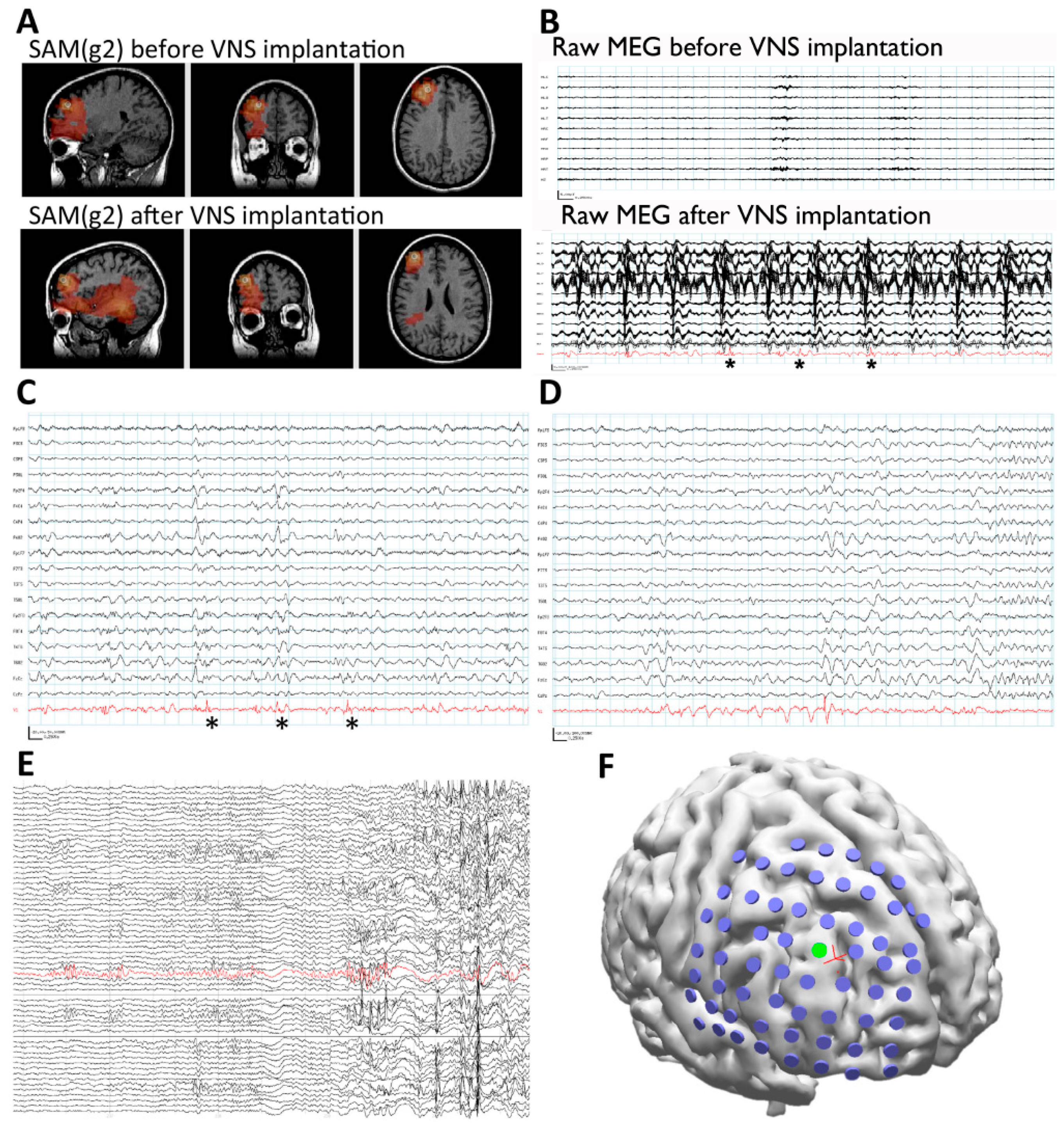
3.4. Case 4: SAM Localization Is Superior to Dipole Analysis
3.5. Case 5: MEG-Only Seizure
4. MEG in Research
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baillet, S.; Mosher, J.C.; Leahy, R.M. Electromagnetic brain mapping. IEEE Signal Process. Mag. 2001, 18, 14–30. [Google Scholar] [CrossRef]
- Caton, R. The Electric Currents of the Brain. BMJ 1875, 2, 278. [Google Scholar] [CrossRef]
- Cohen, D. Magnetoencephalography: Detection of the brain’s electrical activity with a superconducting magnetometer. Science 1972, 175, 664–666. [Google Scholar] [CrossRef] [PubMed]
- Burle, B.; Spieser, L.; Roger, C.; Casini, L.; Hasbroucq, T.; Vidal, F. Spatial and temporal resolutions of EEG: Is it really black and white? A scalp current density view. Int. J. Psychophysiol. 2015, 97, 210–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillebrand, A.; Barnes, G.R.; Hubert, P. Beamformer Analysis of MEG Data. In International Review of Neurobiology; Academic Press: New York, NY, USA, 2005; Volume 68, pp. 149–171. ISBN 0074-7742. [Google Scholar]
- Troebinger, L.; Lopez, J.D.; Lutti, A.; Bradbury, D.; Bestmann, S.; Barnes, G. High precision anatomy for MEG. Neuroimage 2014, 86, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Imai, K.; Otsubo, H.; Sell, E.; Mohamed, I.; Ochi, A.; RamachandranNair, R.; Snead, O.C., 3rd. MEG source estimation from mesio-basal temporal areas in a child with a porencephalic cyst. Acta Neurol. Scand. 2007, 116, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Quraan, M.A.; Moses, S.N.; Hung, Y.; Mills, T.; Taylor, M.J. Detection and localization of hippocampal activity using beamformers with MEG: A detailed investigation using simulations and empirical data. Hum. Brain Mapp. 2011, 32, 812–827. [Google Scholar] [CrossRef] [PubMed]
- Tesche, C.D.; Karhu, J. Somatosensory evoked magnetic fields arising from sources in the human cerebellum. Brain Res. 1997, 744, 23–31. [Google Scholar] [CrossRef]
- Riggs, L.; Moses, S.N.; Bardouille, T.; Herdman, A.T.; Ross, B.; Ryan, J.D. A complementary analytic approach to examining medial temporal lobe sources using magnetoencephalography. Neuroimage 2009, 45, 627–642. [Google Scholar] [CrossRef] [PubMed]
- Stapleton-Kotloski, J.R.; Kotloski, R.J.; Boggs, J.A.; Popli, G.; O’Donovan, C.A.; Couture, D.E.; Cornell, C.; Godwin, D. Localization of Interictal Epileptiform Activity Using Magnetoencephalography with Synthetic Aperture Magnetometry in Patients with a Vagus Nerve Stimulator. Front. Neurol. 2014, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cornwell, B.R.; Carver, F.W.; Coppola, R.; Johnson, L.; Alvarez, R.; Grillon, C. Evoked amygdala responses to negative faces revealed by adaptive MEG beamformers. Brain Res 2008, 1244, 103–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hung, Y.; Smith, M.L.; Bayle, D.J.; Mills, T.; Cheyne, D.; Taylor, M.J. Unattended emotional faces elicit early lateralized amygdala-frontal and fusiform activations. Neuroimage 2010, 50, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Ioannides, A.A.; Corsi-Cabrera, M.; Fenwick, P.B.; del Rio Portilla, Y.; Laskaris, N.A.; Khurshudyan, A.; Theofilou, D.; Shibata, T.; Uchida, S.; Nakabayashi, T.; et al. MEG tomography of human cortex and brainstem activity in waking and REM sleep saccades. Cereb. Cortex 2004, 14, 56–72. [Google Scholar] [CrossRef] [PubMed]
- Balderston, N.L.; Schultz, D.H.; Baillet, S.; Helmstetter, F.J. How to Detect Amygdala Activity with Magnetoencephalography using Source Imaging. JoVE 2013, e50212. [Google Scholar] [CrossRef] [PubMed]
- Dumas, T.; Dubal, S.; Attal, Y.; Chupin, M.; Jouvent, R.; Morel, S.; George, N. MEG Evidence for Dynamic Amygdala Modulations by Gaze and Facial Emotions. PLoS ONE 2013, 8, e74145. [Google Scholar] [CrossRef]
- Styliadis, C.; Ioannides, A.A.; Bamidis, P.D.; Papadelis, C. Amygdala responses to valence and its interaction by arousal revealed by MEG. Int. J. Psychophysiol. 2014, 93, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Martin, T.; Houck, J.M.; Bish, J.P.; Kičić, D.; Woodruff, C.C.; Moses, S.N.; Lee, D.C.; Tesche, C.D. MEG reveals different contributions of somatomotor cortex and cerebellum to simple reaction time after temporally structured cues. Hum. Brain Mapp. 2005, 27, 552–561. [Google Scholar] [CrossRef] [PubMed]
- Styliadis, C.; Ioannides, A.A.; Bamidis, P.D.; Papadelis, C. Distinct cerebellar lobules process arousal, valence and their interaction in parallel following a temporal hierarchy. Neuroimage 2015, 110, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.E.; Vrba, J. Functional neuroimaging by synthetic aperture magnetometry (SAM). Recent Adv. Biomagn. 1999, 302–305. [Google Scholar]
- Robinson, S.E.; Vrba, J.; Otsubo, H.; Ishii, R. Finding epileptic loci by nonlinear parameterization of source waveforms. In Proceedings of the 13th International Conference on Biomagnetism, Jena, Germany, 10–14 August 2002; Nowak, H., Haueisein, J., Giessler, F., Huonker, R., Eds.; VDE Verlag: Berlin, Germany, 2002; pp. 220–222. [Google Scholar]
- Ishii, R.; Shinosaki, K.; Ukai, S.; Inouye, T.; Ishihara, T.; Yoshimine, T.; Hirabuki, N.; Asada, H.; Kihara, T.; Robinson, S.E.; et al. Medial prefrontal cortex generates frontal midline theta rhythm. Neuroreport 1999, 10, 675–679. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.E.; Nagarajan, S.S.; Mantle, M.; Gibbons, V.; Kirsch, H. Localization of interictal spikes using SAM(g2) and dipole fit. Neurol. Clin. Neurophysiol. 2004, 2004, 74. [Google Scholar] [PubMed]
- Robinson, S.E. Localization of event-related activity by SAM(erf). Neurol. Clin. Neurophysiol. 2004, 2004, 109. [Google Scholar] [PubMed]
- Bagic, A.; Funke, M.E.; Ebersole, J. American Clinical MEG Society (ACMEGS) position statement: The value of magnetoencephalography (MEG)/magnetic source imaging (MSI) in noninvasive presurgical evaluation of patients with medically intractable localization-related epilepsy. J. Clin. Neurophysiol. 2009, 26, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Oishi, M.; Otsubo, H.; Iida, K.; Suyama, Y.; Ochi, A.; Weiss, S.K.; Xiang, J.; Gaetz, W.; Cheyne, D.; Chuang, S.H.; et al. Preoperative simulation of intracerebral epileptiform discharges: Synthetic aperture magnetometry virtual sensor analysis of interictal magnetoencephalography data. J. Neurosurg. 2006, 105, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Ray, N.; Kringelbach, M.; Jenkinson, N.; Owen, S.; Davies, P.; Wang, S.; De Pennington, N.; Hansen, P.; Stein, J.; Aziz, T. Using magnetoencephalography to investigate brain activity during high frequency deep brain stimulation in a cluster headache patient. Biomed. Imaging Interv. J. 2007, 3, e25. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.S.; Acevedo, C.; Arzimanoglou, A.; Bogacz, A.; Cross, J.H.; Elger, C.E.; Engel, J.; Forsgren, L.; French, J.A.; Glynn, M.; et al. ILAE official report: A practical clinical definition of epilepsy. Epilepsia 2014, 55, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.S.; van Emde Boas, W.; Blume, W.; Elger, C.; Genton, P.; Lee, P.; Engel, J. Epileptic seizures and epilepsy: Definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia 2005, 46, 470–472. [Google Scholar] [CrossRef] [PubMed]
- Hesdorffer, D.C.; Begley, C.E. Surveillance of epilepsy and prevention of epilepsy and its sequelae: Lessons from the Institute of Medicine report. Curr. Opin. Neurol. 2013, 26, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Aaberg, K.M.; Gunnes, N.; Bakken, I.J.; Lund Søraas, C.; Berntsen, A.; Magnus, P.; Lossius, M.I.; Stoltenberg, C.; Chin, R.; Surén, P. Incidence and Prevalence of Childhood Epilepsy: A Nationwide Cohort Study. Pediatrics 2017, 139, e20163908. [Google Scholar] [CrossRef] [PubMed]
- Cowan, L.D.; Bodensteiner, J.B.; Leviton, A.; Doherty, L. Prevalence of the Epilepsies in Children and Adolescents. Epilepsia 2018, 30, 94–106. [Google Scholar] [CrossRef]
- Schmidt, D.; Schachter, S.C. Drug treatment of epilepsy in adults. BMJ 2014, 348, g254. [Google Scholar] [CrossRef] [PubMed]
- Begley, C.E.; Famulari, M.; Annegers, J.F.; Lairson, D.R.; Reynolds, T.F.; Coan, S.; Dubinsky, S.; Newmark, M.E.; Leibson, C.; So, E.L.; et al. The cost of epilepsy in the United States: An estimate from population-based clinical and survey data. Epilepsia 2000, 41, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Wiebe, S.; Blume, W.T.; Girvin, J.P.; Eliasziw, M. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N. Engl. J. Med. 2001, 345, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Jobst, B.C.; Cascino, G.D. Resective Epilepsy Surgery for Drug-Resistant Focal Epilepsy. JAMA 2015, 313, 285. [Google Scholar] [CrossRef] [PubMed]
- Spencer, S.; Huh, L. Outcomes of epilepsy surgery in adults and children. Lancet Neurol. 2008, 7, 525–537. [Google Scholar] [CrossRef]
- Jayakar, P.; Gaillard, W.D.; Tripathi, M.; Libenson, M.H.; Mathern, G.W.; Cross, J.H. Diagnostic test utilization in evaluation for resective epilepsy surgery in children. Epilepsia 2014, 55, 507–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knowlton, R.C.; Razdan, S.N.; Limdi, N.; Elgavish, R.A.; Killen, J.; Blount, J.; Burneo, J.G.; Ver Hoef, L.; Paige, L.; Faught, E.; et al. Effect of epilepsy magnetic source imaging on intracranial electrode placement. Ann. Neurol. 2009, 65, 716–723. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutherling, W.W.; Crandall, P.H.; Cahan, L.D.; Barth, D.S. The magnetic field of epileptic spikes agrees with intracranial localizations in complex partial epilepsy. Neurology 1988, 38, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, H.M.; Papanicolaou, A.C.; Baumann, S.B.; Rogers, R.L.; Brown, L.M. Magnetoencephalographic localization of interictal spike sources. Case report. J. Neurosurg. 1991, 74, 660–664. [Google Scholar] [CrossRef] [PubMed]
- Stefan, H.; Schneider, S.; Feistel, H.; Pawlik, G.; Schüler, P.; Abraham-Fuchs, K.; Schlegel, T.; Neubauer, U.; Huk, W.J. Ictal and Interictal Activity in Partial Epilepsy Recorded with Multichannel Magnetoelectroencephalography: Correlation of Electroencephalography/Electrocorticography, Magnetic Resonance Imaging, Single Photon Emission Computed Tomography, and Positron Em. Epilepsia 1992, 33, 874–887. [Google Scholar] [CrossRef] [PubMed]
- Barth, D.S.; Sutherling, W.; Engel, J.; Beatty, J. Neuromagnetic localization of epileptiform spike activity in the human brain. Science 1982, 218, 891–894. [Google Scholar] [CrossRef] [PubMed]
- Stefan, H.; Hummel, C.; Scheler, G.; Genow, A.; Druschky, K.; Tilz, C.; Kaltenhauser, M.; Hopfengartner, R.; Buchfelder, M.; Romstock, J. Magnetic brain source imaging of focal epileptic activity: A synopsis of 455 cases. Brain 2003, 126, 2396–2405. [Google Scholar] [CrossRef] [PubMed]
- Knake, S.; Halgren, E.; Shiraishi, H.; Hara, K.; Hamer, H.M.; Grant, P.E.; Carr, V.A.; Foxe, D.; Camposano, S.; Busa, E.; et al. The value of multichannel MEG and EEG in the presurgical evaluation of 70 epilepsy patients. Epilepsy Res. 2006, 69, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Pataraia, E.; Simos, P.G.; Castillo, E.M.; Billingsley, R.L.; Sarkari, S.; Wheless, J.W.; Maggio, V.; Maggio, W.; Baumgartner, J.E.; Swank, P.R.; et al. Does magnetoencephalography add to scalp video-EEG as a diagnostic tool in epilepsy surgery? Neurology 2004, 62, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Oishi, M.; Kameyama, S.; Masuda, H.; Tohyama, J.; Kanazawa, O.; Sasagawa, M.; Otsubo, H. Single and multiple clusters of magnetoencephalographic dipoles in neocortical epilepsy: Significance in characterizing the epileptogenic zone. Epilepsia 2006, 47, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Mamelak, A.N.; Lopez, N.; Akhtari, M.; Sutherling, W.W. Magnetoencephalography-directed surgery in patients with neocortical epilepsy. J. Neurosurg. 2002, 97, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Knowlton, R.C.; Laxer, K.D.; Aminoff, M.J.; Roberts, T.P.; Wong, S.T.; Rowley, H.A. Magnetoencephalography in partial epilepsy: Clinical yield and localization accuracy. Ann. Neurol. 1997, 42, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Iida, K.; Otsubo, H.; Matsumoto, Y.; Ochi, A.; Oishi, M.; Holowka, S.; Pang, E.; Elliott, I.; Weiss, S.K.; Chuang, S.H.; et al. Characterizing magnetic spike sources by using magnetoencephalography-guided neuronavigation in epilepsy surgery in pediatric patients. J. Neurosurg. 2005, 102, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Ishii, R.; Canuet, L.; Ochi, A.; Xiang, J.; Imai, K.; Chan, D.; Iwase, M.; Takeda, M.; Snead, O.C., 3rd; Otsubo, H. Spatially filtered magnetoencephalography compared with electrocorticography to identify intrinsically epileptogenic focal cortical dysplasia. Epilepsy Res. 2008, 81, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Knowlton, R.C.; Elgavish, R.; Howell, J.; Blount, J.; Burneo, J.G.; Faught, E.; Kankirawatana, P.; Riley, K.; Morawetz, R.; Worthington, J.; et al. Magnetic source imaging versus intracranial electroencephalogram in epilepsy surgery: A prospective study. Ann. Neurol. 2006, 59, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Xiang, J.; Holowka, S.; Hunjan, A.; Sharma, R.; Otsubo, H.; Chuang, S. Volumetric localization of epileptic activities in tuberous sclerosis using synthetic aperture magnetometry. Pediatr. Radiol. 2006, 36, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Canuet, L.; Ishii, R.; Iwase, M.; Kurimoto, R.; Ikezawa, K.; Azechi, M.; Wataya-Kaneda, M.; Takeda, M. Tuberous sclerosis: Localizing the epileptogenic tuber with synthetic aperture magnetometry with excess kurtosis analysis. J. Clin. Neurosci. 2008, 15, 1296–1298. [Google Scholar] [CrossRef] [PubMed]
- Iida, K.; Otsubo, H.; Mohamed, I.S.; Okuda, C.; Ochi, A.; Weiss, S.K.; Chuang, S.H.; Snead, O.C., 3rd. Characterizing magnetoencephalographic spike sources in children with tuberous sclerosis complex. Epilepsia 2005, 46, 1510–1517. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, I.; Imai, K.; Yamaguchi, Y.; Ochi, A.; Akizuki, Y.; Go, C.; Akiyama, T.; Snead, O.C., 3rd; Rutka, J.T.; Drake, J.M.; et al. Localization of epileptic foci in children with intractable epilepsy secondary to multiple cortical tubers by using synthetic aperture magnetometry kurtosis. J. Neurosurg. Pediatr. 2009, 4, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Hunold, A.; Haueisen, J.; Ahtam, B.; Doshi, C.; Harini, C.; Camposano, S.; Warfield, S.; Grant, P.E.; Okada, Y.; Papadelis, C. Localization of the Epileptogenic Foci in Tuberous Sclerosis Complex: A Pediatric Case Report. Front. Hum. Neurosci. 2014, 8, 175. [Google Scholar] [CrossRef] [PubMed]
- Jansen, F.E.; Huiskamp, G.; van Huffelen, A.C.; Bourez-Swart, M.; Boere, E.; Gebbink, T.; Vincken, K.L.; van Nieuwenhuize, O. Identification of the Epileptogenic Tuber in Patients with Tuberous Sclerosis: A Comparison of High-resolution EEG and MEG. Epilepsia 2006, 47, 108–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fischer, M.J.; Scheler, G.; Stefan, H. Utilization of magnetoencephalography results to obtain favourable outcomes in epilepsy surgery. Brain 2005, 128, 153–157. [Google Scholar] [CrossRef] [PubMed]
- RamachandranNair, R.; Otsubo, H.; Shroff, M.M.; Ochi, A.; Weiss, S.K.; Rutka, J.T.; Snead, O.C., 3rd. MEG predicts outcome following surgery for intractable epilepsy in children with normal or nonfocal MRI findings. Epilepsia 2007, 48, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Leiken, K.; Xiang, J.; Zhang, F.; Shi, J.; Tang, L.; Liu, H.; Wang, X. Magnetoencephalography Detection of High-Frequency Oscillations in the Developing Brain. Front. Hum. Neurosci. 2014, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jirsch, J.D.; Urrestarazu, E.; LeVan, P.; Olivier, A.; Dubeau, F.; Gotman, J. High-frequency oscillations during human focal seizures. Brain 2006, 129, 1593–1608. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.; LeVan, P.; Chander, R.; Hall, J.; Dubeau, F.; Gotman, J. Interictal high-frequency oscillations (80-500 Hz) are an indicator of seizure onset areas independent of spikes in the human epileptic brain. Epilepsia 2008, 49, 1893–1907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zijlmans, M.; Jacobs, J.; Kahn, Y.U.; Zelmann, R.; Dubeau, F.; Gotman, J. Ictal and interictal high frequency oscillations in patients with focal epilepsy. Clin. Neurophysiol. 2011, 122, 664–671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamilia, E.; Madsen, J.R.; Grant, P.E.; Pearl, P.L.; Papadelis, C. Current and Emerging Potential of Magnetoencephalography in the Detection and Localization of High-Frequency Oscillations in Epilepsy. Front. Neurol. 2017, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Nariai, H.; Nagasawa, T.; Juhasz, C.; Sood, S.; Chugani, H.T.; Asano, E. Statistical mapping of ictal high-frequency oscillations in epileptic spasms. Epilepsia 2011, 52, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.; Zijlmans, M.; Zelmann, R.; Chatillon, C.E.; Hall, J.; Olivier, A.; Dubeau, F.; Gotman, J. High-frequency electroencephalographic oscillations correlate with outcome of epilepsy surgery. Ann. Neurol. 2010, 67, 209–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haegelen, C.; Perucca, P.; Chatillon, C.E.; Andrade-Valenca, L.; Zelmann, R.; Jacobs, J.; Collins, D.L.; Dubeau, F.; Olivier, A.; Gotman, J. High-frequency oscillations, extent of surgical resection, and surgical outcome in drug-resistant focal epilepsy. Epilepsia 2013, 54, 848–857. [Google Scholar] [CrossRef] [PubMed]
- Xiang, J.; Liu, Y.; Wang, Y.; Kirtman, E.G.; Kotecha, R.; Chen, Y.; Huo, X.; Fujiwara, H.; Hemasilpin, N.; Lee, K.; et al. Frequency and spatial characteristics of high-frequency neuromagnetic signals in childhood epilepsy. Epileptic Disord. 2009, 11, 113–125. [Google Scholar] [CrossRef] [PubMed]
- van Klink, N.; Hillebrand, A.; Zijlmans, M. Identification of epileptic high frequency oscillations in the time domain by using MEG beamformer-based virtual sensors. Clin. Neurophysiol. 2016, 127, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Papadelis, C.; Tamilia, E.; Stufflebeam, S.; Grant, P.E.; Madsen, J.R.; Pearl, P.L.; Tanaka, N. Interictal High Frequency Oscillations Detected with Simultaneous Magnetoencephalography and Electroencephalography as Biomarker of Pediatric Epilepsy. JoVE 2016, e54883. [Google Scholar] [CrossRef] [PubMed]
- Papadelis, C.; Poghosyan, V.; Fenwick, P.B.C.; Ioannides, A.A. MEG’s ability to localise accurately weak transient neural sources. Clin. Neurophysiol. 2009, 120, 1958–1970. [Google Scholar] [CrossRef] [PubMed]
- Hirata, M.; Kato, A.; Taniguchi, M.; Ninomiya, H.; Cheyne, D.; Robinson, S.E.; Maruno, M.; Kumura, E.; Ishii, R.; Hirabuki, N.; et al. Frequency-dependent spatial distribution of human somatosensory evoked neuromagnetic fields. Neurosci. Lett. 2002, 318, 73–76. [Google Scholar] [CrossRef]
- Gaetz, W.C.; Cheyne, D.O. Localization of human somatosensory cortex using spatially filtered magnetoencephalography. Neurosci. Lett. 2003, 340, 161–164. [Google Scholar] [CrossRef]
- Papadelis, C.; Eickhoff, S.B.; Zilles, K.; Ioannides, A.A. BA3b and BA1 activate in a serial fashion after median nerve stimulation: Direct evidence from combining source analysis of evoked fields and cytoarchitectonic probabilistic maps. Neuroimage 2011, 54, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Papadelis, C.; Leonardelli, E.; Staudt, M.; Braun, C. Can magnetoencephalography track the afferent information flow along white matter thalamo-cortical fibers? Neuroimage 2012, 60, 1092–1105. [Google Scholar] [CrossRef] [PubMed]
- Ioannides, A.A.; Liu, L.; Poghosyan, V.; Saridis, G.A.; Gjedde, A.; Ptito, M.; Kupers, R. MEG reveals a fast pathway from somatosensory cortex to occipital areas via posterior parietal cortex in a blind subject. Front. Hum. Neurosci. 2013, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adjamian, P.; Worthen, S.F.; Hillebrand, A.; Furlong, P.L.; Chizh, B.A.; Hobson, A.R.; Aziz, Q.; Barnes, G.R. Effective electromagnetic noise cancellation with beamformers and synthetic gradiometry in shielded and partly shielded environments. J. Neurosci. Methods 2009, 178, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Gaetz, W.; Cheyne, D. Localization of sensorimotor cortical rhythms induced by tactile stimulation using spatially filtered MEG. Neuroimage 2006, 30, 899–908. [Google Scholar] [CrossRef] [PubMed]
- Cheyne, D.; Bostan, A.C.; Gaetz, W.; Pang, E.W. Event-related beamforming: A robust method for presurgical functional mapping using MEG. Clin. Neurophysiol. 2007, 118, 1691–1704. [Google Scholar] [CrossRef] [PubMed]
- Nevalainen, P.; Pihko, E.; Metsaranta, M.; Sambeth, A.; Wikstrom, H.; Okada, Y.; Autti, T.; Lauronen, L. Evoked magnetic fields from primary and secondary somatosensory cortices: A reliable tool for assessment of cortical processing in the neonatal period. Clin. Neurophysiol. 2011, 123, 2377–2383. [Google Scholar] [CrossRef] [PubMed]
- Hoshiyama, M.; Kakigi, R.; Koyama, S.; Watanabe, S.; Shimojo, M. Activity in Posterior Parietal Cortex Following Somatosensory Stimulation in Man: Magnetoencephalographic Study Using Spatio-Temporal Source Analysis. Brain Topogr. 1997, 10, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Hoshiyama, M.; Kakigi, R.; Berg, P.; Koyama, S.; Kitamura, Y.; Shimojo, M.; Watanabe, S.; Nakamura, A. Identification of motor and sensory brain activities during unilateral finger movement: Spatiotemporal source analysis of movement-associated magnetic fields. Exp. Brain Res. 1997, 115, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Yamada, T.; Goto, A.; Kato, T.; Ito, K.; Abe, Y.; Kachi, T.; Kakigi, R. Somatosensory Homunculus as Drawn by MEG. Neuroimage 1998, 7, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Inui, K.; Wang, X.; Tamura, Y.; Kaneoke, Y.; Kakigi, R. Serial processing in the human somatosensory system. Cereb. Cortex 2004, 14, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Pang, E.W.; Drake, M.; Otsubo, H. Intraoperative Confirmation of Hand Motor Area Identified Preoperatively by Magnetoencephalography. Pediatr. Neurosurg. 2008, 44, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Cheyne, D.; Bakhtazad, L.; Gaetz, W. Spatiotemporal mapping of cortical activity accompanying voluntary movements using an event-related beamforming approach. Hum. Brain Mapp. 2006, 27, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Cheyne, D.; Bells, S.; Ferrari, P.; Gaetz, W.; Bostan, A.C. Self-paced movements induce high-frequency gamma oscillations in primary motor cortex. Neuroimage 2008, 42, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Cheyne, D.; Ferrari, P. MEG studies of motor cortex gamma oscillations: Evidence for a gamma “fingerprint” in the brain? Front. Hum. Neurosci. 2013, 7, 575. [Google Scholar] [CrossRef] [PubMed]
- Shigihara, Y.; Zeki, S. Parallel processing of face and house stimuli by V1 and specialized visual areas: A magnetoencephalographic (MEG) study. Front. Hum. Neurosci. 2014, 8, 901. [Google Scholar] [CrossRef] [PubMed]
- Hirata, M.; Kato, A.; Taniguchi, M.; Saitoh, Y.; Ninomiya, H.; Ihara, A.; Kishima, H.; Oshino, S.; Baba, T.; Yorifuji, S.; et al. Determination of language dominance with synthetic aperture magnetometry: Comparison with the Wada test. Neuroimage 2004, 23, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Chung, C.K. Language lateralization using MEG beta frequency desynchronization during auditory oddball stimulation with one-syllable words. Neuroimage 2008, 42, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, I.S.; Cheyne, D.; Gaetz, W.C.; Otsubo, H.; Logan, W.J.; Carter Snead, O., 3rd; Pang, E.W. Spatiotemporal patterns of oscillatory brain activity during auditory word recognition in children: A synthetic aperture magnetometry study. Int. J. Psychophysiol. 2008, 68, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Papanicolaou, A.C.; Simos, P.G.; Castillo, E.M.; Breier, J.I.; Sarkari, S.; Pataraia, E.; Billingsley, R.L.; Buchanan, S.; Wheless, J.; Maggio, V.; et al. Magnetocephalography: A noninvasive alternative to the Wada procedure. J. Neurosurg. 2004, 100, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Papanicolaou, A.C.; Pazo-Alvarez, P.; Castillo, E.M.; Billingsley-Marshall, R.L.; Breier, J.I.; Swank, P.R.; Buchanan, S.; McManis, M.; Clear, T.; Passaro, A.D. Functional neuroimaging with MEG: Normative language profiles. Neuroimage 2006, 33, 326–342. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Rezaie, R.; Papanicolaou, A.C. Functional Neuroimaging of Language Using Magnetoencephalography. Phys. Life Rev. 2009, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Sutherling, W.W.; Mamelak, A.N.; Thyerlei, D.; Maleeva, T.; Minazad, Y.; Philpott, L.; Lopez, N. Influence of magnetic source imaging for planning intracranial EEG in epilepsy. Neurology 2008, 71, 990–996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knowlton, R.C.; Elgavish, R.A.; Limdi, N.; Bartolucci, A.; Ojha, B.; Blount, J.; Burneo, J.G.; Ver Hoef, L.; Paige, L.; Faught, E.; et al. Functional imaging: I. Relative predictive value of intracranial electroencephalography. Ann. Neurol. 2008, 64, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Knowlton, R.C.; Elgavish, R.A.; Bartolucci, A.; Ojha, B.; Limdi, N.; Blount, J.; Burneo, J.G.; Ver Hoef, L.; Paige, L.; Faught, E.; et al. Functional imaging: II. Prediction of epilepsy surgery outcome. Ann. Neurol. 2008, 64, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, I.S.; Otsubo, H.; Ferrari, P.; Sharma, R.; Ochi, A.; Elliott, I.; Go, C.; Chuang, S.; Rutka, J.; Snead, C., 3rd; Cheyne, D. Source localization of interictal spike-locked neuromagnetic oscillations in pediatric neocortical epilepsy. Clin. Neurophysiol. 2013, 124, 1517–1527. [Google Scholar] [CrossRef] [PubMed]
- Gaetz, W.; Gordon, R.S.; Papadelis, C.; Fujiwara, H.; Rose, D.F.; Edgar, J.C.; Schwartz, E.S.; Roberts, T.P.L. Magnetoencephalography for Clinical Pediatrics: Recent Advances in Hardware, Methods, and Clinical Applications. J. Pediatr. Epilepsy 2015, 4, 139–155. [Google Scholar] [CrossRef]
- Papadelis, C.; Harini, C.; Ahtam, B.; Doshi, C.; Grant, E.; Okada, Y. Current and emerging potential for magnetoencephalography in pediatric epilepsy. J. Pediatr. Epilepsy 2013, 2, 73–85. [Google Scholar] [CrossRef]
- Okada, Y.; Pratt, K.; Atwood, C.; Mascarenas, A.; Reineman, R.; Nurminen, J.; Paulson, D. BabySQUID: A mobile, high-resolution multichannel magnetoencephalography system for neonatal brain assessment. Rev. Sci. Instrum. 2006, 77, 24301. [Google Scholar] [CrossRef]
- Johnson, B.W.; Crain, S.; Thornton, R.; Tesan, G.; Reid, M. Measurement of brain function in pre-school children using a custom sized whole-head MEG sensor array. Clin. Neurophysiol. 2010, 121, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, M.; Yoshimura, Y.; Shitamichi, K.; Ueno, S.; Hirosawa, T.; Munesue, T.; Ono, Y.; Tsubokawa, T.; Haruta, Y.; Oi, M.; et al. A custom magnetoencephalography device reveals brain connectivity and high reading/decoding ability in children with autism. Sci. Rep. 2013, 3, 1139. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.; Paulson, D.; Hirschkoff, G.; Pratt, K.; Mascarenas, A.; Miller, P.; Han, M.; Caffrey, J.; Kincade, C.; Power, W.; et al. Artemis 123: Development of a whole-head infant and young child MEG system. Front. Hum. Neurosci. 2014, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Brock, J.; Johnson, B.W. Face-sensitive brain responses measured from a four-year-old child with a custom-sized child MEG system. J. Neurosci. Methods 2014, 222, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Hara, K.; Lin, F.-H.; Camposano, S.; Foxe, D.M.; Grant, P.E.; Bourgeois, B.F.; Ahlfors, S.P.; Stufflebeam, S.M. Magnetoencephalographic mapping of interictal spike propagation: A technical and clinical report. AJNR. Am. J. Neuroradiol. 2007, 28, 1486–1488. [Google Scholar] [CrossRef] [PubMed]
- Vrba, J.; Robinson, S. Signal processing in magnetoencephalography. Methods 2001, 25, 249–271. [Google Scholar] [CrossRef] [PubMed]
- Vrba, J.; Robinson, S.E. The effect of environmental noise on magnetometer- and gardiometer-based MEG systems. In Proceedings of the Biomag 2000: 12th International Conference on Biomagnetism, Espoo, Finland, 13–17 August 2000; pp. 953–956. [Google Scholar]
- Fife, A.A.; Vrba, J.; Robinson, S.E.; Anderson, G.; Betts, K.; Burbank, M.B.; Cheyne, D.; Cheung, T.; Govorkov, S.; Haid, G.; et al. Synthetic gradiometer systems for MEG. IEEE Trans. Appl. Supercond. 1999, 9, 4063–4068. [Google Scholar] [CrossRef]
- Kirsch, H.E.; Robinson, S.E.; Mantle, M.; Nagarajan, S. Automated localization of magnetoencephalographic interictal spikes by adaptive spatial filtering. Clin. Neurophysiol. 2006, 117, 2264–2271. [Google Scholar] [CrossRef] [PubMed]
- Ukai, S.; Kawaguchi, S.; Ishii, R.; Yamamoto, M.; Ogawa, A.; Mizuno-Matsumoto, Y.; Robinson, S.E.; Fujita, N.; Yoshimine, T.; Shinosaki, K.; et al. SAM(g2) analysis for detecting spike localization: A comparison with clinical symptoms and ECD analysis in an epileptic patient. Neurol. Clin. Neurophysiol. 2004, 2004, 57. [Google Scholar] [PubMed]
- Zhang, R.; Wu, T.; Wang, Y.; Liu, H.; Zou, Y.; Liu, W.; Xiang, J.; Xiao, C.; Yang, L.; Fu, Z. Interictal magnetoencephalographic findings related with surgical outcomes in lesional and nonlesional neocortical epilepsy. Seizure 2011, 20, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Negrillo, A. Influence of Sleep and Sleep Deprivation on Ictal and Interictal Epileptiform Activity. Epilepsy Res. Treat. 2013, 2013, 492524. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.X.; Mosher, J.C.; Leahy, R.M. A sensor-weighted overlapping-sphere head model and exhaustive head model comparison for MEG. Phys. Med. Biol. 1999, 44, 423–440. [Google Scholar] [CrossRef] [PubMed]
- Rowland, J.A.; Stapleton-Kotloski, J.R.; Dobbins, D.L.; Rogers, E.; Godwin, D.W.; Taber, K.H. Increased Small-World Network Topology Following Deployment-Acquired Traumatic Brain Injury Associated with the Development of Post-Traumatic Stress Disorder. Brain Connect. 2018, 8, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Rowland, J.A.; Stapleton-Kotloski, J.R.; Alberto, G.E.; Rawley, J.A.; Kotloski, R.J.; Taber, K.H.; Godwin, D.W. Contrasting Effects of Posttraumatic Stress Disorder and Mild Traumatic Brain Injury on the Whole-Brain Resting-State Network: A Magnetoencephalography Study. Brain Connect. 2017, 7, 45–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rowland, J.A.; Stapleton-Kotloski, J.R.; Alberto, G.E.; Davenport, A.T.; Kotloski, R.J.; Friedman, D.P.; Godwin, D.W.; Daunais, J.B. Changes in nonhuman primate brain function following chronic alcohol consumption in previously naïve animals. Drug Alcohol Depend. 2017, 177, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Taulu, S.; Hari, R. Removal of magnetoencephalographic artifacts with temporal signal-space separation: Demonstration with single-trial auditory-evoked responses. Hum. Brain Mapp. 2009, 30, 1524–1534. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.W.; Godwin, D.W.; Czoty, P.W.; Nader, M.A.; Kraft, R.A.; Buchheimer, N.C.; Daunais, J.B. A MEG investigation of somatosensory processing in the rhesus monkey. Neuroimage 2009, 46, 998–1003. [Google Scholar] [CrossRef] [PubMed] [Green Version]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stapleton-Kotloski, J.R.; Kotloski, R.J.; Popli, G.; Godwin, D.W. Magnetoencephalography: Clinical and Research Practices. Brain Sci. 2018, 8, 157. https://doi.org/10.3390/brainsci8080157
Stapleton-Kotloski JR, Kotloski RJ, Popli G, Godwin DW. Magnetoencephalography: Clinical and Research Practices. Brain Sciences. 2018; 8(8):157. https://doi.org/10.3390/brainsci8080157
Chicago/Turabian StyleStapleton-Kotloski, Jennifer R., Robert J. Kotloski, Gautam Popli, and Dwayne W. Godwin. 2018. "Magnetoencephalography: Clinical and Research Practices" Brain Sciences 8, no. 8: 157. https://doi.org/10.3390/brainsci8080157




