Photoelectrochemical Device Designs toward Practical Solar Water Splitting: A Review on the Recent Progress of BiVO4 and BiFeO3 Photoanodes
Abstract
:Featured Application
Abstract
1. Introduction
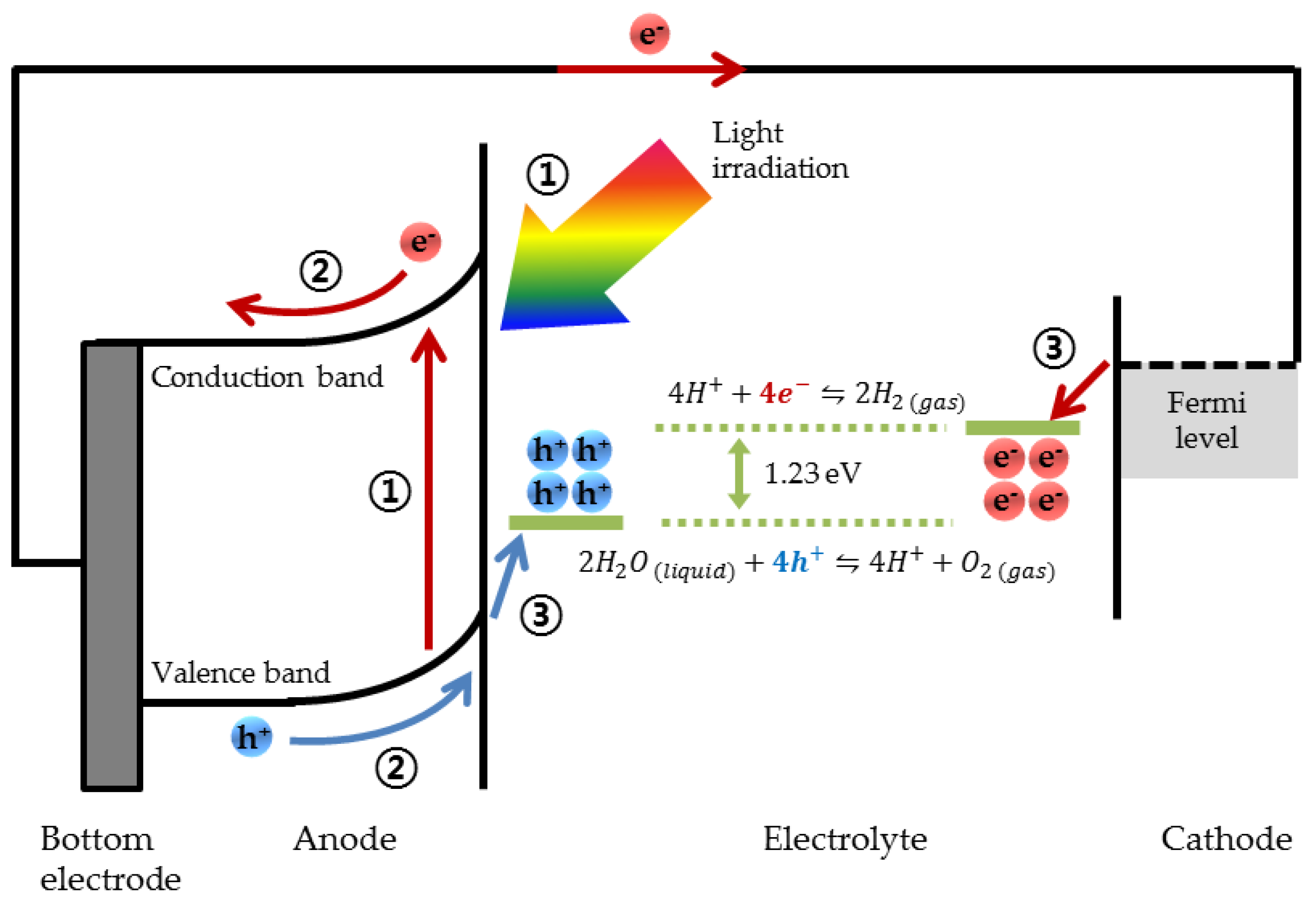
- Absorption of solar radiation by the photoelectrode, followed by charge carrier generation as a result of photoexcitation of electrons in the valence band of photoelectrode;
- Separation and transport of charge carriers to the electrode surface by the internal or external bias applied through the circuit;
- Oxidation of water at the anode by the photoexcited holes. Simultaneously, transport of H+ ions from the anode to the cathode, and transport of the photoexcited electrons to the cathode through an external circuit, followed by the reduction of H+ ions into hydrogen gas at the cathode by the photoexcited electrons.
2. Strategies and Device Designs for Efficient PEC Water Splitting
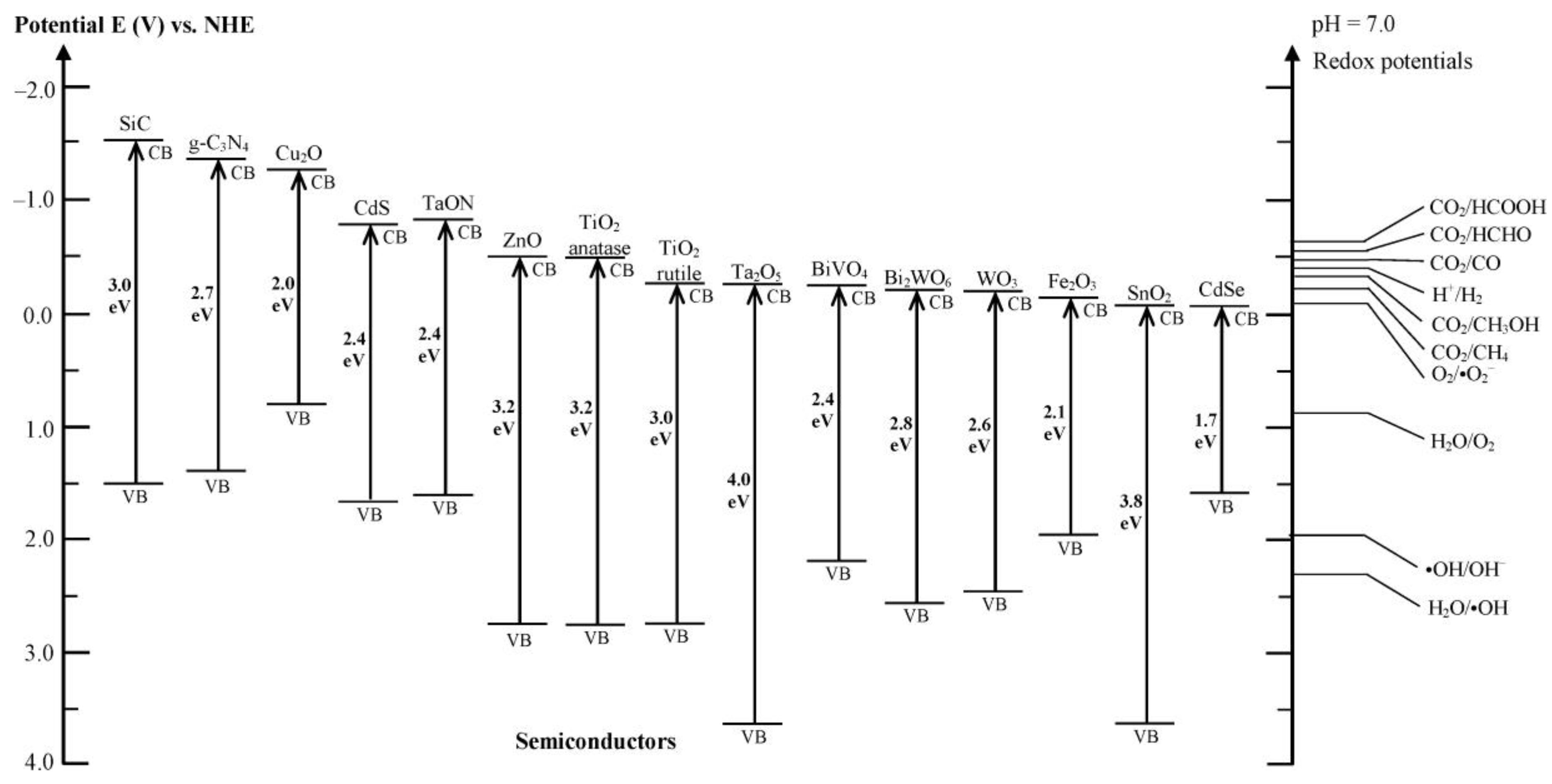
2.1. Absorption of Solar Radiation
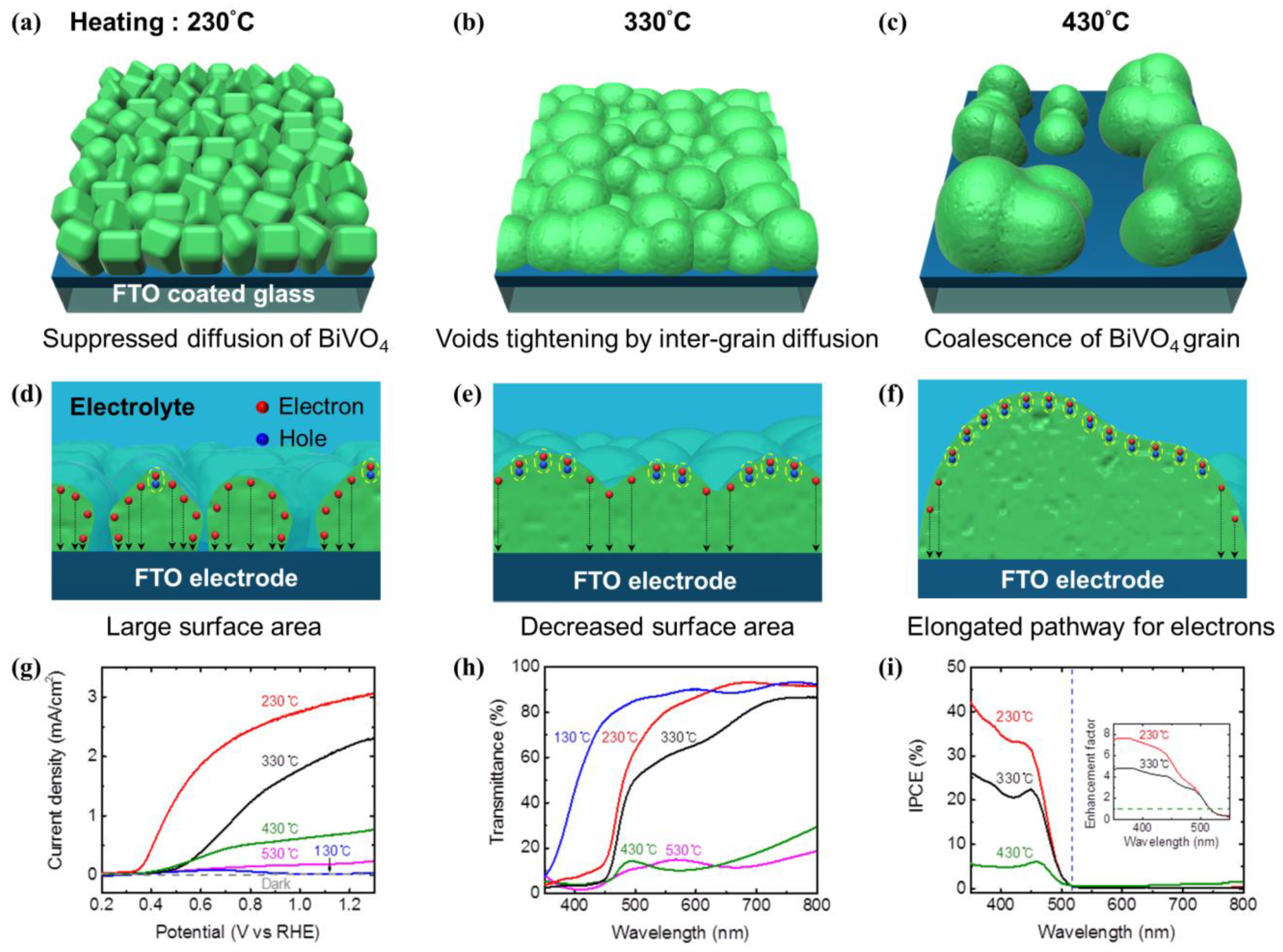
2.2. Separation and Transport of Charge Carrier
2.3. Kinetics of Water Redox Reactions
3. Challenging PEC Device Designs toward Practical Solar Water Splitting
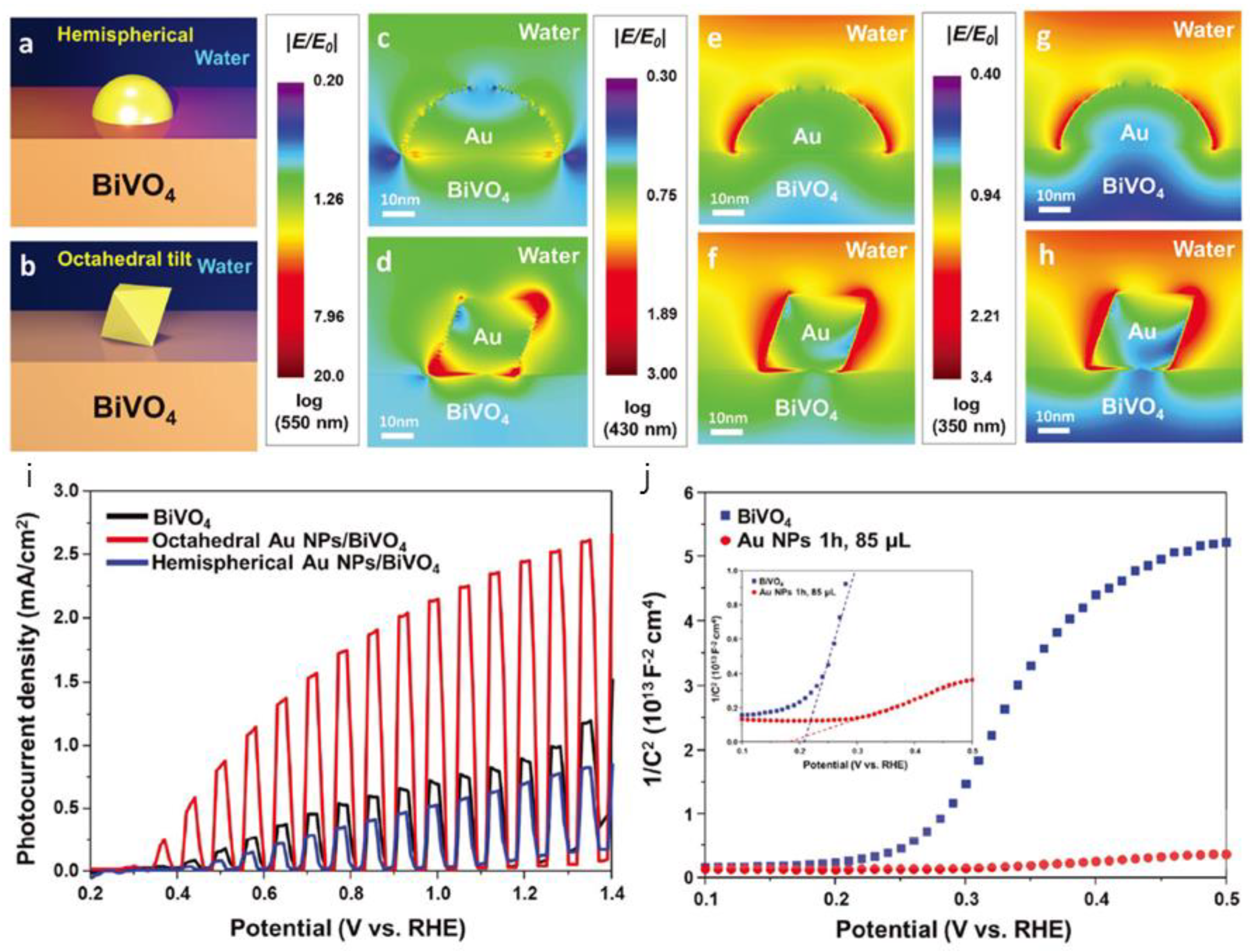
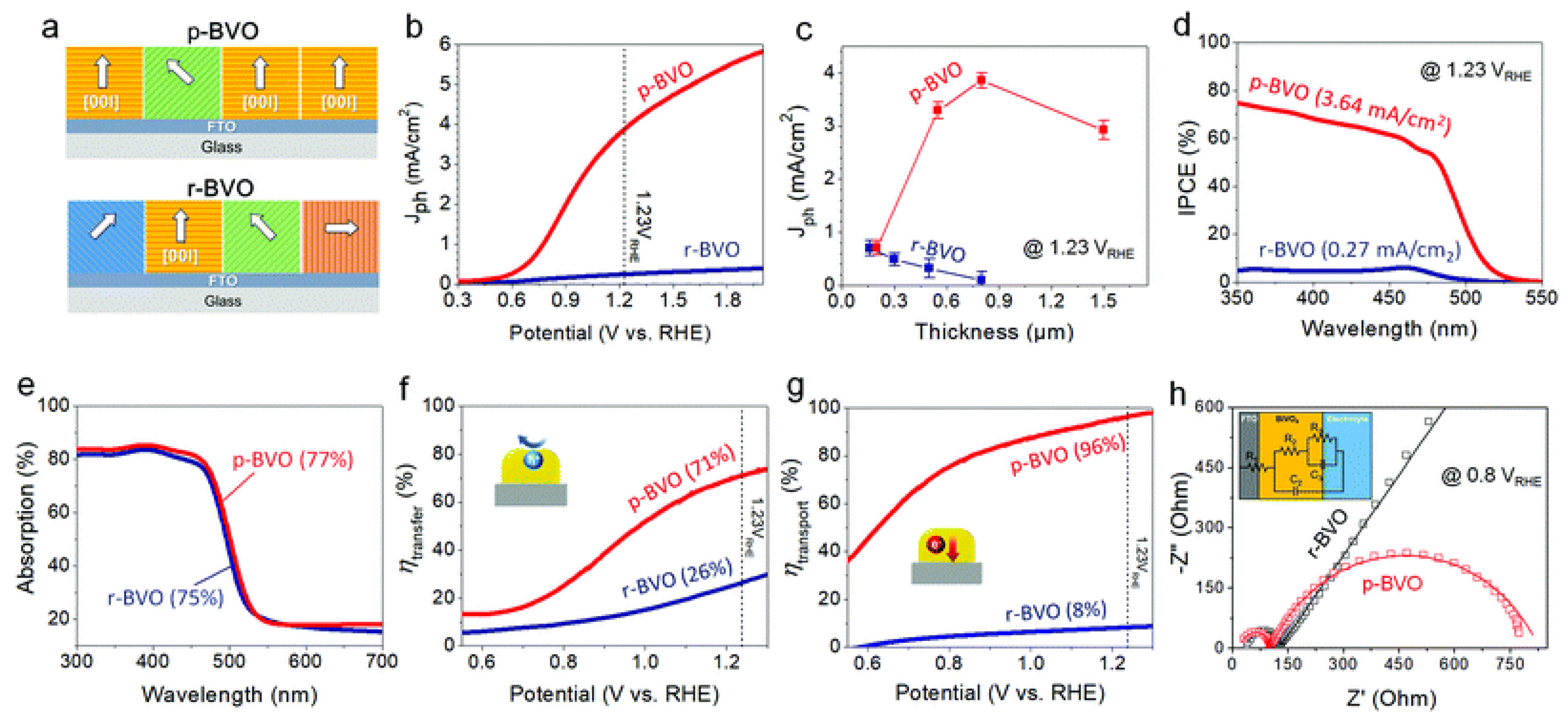
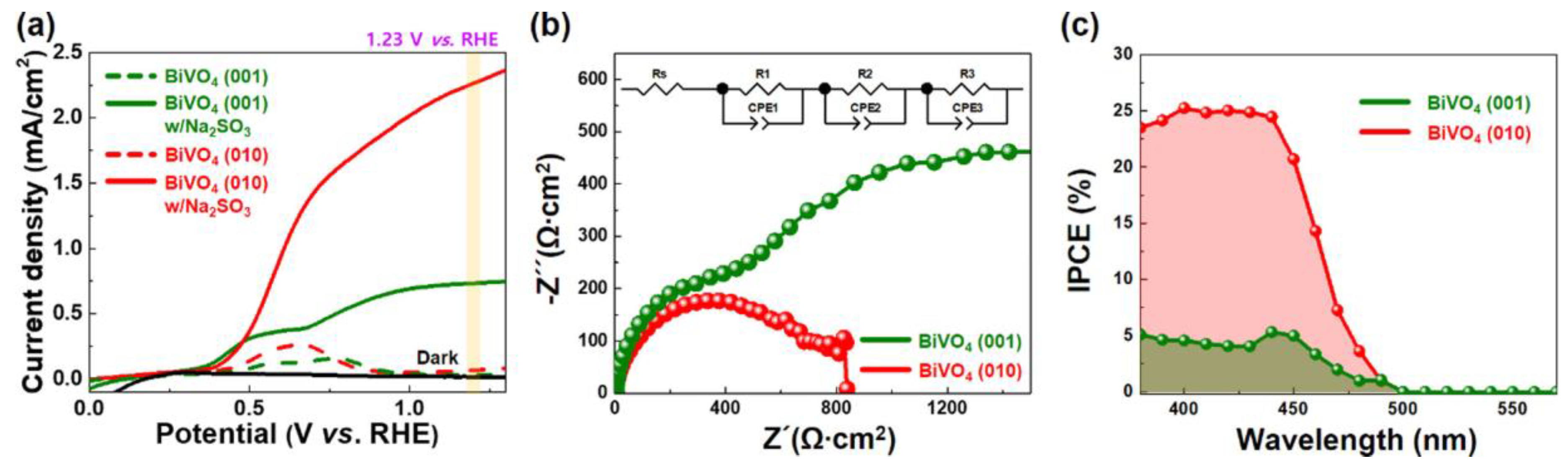
3.1. Application of Facet Engineering in PEC Water Splitting
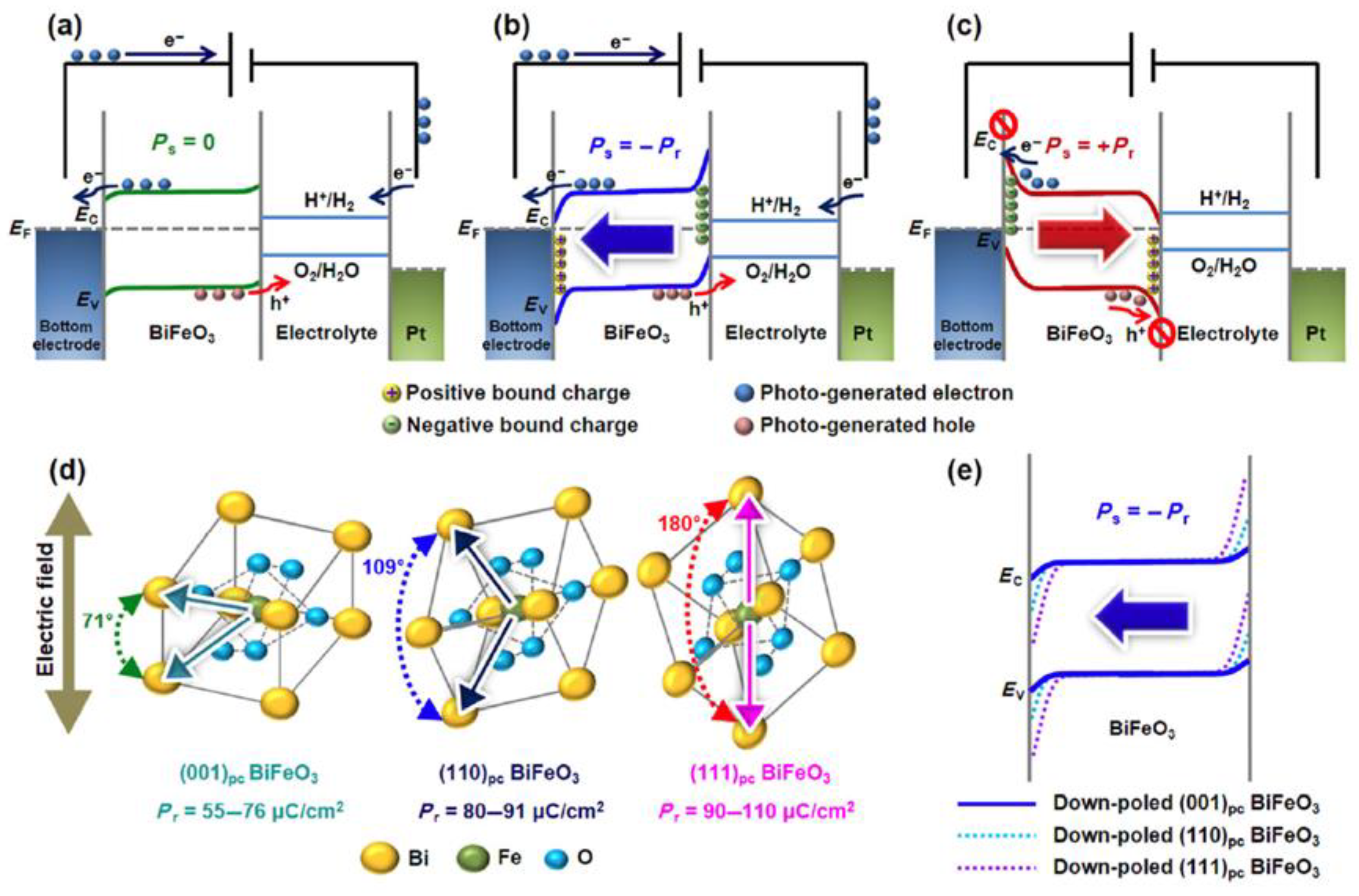
3.2. Development of Ferroelectric Coupled PEC Device
4. Conclusions and Future Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Turner, J.A. Sustainable hydrogen production. Science 2004, 305, 972–974. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Moniz, S.J.; Wang, A.; Zhang, T.; Tang, J. Photoelectrochemical devices for solar water splitting–materials and challenges. Chem. Soc. Rev. 2017, 46, 4645–4660. [Google Scholar] [CrossRef] [PubMed]
- Ong, W.-J.; Tan, L.-L.; Ng, Y.H.; Yong, S.-T.; Chai, S.-P. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: Are we a step closer to achieving sustainability? Chem. Rev. 2016, 116, 7159–7329. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Abdi, F.F.; Firet, N.; van de Krol, R. Efficient BiVO4 thin film photoanodes modified with cobalt phosphate catalyst and W-doping. ChemCatChem 2013, 5, 490–496. [Google Scholar] [CrossRef]
- Abdi, F.F.; Han, L.; Smets, A.H.; Zeman, M.; Dam, B.; Van De Krol, R. Efficient solar water splitting by enhanced charge separation in a bismuth vanadate-silicon tandem photoelectrode. Nat. Commun. 2013, 4, 2195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, W.; Guo, Y.; Guo, B.; Li, H.; Liu, H.; Joel, T.W. Enhanced photovoltaic effect in BiVO4 semiconductor by incorporation with an ultrathin BiFeO3 ferroelectric layer. ACS Appl. Mater. Interfaces 2013, 5, 6925–6929. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Choi, K.-S. Nanoporous BiVO4 photoanodes with dual-layer oxygen evolution catalysts for solar water splitting. Science 2014, 343, 1245026. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Herrmann, L.O.; Baumberg, J.J. Size dependent plasmonic effect on BiVO4 photoanodes for solar water splitting. Sci. Rep. 2015, 5, 16660. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.Y.; Choi, K.S.; Shin, H.-M.; Kim, T.L.; Song, J.; Yoon, S.; Jang, H.W.; Yoon, M.-H.; Jeon, C.; Lee, J. Enhanced photocatalytic performance depending on morphology of bismuth vanadate thin film synthesized by pulsed laser deposition. ACS Appl. Mater. Interfaces 2016, 9, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Grigioni, I.; Stamplecoskie, K.G.; Jara, D.H.; Dozzi, M.V.; Oriana, A.; Cerullo, G.; Kamat, P.V.; Selli, E. Wavelength-dependent ultrafast charge carrier separation in the WO3/BiVO4 coupled system. ACS Energy Lett. 2017, 2, 1362–1367. [Google Scholar] [CrossRef]
- Song, H.; Li, C.; Van, C.N.; Dong, W.; Qi, R.; Zhang, Y.; Huang, R.; Chu, Y.-H.; Duan, C.-G. Role of indium tin oxide electrode on the microstructure of self-assembled WO3-BiVO4 hetero nanostructures. J. Appl. Phys. 2017, 122, 175301. [Google Scholar] [CrossRef]
- Song, J.; Cha, J.; Lee, M.G.; Jeong, H.W.; Seo, S.; Yoo, J.A.; Kim, T.L.; Lee, J.; No, H.; Jeong, S.Y. Template-engineered epitaxial BiVO4 photoanodes for efficient solar water splitting. J. Mater. Chem. A 2017, 5, 18831–18838. [Google Scholar] [CrossRef]
- Wang, S.; Chen, P.; Yun, J.H.; Hu, Y.; Wang, L. An electrochemically treated BiVO4 photoanode for efficient photoelectrochemical water splitting. Angew. Chem. Int. Ed. 2017, 56, 8500–8504. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Shin, S.; Kim, D.H.; Park, I.J.; Kim, J.S.; Huang, P.-S.; Lee, J.-K.; Cho, I.S.; Zheng, X. Boosting the solar water oxidation performance of a BiVO4 photoanode by crystallographic orientation control. Energy Environ. Sci. 2018, 11, 1299–1306. [Google Scholar] [CrossRef]
- Jeong, S.Y.; Shin, H.-M.; Jo, Y.-R.; Kim, Y.J.; Kim, S.; Lee, W.-J.; Lee, G.J.; Song, J.; Moon, B.J.; Seo, S. Plasmonic silver nanoparticle-impregnated nanocomposite BiVO4 photoanode for plasmon-enhanced photocatalytic water splitting. J. Phys. Chem. C 2018, 122, 7088–7093. [Google Scholar] [CrossRef]
- Kim, S.; Yu, Y.; Jeong, S.Y.; Lee, M.G.; Jeong, H.W.; Kwon, Y.M.; Baik, J.M.; Park, H.; Jang, H.W.; Lee, S. Plasmonic gold nanoparticles-decorated BiVO4/ZnO nanowire heterostructure photoanodes for efficient water oxidation. Catal. Sci. Technol. 2018. [Google Scholar] [CrossRef]
- Li, J.; Guo, L.; Zhou, J.; Song, Q.; Liang, Z. Enhancing the photoelectrochemical performance of BiVO4 by decorating only its (040) facet with self-assembled Ag@AgCl QDs. J. Solid State Electron. 2018, 1–10. [Google Scholar] [CrossRef]
- Song, J.; Seo, M.J.; Lee, T.H.; Jo, Y.-R.; Lee, J.; Kim, T.L.; Kim, S.-Y.; Kim, S.-M.; Jeong, S.Y.; An, H. Tailoring crystallographic orientations to substantially enhance charge separation efficiency in anisotropic BiVO4 photoanodes. ACS Catal. 2018, 8, 5952–5962. [Google Scholar] [CrossRef]
- Zhou, C.; Wang, S.; Zhao, Z.; Shi, Z.; Yan, S.; Zou, Z. A facet-dependent schottky-junction electron shuttle in a BiVO4 {010}–Au–Cu2O Z-scheme photocatalyst for efficient charge separation. Adv. Funct. Mater. 2018, 28, 1801214. [Google Scholar] [CrossRef]
- Park, Y.; McDonald, K.J.; Choi, K.-S. Progress in bismuth vanadate photoanodes for use in solar water oxidation. Chem. Soc. Rev. 2013, 42, 2321–2337. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.L.; Choi, M.-J.; Jang, H.W. Boosting interfacial charge transfer for efficient water-splitting photoelectrodes: Progress in bismuth vanadate photoanodes using various strategies. MRS Commun. 2018, 1–14. [Google Scholar] [CrossRef]
- Lee, M.G.; Moon, C.W.; Park, H.; Sohn, W.; Kang, S.B.; Lee, S.; Choi, K.J.; Jang, H.W. Dominance of plasmonic resonant energy transfer over direct electron transfer in substantially enhanced water oxidation activity of BiVO4 by shape-controlled Au nanoparticles. Small 2017, 13, 1701644. [Google Scholar] [CrossRef] [PubMed]
- Abdi, F.F.; Starr, D.E.; Ahmet, I.Y.; van de Krol, R. Photocurrent enhancement by spontaneous formation of a p-n junction in Ca-doped bismuth vanadate photoelectrodes. ChemPlusChem 2018. [Google Scholar] [CrossRef]
- Pendlebury, S.R.; Barroso, M.; Cowan, A.J.; Sivula, K.; Tang, J.; Grätzel, M.; Klug, D.; Durrant, J.R. Dynamics of photogenerated holes in nanocrystalline α-Fe2O3 electrodes for water oxidation probed by transient absorption spectroscopy. Chem. Commun. 2011, 47, 716–718. [Google Scholar] [CrossRef] [PubMed]
- Zhong, D.K.; Cornuz, M.; Sivula, K.; Grätzel, M.; Gamelin, D.R. Photo-assisted electrodeposition of cobalt–phosphate (Co–Pi) catalyst on hematite photoanodes for solar water oxidation. Energy Environ. Sci. 2011, 4, 1759–1764. [Google Scholar] [CrossRef]
- Klahr, B.; Gimenez, S.; Fabregat-Santiago, F.; Bisquert, J.; Hamann, T.W. Photoelectrochemical and impedance spectroscopic investigation of water oxidation with “Co–Pi”-coated hematite electrodes. J. Am. Chem. Soc. 2012, 134, 16693–16700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grave, D.A.; Klotz, D.; Kay, A.; Dotan, H.; Gupta, B.; Visoly-Fisher, I.; Rothschild, A. Effect of orientation on bulk and surface properties of Sn-doped hematite (α-Fe2O3) heteroepitaxial thin film photoanodes. J. Phys. Chem. C 2016, 120, 28961–28970. [Google Scholar] [CrossRef]
- Hajibabaei, H.; Gao, Y.; Hamann, T.W. Advances in Photoelectrochemical Water Splitting: Theory, Experiment and Systems Analysis (Energy and Environment Series), 1st ed.; Royal Society of Chemistry: Cambridge, UK, 2018; pp. 100–127. ISBN 1782629254. [Google Scholar]
- Li, W.; Yang, K.R.; Yao, X.; He, Y.; Dong, Q.; Brudvig, G.W.; Batista, V.S.; Wang, D. Facet-dependent kinetics and energetics of hematite for solar water oxidation reactions. ACS Appl. Mater. Interfaces 2018. [Google Scholar] [CrossRef] [PubMed]
- Grave, D.A.; Yatom, N.; Ellis, D.S.; Toroker, M.C.; Rothschild, A. The “rust” challenge: On the correlations between electronic structure, excited state dynamics, and photoelectrochemical performance of hematite photoanodes for solar water splitting. Adv. Mater. 2018, 1706577. [Google Scholar] [CrossRef] [PubMed]
- Archana, P.S.; Pachauri, N.; Shan, Z.; Pan, S.; Gupta, A. Plasmonic enhancement of photoactivity by gold nanoparticles embedded in hematite films. J. Phys. Chem. C 2015, 119, 15506–15516. [Google Scholar] [CrossRef]
- Quynh, L.T.; Van, C.N.; Bitla, Y.; Chen, J.W.; Do, T.H.; Tzeng, W.Y.; Liao, S.C.; Tsai, K.A.; Chen, Y.C.; Wu, C.L. Self-assembled BiFeO3-ε-Fe2O3 vertical heteroepitaxy for visible light photoelectrochemistry. Adv. Energy Mater. 2016, 6, 1600686. [Google Scholar] [CrossRef]
- Xie, J.; Guo, C.; Yang, P.; Wang, X.; Liu, D.; Li, C.M. Bi-functional ferroelectric BiFeO3 passivated BiVO4 photoanode for efficient and stable solar water oxidation. Nano Energy 2017, 31, 28–36. [Google Scholar] [CrossRef]
- Song, J.; Kim, T.L.; Lee, J.; Cho, S.Y.; Cha, J.; Jeong, S.Y.; An, H.; Kim, W.S.; Jung, Y.-S.; Park, J. Domain-engineered BiFeO3 thin-film photoanodes for highly enhanced ferroelectric solar water splitting. Nano Res. 2018, 11, 642–655. [Google Scholar] [CrossRef]
- Linic, S.; Christopher, P.; Ingram, D.B. Plasmonic-metal nanostructures for efficient conversion of solar to chemical energy. Nat. Mater. 2011, 10, 911–921. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, Y.L.; Liu, R.-S.; Tsai, D.P. Plasmonic photocatalysis. Rep. Prog. Phys. 2013, 76, 046401. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, W. Absorption of colloidal silver in KCl. Z. Phys. 1968, 215, 113–120. [Google Scholar] [CrossRef]
- Russell, B.K.; Mantovani, J.; Anderson, V.; Warmack, R.; Ferrell, T. Experimental test of the mie theory for microlithographically produced silver spheres. Phys. Rev. B 1987, 35, 2151. [Google Scholar] [CrossRef]
- Mock, J.J.; Smith, D.R.; Schultz, S. Local refractive index dependence of plasmon resonance spectra from individual nanoparticles. Nano Lett. 2003, 3, 485–491. [Google Scholar] [CrossRef]
- Park, J.; Kim, H.J.; Nam, S.; Kim, H.; Choi, H.-J.; Jang, Y.J.; Lee, J.S.; Shin, J.; Lee, H.; Baik, J.M. Two-dimensional metal-dielectric hybrid-structured film with titanium oxide for enhanced visible light absorption and photo-catalytic application. Nano Energy 2016, 21, 115–122. [Google Scholar] [CrossRef]
- Li, H.; Li, Z.; Yu, Y.; Ma, Y.; Yang, W.; Wang, F.; Yin, X.; Wang, X. Surface-plasmon-resonance-enhanced photoelectrochemical water splitting from au-nanoparticle-decorated 3D TiO2 nanorod architectures. J. Phys. Chem. C 2017, 121, 12071–12079. [Google Scholar] [CrossRef]
- Awazu, K.; Fujimaki, M.; Rockstuhl, C.; Tominaga, J.; Murakami, H.; Ohki, Y.; Yoshida, N.; Watanabe, T. A plasmonic photocatalyst consisting of silver nanoparticles embedded in titanium dioxide. J. Am. Chem. Soc. 2008, 130, 1676–1680. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, P.-C.; Liu, M.; Fu, X.; Gordiichuk, P.; Yu, Y.; Wolverton, C.; Kang, Y.; Mirkin, C.A. Catalyst design by scanning probe block copolymer lithography. Proc. Natl. Acad. Sci. USA 2018, 115, 3764–3769. [Google Scholar] [CrossRef] [PubMed]
- Low, J.; Yu, J.; Jaroniec, M.; Wageh, S.; Al-Ghamdi, A.A. Heterojunction photocatalysts. Adv. Mater. 2017, 29, 1601694. [Google Scholar] [CrossRef] [PubMed]
- Hurst, J.K. In pursuit of water oxidation catalysts for solar fuel production. Science 2010, 328, 315–316. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Wang, L.-W. Thermodynamic oxidation and reduction potentials of photocatalytic semiconductors in aqueous solution. Chem. Mater. 2012, 24, 3659–3666. [Google Scholar] [CrossRef]
- Abdi, F.F.; van de Krol, R. Nature and light dependence of bulk recombination in Co-Pi-catalyzed BiVO4 photoanodes. J. Phys. Chem. C 2012, 116, 9398–9404. [Google Scholar] [CrossRef]
- Tang, F.; Cheng, W.; Su, H.; Zhao, X.; Liu, Q. Smoothing surface trapping states in 3D coral-like coooh-wrapped-BiVO4 for efficient photoelectrochemical water oxidation. ACS Appl. Mater. Interfaces 2018, 10, 6228–6234. [Google Scholar] [CrossRef] [PubMed]
- Chrisey, D.B.; Hubler, G.K. Pulsed Laser Deposition of Thin Films, 1st ed.; Wiley: New York, NY, USA, 1994; ISBN 0471592188. [Google Scholar]
- Ashfold, M.N.; Claeyssens, F.; Fuge, G.M.; Henley, S.J. Pulsed laser ablation and deposition of thin films. Chem. Soc. Rev. 2004, 33, 23–31. [Google Scholar] [CrossRef] [PubMed]
- MacManus-Driscoll, J.L. Self-assembled heteroepitaxial oxide nanocomposite thin film structures: Designing interface-induced functionality in electronic materials. Adv. Funct. Mater. 2010, 20, 2035–2045. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, Z.; Zou, Z. Electronic structure and optical properties of monoclinic clinobisvanite BiVO4. Phys. Chem. Chem. Phys. 2011, 13, 4746–4753. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhou, X.; Dupuis, M.; Li, C. The nature of photogenerated charge separation among different crystal facets of BiVO4 studied by density functional theory. Phys. Chem. Chem. Phys. 2015, 17, 23503–23510. [Google Scholar] [CrossRef] [PubMed]
- Shirai, K.; Fazio, G.; Sugimoto, T.; Selli, D.; Ferraro, L.; Watanabe, K.; Haruta, M.; Ohtani, B.; Kurata, H.; Di Valentin, C. Water-assisted hole trapping at the highly curved surface of nano-TiO2 photocatalyst. J. Am. Chem. Soc. 2018, 140, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Stoughton, S.; Showak, M.; Mao, Q.; Koirala, P.; Hillsberry, D.; Sallis, S.; Kourkoutis, L.; Nguyen, K.; Piper, L.; Tenne, D. Adsorption-controlled growth of BiVO4 by molecular-beam epitaxy. APL Mater. 2013, 1, 042112. [Google Scholar] [CrossRef]
- Rettie, A.J.; Mozaffari, S.; McDaniel, M.D.; Pearson, K.N.; Ekerdt, J.G.; Markert, J.T.; Mullins, C.B. Pulsed laser deposition of epitaxial and polycrystalline bismuth vanadate thin films. J. Phys. Chem. C 2014, 118, 26543–26550. [Google Scholar] [CrossRef]
- Chien, N.V.; Chang, W.S.; Chen, J.-W.; Tsai, K.-A.; Tzeng, W.-Y.; Lin, Y.-C.; Kuo, H.-H.; Liu, H.-J.; Chang, K.-D.; Chou, W.-C. Heteroepitaxial approach to explore charge dynamics across Au/BiVO4 interface for photoactivity enhancement. Nano Energy 2015, 15, 625–633. [Google Scholar] [CrossRef]
- Chien, N.V.; Do, T.H.; Chen, J.-W.; Tzeng, W.-Y.; Tsai, K.-A.; Song, H.; Liu, H.-J.; Lin, Y.-C.; Chen, Y.-C.; Wu, C.-L. WO3 mesocrystal-assisted photoelectrochemical activity of BiVO4. NPG Asia Mater. 2017, 9, e357. [Google Scholar] [CrossRef]
- Zhang, W.; Yan, D.; Tong, X.; Liu, M. Ultrathin lutetium oxide film as an epitaxial hole-blocking layer for crystalline bismuth vanadate water splitting photoanodes. Adv. Funct. Mater. 2018, 28, 1705512. [Google Scholar] [CrossRef]
- Zhang, W.; Yan, D.; Li, J.; Wu, Q.; Cen, J.; Zhang, L.; Orlov, A.; Xin, H.; Tao, J.; Liu, M. Anomalous conductivity tailored by domain-boundary transport in crystalline bismuth vanadate photoanodes. Chem. Mater. 2018, 30, 1677–1685. [Google Scholar] [CrossRef]
- Rettie, A.J.; Lee, H.C.; Marshall, L.G.; Lin, J.-F.; Capan, C.; Lindemuth, J.; McCloy, J.S.; Zhou, J.; Bard, A.J.; Mullins, C.B. Combined charge carrier transport and photoelectrochemical characterization of BiVO4 single crystals: Intrinsic behavior of a complex metal oxide. J. Am. Chem. Soc. 2013, 135, 11389–11396. [Google Scholar] [CrossRef] [PubMed]
- Rettie, A.J.; Chemelewski, W.D.; Lindemuth, J.; McCloy, J.S.; Marshall, L.G.; Zhou, J.; Emin, D.; Mullins, C.B. Anisotropic small-polaron hopping in W:BiVO4 single crystals. Appl. Phys. Lett. 2015, 106, 022106. [Google Scholar] [CrossRef]
- Pintilie, L.; Alexe, M. Metal-ferroelectric-metal heterostructures with schottky contacts. I. Influence of the ferroelectric properties. J. Appl. Phys. 2005, 98, 124103. [Google Scholar] [CrossRef]
- Pantel, D.; Chu, Y.-H.; Martin, L.W.; Ramesh, R.; Hesse, D.; Alexe, M. Switching kinetics in epitaxial BiFeO3 thin films. J. Appl. Phys. 2010, 107, 084111. [Google Scholar] [CrossRef]
- Hauser, A.; Zhang, J.; Mier, L.; Ricciardo, R.; Woodward, P.; Gustafson, T.; Brillson, L.; Yang, F. Characterization of electronic structure and defect states of thin epitaxial BiFeO3 films by UV-visible absorption and cathodoluminescence spectroscopies. Appl. Phys. Lett. 2008, 92, 222901. [Google Scholar] [CrossRef]
- Yi, H.; Choi, T.; Choi, S.; Oh, Y.S.; Cheong, S.W. Mechanism of the switchable photovoltaic effect in ferroelectric BiFeO3. Adv. Mater. 2011, 23, 3403–3407. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhou, Y.; You, L.; Wang, J.; Shen, M.; Fang, L. Enhanced ferroelectric photoelectrochemical properties of polycrystalline BiFeO3 film by decorating with ag nanoparticles. Appl. Phys. Lett. 2016, 108, 022902. [Google Scholar] [CrossRef]
- Ji, W.; Yao, K.; Lim, Y.-F.; Liang, Y.C.; Suwardi, A. Epitaxial ferroelectric BiFeO3 thin films for unassisted photocatalytic water splitting. Appl. Phys. Lett. 2013, 103, 062901. [Google Scholar] [CrossRef]
- Rong, N.; Chu, M.; Tang, Y.; Zhang, C.; Cui, X.; He, H.; Zhang, Y.; Xiao, P. Improved photoelectrocatalytic properties of Ti-doped BiFeO3 films for water oxidation. J. Mater. Sci. 2016, 51, 5712–5723. [Google Scholar] [CrossRef]
- Moniz, S.J.; Quesada-Cabrera, R.; Blackman, C.S.; Tang, J.; Southern, P.; Weaver, P.M.; Carmalt, C.J. A simple, low-cost CVD route to thin films of BiFeO3 for efficient water photo-oxidation. J. Mater. Chem. A 2014, 2, 2922–2927. [Google Scholar] [CrossRef]
- Cao, D.; Wang, Z.; Wen, L.; Mi, Y.; Lei, Y. Switchable charge-transfer in the photoelectrochemical energy-conversion process of ferroelectric BiFeO3 photoelectrodes. Angew. Chem. Int. Ed. 2014, 53, 11027–11031. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-L.; Chang, W.S.; Chien, N.V.; Liu, H.-J.; Tsai, K.-A.; Chen, J.-W.; Kuo, H.-H.; Tzeng, W.-Y.; Chen, Y.-C.; Wu, C.-L. Tunable photoelectrochemical performance of Au/BiFeO3 heterostructure. Nanoscale 2016, 8, 15795–15801. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wang, J. Orientation dependence of ferroelectric behavior of BiFeO3 thin films. J. Appl. Phys. 2009, 106, 104111. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Wuttig, M.; Ramesh, R.; Wang, N.; Ruette, B.; Pyatakov, A.P.; Zvezdin, A.; Viehland, D. Dramatically enhanced polarization in (001), (101), and (111) BiFeO3 thin films due to epitiaxial-induced transitions. Appl. Phys. Lett. 2004, 84, 5261–5263. [Google Scholar] [CrossRef]
- Sone, K.; Naganuma, H.; Miyazaki, T.; Nakajima, T.; Okamura, S. Crystal structures and electrical properties of epitaxial BiFeO3 thin films with (001), (110), and (111) orientations. Jpn. J. Appl. Phys. 2010, 49, 09MB03. [Google Scholar] [CrossRef]
- Baek, S.H.; Folkman, C.M.; Park, J.W.; Lee, S.; Bark, C.W.; Tybell, T.; Eom, C.B. The nature of polarization fatigue in BiFeO3. Adv. Mater. 2011, 23, 1621–1625. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Kim, D.; Baek, S.; Eom, C.; Zavaliche, F.; Yang, S.; Ramesh, R.; Chen, Y.; Pan, X.; Ke, X. Synthesis and ferroelectric properties of epitaxial BiFeO3 thin films grown by sputtering. Appl. Phys. Lett. 2006, 88, 242904. [Google Scholar] [CrossRef]
- Yang, W.; Yu, Y.; Starr, M.B.; Yin, X.; Li, Z.; Kvit, A.; Wang, S.; Zhao, P.; Wang, X. Ferroelectric polarization-enhanced photoelectrochemical water splitting in TiO2–BaTiO3 core–shell nanowire photoanodes. Nano Lett. 2015, 15, 7574–7580. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, S.Y.; Song, J.; Lee, S. Photoelectrochemical Device Designs toward Practical Solar Water Splitting: A Review on the Recent Progress of BiVO4 and BiFeO3 Photoanodes. Appl. Sci. 2018, 8, 1388. https://doi.org/10.3390/app8081388
Jeong SY, Song J, Lee S. Photoelectrochemical Device Designs toward Practical Solar Water Splitting: A Review on the Recent Progress of BiVO4 and BiFeO3 Photoanodes. Applied Sciences. 2018; 8(8):1388. https://doi.org/10.3390/app8081388
Chicago/Turabian StyleJeong, Sang Yun, Jaesun Song, and Sanghan Lee. 2018. "Photoelectrochemical Device Designs toward Practical Solar Water Splitting: A Review on the Recent Progress of BiVO4 and BiFeO3 Photoanodes" Applied Sciences 8, no. 8: 1388. https://doi.org/10.3390/app8081388




