Ultrastructural Changes of Neuroendocrine Pheochromocytoma Cell Line PC-12 Exposed In Vitro to Rotenone
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Cell Viability
2.3. Transmission Electron Microscopy
2.4. Morphometric Analysis
2.5. Statistical Analysis
3. Results
3.1. Effect on Viability
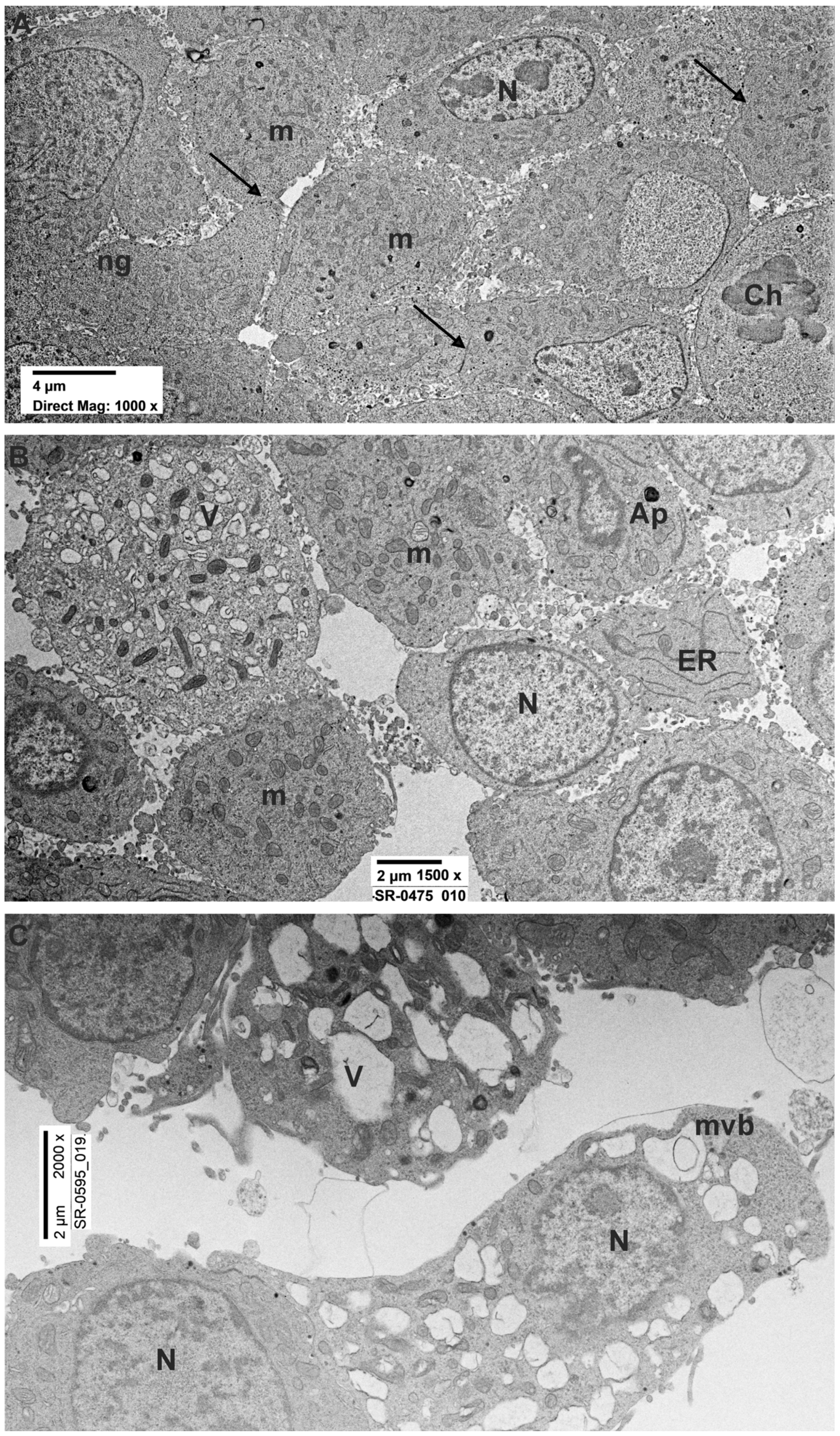
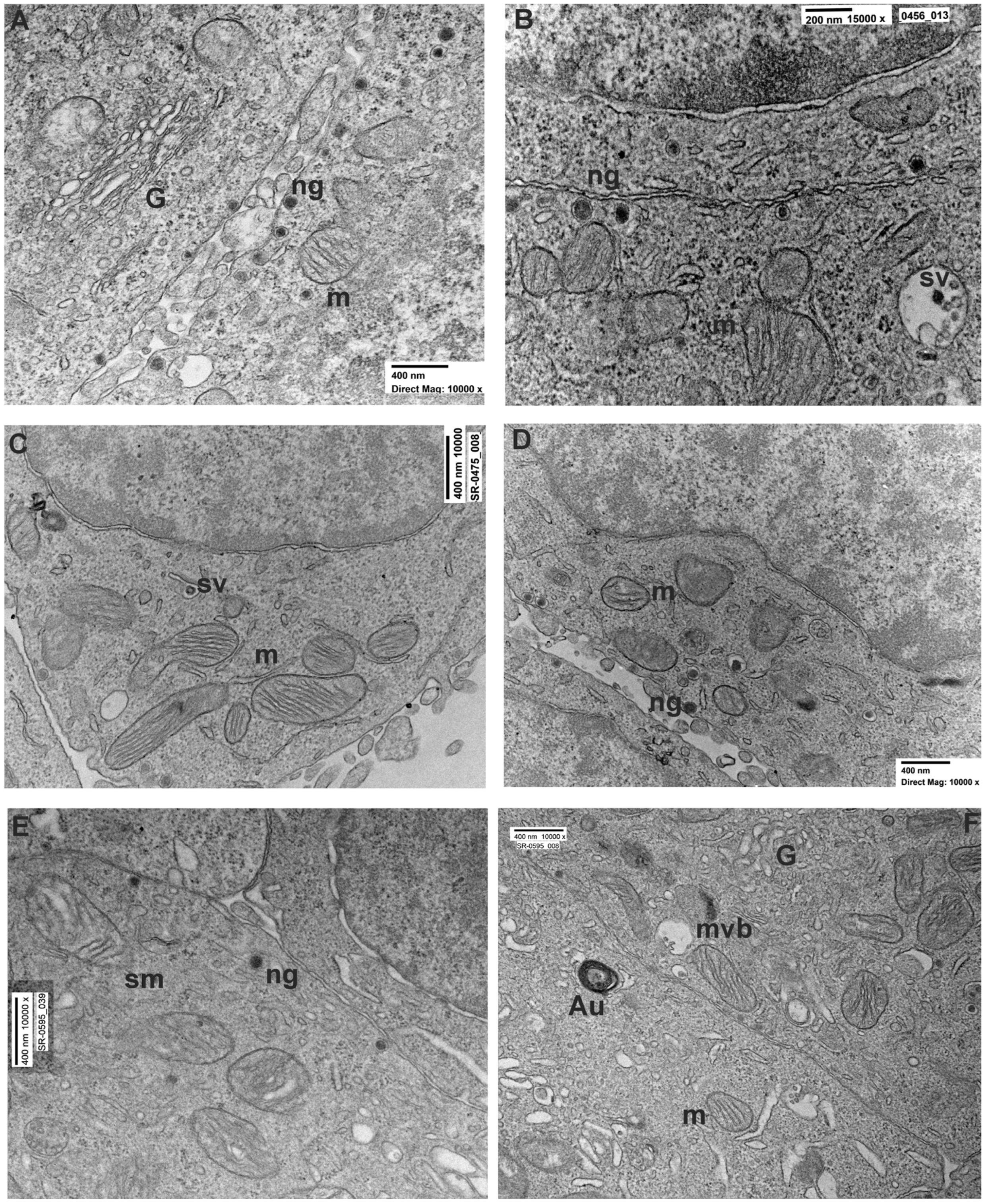
3.2. Ultrastructural Evaluation
3.2.1. PC12 Control
3.2.2. PC12 Treated with Rotenone 0.5 µM for 24 h
3.2.3. PC12 Treated with Rotenone 0.5 µM for 48 h
3.2.4. Morphometric Analysis
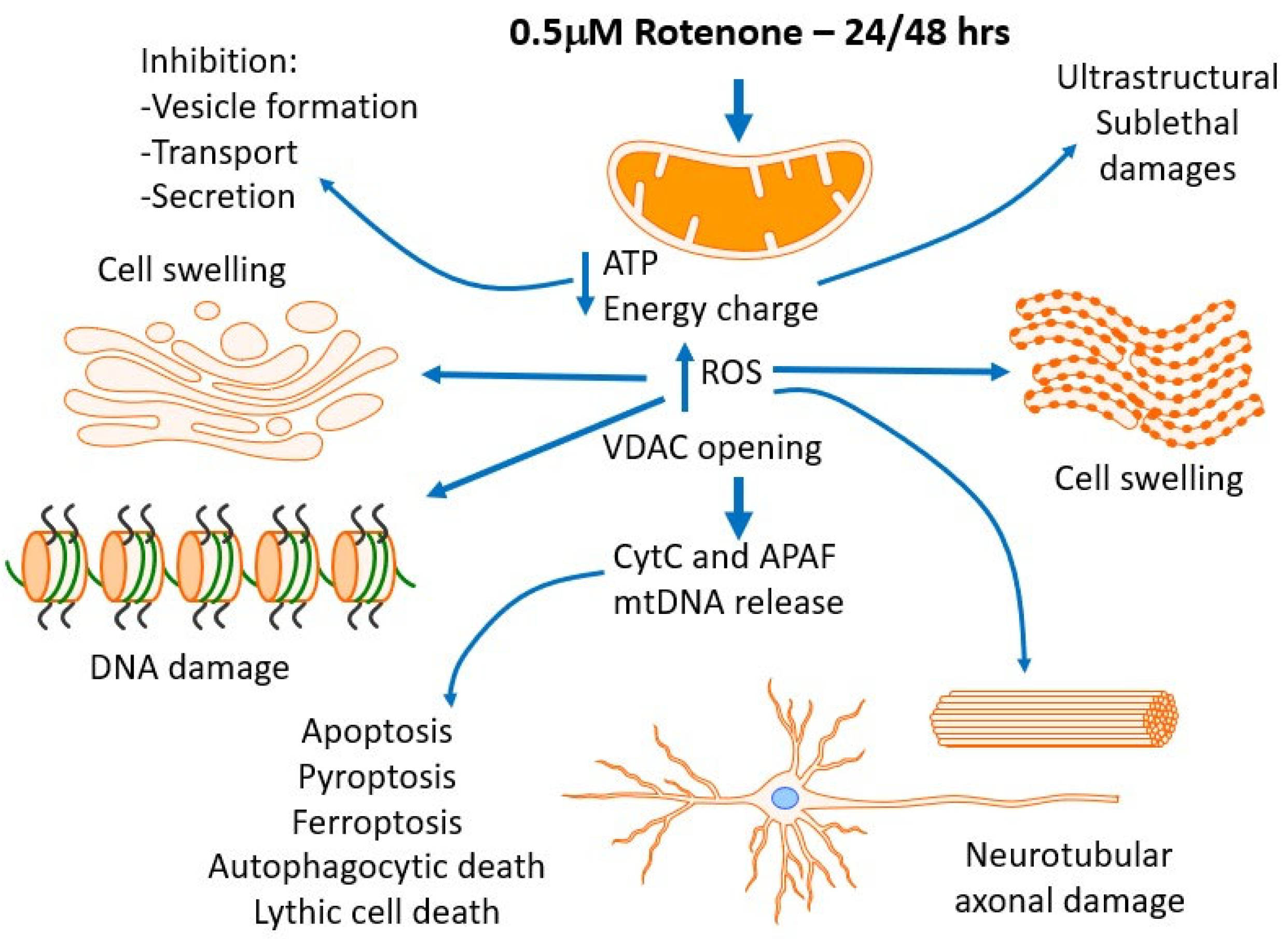
4. Discussion
4.1. Mitochondrial Disfunction
4.2. Effects of ROS and Oxidations of Other Biological Targets—Metabolic Effects
4.3. Possible Impact on Granule/Vesicle/Multivesicular Bodies Formation, Transport and Secretion, and Autophagocytosis
4.4. Cell-to-Cell Contacts in Confluent Cultures
4.5. In Vivo/In Vitro Mechanisms and Limitations of This Paper
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sherer, T.B.; Betarbet, R.; Stout, A.K.; Lund, S.; Baptista, M.; Panov, A.V.; Cookson, M.R.; Greenamyre, J.T. An In Vitro Model of Parkinson’s Disease: Linking Mitochondrial Impairment to Altered α-Synuclein Metabolism and Oxidative Damage. J. Neurosci. 2002, 22, 7006–7015. [Google Scholar] [CrossRef] [PubMed]
- Tanner, C.M.; Kamel, F.; Ross, G.W.; Hoppin, J.A.; Goldman, S.M.; Korell, M.; Marras, C.; Bhudhikanok, G.S.; Kasten, M.; Chade, A.R.; et al. Rotenone, Paraquat, and Parkinson’s Disease. Environ. Health Perspect. 2011, 119, 866–872. [Google Scholar] [CrossRef]
- Sharma, N.; Jamwal, S.; Kumar, P. Beneficial Effect of Antidepressants against Rotenone Induced Parkinsonism like Symptoms in Rats. Pathophysiology 2016, 23, 123–134. [Google Scholar] [CrossRef]
- Dodiya, H.B.; Forsyth, C.B.; Voigt, R.M.; Engen, P.A.; Patel, J.; Shaikh, M.; Green, S.J.; Naqib, A.; Roy, A.; Kordower, J.H.; et al. Chronic Stress-Induced Gut Dysfunction Exacerbates Parkinson’s Disease Phenotype and Pathology in a Rotenone-Induced Mouse Model of Parkinson’s Disease. Neurobiol. Dis. 2020, 135, 104352. [Google Scholar] [CrossRef]
- Gosselin, R.E.; Smith, R.P.; Hodge, H.C. Clinical Toxicology of Commercial Products, 5th ed.; Williams & Wilkins: Baltimore, MD, USA; London, UK, 1984; pp. 111–366. [Google Scholar]
- Sai, Y.; Wu, Q.; Le, W.; Ye, F.; Li, Y.; Dong, Z. Rotenone-Induced PC12 Cell Toxicity Is Caused by Oxidative Stress Resulting from Altered Dopamine Metabolism. Toxicol. In Vitro 2008, 22, 1461–1468. [Google Scholar] [CrossRef]
- Betarbet, R.; Sherer, T.B.; MacKenzie, G.; Garcia-Osuna, M.; Panov, A.V.; Greenamyre, J.T. Chronic Systemic Pesticide Exposure Reproduces Features of Parkinson’s Disease. Nat. Neurosci. 2000, 3, 1301–1306. [Google Scholar] [CrossRef]
- Radad, K.; Al-Shraim, M.; Al-Emam, A.; Wang, F.; Kranner, B.; Rausch, W.-D.; Moldzio, R. Rotenone: From Modelling to Implication in Parkinson’s Disease. Folia Neuropathol. 2019, 57, 317–326. [Google Scholar] [CrossRef]
- Heinz, S.; Freyberger, A.; Lawrenz, B.; Schladt, L.; Schmuck, G.; Ellinger-Ziegelbauer, H. Mechanistic Investigations of the Mitochondrial Complex I Inhibitor Rotenone in the Context of Pharmacological and Safety Evaluation. Sci. Rep. 2017, 7, 45465. [Google Scholar] [CrossRef]
- Grivennikova, V.G.; Maklashina, E.O.; Gavrikova, E.V.; Vinogradov, A.D. Interaction of the Mitochondrial NADH-Ubiquinone Reductase with Rotenone as Related to the Enzyme Active/Inactive Transition. Biochim. Biophys. Acta BBA Bioenerg. 1997, 1319, 223–232. [Google Scholar] [CrossRef]
- Tafani, M.; Sansone, L.; Limana, F.; Arcangeli, T.; De Santis, E.; Polese, M.; Fini, M.; Russo, M.A. The Interplay of Reactive Oxygen Species, Hypoxia, Inflammation, and Sirtuins in Cancer Initiation and Progression. Oxidative Med. Cell. Longev. 2016, 2016, 3907147. [Google Scholar] [CrossRef]
- Betarbet, R.; Canet-Aviles, R.M.; Sherer, T.B.; Mastroberardino, P.G.; McLendon, C.; Kim, J.-H.; Lund, S.; Na, H.-M.; Taylor, G.; Bence, N.F.; et al. Intersecting Pathways to Neurodegeneration in Parkinson’s Disease: Effects of the Pesticide Rotenone on DJ-1, α-Synuclein, and the Ubiquitin–Proteasome System. Neurobiol. Dis. 2006, 22, 404–420. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.E.; Stringer, A.; Bobrovskaya, L. Rotenone Induces Gastrointestinal Pathology and Microbiota Alterations in a Rat Model of Parkinson’s Disease. Neurotoxicology 2018, 65, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Cannon, J.R.; Tapias, V.; Na, H.M.; Honick, A.S.; Drolet, R.E.; Greenamyre, J.T. A Highly Reproducible Rotenone Model of Parkinson’s Disease. Neurobiol. Dis. 2009, 34, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Peng, K.; Yang, L.; Wang, J.; Ye, F.; Dan, G.; Zhao, Y.; Cai, Y.; Cui, Z.; Ao, L.; Liu, J.; et al. The Interaction of Mitochondrial Biogenesis and Fission/Fusion Mediated by PGC-1α Regulates Rotenone-Induced Dopaminergic Neurotoxicity. Mol. Neurobiol. 2017, 54, 3783–3797. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, E.; Kohno, K.; Arai, N.; Harashima, A.; Ariyasu, T.; Ushio, S. Adenosine N1-Oxide Exerts Anti-Inflammatory Effects through the PI3K/Akt/GSK-3β Signaling Pathway and Promotes Osteogenic and Adipocyte Differentiation. Biol. Pharm. Bull. 2019, 42, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Fu, R.; Duan, D.; Li, Z.; Li, B.; Ming, Y.; Li, L.; Ni, R.; Chen, J. Cyclovirobuxine D Induces Apoptosis and Mitochondrial Damage in Glioblastoma Cells Through ROS-Mediated Mitochondrial Translocation of Cofilin. Front. Oncol. 2021, 11, 656184. [Google Scholar] [CrossRef] [PubMed]
- Lin, V.Y.; Fain, M.D.; Jackson, P.L.; Berryhill, T.F.; Wilson, L.S.; Mazur, M.; Barnes, S.J.; Blalock, J.E.; Raju, S.V.; Rowe, S.M. Vaporized E-Cigarette Liquids Induce Ion Transport Dysfunction in Airway Epithelia. Am. J. Respir. Cell Mol. Biol. 2019, 61, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Xing, H.; Wang, X.; Liu, Y.; Li, B.; Zhang, L.; Li, Z.; Duan, D.; Chen, J. Neuroprotective Effects of Tetrahydroxystilbene Glucoside against Rotenone-Induced Toxicity in PC12 Cells. Biol. Pharm. Bull. 2022, 45, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Sherer, T.B.; Betarbet, R.; Testa, C.M.; Seo, B.B.; Richardson, J.R.; Kim, J.H.; Miller, G.W.; Yagi, T.; Matsuno-Yagi, A.; Greenamyre, J.T. Mechanism of Toxicity in Rotenone Models of Parkinson’s Disease. J. Neurosci. 2003, 23, 10756–10764. [Google Scholar] [CrossRef]
- Belcastro, V.; Tozzi, A.; Tantucci, M.; Costa, C.; Di Filippo, M.; Autuori, A.; Picconi, B.; Siliquini, S.; Luchetti, E.; Borsini, F.; et al. A2A Adenosine Receptor Antagonists Protect the Striatum against Rotenone-Induced Neurotoxicity. Exp. Neurol. 2009, 217, 231–234. [Google Scholar] [CrossRef]
- Rocha, S.M.; Bantle, C.M.; Aboellail, T.; Chatterjee, D.; Smeyne, R.J.; Tjalkens, R.B. Rotenone Induces Regionally Distinct α-Synuclein Protein Aggregation and Activation of Glia Prior to Loss of Dopaminergic Neurons in C57Bl/6 Mice. Neurobiol. Dis. 2022, 167, 105685. [Google Scholar] [CrossRef] [PubMed]
- Tischler, A.S. Chromaffin Cells as Models of Endocrine Cells and Neurons. Ann. N. Y. Acad. Sci. 2002, 971, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Deng, T.; Zhai, Z.; Sun, T.; Xu, Y. The Cellular Model for Alzheimer’s Disease Research: PC12 Cells. Front. Mol. Neurosci. 2023, 15, 1016559. [Google Scholar] [CrossRef] [PubMed]
- Sansone, L.; De Iure, A.; Cristina, M.; Belli, M.; Vitiello, L.; Marcolongo, F.; Rosellini, A.; Macera, L.; Spezia, P.G.; Tomino, C.; et al. Nicotine in Combination with SARS-CoV-2 Affects Cells Viability, Inflammatory Response and Ultrastructural Integrity. Int. J. Mol. Sci. 2022, 23, 9488. [Google Scholar] [CrossRef] [PubMed]
- Belli, M.; Rinaudo, P.; Palmerini, M.G.; Ruggeri, E.; Antonouli, S.; Nottola, S.A.; Macchiarelli, G. Pre-Implantation Mouse Embryos Cultured In Vitro under Different Oxygen Concentrations Show Altered Ultrastructures. Int. J. Environ. Res. Public Health 2020, 17, 3384. [Google Scholar] [CrossRef]
- Gatti, M.; Belli, M.; De Rubeis, M.; Khalili, M.A.; Familiari, G.; Nottola, S.A.; Macchiarelli, G.; Hajderi, E.; Palmerini, M.G. Ultrastructural Evaluation of Mouse Oocytes Exposed In Vitro to Different Concentrations of the Fungicide Mancozeb. Biology 2023, 12, 698. [Google Scholar] [CrossRef] [PubMed]
- Palmerini, M.G.; Belli, M.; Nottola, S.A.; Miglietta, S.; Bianchi, S.; Bernardi, S.; Antonouli, S.; Cecconi, S.; Familiari, G.; Macchiarelli, G. Mancozeb Impairs the Ultrastructure of Mouse Granulosa Cells in a Dose-Dependent Manner. J. Reprod. Dev. 2018, 64, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Huang, J.; Chen, R.; Zhang, T.; Shen, L.; Yang, J.; Sun, X. Protection against Neurotoxicity by an Autophagic Mechanism. Braz. J. Med. Biol. Res. 2012, 45, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Van Rossum, G.D.V.; Russo, M.A. Requirement of Cl− and Na+ for the Ouabain-Resistant Control of Cell Volume in Slices of Rat Liver. J. Membr. Biol. 1984, 77, 63–76. [Google Scholar] [CrossRef]
- Russo, M.A.; Ernst, S.A.; Kapoor, S.C.; Van Rossum, G.D.V. Morphological and Physiological Studies of Rat Kidney Cortex Slices Undergoing Isosmotic Swelling and Its Reversal: A Possible Mechanism for Ouabain-Resistant Control of Cell Volume. J. Membr. Biol. 1985, 85, 1–24. [Google Scholar] [CrossRef]
- Russo, M.A.; van Rossum, G.D. The Basis for the Cellular Damage Induced by Ethacrynic Acid in Liver Slices In Vitro. Comparison of Structure and Function. Lab. Investig. J. Tech. Methods Pathol. 1986, 54, 695–707. [Google Scholar]
- Xue, W.; Men, S.; Liu, R. Rotenone Restrains the Proliferation, Motility and Epithelial-Mesenchymal Transition of Colon Cancer Cells and the Tumourigenesis in Nude Mice via PI3K/AKT Pathway. Clin. Exp. Pharmacol. Physiol. 2020, 47, 1484–1494. [Google Scholar] [CrossRef]
- Hackenbrock, C.R. Ultrastructural Bases For Metabolically Linked Mechanical Activity In Mitochondria. J. Cell Biol. 1968, 37, 345–369. [Google Scholar] [CrossRef] [PubMed]
- Arnold, B.; Cassady, S.J.; VanLaar, V.S.; Berman, S.B. Integrating Multiple Aspects of Mitochondrial Dynamics in Neurons: Age-Related Differences and Dynamic Changes in a Chronic Rotenone Model. Neurobiol. Dis. 2011, 41, 189–200. [Google Scholar] [CrossRef]
- Chiaradia, E.; Renzone, G.; Scaloni, A.; Caputo, M.; Costanzi, E.; Gambelunghe, A.; Muzi, G.; Avellini, L.; Emiliani, C.; Buratta, S. Protein Carbonylation in Dopaminergic Cells Exposed to Rotenone. Toxicol. Lett. 2019, 309, 20–32. [Google Scholar] [CrossRef]
- Gómez, A.; Ferrer, I. Increased Oxidation of Certain Glycolysis and Energy Metabolism Enzymes in the Frontal Cortex in Lewy Body Diseases. J. Neurosci. Res. 2009, 87, 1002–1013. [Google Scholar] [CrossRef]
- Gibson, G.E.; Kingsbury, A.E.; Xu, H.; Lindsay, J.G.; Daniel, S.; Foster, O.J.F.; Lees, A.J.; Blass, J.P. Deficits in a Tricarboxylic Acid Cycle Enzyme in Brains from Patients with Parkinson’s Disease. Neurochem. Int. 2003, 43, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Pathak, D.; Berthet, A.; Nakamura, K. Energy Failure: Does It Contribute to Neurodegeneration? Ann. Neurol. 2013, 74, 506–516. [Google Scholar] [CrossRef]
- Morel, N.; Poëa-Guyon, S. The Membrane Domain of Vacuolar H+ATPase: A Crucial Player in Neurotransmitter Exocytotic Release. Cell. Mol. Life Sci. 2015, 72, 2561–2573. [Google Scholar] [CrossRef]
- Colacurcio, D.J.; Nixon, R.A. Disorders of Lysosomal Acidification—The Emerging Role of v-ATPase in Aging and Neurodegenerative Disease. Ageing Res. Rev. 2016, 32, 75–88. [Google Scholar] [CrossRef]
- Matsuda, N.; Tanaka, K. Does Impairment of the Ubiquitin-Proteasome System or the Autophagy-Lysosome Pathway Predispose Individuals to Neurodegenerative Disorders Such as Parkinson’s Disease? J. Alzheimers Dis. 2010, 19, 1–9. [Google Scholar] [CrossRef]
- Dehay, B.; Martinez-Vicente, M.; Caldwell, G.A.; Caldwell, K.A.; Yue, Z.; Cookson, M.R.; Klein, C.; Vila, M.; Bezard, E. Lysosomal Impairment in Parkinson’s Disease. Mov. Disord. 2013, 28, 725–732. [Google Scholar] [CrossRef]
- Peggion, C.; Calì, T.; Brini, M. Mitochondria Dysfunction and Neuroinflammation in Neurodegeneration: Who Comes First? Antioxidants 2024, 13, 240. [Google Scholar] [CrossRef]
- Singh, A.; Strobbe, D.; Campanella, M. Pyroptosis Targeting via Mitochondria: An Educated Guess to Innovate COVID-19 Therapies. Br. J. Pharmacol. 2022, 179, 2081–2085. [Google Scholar] [CrossRef]
- Dai, E.; Chen, X.; Linkermann, A.; Jiang, X.; Kang, R.; Kagan, V.E.; Bayir, H.; Yang, W.S.; Garcia-Saez, A.J.; Ioannou, M.S.; et al. A Guideline on the Molecular Ecosystem Regulating Ferroptosis. Nat. Cell Biol. 2024. [Google Scholar] [CrossRef]
- Denton, D.; Kumar, S. Autophagy-Dependent Cell Death. Cell Death Differ. 2019, 26, 605–616. [Google Scholar] [CrossRef]
- Hu, R.; Cao, Q.; Sun, Z.; Chen, J.; Zheng, Q.; Xiao, F. A Novel Method of Neural Differentiation of PC12 Cells by Using Opti-MEM as a Basic Induction Medium. Int. J. Mol. Med. 2017, 41, 195–201. [Google Scholar] [CrossRef]
| Treatment | Cell Death (%) |
|---|---|
| Control 24 h | 7.23 ± 0.95 |
| Rotenone 24 h | 14.70 ± 1.85 |
| Control 48 h | 4.65 ± 0.92 |
| Rotenone 48 h | 16.71 ± 0.40 |
| Rotenone | |||
|---|---|---|---|
| Control | 0.5 µM for 24 h | 0.5 µM for 48 h | |
| Mitochondria | Round/ovoid shape with electron-dense cristae. Uniformly distributed. | Round/elongated shape with electron-dense cristae. Uniformly distributed. | Round/elongated shape with less electron-dense cristae. Uniformly distributed. |
| Nuclei | Roundish, delimited by an intact electron-dense nuclear membrane. | Roundish, delimited by an intact electron-dense nuclear membrane. | Roundish or irregular, delimited by an intact electron-dense nuclear membrane. Presence of nuclear invagination. |
| Nuclear content of Hetero Chromatin | Regularly distributed in the nucleus with minor condensation close the nuclear envelope. | Regularly distributed in the nucleus with minor condensation close the nuclear envelope. | Regularly distributed in the nucleus with minor condensation close the nuclear envelope. |
| ER | Usually found in form of isolated cisternae. | Usually found in form of isolated cisternae. | Usually found in form of isolated cisternae |
| Neuropeptide granules | Numerous. Mainly distributed in peripheral area of the cell. | Less numerous. Mainly distributed in peripheral area of the cell. | Rare. Mainly distributed in peripheral area of the cell. |
| Intercellular connection | Numerous. | Less numerous. | Less numerous. |
| Autophagosomes and vacuoles | Rare. | Occasionally present. | Present. |
| Rotenone | |||
|---|---|---|---|
| Control | 0.5 µM for 24 h | 0.5 µM for 48 h | |
| Numerical density of mitochondria (per cell) | 24.8 ± 5.4 a | 22.7 ± 5.8 b | 20 ± 5.1 a |
| Mitochondrial length (µm) | 0.418 ± 0.183 a | 0.484 ± 0.173 b | 0.478 ± 0.161 c |
| Numerical density of neuropeptide granules (per cell) | 44.3 ± 16.5 a,b | 15.7 ± 11.4 a | 9.3 ± 14.3 b |
| Numerical density of neuropeptides granules on cell borders (per linear surface) | 11.1 ± 3.2 a,b | 5.2 ± 2.4 a | 3.1 ± 1.7 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belli, M.; Cristina, M.; Calabrese, V.; Russo, M.; Granato, M.; Russo, M.A.; Sansone, L. Ultrastructural Changes of Neuroendocrine Pheochromocytoma Cell Line PC-12 Exposed In Vitro to Rotenone. Brain Sci. 2024, 14, 476. https://doi.org/10.3390/brainsci14050476
Belli M, Cristina M, Calabrese V, Russo M, Granato M, Russo MA, Sansone L. Ultrastructural Changes of Neuroendocrine Pheochromocytoma Cell Line PC-12 Exposed In Vitro to Rotenone. Brain Sciences. 2024; 14(5):476. https://doi.org/10.3390/brainsci14050476
Chicago/Turabian StyleBelli, Manuel, Mario Cristina, Valeria Calabrese, Marta Russo, Marisa Granato, Matteo Antonio Russo, and Luigi Sansone. 2024. "Ultrastructural Changes of Neuroendocrine Pheochromocytoma Cell Line PC-12 Exposed In Vitro to Rotenone" Brain Sciences 14, no. 5: 476. https://doi.org/10.3390/brainsci14050476






