Influence of Wood Biochar on Phenanthrene Catabolism in Soils
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Soil Preparation for Aging and Spiking
| Soil Property | Myerscough | Kettering |
|---|---|---|
| pH | 6.53 | 7.4 |
| Moisture content (%) | 21.07 | 30.0 |
| Water holding capacity (%) | 35.02 | 40.3 |
| Clay (%) | 19.50 | 23.0 |
| Silt (%) | 20.00 | 35.0 |
| Sand (%) | 60.40 | 42.0 |
| Total organic matter (%) | 2.70 | 5.0 |
| Temperature (°C) | Particle Size (mm) | pH | Ash Content (%) | Pore Volume (mL g−1) | Liquid Quantity (μL g−1) |
|---|---|---|---|---|---|
| 450–500 | ≤2 | 9.6 | 13.7 | 1.39 | 44 |
| 450–500 | 3–7 | 9.6 | 14.4 | 2.20 | 60 |
2.3. Measuring 14C-Phenanthrene Mineralisation by Indigenous Soil Microorganisms
2.4. Statistical Analyses
3. Results
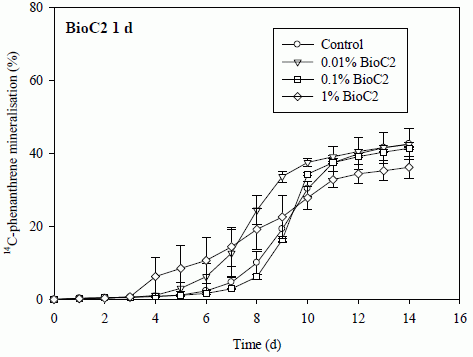
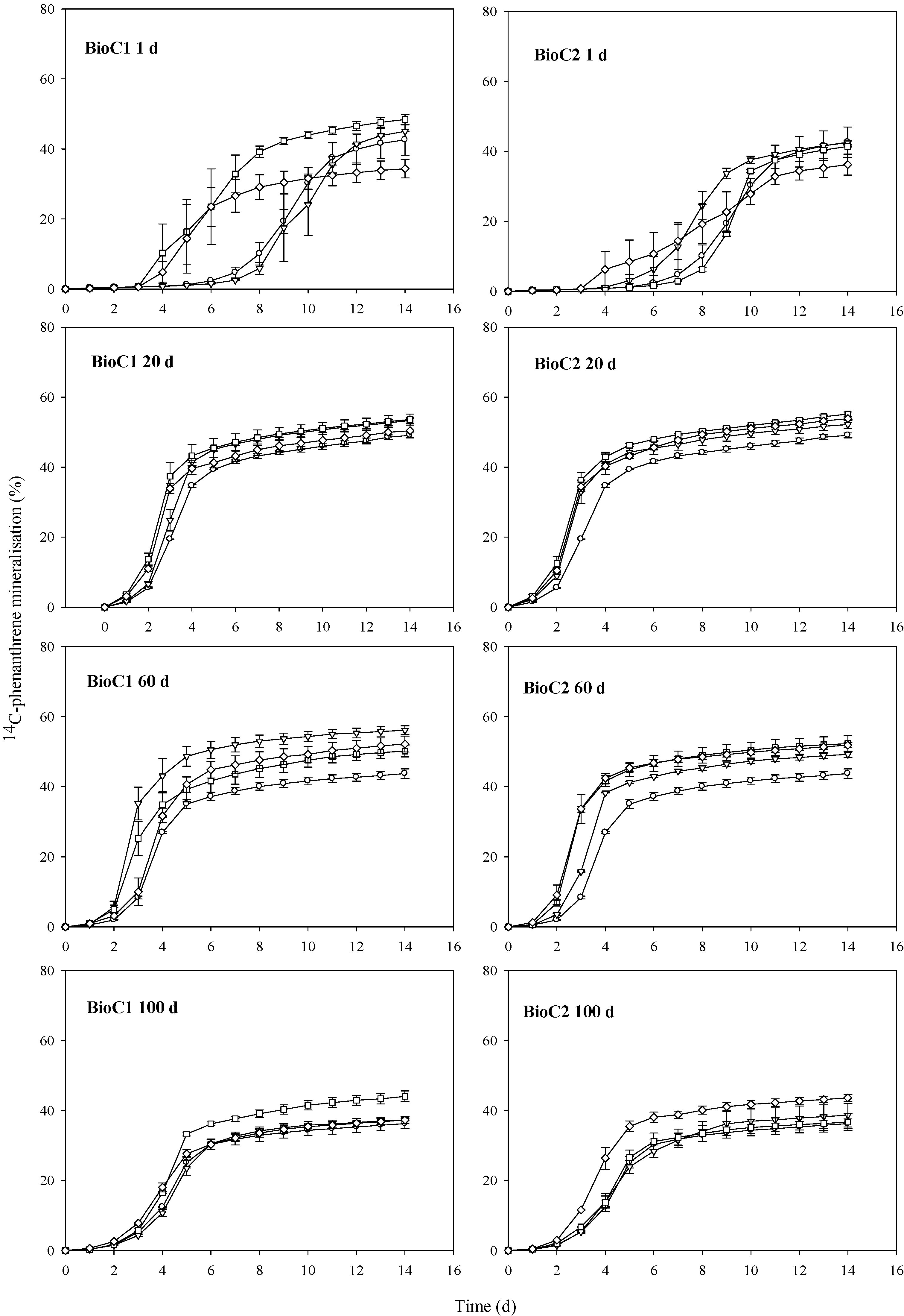
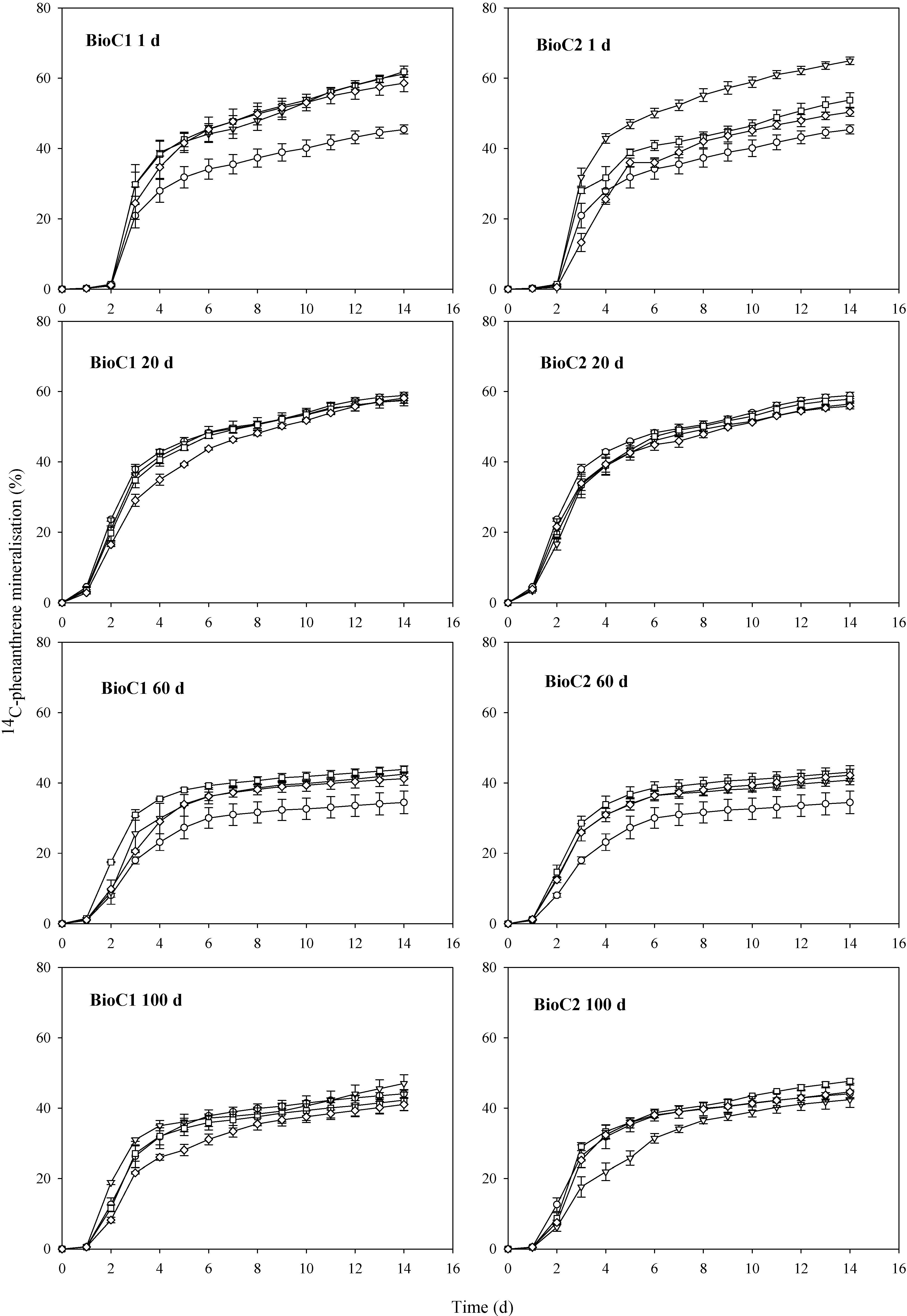
3.1. Effects of Biochar Amendment on Mineralisation of 14C-Phenanthrene
3.1.1. Impact of Biochar on the Lag Phase Prior to Mineralisation of 14C-Phenanthrene in Soil
| Aging Period (d) | Soil | Amendment (%) | Lag Phase (d) | Maximum Rates (% d−1) | Total Extent(%) |
|---|---|---|---|---|---|
| 1 | Myerscough | 0 | 7.09 ± 1.86 aA | 17.69 ± 0.98 aA | 42.59 ± 4.30 aA |
| 0.01 | 7.80 ± 1.29 aA | 25.09 ± 3.03 aA | 45.07 ± 0.03 aB | ||
| 0.1 | 5.48 ± 0.87 aA | 26.04 ± 3.10 aA | 48.44 ± 1.47 aB | ||
| 1 | 4.94 ± 0.64 aA | 29.67 ± 3.13 aA | 34.34 ± 2.62 aB | ||
| Kettering | 0 | 3.31 ± 0.10 aB | 20.00 ± 1.95 aA | 45.39 ± 1.28 bA | |
| 0.01 | 3.15 ± 0.03 aB | 32.73 ± 4.08 aA | 61.16 ± 0.89 aA | ||
| 0.1 | 3.13 ± 0.03 aA | 28.62 ± 3.79 aA | 61.86 ± 1.57 aA | ||
| 1 | 3.18 ± 0.04 aA | 26.76 ± 3.72 aA | 58.58 ± 2.43 aA | ||
| 20 | Myerscough | 0 | 2.93 ± 0.08 aA | 15.29 ± 0.62 aA | 49.08 ± 0.70 aB |
| 0.01 | 2.69 ± 0.09 aA | 20.33 ± 1.80 aA | 53.31 ± 0.80 aA | ||
| 0.1 | 2.21 ± 0.07 bB | 21.77 ± 1.50 aA | 53.58 ± 1.59 aA | ||
| 1 | 2.36 ± 0.06 bB | 21.68 ± 0.63 aA | 50.40 ± 2.01 aB | ||
| Kettering | 0 | 2.03 ± 0.01 aB | 19.15 ± 0.73 aA | 58.92 ± 0.90 aA | |
| 0.01 | 2.08 ± 0.06 aB | 16.87 ± 2.66 aA | 57.45 ± 0.80 aA | ||
| 0.1 | 2.10 ± 0.02 aA | 16.28 ± 1.92 aA | 57.64 ± 1.68 aA | ||
| 1 | 2.66 ± 0.41 aA | 13.65 ± 0.92 aB | 58.14 ± 0.71 aA | ||
| 60 | Myerscough | 0 | 3.56 ± 0.10 aA | 16.40 ± 2.10 bA | 43.69 ± 1.36 aA |
| 0.01 | 3.83 ± 0.16 aA | 29.50 ± 3.09 aA | 56.08 ± 1.26 aA | ||
| 0.1 | 2.96 ± 0.07 aA | 29.02 ± 2.72 aA | 50.15 ± 1.70 aA | ||
| 1 | 3.18 ± 0.07 aA | 21.61 ± 1.87 bA | 52.16 ± 2.28 aA | ||
| 60 | Kettering | 0 | 2.62 ± 0.05 aB | 10.09 ± 0.39 aA | 34.46 ± 3.21 aA |
| 0.01 | 2.52 ± 0.25 aA | 15.26 ± 1.72 aB | 42.49 ± 0.72 aB | ||
| 0.1 | 2.50 ± 0.28 aA | 12.27 ± 1.62 aB | 43.85 ± 1.03 aA | ||
| 1 | 2.48 ± 0.04 aB | 10.66 ± 0.30 aB | 41.23 ± 1.87 aB | ||
| 100 | Myerscough | 0 | 3.92 ± 0.02 aA | 12.79 ± 1.46 aA | 36.26 ± 1.40 aA |
| 0.01 | 4.12 ± 0.07 aA | 12.79 ± 1.36 aA | 37.29 ± 1.07 aA | ||
| 0.1 | 3.78 ± 0.15 aA | 17.78 ± 1.03 aA | 44.08 ± 1.52 aA | ||
| 1 | 3.46 ± 0.04 aA | 10.23 ± 1.01 aA | 37.29 ± 0.83 aA | ||
| Kettering | 0 | 2.38 ± 0.06 aB | 13.70 ± 1.27 aA | 44.07 ± 1.08 aA | |
| 0.01 | 2.26 ± 0.01 aB | 17.02 ± 1.23 aA | 47.02 ± 2.46 aA | ||
| 0.1 | 2.41 ± 0.04 aB | 15.55 ± 3.14 aA | 42.26 ± 2.99 aA | ||
| 1 | 2.51 ± 0.07 aB | 10.76 ± 2.63 aA | 41.03 ± 1.71 aA |
| Aging Period (d) | Soil | Amendment (%) | Lag Phase (d) | Maximum Rates (% d−1) | Total Extent (%) |
|---|---|---|---|---|---|
| 1 | Myerscough | 0 | 7.09 ± 1.86 aA | 17.69 ± 0.98 aA | 42.59 ± 4.30 aA |
| 0.01 | 7.01 ± 0.70 aA | 18.95 ± 2.87 aB | 42.45 ± 0.50 aB | ||
| 0.1 | 8.66 ± 0.10 aA | 17.95 ± 0.91 aB | 41.39 ± 0.20 aB | ||
| 1 | 7.08 ± 0.02 aA | 15.49 ± 2.20 aB | 36.21 ± 3.00 aB | ||
| Kettering | 0 | 3.31 ± 0.10 aB | 20.00± 1.95 bA | 45.39 ± 1.28 bA | |
| 0.01 | 3.27 ± 0.17 aB | 30.75 ± 2.98 aA | 64.92 ± 1.07 aA | ||
| 0.1 | 3.18 ± 0.06 aB | 26.64 ± 0.70 aA | 53.76 ± 2.06 aA | ||
| 1 | 3.35 ± 0.55 aB | 14.72 ± 1.68 bA | 50.32 ± 1.20 bA | ||
| 20 | Myerscough | 0 | 2.93 ± 0.08 aA | 15.29 ± 0.62 aA | 49.08 ± 0.70 bB |
| 0.01 | 2.43 ± 0.09 bA | 24.10 ± 2.39 aA | 52.19 ± 1.08 aB | ||
| 0.1 | 2.24 ± 0.11 bA | 23.84 ± 1.22 aA | 55.17 ± 0.61 aA | ||
| 1 | 2.38 ± 0.12 bA | 22.21 ± 1.82 aA | 53.84 ± 0.19 aA | ||
| Kettering | 0 | 2.03 ± 0.08 bB | 19.15 ± 0.73 aA | 58.92 ± 0.90 aA | |
| 0.01 | 2.12 ± 0.01 aB | 17.62 ± 0.04 aB | 56.48 ± 0.88 aA | ||
| 0.1 | 2.09 ± 0.03 aA | 15.89 ± 1.11 aB | 57.83 ± 0.79 aA | ||
| 1 | 2.07 ± 0.01 aB | 13.39 ± 1.07 aB | 55.86 ± 0.78 aA | ||
| 60 | Myerscough | 0 | 3.56 ± 0.10 aA | 16.40 ± 2.10 bA | 43.69 ± 1.36 aA |
| 0.01 | 3.13 ± 0.02 aA | 22.23 ± 0.22 aA | 49.20 ± 0.87 aA | ||
| 0.1 | 2.54 ± 0.20 bA | 24.76 ± 1.91 aA | 52.26 ± 2.28 aA | ||
| 1 | 2.61 ± 0.16 bA | 24.46 ± 1.82 aA | 51.82 ± 0.73 aA | ||
| 60 | Kettering | 0 | 2.62 ± 0.05 aB | 10.09 ± 0.39 bA | 34.46 ± 3.21 aA |
| 0.01 | 2.40 ± 0.07 bB | 9.72 ± 0.55 bB | 40.82 ± 1.31 aB | ||
| 0.1 | 2.30 ± 0.05 bA | 13.90 ± 0.80 aB | 43.07 ± 1.85 aB | ||
| 1 | 2.34 ± 0.03 bA | 13.50 ± 0.62 aB | 42.23 ± 1.71 aB | ||
| 100 | Myerscough | 0 | 3.92 ± 0.02 aA | 12.79 ± 1.46 aA | 36.26 ± 1.40 aA |
| 0.01 | 3.90 ± 0.10 aA | 9.75 ± 1.21 aA | 38.69 ± 3.37 aA | ||
| 0.1 | 3.63 ± 0.03 bA | 12.79 ± 1.46 aB | 36.70 ± 2.45 aB | ||
| 1 | 3.25 ± 0.02 bA | 14.66 ± 2.15 aA | 43.61 ± 0.95 aA | ||
| Kettering | 0 | 2.38 ± 0.06 aB | 13.70 ± 1.27 aA | 44.07 ± 1.08 aA | |
| 0.01 | 2.62 ± 0.18 aB | 14.96 ± 3.51 aA | 42.40 ± 2.22 aA | ||
| 0.1 | 2.57 ± 0.11 aB | 20.33 ± 0.37 aA | 47.66 ± 0.61 aA | ||
| 1 | 2.55 ± 0.09 aB | 15.81 ± 1.49 aA | 44.60 ± 1.99 aA |
3.1.2. Impact of Biochar on the Maximum Rates of 14C-Phenanthrene Mineralisation in Soil
3.1.3. Impact of Biochar on the Total Extents of 14C-Phenanthrene Mineralisation
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Rhodes, A.H.; McAllister, L.E.; Chen, R.; Semple, K.T. Impact of activated charcoal on the mineralisation of 14C-phenanthrene in soils. Chemosphere 2010, 79, 463–469. [Google Scholar] [CrossRef]
- Semple, K.T.; Morriss, A.W.J.; Paton, G.I. Bioavailability of hydrophobic organic contaminants in soils: Fundamental concepts and techniques for analysis. Eur. J. Soil Sci. 2003, 564, 1–10. [Google Scholar]
- Aeolin, C.M.; Bradley, D.C.; Pfaender, F.K. Adaptation of aquifer microbial communities to the biodegradation of xenobiotic compounds: Influence of substrate concentration and pre-exposure. Environ. Tox. Chem. 1989, 8, 75–86. [Google Scholar] [CrossRef]
- Johnson, A.R.; Karlson, U. PAH degradation capacity of soil microbial communities—Does it depend on PAH exposure? Microbial. Ecol. 2005, 50, 488–495. [Google Scholar] [CrossRef]
- Macleod, C.J.; Semple, K.T. The adaptation of two similar soils to pyrene catabolism. Environ. Pollut. 2002, 119, 357–364. [Google Scholar] [CrossRef]
- Brandt, B.W.; Kelpin, F.D.; van Leeuwen, I.M.; Kooijman, S.A. Modelling microbial adaptation to changing availability of substrates. Water Res. 2004, 38, 1003–1013. [Google Scholar] [CrossRef]
- Tian, Y.; Zheng, T.; Wang, X. PAHs contamination and PAH-degrading bacteria in Xiamen Western Sea. Chem. Speciat. Bioavailab. 2002, 14, 25–33. [Google Scholar] [CrossRef]
- Semple, K.T.; Doick, K.J.; Wick, L.Y.; Harms, H. Microbial interactions with organic contaminants in soil: Definitions, processes and measurements. Environ. Pollut. 2007, 150, 166–176. [Google Scholar] [CrossRef]
- Couling, N.R.; Towell, M.G.; Semple, K.T. Reprinted from Biodegradation of PAHs in soil: Influence of chemical structure, concentration and multiple amendment. Environ. Pollut. 2010, 158, 3411–3420. [Google Scholar] [CrossRef]
- Lee, P.H.; Doik, K.J.; Semple, K.T. The development of phenanthrene catabolism in soil amended with transformer oil. FEMS Microbiol. Lett. 2003, 228, 217–223. [Google Scholar] [CrossRef]
- Gourlay, C.; Tusseau-Vuillemin, M.H.; Mouchel, J.M.; Garric, J. The ability of dissolved organic matter (DOM) to influence benzo[a]pyrene bioavailability increases with DOM biodegradation. Ecotoxicol. Environ. Saf. 2005, 61, 74–82. [Google Scholar] [CrossRef]
- Braida, W.J.; Pignatello, J.J.; Lu, Y.; Ravikovitch, P.I.; Neimark, A.V.; Xing, B. Sorption hysteresis of benzene in charcoal particles. Environ. Sci. Technol. 2003, 37, 409–417. [Google Scholar] [CrossRef]
- Song, J.; Peng, P. Characterisation of black carbon materials by pyrolysis-gas chromatography-mass spectrometry. J. Anal. Appl. Pyrolysis 2010, 87, 129–137. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L. Effect of biochar and greenwaste compost amendments on mobility, bioavailability and toxicity of inorganic and organic contaminants in a multi-element polluted soil. Environ. Pollut. 2010, 158, 2282–2287. [Google Scholar] [CrossRef]
- Accardi-Dey, A.; Gschwend, P.M. Assessing the combined roles of natural organic matter and black carbon as sorbents in sediments. Environ. Sci. Technol. 2002, 3, 21–29. [Google Scholar] [CrossRef]
- Cao, X.; Ma, L.; Gao, B.; Harris, W. Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ. Sci. Technol. 2009, 43, 3285–3291. [Google Scholar] [CrossRef]
- Chen, B.; Chen, Z. Sorption of naphthalene and 1-naphthol by biochar of orange peels with different pyrolytic temperatures. Chemosphere 2009, 76, 127–133. [Google Scholar] [CrossRef]
- Huang, W.; Peng, P.; Yu, Z.; Fu, J. Effects of organic matter heterogeneity on sorption and desorption of organic contaminants by soils and sediments. Appl. Geochem. 2003, 18, 955–972. [Google Scholar] [CrossRef]
- Kasozi, G.N.; Zimmerman, A.R.; Nkedi-Kizza, P.; Gao, B. Catechol and humic acid sorption onto a range of laboratory produced black carbons (biochars). Environ. Sci. Technol. 2010, 44, 6189–6195. [Google Scholar] [CrossRef]
- Cornelissen, G.; Gustafsson, O. Sorption of phenanthrene to environmental black carbon in sediment with and without organic matter and native sorbates. Environ. Sci. Technol. 2004, 38, 148–155. [Google Scholar] [CrossRef]
- Bushnaf, K.M.; Puricelli, S.; Saponaro, S.; Werner, D. Effect of biochar on the fate of volatile petroleum hydrocarbons in an aerobic sandy soil. J. Contam. Hydrol. 2011, 126, 208–215. [Google Scholar] [CrossRef]
- Boucard, T.K.; Bardgett, R.D.; Jones, K.C.; Semple, K.T. Reprinted from influence of plants on the chemical extractability and biodegradability of 2,4-dichlorophenol in soil. Environ. Pollut. 2005, 133, 53–62. [Google Scholar] [CrossRef]
- Doick, K.J.; Lee, P.H.; Semple, K.T. Assessment of spiking procedures for the introduction of a phenanthrene-LNAPL mixture into a field-wet soil. Environ. Pollut. 2003, 126, 399–406. [Google Scholar] [CrossRef]
- Webber, J.B.W.; Corbett, P.; Semple, K.T.; Ogbonnaya, U.; Teel, W.S.; Masiello, C.A.; Fisher, Q.J.; Valenza, J.J., II; Song, Y.Q.; Hu, Q. An NMR study of porous rock and biochar containing organic material. Micropor. Mesopor. Mat. 2013, 178, 94–98. [Google Scholar] [CrossRef]
- Novak, J.M.; Lima, I.; Xing, B.; Gaskin, J.W.; Steiner, C.; Das, K.C.; Ahmedna, M.; Rehrah, D.; Watts, D.W.; Busscher, W.J.; et al. Characterization of designer biochar produced at different temperatures and their effects on loamy sand. Ann. Environ. Sci. 2009, 3, 195–206. [Google Scholar]
- Cao, X.; Harris, W. Properties of dairy-manure derived biochar pertinent to its potential use in remediation. Bioresour. Technol. 2010, 100, 5222–5228. [Google Scholar]
- Reid, B.J.; MacLeod, C.J.A.; Lee, P.H.; Morriss, A.W.J.; Stokes, J.D.; Semple, K.T. A simple 14C-respirometric method for assessing microbial catabolic potential and contaminant bioavailability. FEMS Microbiol. Lett. 2001, 196, 141–146. [Google Scholar] [CrossRef]
- Doick, K.J.; Semple, K.T. The effect of soil: Water ratios on the mineralization of phenanthrene: LNAPL mixtures in soil. FEMS Microbiol. Lett. 2003, 220, 29–33. [Google Scholar] [CrossRef]
- Top, E.M.; Springael, D. The role of mobile genetic elements in bacterial adaptation to xenobiotic organic compounds. Curr. Opin. Biotechnol. 2003, 14, 262–269. [Google Scholar]
- Rhodes, A.H.; Hofman, J.; Semple, K.T. Development of phenanthrene catabolism in natural and artificial soils. Environ. Pollut. 2008, 152, 424–430. [Google Scholar] [CrossRef]
- Lehmann, J.; Rillig, M.C.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowley, D. Biochar effects on soil biota—A review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Gundale, M.J.; Deluca, T.H. Temperate and source material influence ecological attributes of ponderosa pine and Douglas-fir charcoal. For. Ecol. Manag. 2006, 231, 86–93. [Google Scholar]
- Kolb, S.E.; Fermanich, K.J.; Dornbush, M.E. Effect of charcoal quantity on microbial biomass and activity in temperate soils. Soil Sci. Soc. Am. J. 2009, 73, 1173–1181. [Google Scholar]
- Smith, J.L.; Collins, H.P.; Bailey, V.L. The effect of young biochar on soil respiration. Soil Biol. Biochem. 2010, 42, 2345–2347. [Google Scholar]
- Pietikäinen, J.; Kiikkila, O.; Fritze, H. Charcoal as a habitat for microbes and its effects on microbial community of the underlying humus. Oikos 2000, 89, 231–242. [Google Scholar]
- Demeyer, A.; Voundi Nkana, J.C.; Verloo, M.G. Characteristics of wood ash and influence on soil properties and nutrient uptake: An overview. Bioresour. Technol. 2001, 77, 287–295. [Google Scholar]
- Steinbeiss, S.; Gleixner, G.; Antonietti, M. Effect of biochar amendment on soil carbon balance and soil microbial activity. Soil Biol. Biochem. 2009, 41, 1301–1310. [Google Scholar] [CrossRef]
- Kogel-Knabner, I. The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil Biol. Biochem. 2002, 34, 139–162. [Google Scholar] [CrossRef]
- Luo, Y.; Durenkamp, M.; de Nobili, M.; Lin, Q.; Devonshire, B.J.; Brookes, P.C. Microbial biomass growth, following incorporation of biochars produced at 350 °C or 700 °C, in a silty-clay loam soil of high and low pH. Soil Biol. Biochem. 2013, 57, 513–523. [Google Scholar] [CrossRef]
- Thies, J.E.; Rillig, M.C. Characteristics of Biochar: Biological Properties. In Biochar for Environmental Management; Lehmann, J., Joseph, S., Eds.; Earthscan: Sterling, VA., USA, 2009; pp. 85–105. [Google Scholar]
- Steiner, C.; Das, K.C.; Garcia, M.; Forster, B.; Zech, W. Charcoal and smoke extract stimulate the soil microbial community in a highly weathered xanthic Ferralsol. Pedobiologia 2008, 51, 359–366. [Google Scholar]
- Pignatello, J.J.; Kwon, S.; Lu, Y. Effect of natural organic substances on the surface and adsorptive properties of environmental black carbon (char): Attenuation of surface activity by humic and fulvic acids. Environ. Sci. Technol. 2006, 40, 7757–7763. [Google Scholar] [CrossRef]
- Kwon, S.; Pignatello, J.J. Effect of natural organic substances on the surface and adsorptive properties of environmental black carbon (char): Pseudopore blockage by model lipid components and its implication for N2-probed surface properties of natural sorbents. Environ. Sci. Technol. 2005, 39, 7932–7939. [Google Scholar] [CrossRef]
- Lou, L.; Liu, F.; Yue, Q.; Chen, F.; Yang, Q.; Hu, B.; Chen, Y. Influence of humic acid on the sorption of pentachlorophenol by aged sediment amended with rice-straw biochar. Appl. Geochem. 2013, 33, 76–83. [Google Scholar]
- Harms, H.; Bosma, T.N.P. Mass transfer limitation of microbial growth and pollutant degradation. J. Ind. Microbiol. Biotechnol. 1997, 18, 97–105. [Google Scholar] [CrossRef]
- Rhodes, A.H.; Carlin, A.; Semple, K.T. Impact of black carbon in the extraction and mineralisation of phenanthrene in soil. Environ. Sci. Technol. 2008, 42, 740–745. [Google Scholar]
- Zhou, Z.; Sun, H.; Zhang, W. Desorption of polycyclic aromatic hydrocarbons from aged and unaged charcoals with and without modification of humic acids. Environ. Pollut. 2010, 158, 1916–1921. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ogbonnaya, U.; Oyelami, A.; Matthews, J.; Adebisi, O.; Semple, K.T. Influence of Wood Biochar on Phenanthrene Catabolism in Soils. Environments 2014, 1, 60-74. https://doi.org/10.3390/environments1010060
Ogbonnaya U, Oyelami A, Matthews J, Adebisi O, Semple KT. Influence of Wood Biochar on Phenanthrene Catabolism in Soils. Environments. 2014; 1(1):60-74. https://doi.org/10.3390/environments1010060
Chicago/Turabian StyleOgbonnaya, Uchenna, Ayodeji Oyelami, Justin Matthews, Olusoji Adebisi, and Kirk T. Semple. 2014. "Influence of Wood Biochar on Phenanthrene Catabolism in Soils" Environments 1, no. 1: 60-74. https://doi.org/10.3390/environments1010060