Spatial Working Memory Deficits in Male Rats Following Neonatal Hypoxic Ischemic Brain Injury Can Be Attenuated by Task Modifications
Abstract
:1. Introduction
2. Experimental Section
2.1. Subjects (Studies 1 and 2)
2.2. Surgical Procedure (Studies 1 and 2)
2.3. Behavioral Testing (Study 1)
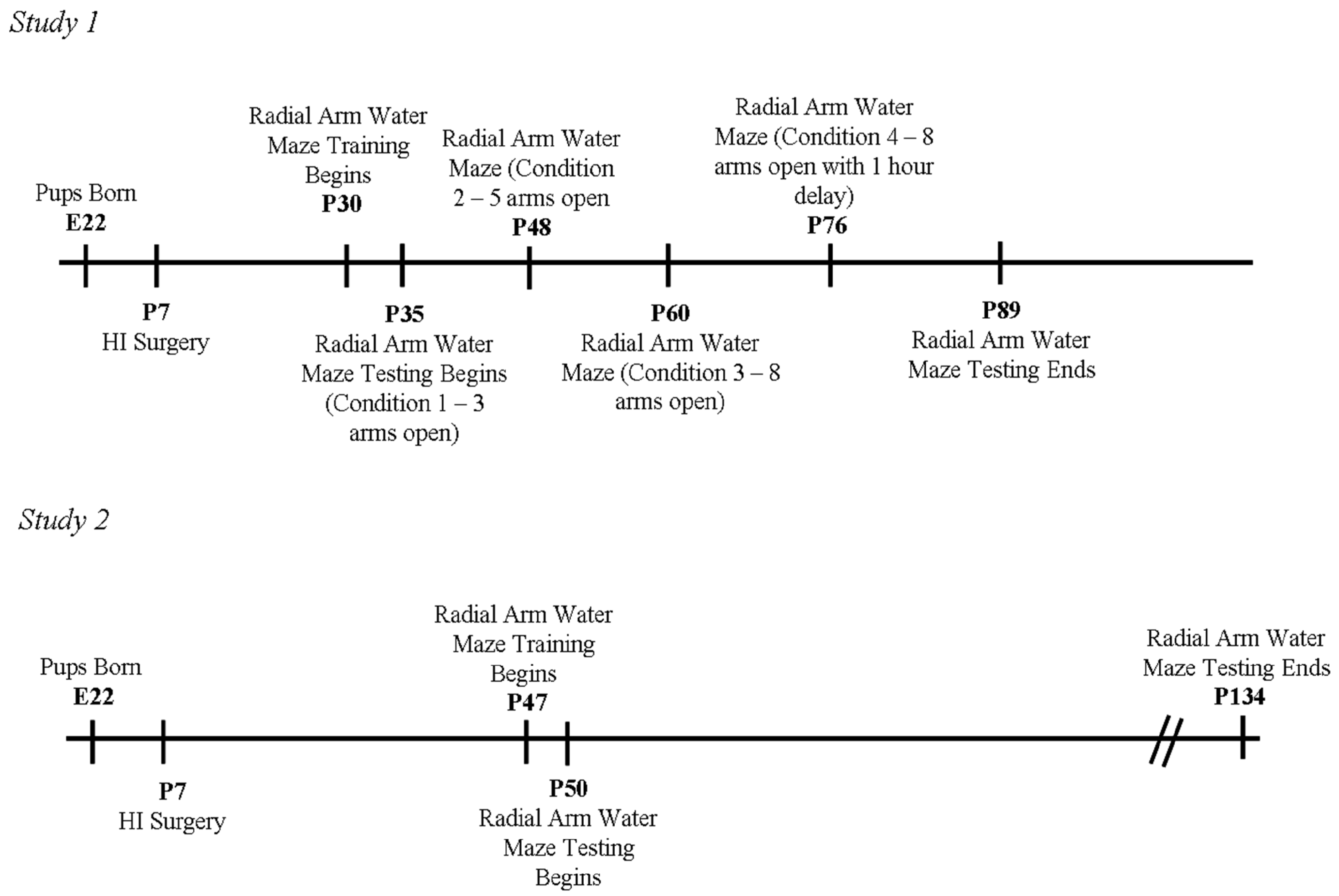
2.3.1. Weeks 1 and 2 (Condition 1—3 Arms Open)
2.3.2. Weeks 3 and 4 (Condition 2—5 Arms Open)
2.3.3. Weeks 5 and 6 (Condition 3—8 Arms Open)
2.3.4. Weeks 7 and 8 (Condition 4—8 Arms Open with 1 h Delay)
2.4. Behavioral Testing (Study 2)
2.5. Histology (Studies 1 and 2)
2.6. Statistical Analyses (Studies 1 and 2)
2.6.1. Study 1 and 2, Anatomical Analyses
2.6.2. Study 1, Error and Latency Analyses
2.6.3. Study 2, Error and Latency Analyses
2.6.4. Studies 1 and 2, Cross-Study Error Analysis—Weeks 7 and 8 Only
3. Results and Discussion
3.1. Anatomic Results (Studies 1 and 2)
3.1.1. Study 1 Anatomy
3.1.2. Study 2 Anatomy
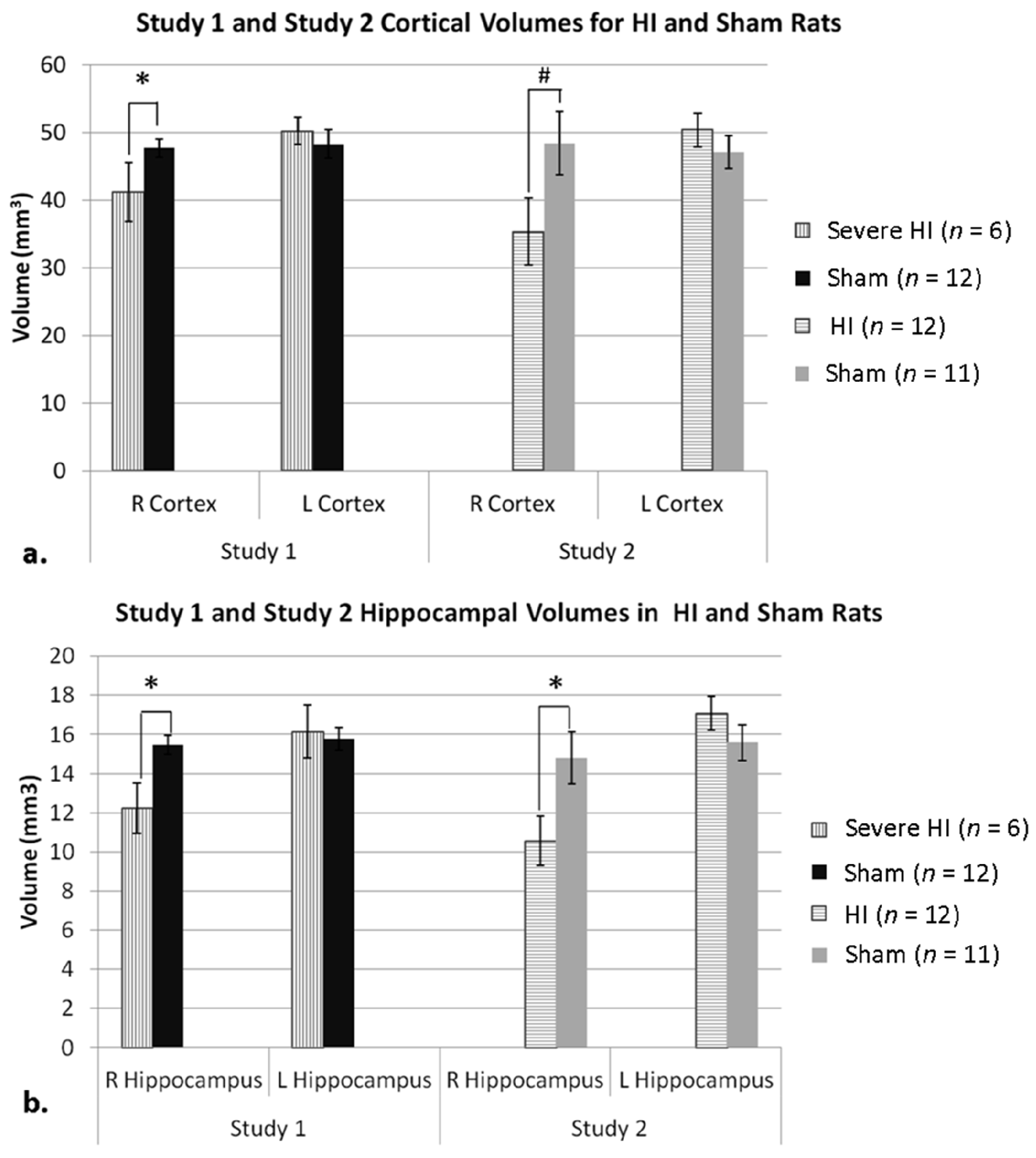
3.1.3. Study 1 Anatomy Re-Analysis—Sub-Grouping by Cortical Atrophy Measures
3.2. Behavioral Results (Study 1 and 2)
3.2.1. Study 1 and 2, Analysis of Control Trial Errors
3.2.2. Study 1, Analysis of Overall Errors
3.2.3. Study 1, Analysis of Errors within Condition
3.2.4. Study 1, Analysis of Errors for Each Week, by Treatment
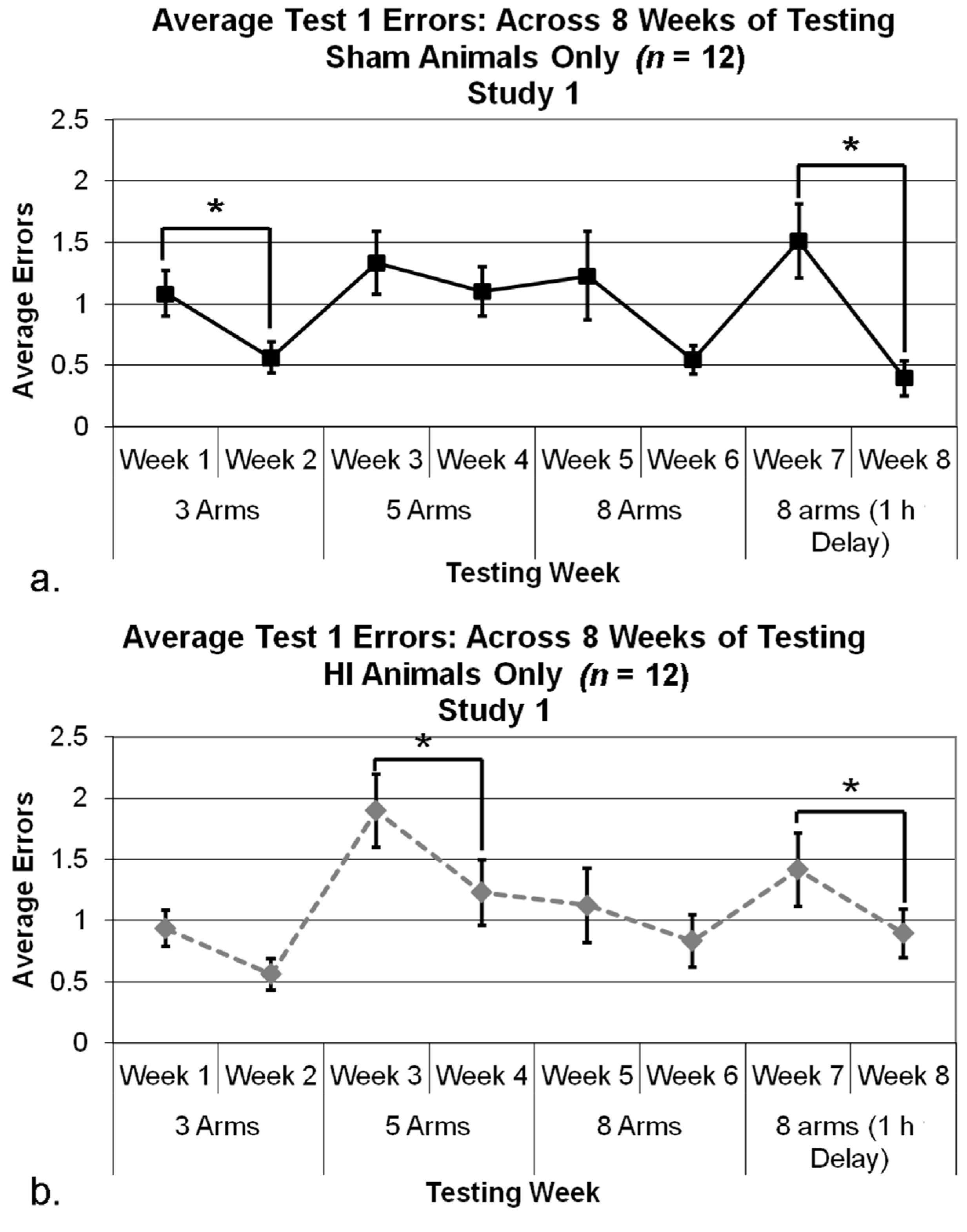
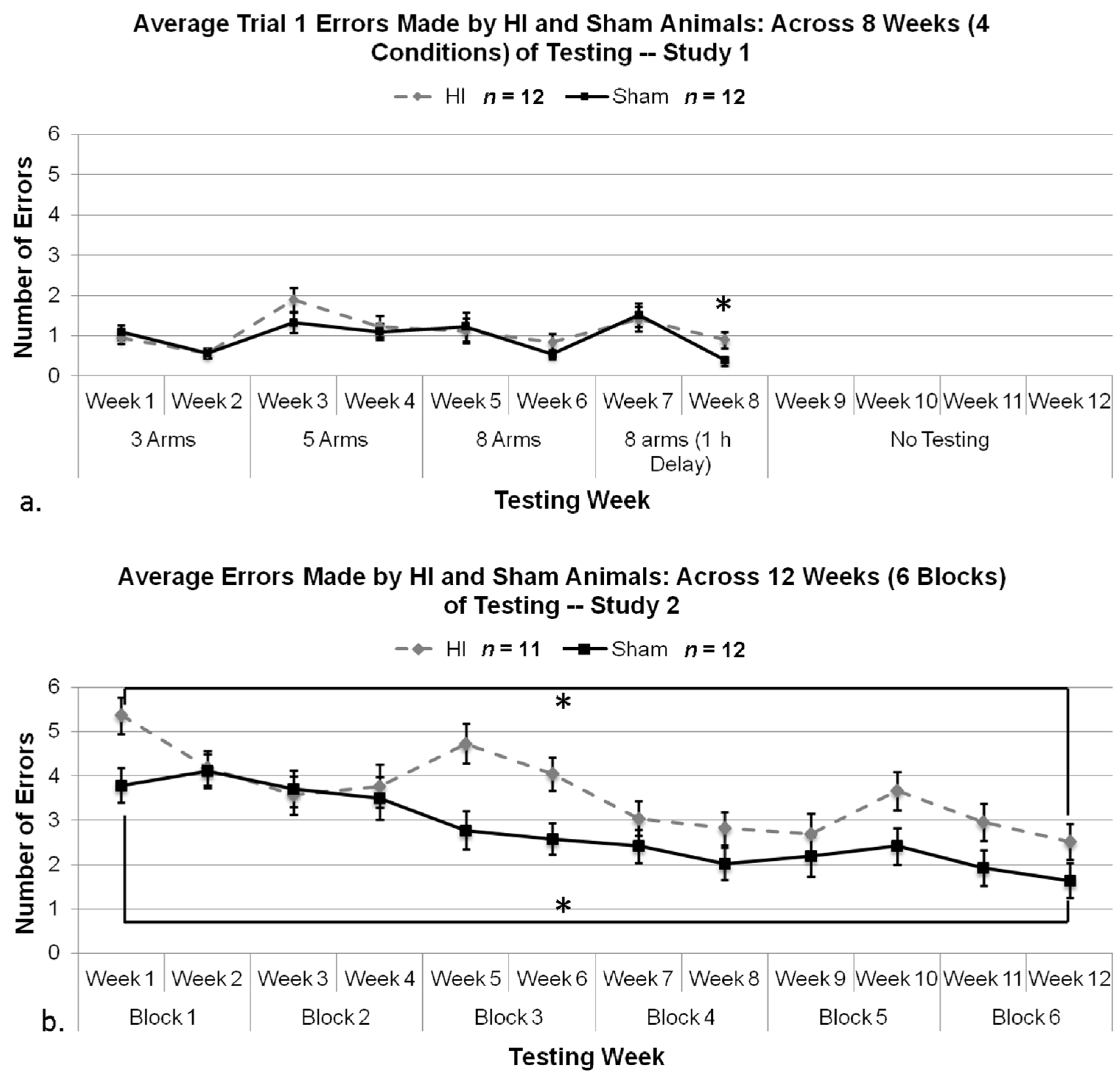
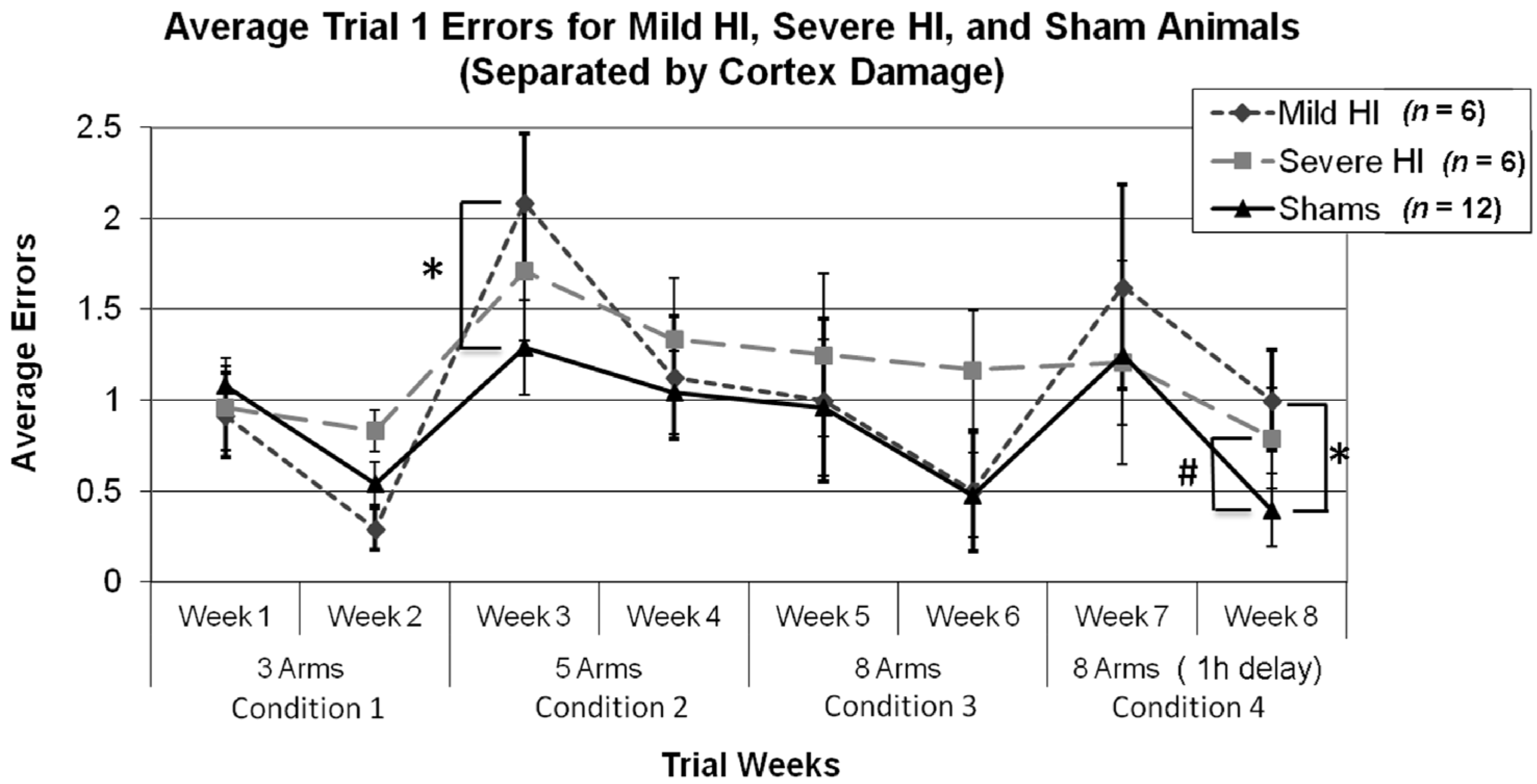
3.2.5. Study 1, Re-Analysis of Errors for Each Week, by Treatment (Severe and Mild HI Subsets)
3.2.6. Study 2, Analysis of Errors Overall
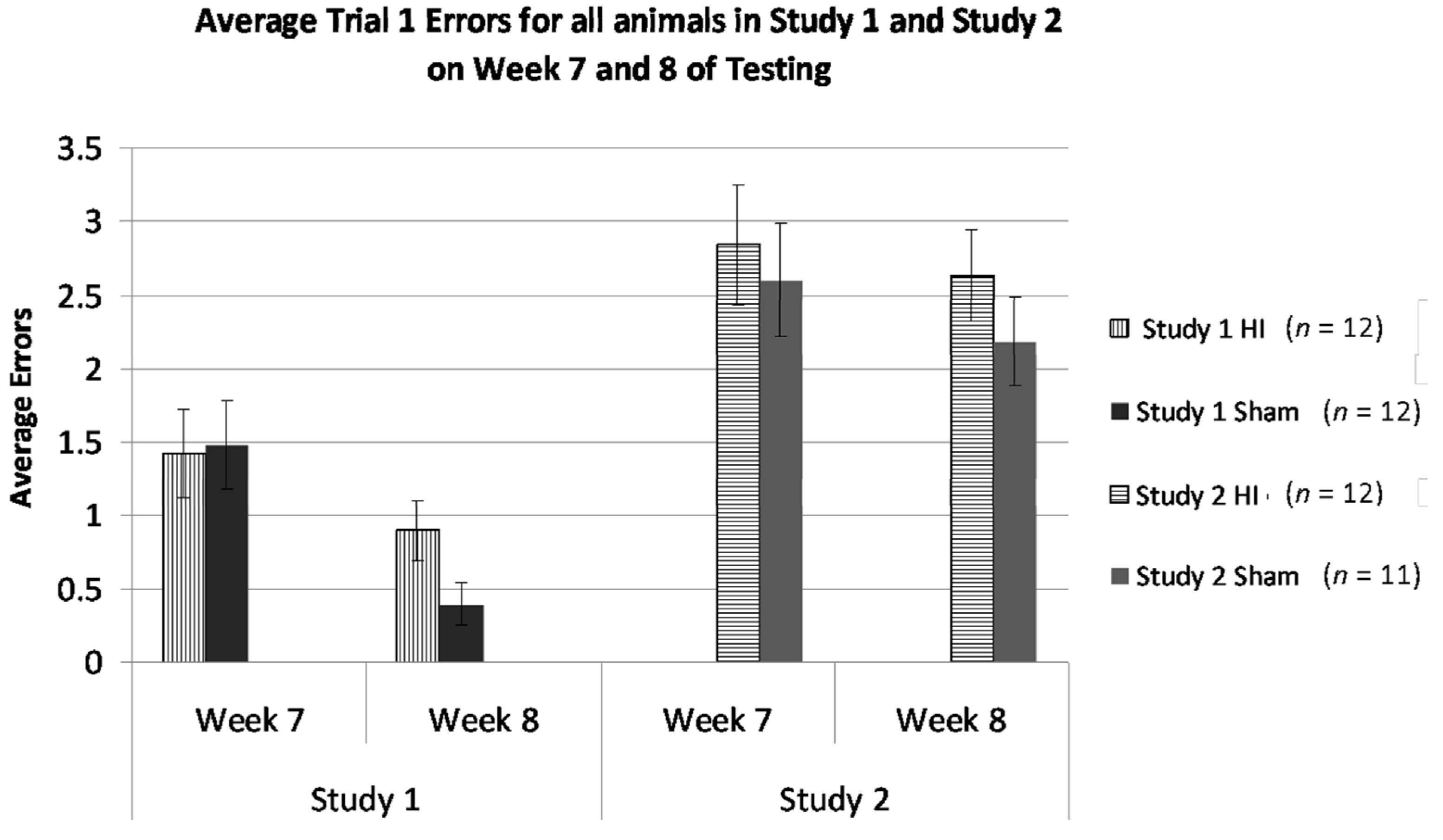
3.2.7. Study 1 and 2, Analysis of Errors in Weeks 7 and 8 by Treatment and Study
3.2.8. Study 1, Analysis of Mean Latency per Choice, by Treatment

3.2.9. Study 2, Analysis of Mean Latency per Choice, by Treatment
3.3. Discussion
3.3.1. HI Rats Still Show Learning, Even When the Task is Difficult
3.3.2. HI Deficits are More Robust on High Memory Demand Tasks
3.3.3. HI Rats Showed Reduced Latencies Compared to Shams Despite More Errors Made, and This Impulsivity was Strongly Related to Task Difficulty
3.3.4. HI Rats Learn Much Better When Introduced to a High Memory-Demand Task through Gradual Training, as Opposed to Immediate Introduction
3.3.5. Underlying Neuropathology for Memory Deficits and Impulsivity
4. Conclusions
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Fatemi, A.; Wilson, M.A.; Johnston, M.V. Hypoxic-ischemic encephalopathy in the term infant. Clin. Perinatol. 2009, 36, 835–858. [Google Scholar] [CrossRef]
- Vannucci, S.J.; Hagberg, H. Hypoxia-ischemia in the immature brain. J. Exp. Biol. 2004, 207, 3149–3154. [Google Scholar] [CrossRef]
- McLean, C.; Ferriero, D. Mechanisms of hypoxic-ischemic injury in the term infant. Semin. Perinatol. 2004, 28, 425–432. [Google Scholar] [CrossRef]
- Qureshi, A.M.; ur Rehman, A.; Siddiqi, T.S. Hypoxic ischemic encephalopathy in neonates. J. Ayub Med. Coll. Abbottabad 2010, 22, 190–193. [Google Scholar]
- De Vries, L.S.; Cowan, F.M. Evolving understanding of hypoxic-ischemic encephalopathy in the term infant. Semin. Pediatr. Neurol. 2009, 16, 216–225. [Google Scholar] [CrossRef]
- Volpe, J.J. Perinatal brain injury: From pathogenesis to neuroprotection. Ment. Retard. Dev. Disabil. Res. Rev. 2001, 7, 56–64. [Google Scholar] [CrossRef]
- Takeoka, M.; Soman, T.B.; Yoshii, A.; Caviness, V.S., Jr.; Gonzalez, R.G.; Grant, P.E.; Krishnamoorthy, K.S. Diffusion-weighted images in neonatal cerebral hypoxic-ischemic injury. Pediatr. Neurol. 2002, 26, 274–281. [Google Scholar] [CrossRef]
- Barrett, R.D.; Bennet, L.; Davidson, J.; Dean, J.M.; George, S.; Emerald, B.S.; Gunn, A.J. Destruction and reconstruction: Hypoxia and the developing brain. Birth Defects Res. C Embryo Today 2007, 81, 163–176. [Google Scholar] [CrossRef]
- Volpe, J.J. Brain injury in premature infants: A complex amalgam of destructive and developmental disturbances. Lancet Neurol. 2009, 8, 110–124. [Google Scholar] [CrossRef]
- Du Plessis, A.J.; Volpe, J.J. Perinatal brain injury in the preterm and term newborn. Curr. Opin. Neurol. 2002, 15, 151–157. [Google Scholar] [CrossRef]
- Grant, P.E.; Yu, D. Acute injury to the immature brain with hypoxia with or without hypoperfusion. Magn. Reson. Imaging Clin. N. Am. 2006, 14, 271–285. [Google Scholar] [CrossRef]
- Yakovlev, A.G.; Faden, A.I. Mechanisms of neural death: Implications for development of neuroprotective treatment strategies. NeuroRx 2004, 1, 5–16. [Google Scholar] [CrossRef]
- Kitanaka, C.; Kuchino, Y. Caspase-independent programmed cell death with necrotic morphology. Cell Death Differ. 1999, 6, 508–515. [Google Scholar]
- Back, S.A.; Riddle, A.; Dean, J.; Hohimer, A.R. The instrumented fetal sheep as a model of cerebral white matter injury in the premature infant. Neurotherapeutics 2012, 9, 359–370. [Google Scholar] [CrossRef]
- Perlman, J.M. White matter injury in the preterm infant: An important determination of abnormal neurodevelopment outcome. Early Hum. Dev. 1998, 53, 99–120. [Google Scholar] [CrossRef]
- Inder, T.E.; Anderson, N.J.; Spencer, C.; Wells, S.; Volpe, J.J. White matter injury in the premature infant: A comparison between serial cranial sonographic and MRI findings at term. AJNR Am. J. Neuroradiol. 2003, 24, 805–809. [Google Scholar]
- Alvarez-Diaz, A.; Hilario, E.; de Cerio, F.G.; Valls-i-Soler, A.; Alvarez-Diaz, F.J. Hypoxic-ischemic injury in the immature brain—Key vascular and cellular players. Neonatology 2007, 92, 227–235. [Google Scholar] [CrossRef]
- Vannucci, R.C. Hypoxic-ischemic encephalopathy. Am. J. Perinatol. 2000, 17, 113–120. [Google Scholar] [CrossRef]
- Hayakawa, M.; Ito, Y.; Saito, S.; Mitsuda, N.; Hosono, S.; Yoda, H.; Cho, K.; Otsuki, K.; Ibara, S.; Terui, K.; et al.; Executive committee, Symposium on Japan Society of Perinatal and Neonatal Medicine Incidence and prediction of outcome in hypoxic-ischemic encephalopathy in japan. Pediatr. Int. 2013. [Google Scholar] [CrossRef]
- Vannucci, R.C.; Vannucci, S.J. Perinatal hypoxic-ischemic brain damage: Evolution of an animal model. Dev. Neurosci. 2005, 27, 81–86. [Google Scholar] [CrossRef]
- Huang, B.Y.; Castillo, M. Hypoxic-ischemic brain injury: Imaging findings from birth to adulthood. Radiographics 2008, 28, 417–439. [Google Scholar] [CrossRef]
- Nosarti, C.; Giouroukou, E.; Healy, E.; Rifkin, L.; Walshe, M.; Reichenberg, A.; Chitnis, X.; Williams, S.C.; Murray, R.M. Grey and white matter disruption in very preterm adolescents mediates neurodevelopmental outcome. Brain 2008, 131, 205–217. [Google Scholar]
- Izbudak, I.; Grant, P.E. MR imaging of the term and preterm neonate with diffuse brain injury. Magn. Reson. Imaging Clin. N. Am. 2011, 19, 709–731. [Google Scholar] [CrossRef]
- Jyoti, R.; OʼNeil, R.; Hurrion, E. Predicting outcome in term neonates with hypoxic-ischaemic encephalopathy using simplified MR criteria. Pediatr. Radiol. 2006, 36, 38–42. [Google Scholar] [CrossRef]
- Martinez-Biarge, M.; Diez-Sebastian, J.; Kapellou, O.; Gindner, D.; Allsop, J.M.; Rutherford, M.A.; Cowan, F.M. Predicting motor outcome and death in term hypoxic-ischemic encephalopathy. Neurology 2011, 76, 2055–2061. [Google Scholar] [CrossRef]
- Martinez-Biarge, M.; Bregant, T.; Wusthoff, C.J.; Chew, A.T.; Diez-Sebastian, J.; Rutherford, M.A.; Cowan, F.M. White matter and cortical injury in hypoxic-ischemic encephalopathy: Antecedent factors and 2-year outcome. J. Pediatr. 2012, 161, 799–807. [Google Scholar] [CrossRef]
- Cserjesi, R.; van Braeckel, K.N.; Butcher, P.R.; Kerstjens, J.M.; Reijneveld, S.A.; Bouma, A.; Geuze, R.H.; Bos, A.F. Functioning of 7-year-old children born at 32 to 35 weeks’ gestational age. Pediatrics 2012, 130, e838–e846. [Google Scholar] [CrossRef]
- Kent, A.L.; Wright, I.M.; Abdel-Latif, M.E.; New South Wales and Australian Capital Territory Neonatal Intensive Care Units Audit Group. Mortality and adverse neurologic outcomes are greater in preterm male infants. Pediatrics 2012, 129, 124–131. [Google Scholar] [CrossRef]
- Potjik, M.R.; de Winter, A.F.; Bos, A.F.; Kerstjens, J.M.; Reijneveld, S.A. Higher rates of behavioural and emotional problems at preschool age in children born moderately preterm. Arch. Dis. Child. 2012, 97, 112–117. [Google Scholar] [CrossRef]
- Fotopoulos, S.; Pavlou, K.; Skouteli, H.; Papassotiriou, I.; Lipsou, N.; Xanthou, M. Early markers of brain damage in premature low-birth-weight neonates who suffered from perinatal asphyxia and/or infection. Biol. Neonate 2001, 79, 213–218. [Google Scholar] [CrossRef]
- Van Handel, M.; Swaab, H.; de Vries, L.S.; Jongmans, M.J. Long-term cognitive and behavioral consequences of neonatal encephalopathy following perinatal asphyxia: A review. Eur. J. Pediatr. 2007, 166, 645–654. [Google Scholar] [CrossRef]
- Casiro, O.G.; Moddemann, D.M.; Stanwick, R.S.; Panikkar-Thiessen, V.K.; Cowan, H.; Cheang, M.S. Language development of very low birth weight infants and full term controls at 12 months of age. Early Hum. Dev. 1990, 24, 65–77. [Google Scholar] [CrossRef]
- Marlow, N.; Rose, A.S.; Rands, C.E.; Draper, E.S. Neuropsychological and educational problems at school age associated with neonatal encephalopathy. Arch. Dis. Child. Fetal Neonatal Ed. 2005, 90, F380–F387. [Google Scholar] [CrossRef]
- Fitch, R.H.; Tallal, P. Neural mechanisms of language-based learning impairments: Insights from human populations and animal models. Behav. Cogn. Neurosci. Rev. 2003, 2, 155–178. [Google Scholar] [CrossRef]
- Jansson-Verkasalo, E.; Korpilahti, P.; Jantti, V.; Valkama, M.; Vainionpaa, L.; Alku, P.; Suominen, K.; Naatanen, R. Neurophysiologic correlates of deficient phonological representations and object naming in prematurely born children. Clin. Neurophysiol. 2004, 115, 179–187. [Google Scholar] [CrossRef]
- Jansson-Verkasalo, E.; Valkama, M.; Vainionpaa, L.; Paakko, E.; Ilkko, E.; Lehtihalmes, M. Language development in very low birth weight preterm children: A follow-up study. Folia Phoniatr. Logop. 2004, 56, 108–119. [Google Scholar] [CrossRef]
- Steinman, K.J.; Gorno-Tempini, M.L.; Glidden, D.V.; Kramer, J.H.; Miller, S.P.; Barkovich, A.J.; Ferriero, D.M. Neonatal watershed brain injury on magnetic resonance imaging correlates with verbal IQ at 4 years. Pediatrics 2009, 123, 1025–1030. [Google Scholar] [CrossRef]
- Gadian, D.G.; Aicardi, J.; Watkins, K.E.; Porter, D.A.; Mishkin, M.; Vargha-Khadem, F. Developmental amnesia associated with early hypoxic-ischaemic injury. Brain 2000, 123, 499–507. [Google Scholar] [CrossRef]
- Curtis, W.J.; Zhuang, J.; Townsend, E.L.; Hu, X.; Nelson, C.A. Memory in early adolescents born prematurely: A functional magnetic resonance imaging investigation. Dev. Neuropsychol. 2006, 29, 341–377. [Google Scholar] [CrossRef]
- Baron, I.S.; Brandt, J.; Ahronovich, M.D.; Baker, R.; Erickson, K.; Litman, F.R. Selective deficit in spatial location memory in extremely low birth weight children at age six: The PETIT study. Child Neuropsychol. 2012, 18, 299–311. [Google Scholar] [CrossRef]
- Sommerfelt, K.; Markestad, T.; Ellersten, B. Neuropsychological performance in low birth weight preschoolers: A population-based, controlled study. Eur. J. Pediatr. 1998, 157, 53–58. [Google Scholar] [CrossRef]
- Lou, H.C. Etiology and pathogenesis of attention-deficit hyperactivity disorder (ADHD): Significance of prematurity and perinatal hypoxic-haemodynamic encephalopathy. Acta Paediatr. 1996, 85, 1266–1271. [Google Scholar] [CrossRef]
- Lindstrom, K.; Lindblad, F.; Hjern, A. Preterm birth and attention-deficit/hyperactivity disorder in schoolchildren. Pediatrics 2011, 127, 858–865. [Google Scholar] [CrossRef]
- Sun, J.; Buys, N. Early executive function deficit in preterm children and its association with neurodevelopmental disorders in childhood: A literature review. Int. J. Adolesc. Med. Health 2012, 24, 291–299. [Google Scholar]
- De Kieviet, J.F.; van Elburg, R.M.; Lafeber, H.N.; Ooosterlaan, J. Attention problems of very preterm children compared with age-matches term controls at school-age. J. Pediatr. 2012, 161, 824–829. [Google Scholar] [CrossRef]
- Scott, M.N.; Taylor, H.G.; Fristad, M.A.; Klein, N.; Espy, K.A.; Minich, N.; Hack, M. Behavior disorders in extremely preterm/extremely low birth weight children in kindergarten. J. Dev. Behav. Pediatr. 2012, 33, 202–213. [Google Scholar] [CrossRef]
- Shum, D.; Neulinger, K.; OʼCallaghan, M.; Mohay, H. Attentional problems in children born very preterm or with extremely low birth weight at 7–9 years. Arch. Clin. Neuropsychol. 2008, 23, 103–112. [Google Scholar] [CrossRef]
- Galera, C.; Cote, S.M.; Bouvard, M.P.; Pingault, J.B.; Melchoir, M.; Michel, G.; Boivin, M.; Tremblay, R.E. Early risk factors for hyperactivity-impulsivity and inattention trajectories form age 17 months to 8 years. Arch. Gen. Psychiatry 2011, 68, 1267–1275. [Google Scholar] [CrossRef]
- Espy, K.A.; Senn, T.E.; Charak, D.A.; Tyler, J.; Wiebe, S.A. Perinatal pH and neuropsychological outcomes at age 3 years in children born preterm: An exploratory study. Dev. Neuropsychol. 2007, 32, 669–682. [Google Scholar] [CrossRef]
- Mulder, H.; Pitchford, N.J.; Marlow, N. Inattentive behaviour is associated with poor working memory and slow processing speed in very pre-term children in middle childhood. Br. J. Educ. Psychol. 2011, 81, 147–160. [Google Scholar] [CrossRef]
- Allen, M.C. Neurodevelopmental outcomes of preterm infants. Curr. Opin. Neurol. 2008, 21, 123–128. [Google Scholar] [CrossRef]
- Marlow, N.; Hennessy, E.M.; Bracewell, M.A.; Wolke, D.; EPICure Study Group. Motor and executive function at 6 years of age after extremely preterm birth. Pediatrics 2007, 120, 793–804. [Google Scholar] [CrossRef]
- Espy, K.A.; Stalets, M.M.; McDiarmid, M.M.; Senn, T.E.; Cwik, M.F.; Hamby, A. Executive functions in preschool children born preterm: Application of cognitive neuroscience paradigms. Child Neuropsychol. 2002, 8, 83–92. [Google Scholar]
- Aarnoudse-Moens, C.S.; Weisglas-Kuperus, N.; van Goudoever, J.B.; Oosterlaan, J. Meta-analysis of neurobehavioral outcomes in very preterm and/or very low birth weight children. Pediatrics 2009, 124, 717–728. [Google Scholar] [CrossRef]
- Isaacs, E.B.; Edmonds, C.J.; Chong, W.K.; Lucas, A.; Morley, R.; Gadian, D.G. Brain morphometry and IQ measurements in preterm children. Brain 2004, 127, 2595–2607. [Google Scholar] [CrossRef]
- Luu, T.M.; Ment, L.; Allan, W.; Schneider, K.; Vohr, B.R. Executive and memory function in adolescents born very preterm. Pediatrics 2011, 127, e639–e646. [Google Scholar] [CrossRef]
- Potharst, E.S.; van Wassenaer-Leemhuis, A.G.; Houtzager, B.A.; Livesey, D.; Kok, J.H.; Last, B.F.; Oosterlaan, J. Perinatal risk factors for neurocognitive impairments in preschool children born very preterm. Dev. Med. Child Neurol. 2013, 55, 178–184. [Google Scholar] [CrossRef]
- Bhutta, A.T.; Cleves, M.A.; Casey, P.H.; Cradock, M.M.; Anand, K.J. Cognitive and behavioral outcomes of school-aged children who were born preterm: A meta-analysis. J. Am. Med. Assoc. 2002, 288, 728–737. [Google Scholar] [CrossRef]
- Botting, N.; Powls, A.; Cooke, R.W.; Marlow, N. Attention deficit hyperactivity disorders and other psychiatric outcomes in very low birth weight children at 12 years. J. Child Psychol. Psychiatry 1997, 38, 931–941. [Google Scholar] [CrossRef]
- Breslau, N.; Chilcoat, H.D. Psychiatric sequelae of low birth weight at 11 years of age. Biol. Psychiatry 2000, 47, 1005–1011. [Google Scholar] [CrossRef]
- Burka, S.L.; Lipsitt, L.P.; Tsuang, M.T. Emotional and behavioural development of low birth-weight infants. Annu. Adv. Appl. Dev. Psychol. 1992, 187–214. [Google Scholar]
- Nosarti, C.; Allin, M.P.; Frangou, S.M.; Rifkin, L.; Murray, R.M. Hyperactivity in adolescents born very preterm is associated with decreased caudate volume. Biol. Psychiatry 2005, 57, 661–666. [Google Scholar] [CrossRef]
- Perricone, G.; Morales, M.R.; Anzalone, G. Neurodevelopmental outcomes of moderately preterm birth: Precursors of attention deficit hyperactivity disorderat preschool age. Springerplus 2013. [Google Scholar] [CrossRef]
- Edgin, J.O.; Inder, T.E.; Anderson, P.J.; Hood, K.M.; Clark, C.A.; Woodward, L.J. Executive functioning in preschool children born very preterm: Relationship with early white matter pathology. J. Int. Neuropsychol. Soc. 2008, 14, 90–101. [Google Scholar]
- Ford, R.M.; Neulinger, K.; O’Callaghan, M.; Mohay, H.; Gray, P.; Shum, D. Executive function in 7–9-year-old children born extremely preterm or with extremely low birth weight: Effects of biomedical history, age at assessment, and socioeconomic status. Arch. Clin. Neuropsychol. 2011, 26, 632–644. [Google Scholar] [CrossRef]
- Clark, C.A.; Woodward, L.J. Neonatal cerebral abnormalities and later verbal and visuospatial working memory abilities of children born very preterm. Dev. Neuropsychol. 2010, 35, 622–642. [Google Scholar] [CrossRef]
- Farooqi, A.; Hagglof, B.; Serenius, F. Behaviours related to executive functions and learning skills at 11 years of age after extremely preterm birth: A Swedish national prospective follow-up study. Acta Paediatr. 2013, 102, 625–634. [Google Scholar] [CrossRef]
- Luciana, M.; Lindeke, L.; Georgieff, M.; Mills, M.; Nelson, C.A. Neurobehavioral evidence for working-memory deficits in school-aged children with histories of prematurity. Dev. Med. Child Neurol. 1999, 41, 521–533. [Google Scholar] [CrossRef]
- Beauchamp, M.H.; Thompson, D.K.; Howard, K.; Doyle, L.W.; Egan, G.F.; Inder, T.E.; Anderson, P.J. Preterm infant hippocampal volumes correlate with later working memory deficits. Brain 2008, 131, 2986–2994. [Google Scholar]
- Vannucci, R.C.; Vannucci, S.J. A model of perinatal hypoxic-ischemic brain damage. Ann. N. Y. Acad. Sci. 1997, 835, 234–249. [Google Scholar] [CrossRef]
- Vannucci, R.C.; Connor, J.R.; Mauger, D.T.; Palmer, C.; Smith, M.B.; Towfighi, J.; Vannucci, S.J. Rat model of perinatal hypoxic-ischemic brain damage. J. Neurosci. Res. 1999, 55, 158–163. [Google Scholar] [CrossRef]
- Towfighi, J. Neuropathology of remote hypoxic-ischemic damage in the immature rat. Acta Neuropathol. 1991, 81, 578–587. [Google Scholar] [CrossRef]
- Bhutta, A.T. Abnormal cognition and behavior in preterm neonates linked to smaller brain volumes. Trends Neurosci. 2001, 24, 129–130. [Google Scholar] [CrossRef]
- Smith, A.L.; Alexander, M.; Rosenkrantz, T.S.; Sadek, M.L.; Fitch, R.H. Sex differences in behavioral outcome following neonatal hypoxia ischemia: Insights from a clinical meta-analysis and a rodent model of induced hypoxic ischemic brain injury. Exp. Neurol. 2014, 254C, 54–67. [Google Scholar]
- McClure, M.M.; Threlkeld, S.W.; Fitch, R.H. The effects of erythropoietin on auditory processing following neonatal hypoxic-ischemic injury. Brain Res. 2006, 1087, 190–195. [Google Scholar]
- McClure, M.M.; Threlkeld, S.W.; Fitch, R.H. Auditory processing and learning/memory following erythropoietin administration in neonatally hypoxic-ischemic injured rats. Brain Res. 2007, 1132, 203–209. [Google Scholar]
- Hill, C.A.; Threlkeld, S.W.; Fitch, R.H. Early testosterone modulated sex differences in behavioral outcome following neonatal hypoxia ischemia in rats. Int. J. Dev. Neurosci. 2011, 29, 381–388. [Google Scholar]
- Alexander, M.L.; Smith, A.L.; Rosenkrantz, T.S.; Fitch, R.H. Therapeutic effect of caffeine treatment immediately following neonatal hypoxic-ischemic injury on spatial memory in male rats. Brain Sci. 2013, 3, 177–190. [Google Scholar] [CrossRef]
- Kumral, A.; Uysal, N.; Tugyan, K.; Sonmez, A.; Yilmaz, O.; Gokmen, N; Kiray, M.; Genc, S.; Duman, N.; Koroglu, T.F.; et al. Erythropoietin improves long-term spatial memory deficits and brain injury following neonatal hypoxia-ischemia in rats. Behav. Brain Res. 2004, 153, 77–86. [Google Scholar] [CrossRef]
- Wagner, B.P.; Nedelcu, J.; Martin, E. Delayed postischemic hypothermia improves long-term behavioral outcome after cerebral hypoxia-ischemia in neonatal rats. Pediatr. Res. 2002, 51, 354–360. [Google Scholar] [CrossRef]
- Arteni, N.S.; Pereira, L.O.; Rodrigues, A.L.; Lavinsky, D.; Achaval, M.E.; Netto, C.A. Lateralized and sex-dependent behavioral and morphological effects of unilateral neonatal cerebral hypoxia-ischemia in the rat. Behav. Brain Res. 2010, 210, 92–98. [Google Scholar] [CrossRef]
- Pereira, L.O.; Arteni, N.S.; Peterson, R.C.; da Rocha, A.P.; Achaval, M.; Netto, C.A. Effects of daily environmental enrichment on memory deficits and brain injury following neonatal hypoxia-ischemia in the rat. Neurobiol. Learn. Mem. 2007, 87, 101–108. [Google Scholar] [CrossRef]
- Chou, I.C.; Trakht, T.; Signori, C.; Smith, J.; Felt, B.T.; Vazquez, D.M.; Barks, J.D. Behavioral/environmental intervention improves learning after cerebral hypoxia-ischemia in rats. Stroke 2001, 32, 2192–2197. [Google Scholar] [CrossRef]
- Almli, C.R. BDNF protects against spatial memory deficits following neonatal hypoxia-ischemia. Exp. Neurol. 2000, 166, 99–114. [Google Scholar] [CrossRef]
- Mishima, K.; Ikeda, T.; Yoshikawa, T.; Aoo, N.; Egashira, N.; Xia, Y.X.; Ikenoue, T.; Iwasaki, K.; Fujiwara, M. Effects of hypothermia and hyperthermia on attentional and spatial learning deficits following neonatal hypoxia-ischemic insult in rats. Behav. Brain Res. 2004, 151, 209–217. [Google Scholar] [CrossRef]
- Ikeda, T.; Mishima, K.; Yoshikawa, T.; Iwasaki, K.; Fujiwara, M.; Xia, Y.X.; Ikenoue, T. Selective and long-term learning impairment following neonatal hypoxic-ischemic brain insult in rats. Behav. Brain Res. 2001, 118, 17–25. [Google Scholar] [CrossRef]
- Ikeda, T.; Mishima, K.; Aoo, N.; Egashira, N.; Iwasaki, K.; Fujiwara, M.; Ikenoue, T. Combination treatment of neonatal rats with hypoxia-ischemia and endotoxin induces long-lasting memory and learning impairment that is associated with extended cerebral damage. Am. J. Obstet. Gynecol. 2004, 191, 2132–2141. [Google Scholar] [CrossRef]
- Balduini, W.; de Angelis, V.; Mazzoni, E.; Cimino, M. Long-lasting behavioral alterations following a hypoxic/ischemic brain injury in neonatal rats. Brain Res. 2000, 859, 318–325. [Google Scholar] [CrossRef]
- Mishima, K.; Ikeda, T.; Aoo, N.; Takai, N.; Takahashi, S.; Egashira, N.; Ikenoue, T.; Iwasaki, K.; Fujiwara, M. Hypoxia-ischemic insult in neonatal rats induced slowly progressive brain damage related to memory impairment. Neurosci. Lett. 2005, 376, 194–199. [Google Scholar] [CrossRef]
- Oorschot, D.E.; Voss, L.; Covey, M.V.; Bilkey, D.K.; Saunders, S.E. ADHD-like hyperactivity, with no attention deficit, in adult rats after repeated hypoxia during the equivalent of extreme prematurity. J. Neurosci. Methods 2007, 166, 315–322. [Google Scholar] [CrossRef]
- Dudchenko, P.A. An overview of the tasks used to test working memory in rodents. Neurosci. Biobehav. Rev. 2004, 28, 699–709. [Google Scholar] [CrossRef]
- Olton, D.S.; Samuelson, R.J. Remembrance of places passed: Spatial memory in rats. J. Exp. Psychol. Anim. Behav. Process. 1976, 97–116. [Google Scholar] [CrossRef]
- Olton, D.S.; Paras, B.C. Spatial memory and hippocampal function. Neuropsychologia 1978, 17, 669–682. [Google Scholar] [CrossRef]
- Honig, W.K. Studies of working memory in the pigeon. Cogn. Process. Anim. Behav. 1978, 211–248. [Google Scholar]
- Chrobak, J.J.; Hinman, J.R.; Sabolek, H.R. Revealing past memories: Proactive interference and ketamine-induced memory deficits. J. Neurosci. 2008, 28, 4512–4520. [Google Scholar] [CrossRef]
- Hill, C.A.; Alexander, M.L.; McCullough, L.D.; Fitch, R.H. Inhibition of X-linked inhibitor of apoptosis with embelin differentially affects male versus female behavioral outcome following neonatal hypoxia-ischemia in rats. Dev. Neurosci. 2011, 33, 494–504. [Google Scholar] [CrossRef]
- Koss, W.A.; Franklin, A.D.; Juraska, J.M. Delayed alternation in adolescent and adult male and female rats. Dev. Psychobiol. 2011, 53, 724–731. [Google Scholar] [CrossRef]
- Shukitt-Hale, B.; McEwen, J.J.; Szprengiel, A.; Joseph, J.A. Effect of age on the radial arm water maze—a test of spatial learning and memory. Neurobiol. Aging 2004, 25, 223–229. [Google Scholar] [CrossRef]
- Bulhuis, J.J.; Buresova, O.; Bures, J. Persistance of working memory of rats in an aversively motivated radial maze task. Behav. Brain Res. 1985, 15, 43–49. [Google Scholar] [CrossRef]
- Strijkstra, A.M.; Bolhuis, J.J. Memory persistence of rats in a radial maze varies with training procedure. Behav. Neural Biol. 1987, 47, 158–166. [Google Scholar] [CrossRef]
- McClure, M.M.; Peiffer, A.M.; Rosen, G.D.; Fitch, R.H. Auditory processing deficits in rats with neonatal hypoxic-ischemic injury. Int. J. Dev. Neurosci. 2005, 23, 351–362. [Google Scholar] [CrossRef]
- Aarnoudse-Moens, C.S.; Weisglas-Kuperus, N.; Duivenvoorden, H.J.; van Goudoever, J.B.; Oosterlaan, J. Executive function and IQ predict mathematical and attention problems in very preterm children. PLoS One 2013, 8, e55994. [Google Scholar] [CrossRef] [Green Version]
- Tanis, J.C.; van der Ree, M.H.; Roze, E.; Huis inʼt Veld, A.E.; van den Berg, P.P.; van Braeckel, K.N.; Bos, A.F. Functional outcome of very preterm-born and small-for-gestational-age children at school age. Pediatr. Res. 2012, 72, 641–648. [Google Scholar] [CrossRef]
- Bos, A.F.; Roze, E. Neurodevelopmental outcome in preterm infants. Dev. Med. Child Neurol. 2011, 53, 35–39. [Google Scholar] [CrossRef]
- Anderson, P.J.; Doyle, L.W.; Victorian Infant Collaborative Study. Executive functioning in school-aged children who were born very preterm or with extremely low birth weight in the 1990s. Pediatrics 2004, 114, 50–57. [Google Scholar] [CrossRef]
- Pugliese, M.; Rossi, C.; Guidotti, I.; Gallo, C.; Della Casa, E.; Bertoncelli, N.; Coccolini, E.; Ferrari, F. Preterm birth and developmental problems in infancy and preschool age Part II: Cognitive, neuropsychological and behavioral outcomes. J. Matern. Fetal Neonatal Med. 2013, 26, 1653–1657. [Google Scholar] [CrossRef]
- Hutchinson, E.A.; de Luca, C.R.; Doyle, L.W.; Roberts, G.; Anderson, P.J.; Victorian Infant Collaborative Study Group. School-age outcomes of extremely preterm or low birth weight children. Pediatrics 2013, 131, e1053–e1061. [Google Scholar] [CrossRef]
- Visual Statistician Calulcator. Available online: http://web.mst.edu/~psyworld/tukeyscalculator.htm#1 (accessed on 4 March 2014).
- Briscoe, J.; Gathercole, S.E.; Marlow, N. Short-term memory and language outcomes after extreme prematurity at birth. J. Speech Lang. Hear. Res. 1998, 41, 654–666. [Google Scholar]
- Badawi, N.; Keogh, J.M.; Dixon, G.; Kurinczuk, J.J. Developmental outcomes of newborn encephalopathy in the term infant. Indian J. Pediatr. 2001, 68, 527–530. [Google Scholar] [CrossRef]
- Getahun, D.; Rhoads, G.G.; Demissie, K.; Lu, S.E.; Quinn, V.P.; Fassett, M.J.; Wing, D.A.; Jacobsen, S.J. In utero exposure to ischemic-hypoxic conditions andattention-deficit/hyperactivity disorder. Pediatrics 2013, 131, e53–e61. [Google Scholar] [CrossRef]
- Rose, S.A.; Feldman, J.F.; Jankowski, J.J.; van Rossem, R. The structure of memory in infants and toddlers: An SEM study with full-terms and preterms. Dev. Sci. 2011, 14, 83–91. [Google Scholar] [CrossRef]
- De Haan, M.; Bauer, P.J.; Georgieff, M.K.; Nelson, C.A. Explicit memory in low-risk infants aged 19-months born between 27 and 42 weeks of gestation. Dev. Med. Child Neurol. 2000, 42, 304–312. [Google Scholar] [CrossRef]
- Ritter, B.C.; Nelle, M.; Perrig, W.; Steinlin, M.; Everts, R. Executive functions of children born very preterm—Deficit or delay? Eur. J. Pediatr. 2013, 172, 473–483. [Google Scholar] [CrossRef]
- Ment, L.R.; Vohr, B.; Allan, W.; Katz, K.H.; Schneider, K.C.; Westerveld, M. Change in cognitive function over time in very low-birth-weight infants. J. Am. Med. Assoc. 2003, 289, 705–711. [Google Scholar] [CrossRef]
- Tideman, E. Longitudinal follow-up of children born preterm: Cognitive development at age 19. Early Hum. Dev. 2000, 58, 81–90. [Google Scholar] [CrossRef]
- Barde, L.H.; Yeatman, J.D.; Lee, E.S.; Glover, G.; Feldman, H.M. Differences in neural activation between preterm and full term born adolescents on a sentence comprehension task: Implications for educational accommodations. Dev. Cogn. Neurosci. 2012, 2, 114–128. [Google Scholar] [CrossRef]
- Pritchard, V.E.; Clark, C.A.; Liberty, K.; Champion, P.R.; Wilson, K.; Woodward, L.J. Early school-based learning difficulties in children born very preterm. Early Hum. Dev. 2009, 85, 215–224. [Google Scholar] [CrossRef]
- Nosarti, C.; Rubia, K.; Smith, A.B.; Frearson, S.; Williams, S.C.; Rifkin, L.; Murray, R.M. Altered functional neuroanatomy of response inhibition in adolescent males who were born very preterm. Dev. Med. Child Neurol. 2006, 48, 265–271. [Google Scholar] [CrossRef]
- Curtis, W.J.; Lindeke, L.L.; Georgieff, M.K.; Nelson, C.A. Neurobehavioural functioning in neonatal intensive care unit graduates in late childhood and early adolescence. Brain 2002, 125, 1646–1659. [Google Scholar] [CrossRef]
- Vicari, S.; Caravale, B.; Carlesimo, G.A.; Casadei, A.M.; Allemand, F. Spatial working memory deficits in children at ages 3–4 who were low birth weight, preterm infants. Neuropsychology 2004, 18, 673–678. [Google Scholar] [CrossRef]
- Sansavini, A.; Guarini, A.; Caselli, M.C. Preterm birth: Neuropsychological profiles and atypical developmental pathways. Dev. Disabil. Res. Rev. 2011, 17, 102–113. [Google Scholar] [CrossRef]
- Arteni, N.S.; Salgueiro, J.; Torres, I.; Achaval, M.; Netto, C.A. Neonatal cerebral hypoxia-ischemia causes lateralized memory impairments in the adult rat. Brain Res. 2003, 973, 171–178. [Google Scholar] [CrossRef]
- Cherkes-Julkowski, M. Learning disability, attention-deficit disorder, and language impairment as outcomes of prematurity: A longitudinal descriptive study. Educ. Psychol. 1998, 31, 294–306. [Google Scholar]
- Donders, J.; Hoffman, N.M. Gender differences in learning and memory after pediatric traumatic brain injury. Neuropsychology 2002, 16, 491–499. [Google Scholar] [CrossRef]
- Lauterbach, M.D.; Raz, S.; Sander, C.J. Neonatal hypoxic risk in preterm birth infants: The influence of sex and severity of respiratory distress on cognitive recovery. Neuropsychology 2001, 15, 411–420. [Google Scholar] [CrossRef]
- Rutter, M.; Caspi, A.; Moffitt, T.E. Using sex differences in psychopathology to study causal mechanisms: Unifying issues and research strategies. J. Child Psychol. Psychiatry 2003, 44, 1092–1115. [Google Scholar] [CrossRef]
- Gualtieri, C.T.; Ondrusek, M.G.; Finley, C. Attention deficit disorders in adults. Clin. Neuropharmacol. 1985, 8, 343–356. [Google Scholar] [CrossRef]
- Spencer, T.J. Attention-deficit/hyperactivity disorder: Diagnosis, lifespan, comorbidities, and neurobiology. Ambul. Pediatr. 2007, 7, 73–81. [Google Scholar] [CrossRef]
- Biederman, J. Growth deficits and attention-deficit/hyperactivity disorder revisited: Impact of gender, development, and treatment. Pediatrics 2003, 111, 1010–1016. [Google Scholar] [CrossRef]
- Biederman, J.; Mick, E.; Faraone, S.V.; Braaten, E.; Doyle, A.; Spencer, T.; Wilens, T.E.; Frazier, E.; Johnson, M.A. Influence of gender on attention deficit hyperactivity disorder in children referred to a psychiatric clinic. Am. J. Psychiatry 2002, 159, 36–42. [Google Scholar] [CrossRef]
- Rucklidge, J.J. Gender differences in attention-deficit/hyperactivity disorder. Psychiatr. Clin. N. Am. 2010, 33, 357–373. [Google Scholar] [CrossRef]
- Johnston, M.V. Excitotoxicity in perinatal brain injury. Brain Pathol. 2005, 25, 234–240. [Google Scholar]
- Klingberg, T.; Forssberg, H.; Westerberg, H. Increased brain activity in frontal and parietal cortex underlies the development of visuospatial working memory capacity during childhood. J. Cogn. Neurosci. 2002, 14, 1–10. [Google Scholar] [CrossRef]
- Thomas, K.M.; King, S.W.; Franzen, P.L.; Welsh, T.F.; Berkowitz, A.L.; Noll, D.C.; Birmaher, V.; Casey, B.J. A developmental functional MRI study of spatial working memory. Neuroimage 1999, 10, 327–338. [Google Scholar] [CrossRef]
- Martel, M.M.; Nigg, J.T.; Wong, M.M.; Fitzgerald, H.E.; Jester, J.M.; Puttler, L.I.; Glass, J.M.; Adams, K.M.; Zucker, R.A. Childhood and adolescent resiliency, regulation, and executive functioning in relation to adolescent problems and competence in a high-risk sample. Dev. Psychopathol. 2007, 19, 541–563. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Smith, A.L.; Hill, C.A.; Alexander, M.; Szalkowski, C.E.; Chrobak, J.J.; Rosenkrantz, T.S.; Fitch, R.H. Spatial Working Memory Deficits in Male Rats Following Neonatal Hypoxic Ischemic Brain Injury Can Be Attenuated by Task Modifications. Brain Sci. 2014, 4, 240-272. https://doi.org/10.3390/brainsci4020240
Smith AL, Hill CA, Alexander M, Szalkowski CE, Chrobak JJ, Rosenkrantz TS, Fitch RH. Spatial Working Memory Deficits in Male Rats Following Neonatal Hypoxic Ischemic Brain Injury Can Be Attenuated by Task Modifications. Brain Sciences. 2014; 4(2):240-272. https://doi.org/10.3390/brainsci4020240
Chicago/Turabian StyleSmith, Amanda L., Courtney A. Hill, Michelle Alexander, Caitlin E. Szalkowski, James J. Chrobak, Ted S. Rosenkrantz, and R. Holly Fitch. 2014. "Spatial Working Memory Deficits in Male Rats Following Neonatal Hypoxic Ischemic Brain Injury Can Be Attenuated by Task Modifications" Brain Sciences 4, no. 2: 240-272. https://doi.org/10.3390/brainsci4020240



