Neural Responses to Consciously and Unconsciously Perceived Emotional Faces: A Spinal fMRI Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Stimulus Materials
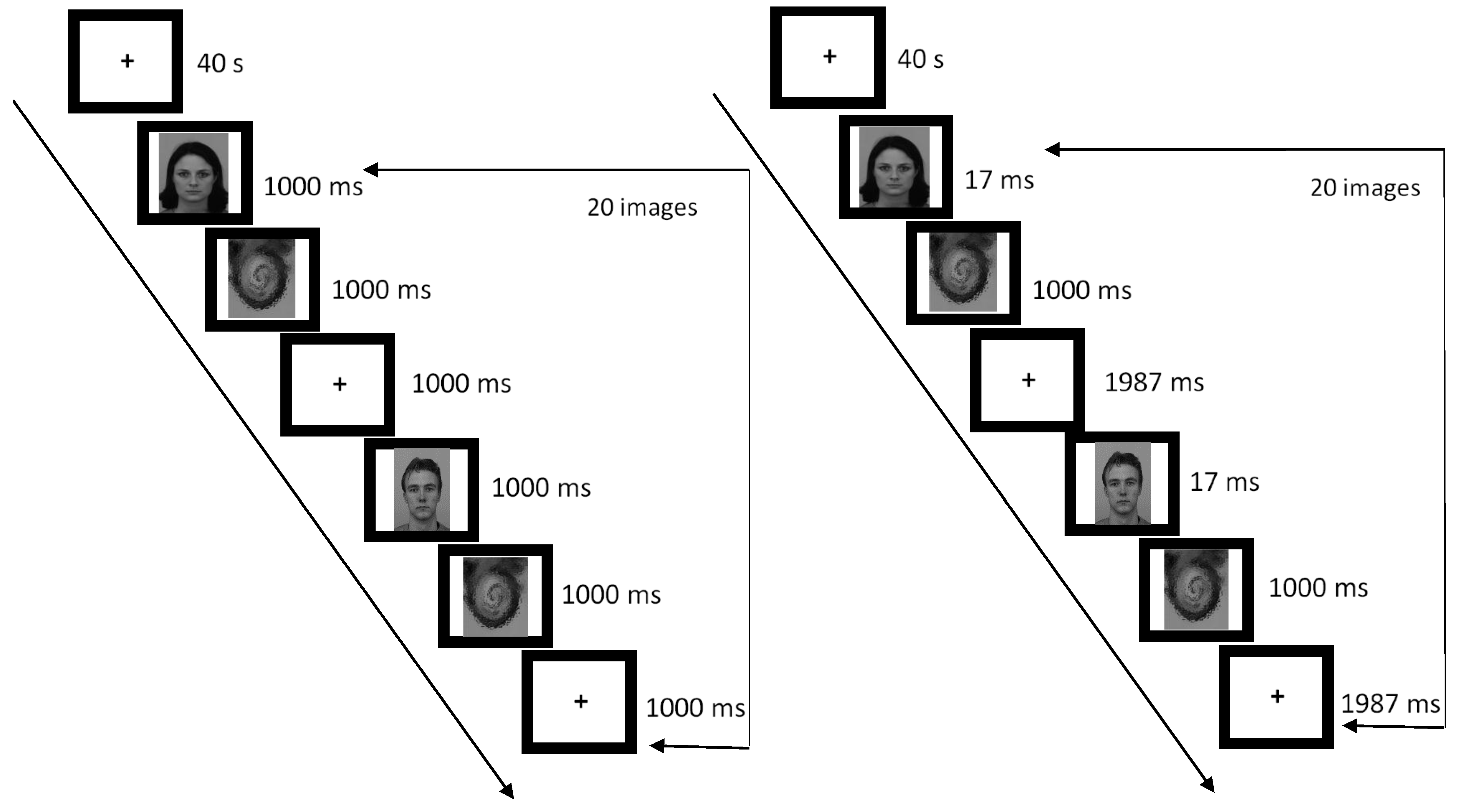
2.3. Study Design and Procedure
2.4. Data Acquisition and Scanning Parameters
3. Results
3.1. Conscious vs. Unconscious Perception
3.2. Perceiving Different Emotions: Conscious Perception
3.3. Perceiving Different Emotions: Unconscious Perception
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davidson, R.J.; Irwin, W. The functional neuroanatomy of emotion and affective style. Trends Cogn. Sci. 1999, 3, 11–21. [Google Scholar] [CrossRef]
- De Gelder, B. Towards the neurobiology of emotional body language. Nat. Rev. Neurosci. 2006, 7, 242–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LeDoux, J.E. Emotion circuits in the brain. Annu. Rev. Neurosci. 2000, 23, 155–184. [Google Scholar] [CrossRef] [PubMed]
- Surguladze, S.A.; Brammer, M.J.; Young, A.W.; Andrew, C.; Travis, M.J.; Williams, S.C.R.; Phillips, M.L. A preferential increase in the extrastriate response to signals of danger. NeuroImage 2003, 19, 1317–1328. [Google Scholar] [CrossRef]
- Morris, J.S.; Friston, K.J.; Buchel, C.; Frith, C.D.; Young, A.W.; Calder, A.J.; Dolan, R.J. A neuromodulatory role for the human amygdala in processing emotional facial expressions. Brain 1998, 121, 47–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koganemaru, S.; Domen, K.; Fukuyama, H.; Mima, T. Negative emotion can enhance human motor cortical plasticity. Eur. J. Neurosci. 2012, 35, 1637–1645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveri, M.; Babiloni, C.; Filippi, M.M.; Caltagirone, C.; Babiloni, F.; Cicinelli, P.; Traversa, R.; Palmieri, M.G.; Rossini, P.M. Influence of the supplementary motor area on primary motor cortex excitability during movements triggered by neutral or emotionally unpleasant visual cues. Exp. Brain Res. 2003, 149, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.G.; De Oliveira, L.; Erthal, F.S.; Joffily, M.; Mocaiber, I.F.; Volchan, E.; Pessoa, L. Emotion affects action: Midcingulate cortex as a pivotal node of interaction between negative emotion and motor signals. Cogn. Affect. Behav. Neurosci. 2010, 10, 94–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schienle, A.; Schafer, A.; Walter, B.; Stark, R.; Vaitl, D. Brain activation of spider phobics towards disorder-relevant, generally disgust- and fear-inducing pictures. Neurosci. Lett. 2005, 388, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.M.; Lipp, O.V.; Marinovic, W.; Wallis, G.; Riek, S. Increased corticospinal excitability induced by unpleasant visual stimuli. Neurosci. Lett. 2010, 48, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Coombes, S.A.; Tandonnet, C.; Fujiyama, H.; Janelle, C.M.; Cauraugh, J.H.; Summers, J.J. Emotion and motor preparation: A transcranial magnetic stimulation study of corticospinal motor tract excitability. Cogn. Affect. Behav. Neurosci. 2009, 9, 380–388. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajcak, G.; Molnar, C.; George, M.S.; Bolger, K.; Koola, J.; Nahas, Z. Emotion facilitates action: A transcranial magnetic stimulation study of motor cortex excitability during picture viewing. Psychophysiology 2007, 44, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Schutter, D.; Hofman, D.; Van Honk, J. Fearful faces selectively increase corticospinal motor tract excitability: A transcranial magnetic stimulation study. Psychophysiology 2008, 45, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, A.M.; Van den Wildenberg, W.P.; Van Stegeren, A.H.; Hajcak, G.; Ridderinkhof, K.R. Emotional stimuli modulate readiness for action: A transcranial magnetic stimulation study. Cogn. Affect. Behav. Neurosci. 2010, 10, 174–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McIver, T.A.; Kornelsen, J.; Smith, S.D. Limb-specific emotional modulation of cervical spinal cord neurons. Cogn. Affect. Behav. Neurosci. 2013, 13, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.D.; Kornelsen, J. Emotion-dependent responses in spinal cord neurons: A spinal fMRI study. NeuroImage 2011, 58, 269–274. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kornelsen, J.; Smith, S.D.; McIver, T.A. A neural correlate of visceral emotional responses: Evidence from fMRI of the thoracic spinal cord. Soc. Cogn. Affect. Neurosci. 2015, 10, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.D.; Kornelsen, J.; McIver, T.A. Generating facial expressions of disgust activates neurons in the thoracic spinal cord: An fMRI study. Soc. Neurosci. 2017, 13, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Lang, P.J.; Bradley, M.M.; Cuthbert, B.N. International Affective Picture System (IAPS): Affective Ratings of Pictures and Instruction Manual. In Technical Report A-8; University of Florida: Gainesville, FL, USA, 2008. [Google Scholar]
- Kolesar, T.A.; Kornelsen, J.; Smith, S.D. Separating neural activity associated with emotion and implied motion: An fMRI study. Emotion 2017, 17, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Frijda, N.H. Studies in Emotion and Social Interaction. The Emotions; Editions de la Maison des Sciences de l’Homme: Paris, France; Cambridge University Press: Cambridge, UK, 1986. [Google Scholar]
- Lang, P.J.; Bradley, M.M. Emotion and the motivational brain. Biol. Psychol. 2010, 84, 437–450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, Y.; Liu, B.; Xu, J.; Zhang, G.; Li, X.; Wang, P.; Wang, B. Decoding facial expressions based on face-selective and motion-sensitive areas. Hum. Brain Mapp. 2017, 38, 3113–3125. [Google Scholar] [CrossRef] [PubMed]
- Balconi, M.; Bortolotti, A. Conscious and unconscious face recognition is improved by high-frequency rTMS on pre-motor cortex. Conscious. Cogn. 2013, 22, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Pichon, S.; de Gelder, B.; Grèzes, J. Two different faces of threat. Comparing the neural systems for recognizing fear and anger in dynamic body expressions. NeuroImage 2009, 47, 1873–1883. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, J.S.; Frith, C.D.; Perrett, D.I.; Rowland, D.; Young, A.W.; Calder, A.J.; Dolan, R.J. A differential neural response in the human amygdala to fearful and happy facial expressions. Nature 1996, 383, 812–815. [Google Scholar] [CrossRef] [PubMed]
- Vuilleumier, P.; Armony, J.L.; Driver, J.; Dolan, R.J. Distinct spatial frequency sensitivities for processing faces and emotional expressions. Nat. Neurosci. 2006, 6, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Costafreda, S.G.; Brammer, M.J.; David, A.S.; Fu, C.H. Predictors of amygdala activation during the processing of emotional stimuli: A meta-analysis of 385 PET and fMRI studies. Brain Res. Rev. 2008, 58, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Coll, S.Y.; Ceravolo, L.; Frühholz, S.; Grandjean, D. The behavioral and neural binding phenomena during visuomotor integration of angry facial expressions. Sci. Rep.-UK 2018, 8, 6887. [Google Scholar] [CrossRef] [PubMed]
- Fusar-Poli, P.; Placentino, A.; Carletti, F.; Landi, P.; Allen, P.; Politi, P. Functional atlas of emotional faces processing: A voxel-based meta-analysis of 105 functional magnetic resonance imaging studies. J. Psychiatry. Neurosci. 2009, 34, 418–432. [Google Scholar] [PubMed]
- Gläscher, J.; Adolphs, R. Processing of the arousal of subliminal and supraliminal emotional stimuli by the human amygdala. J. Neurosci. 2003, 23, 10274–10282. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.S.; Öhman, A.; Dolan, R.J. Conscious and unconscious emotional learning in the human amygdala. Nature 1998, 393, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Bulut, N.S.; Würz, A.; Küpeli, N.Y.; Bulut, G.C.; Sungur, M.Z. Heart rate variability response to affective pictures processed in and outside of conscious awareness: Three consecutive studies of emotional regulation. Int. J. Psychophysiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Shields, R.W., Jr. Functional anatomy of the autonomic nervous system. J. Clin. Neurophysiol. 1993, 10, 2–13. [Google Scholar] [CrossRef] [PubMed]
- Esteves, F.; Öhman, A. Masking the face: Recognition of emotional facial expressions as a function of the parameters of backward masking. Scand. J. Psychol. 1993, 34, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lundqvist, D.; Flykt, A.; Öhman, A. The Karolinska Directed Emotional Faces—KDEF [CD-ROM]; Department of Clinical Neuroscience, Psychology Section, Karolinska Institutet: Stockholm, Sweden, 1998; ISBN 91-630-7164-9. [Google Scholar]
- Stroman, P.W. Magnetic resonance imaging of neuronal function in the spinal cord: Spinal FMRI. Clin. Med. Res. 2005, 3, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Stroman, P.W.; Bosma, R.L.; Tsyben, A. Somatotopic arrangement of thermal sensory regions in healthy human spinal cord determined by means of spinal cord functional MRI. Magn. Reson. Med. 2011, 68, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.B., Jr.; Kleck, R.E. Effects of direct and averted gaze on the perception of facially communicated emotion. Emotion 2005, 5, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Cristinzio, C.; N’Diaye, K.; Seeck, M.; Vuilleumier, P.; Sander, D. Integration of gaze direction and facial expression in patients with unilateral amygdala damage. Brain 2010, 133, 248–261. [Google Scholar] [CrossRef] [PubMed]
- Hess, U.; Adams, R.; Kleck, R. Looking at you or looking elsewhere: The influence of head orientation on the signal value of emotional facial expressions. Motiv. Emot. 2007, 31, 137–144. [Google Scholar] [CrossRef]
- Hortensius, R.; De Gelder, B.; Schutter, D.J.L.G. When anger dominates the mind: Increased motor corticospinal excitability in the face of threat. Psychophysiology 2016, 53, 1307–1316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huis in ’t Veld, E.M.; Van Boxtel, G.J.; De Gelder, B. The Body Action Coding System I: Muscle activations during the perception and expression of emotion. Soc. Neurosci. 2014, 9, 249–264. [Google Scholar] [CrossRef] [PubMed]
- Drake, R.L.; Vogl, W.; Mitchell, A.W.M. Gray’s Anatomy for Students; Elsevier: New York, NY, USA, 2005. [Google Scholar]
- Hariri, A.R.; Tessitore, A.; Mattay, V.S.; Fera, F.; Weinberger, D.R. The amygdala response to emotional stimuli: A comparison of faces and scenes. Neuroimage 2002, 17, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Alpers, G.W.; Adolph, D.; Pauli, P. Emotional scenes and facial expressions elicit different psychophysiological responses. Int. J. Psychophysiol. 2011, 80, 173–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tettamanti, M.; Rognoni, E.; Cafiero, R.; Costa, T.; Galati, D.; Perani, D. Distinct pathways of neural coupling for different basic emotions. Neuroimage 2012, 59, 1804–1817. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, A.; Edlow, B.L.; Immordino-Yang, M.H. The brainstem in emotion: A review. Front. Neuroanat. 2017, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Lohse, M.; Overgaard, M. Emotional priming depends on the degree of conscious experience. Neuropsychologia 2017. [Google Scholar] [CrossRef] [PubMed]
- Carr, T.H.; Dagenback, D. Semantic priming and repetition priming from masked words: Evidence for a center-surround attentional mechanism in perceptual recognition. J. Exp. Psychol. Learn. 1990, 16, 341–350. [Google Scholar] [CrossRef]
- Smith, S.D.; Bulman-Fleming, M.B. A hemispheric asymmetry for the unconscious perception of emotion. Brain Cogn. 2004, 55, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Filkowski, M.M.; Haas, B.W. Rethinking the use of neutral faces as a baseline in fMRI neuroimaging studies of Axis-I psychiatric disorders. J. Neuroimaging 2017, 27, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.L.; Zinbarg, R.E. Interpreting neutral faces as threatening is a default mode for socially anxious individuals. J. Abnorm. Psychol. 2008, 117, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.L.; Cottrell, G.W.; Gosselin, F.; Schyns, P.G. Transmitting and decoding facial expressions. Psychol. Sci. 2005, 1, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Curby, K.M.; Johnson, K.J.; Tyson, A. Face to face with emotion: Holistic face processing is modulated by emotional state. Cogn. Emot. 2012, 26, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Dimburg, U.; Thunberg, M.; Elmehad, K. Unconscious facial reactions to emotional facial expressions. Psychol. Sci. 2000, 11, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Barrett, L.F. The theory of constructed emotion: An active inference account of interoception and categorization. Soc. Cogn. Affect. Neurosci. 2017, 12, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Kret, M.E.; Pichon, S.; Grèzes, J.; De Gelder, B. Similarities and differences in perceiving threat from dynamic faces and bodies. An fMRI study. NeuroImage 2011, 54, 1755–1762. [Google Scholar] [CrossRef] [PubMed] [Green Version]

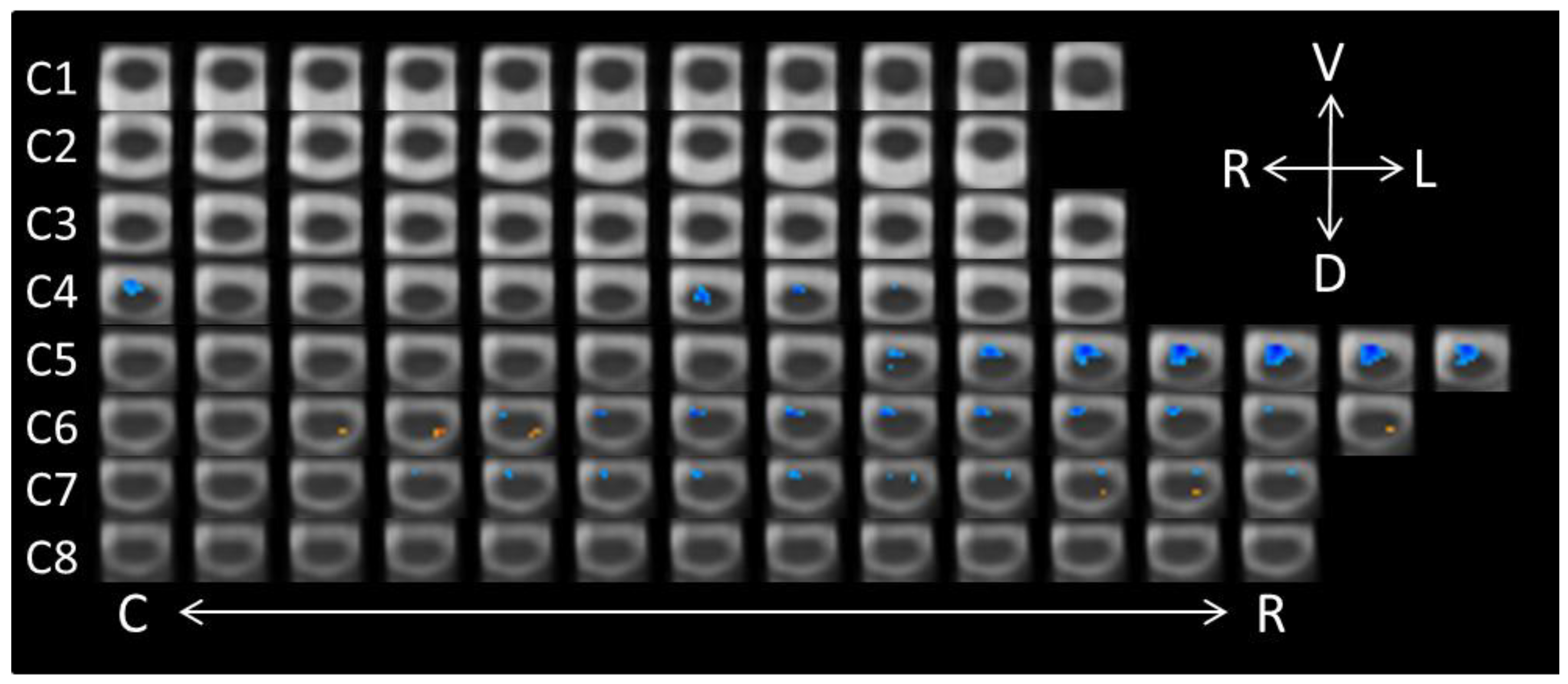

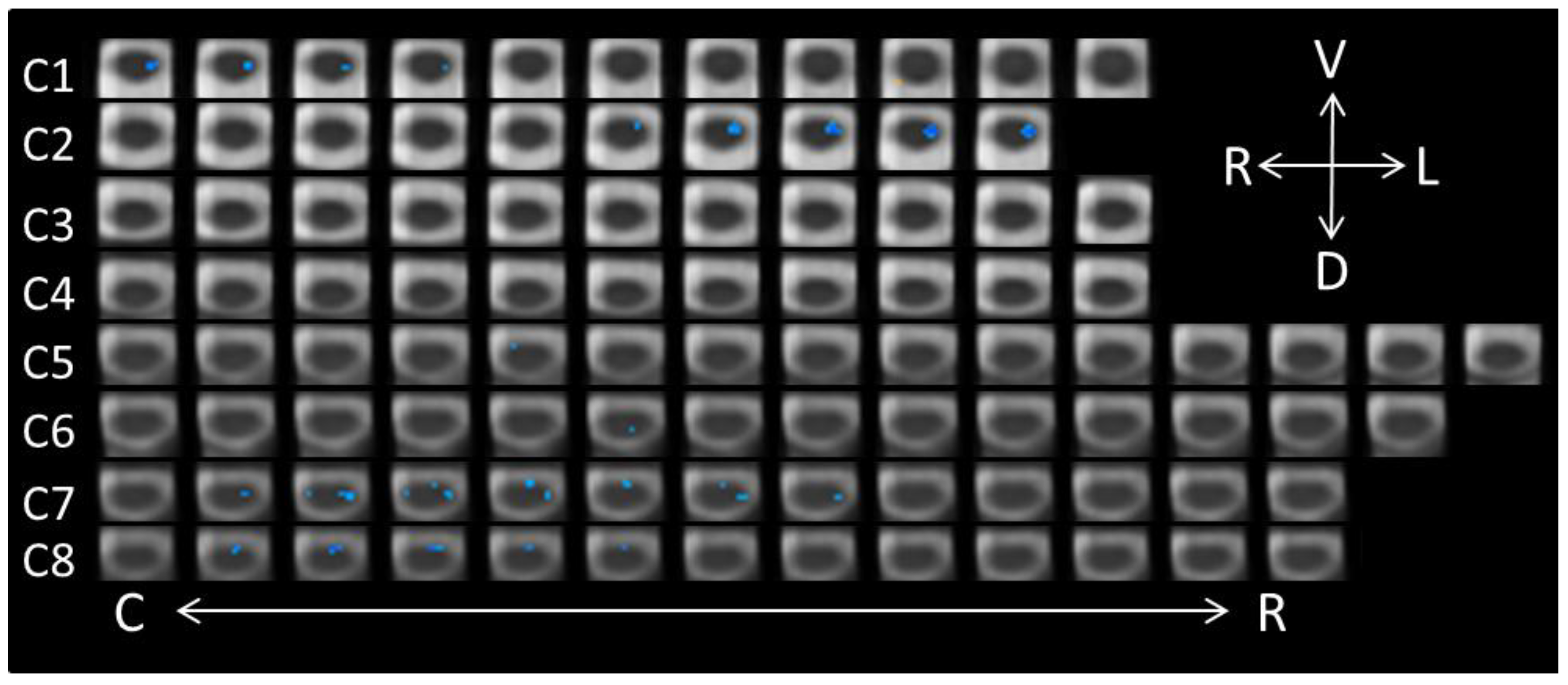
| Spinal Cord Segment | Side | Dorsal/Ventral | Voxels |
|---|---|---|---|
| Neutral Conscious > Neutral Unconscious | |||
| C1 | Right | Dorsal | 12 |
| Neutral Unconscious > Neutral Conscious | |||
| C1 | Right | Medial | 14 |
| C2 | Right | Dorsal | 3 |
| Right | Ventral | 4 | |
| C4/C5 | Left | Ventromedial | 8 |
| C5/C6 | Right/Medial | Ventral | 101 |
| C7 | Left | Ventral | 3 |
| C8 | Right | Dorsal | 4 |
| Medial/Right | Ventral | 10 | |
| Anger Conscious > Anger Unconscious | |||
| C3 | Left | Ventral | 7 |
| C5 | Left | Medial | 5 |
| C6 | Left | Ventral | 27 |
| C7 | Medial/Right | Ventral | 15 |
| Anger Unconscious > Anger Conscious | |||
| No active voxels | |||
| Fear Conscious > Fear Unconscious | |||
| C1/C2 | Left | Medial | 3 |
| C6 | Left | Dorsal | 5 |
| Fear Unconscious > Fear Conscious | |||
| C1 | Medial | Ventral | 3 |
| C4 | Right | Ventral | 3 |
| C6 | Right | Ventral | 3 |
| Spinal Cord Segment | Side | Dorsal/Ventral | Voxels |
|---|---|---|---|
| Anger > Neutral | |||
| C1 | Right | Medial | 9 |
| C2 | Right | Dorsomedial | 15 |
| C4 | Left/Medial | Ventral | 5 |
| C4/C5 | Left | Medial | 19 |
| C5/C6 | Right/Medial/Left | Ventromedial | 46 |
| C7 | Left/Medial/Right | Ventral | 32 |
| C8 | Medial/Right | Ventral | 29 |
| Neutral > Anger | |||
| No active voxels | |||
| Fear > Neutral | |||
| No active voxels | |||
| Neutral > Fear | |||
| C4 | Right | Ventromedial | 7 |
| C5 | Right | Dorsomedial | 27 |
| C6/C7 | Left | Ventral | 5 |
| Anger > Fear | |||
| C4 | Medial | Ventromedial | 18 |
| C5/C4 | Right | Ventral | 161 |
| C6 | Right | Ventral | 46 |
| C7 | Right | Ventral | 18 |
| Left | Ventral | 10 | |
| Fear > Anger | |||
| C6 | Left | Dorsal | 9 |
| C7 | Left | Dorsal | 3 |
| Spinal Cord Segment | Side | Dorsal/Ventral | Voxels |
|---|---|---|---|
| Anger > Neutral | |||
| C1 | Right | Dorsal | 19 |
| Medial | Dorsal | 3 | |
| Neutral > Anger | |||
| C3 | Left | Ventral | 4 |
| C5/C6 | Medial | Ventral | 31 |
| C6 | Left | Dorsomedial | 6 |
| C7/C8 | Left/Medial/Right | Ventral | 39 |
| Fear > Neutral | |||
| No active voxels | |||
| Neutral > Fear | |||
| C1/C2 | Left | Ventromedial | 45 |
| C7 | Left | Medial | 5 |
| C7 | Medial | Ventral | 9 |
| Left | Medial | 13 | |
| C8 | Medial/Left | Ventral | 14 |
| Anger > Fear | |||
| C3/C4 | Right | Medial | 4 |
| Fear > Anger | |||
| No active voxels | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilson, A.D.; Kolesar, T.A.; Kornelsen, J.; Smith, S.D. Neural Responses to Consciously and Unconsciously Perceived Emotional Faces: A Spinal fMRI Study. Brain Sci. 2018, 8, 156. https://doi.org/10.3390/brainsci8080156
Wilson AD, Kolesar TA, Kornelsen J, Smith SD. Neural Responses to Consciously and Unconsciously Perceived Emotional Faces: A Spinal fMRI Study. Brain Sciences. 2018; 8(8):156. https://doi.org/10.3390/brainsci8080156
Chicago/Turabian StyleWilson, Alyssia D., Tiffany A. Kolesar, Jennifer Kornelsen, and Stephen D. Smith. 2018. "Neural Responses to Consciously and Unconsciously Perceived Emotional Faces: A Spinal fMRI Study" Brain Sciences 8, no. 8: 156. https://doi.org/10.3390/brainsci8080156




