3.1. Field and Core Observations
Observations made at quarries and outcrops clearly indicate that the regional distribution of MVT mineralization extends throughout the study area northward into the state of Michigan. The most abundant minerals found in vugs and fractures tended to be dolomite and calcite, with quartz present locally. Sulfide mineralization was dominated by pyrite and marcasite, but sphalerite, chalcopyrite, and galena were also observed in hand specimens. Additional macroscopic minerals included fluorite, celestine, strontianite, and barite.
The distribution of sulfide minerals (and related weathering products) in the host sedimentary rocks was heterogeneous, but stratigraphically predictable. Throughout northeastern Wisconsin, the stratigraphic unit with the most sulfide mineralization was the top of the Ordovician Ancell Group (dominated by the St. Peter Sandstone in this portion of the state). This zone of naturally enriched iron sulfide mineralization, known as the Sulfide Cement Horizon (SCH), occurs across eastern Wisconsin from the Illinois border in the south to the Michigan border in the north ([
13,
23], this study). In the study area, the SCH was exposed on the quarry floors or in sump trenches at several quarries in Shawano, Oconto, Marinette, Outagamie, and Winnebago counties and areas southwest of the study area (
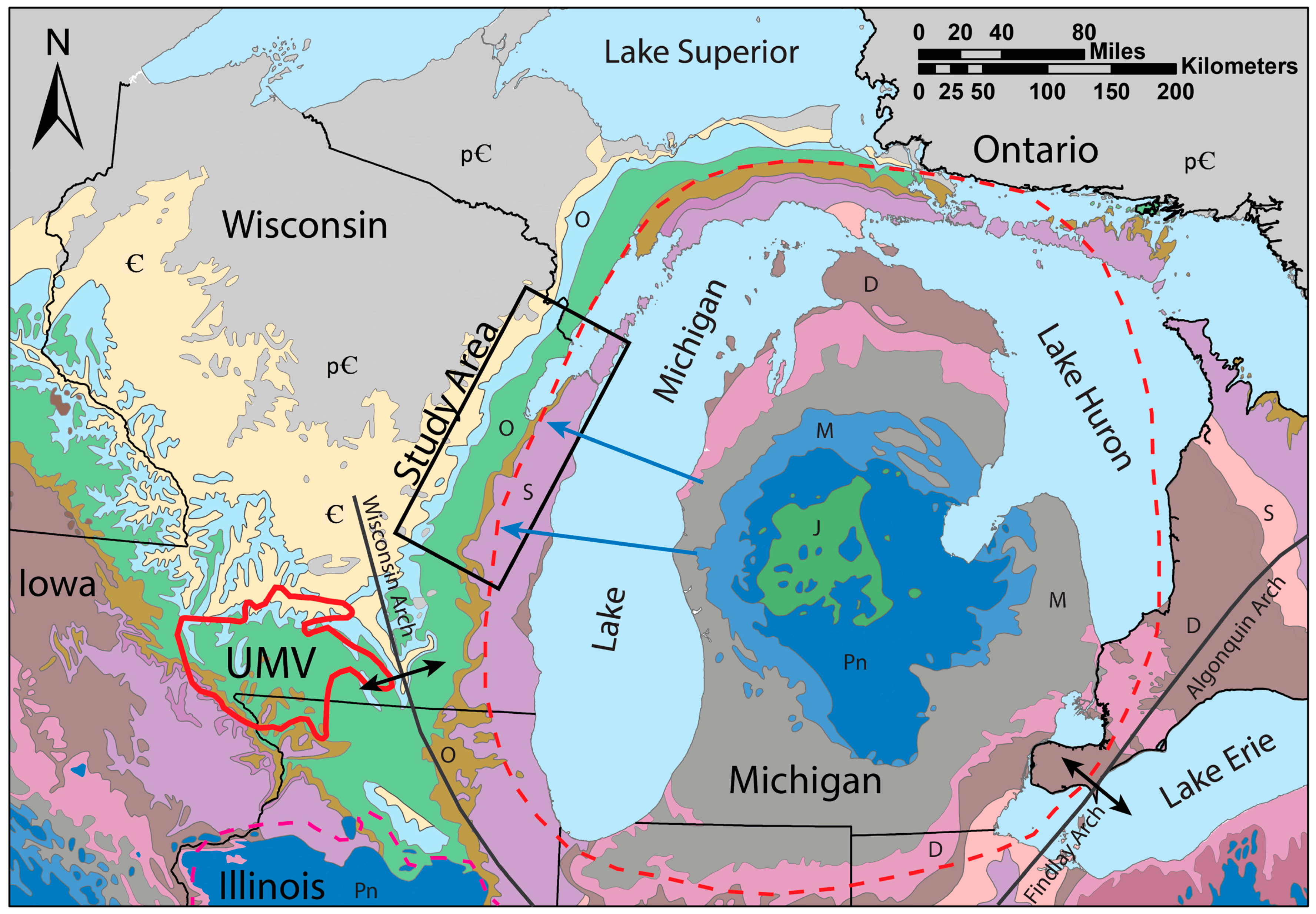
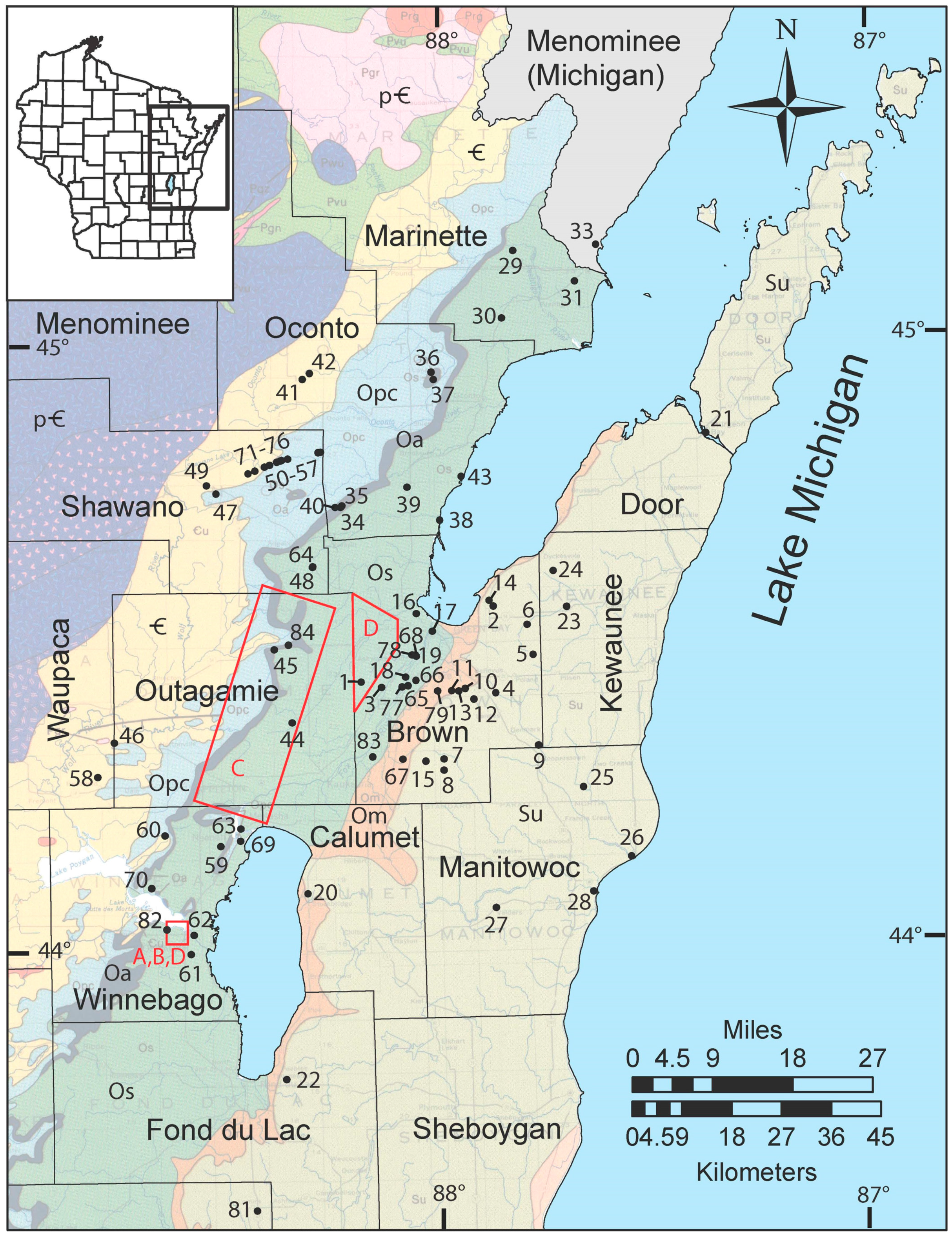
Figure 5). No distinct ore deposits made any one part of the study area more concentrated than another. Cores and cuttings also show that the SCH is present down dip to the east of the outcrop belt, from near the surface in quarries to depths of several hundred meters (
Figure 2 and
Figure 4). Nodules and intergranular cements of pyrite and marcasite were abundant where this group of rocks is rarely exposed. In one quarry near the city of Oshkosh in Winnebago County, a pink coating of amorphous material similar to erythrite (hydrated cobalt arsenate) and annabergite (hydrated nickel arsenate) was observed as a weathering product near the SCH in the St. Peter Sandstone, and was confirmed by SEM-EDS analysis. Another quarry in Green Lake County (southwest of the study area) contained primarily calcite and sphalerite cement in the SCH. A thin (~10–50 cm) layer of dark brown shale above the St. Peter Sandstone was interpreted to be the Glenwood Shale and was present at many locations. It contained abundant fine-grained sulfides, including pyritized arthropod and bryozoan fossils in some quarries.
The next most mineralized stratigraphic zone occurred near the contact between the Silurian Mayville Dolomite and the underlying Ordovician Maquoketa Shale and Neda formations (
Figure 6). Where present, the Upper Ordovician Neda Ironstone and related strata appeared to have sulfides associated with the upper extent of those units. In places, the mass of sulfides was striking, with large (10–40 cm thick) accumulations of pyrite easily identified, especially in central Brown County along portions of the Niagara Escarpment. This sulfide mineralization typically extended upward into the lower few meters of the Silurian Mayville Dolomite. In at least one case, a local aggregate producer had problems with the “aesthetic” quality of its product due to rust stains from the weathering of pyrite fragments in asphalt and concrete products. This interval was best exposed in quarries and natural outcrops along Scray Hill in the towns of Ledgeview and Glenmore in central Brown County. Significant, but lesser amounts of pyrite were present at this contact and within the upper part of the Maquoketa Formation where the Neda Formation is not present.
The Ordovician Sinnipee Group carbonates contained significant quantities of metal sulfides, even at stratigraphic intervals farther away from the SCH in the Ancell Group. Sulfide mineralization was locally abundant along bedding planes, in vertical fractures and joints, and as vug fillings and intercrystalline cements. The mineralization was recognized throughout the Sinnipee Group, and some vertical fractures were mineralized by pyrite, K-silicates, and calcite cements throughout over 15 m of vertical exposure.
Some mineralization occurred within the Prairie du Chien Group and the Cambrian sandstones. Chalcopyrite, quartz, pyrite, and sphalerite were observed in the Prairie du Chien Group, with one notable location in western Outagamie County (Location 46) containing smoky quartz, amethyst, galena and malachite. Pyrite and marcasite nodules were observed in drill cores and cuttings from the Cambrian sandstones, with chalcopyrite, sphalerite, and galena observed locally. The top of the Jordan Sandstone (the uppermost Cambrian unit in northeastern Wisconsin) has also been observed to host zones enriched in pyrite and marcasite [
34].
The carbonate units with the least amount of sulfide mineralization were the Silurian dolostones, which represent the youngest strata in the study area. Cores, abundant quarry exposures, and outcrops of the Silurian dolostones indicate that most sulfides are restricted to the lower few meters of the Mayville Formation. However, trace amounts of iron sulfides were present in various forms throughout the Silurian section. The presence of iron-stained vertical joints suggests that this unit, which is regionally karsted, has had much of its sulfide mineralization removed through near-surface weathering during the Cenozoic Era.
3.2. Petrographic Analysis Using SEM with EDS
Samples of polished rock slabs, polished thin sections, crystal-lined vugs, and well-cuttings chips were analyzed to determine minerals present, to outline mineral paragenesis, and to identify whether or not separate mineral phases were present that could be identified as the source of certain heavy metals present in regional groundwater.
Separate mineral phases containing Fe, Zn, Pb, Cu, and other metals were observed in samples from many locations, especially where parts of the middle and lower Ordovician section are exposed (
Figure 7,
Figure 8,
Figure 9 and
Figure 10). Typically, pyrite and marcasite (FeS
2) were the dominant sulfide phases present. Pyrite exhibited several crystal habits, including cubes, octahedrons, and pyritohedrons up to 1 cm in length. Marcasite occured as bladed—and sometimes cockscomb—habits with a greenish or bluish metallic, iridescent coating of unknown origin. Sphalerite (ZnS) was the next most commonly observed sulfide phase, with generally rounded, irregular crystals up to 5 cm. Galena (PbS) was far less common, and was typically found as small cubic crystals a few millimeters in size, but crystals up to 1 cm were also found in the southern part of the study area. Chalcopyrite crystals are 1 to 3 mm in size and display a typical disphenoid crystal form.
Two copper-bearing minerals were observed in the study area. The crystal morphology and EDS spectrum of the most common Cu-bearing phase suggests that it is chalcopyrite (CuFeS
2). This has been observed at several locations in Oconto, Winnebago, and Outagamie counties (
Figure 3 and
Figure 9). Botryoidal and fibrous malachite has been observed in growths up to 3 mm long at one location in western Outagamie County in association with pyrite and chalcopyrite (
Figure 9). Malachite has also been observed in association with chalcopyrite south of the study area at the Morris Pit in Green Lake County, Wisconsin [
50].
The vast majority of pyrite and marcasite analyzed from the region had no detectable peaks for Ni, Co, or As using SEM-EDS techniques. However, detections of Ni, Co, and As in sulfide minerals were made in some cases (
Figure 10;
Table 1). Detection of a discrete As peak was limited to one specimen, with values of ~2 to 4 weight percent (~0.5 to 1 mole percent), based on EDS analysis. Although this is higher than the As concentration described by Thornburg and Sahai [
30] for As-bearing pyrite in which isomorphic substitution of As was interpreted, it is still likely that the As occurs in a similar form here, and not in a discrete mineral phase such as arsenopyrite.
Despite its occurrence in southern parts of Wisconsin [
8,
14,
51,
52], millerite (NiS) has not been documented in this study area of northeastern Wisconsin. Additional references to minor amounts of millerite, bravoite, violarite, honessite, cobaltite and smaltite are given for the Upper Mississippi Valley lead-zinc district in southwestern Wisconsin by Heyl
et al. [
8,
52].
Detection of numerous microscopic crystals of sulfide minerals enriched in Ni and Co was made in samples from Brown and Oconto counties (
Figure 10;
Table 1). These microscopic occurrences are the first examples of Ni- and Co-bearing minerals found in northeastern Wisconsin. Crystals contain widely varying proportions of Ni-Co-Fe, based on SEM-EDS analysis.
Figure 11 shows normalized mole percent values for the sulfide minerals containing Ni, Co, and Fe listed in
Table 1. Nickel proportions up to 89 mole percent and cobalt proportions up to 24.6 mole percent suggest that the occurrence of these elements is not simply restricted to isomorphous substitution within pyrite, as appears to be the case with arsenic from a few locations in the study area [
30]. Based upon crystal morphology and SEM-EDS composition, bravoite and vaesite are the most likely mineral species present for the Ni and Co-rich crystals.
The precise mineralogy of these Ni and Co phases is challenging to determine due to the size of the crystals (tens of micrometers). Crystal morphology varied from indistinct to cubic to cube-pyritohedron forms (
Figure 10), which is possible in the solid solution series of the pyrite group. In cases where the crystal approaches the NiS
2 end-member composition, the mineral vaesite (NiS
2) is the most likely candidate [
53]. Other crystals with elevated Co and Fe are best described as bravoite ((Ni-Co-Fe)S
2). In the absence of X-ray data, strict identification of these minerals is not possible, but their compositions are shown in
Figure 11.
In contrast to the above metals, none of the samples examined with the SEM displayed separate mineral phases for arsenic, such as arsenopyrite. While the presence of arsenopyrite cannot be ruled out in these rocks, it seems likely that As is substituting for Fe as a trace element in the iron sulfides, as has been suggested by Thornburg and Sahai [
30]. For a limited sample set, they concluded that As was present in isomorphous substitution with pyrite/marcasite, which can be released by oxidation and subsequently sorb onto ferric oxyhydroxides.
Some fractures and vugs contained a complex microscopic array of minerals in the MVT assemblage (e.g.,
Figure 9B). In one 2 mm-wide fracture intercepted by a drill core, sphalerite, galena, fluorite, dolomite, marcasite, Sr-barite, and calcite were observed in close association with each other.
With only one exception near the Wisconsin–Michigan border (Locality 33), all galena observed in northeastern Wisconsin falls south of an east–west regional fault that is possibly associated with a crustal boundary in the Precambrian basement rocks known as the Spirit Lake Tectonic Zone [
25]. The reason for this distribution is unknown, but it might be related to the metals concentrations in Precambrian bedrock through which hydrothermal deep groundwater likely passed [
54,
55].
3.3. Paragenetic Sequence of Mineralization
Table 2 lists the diagenetic minerals observed in Paleozoic sedimentary rocks in the region. Although no single diagram can precisely synthesize the full complexity of the mineral paragenesis in the study area,
Figure 12 illustrates the general mineralization sequence in northeastern Wisconsin.
In general, pervasive dolomitization and silicate mineralization are the earliest events. Early quartz appears to have formed at temperatures ≤ ~50 °C, due to the presence of all-liquid fluid inclusion assemblages entrapped within quartz overgrowths. Later quartz and the majority of the dolomite were formed from a Na-Ca-Mg-Cl brine at temperatures of ~80–100 °C as temperatures warmed [
23]. Iron sulfides occurred throughout much of the sequence, but the main stage of MVT mineralization included sphalerite, galena, chalcopyrite, pyrite, marcasite, and Ni-Co-Fe-sulfide minerals (
Figure 8,
Figure 9 and
Figure 10). The MVT mineralization was intergrown with two episodes of dolomite, with most MVT minerals closely associated with late planar dolomite (
Figure 7 and
Figure 8). Middle and late-stage dolomite, as well as MVT minerals, appear to have formed by similar brines at temperatures of 80–110 °C during what appears to be peak heating. Siderite was not documented in the region, although some dolomite was ferroan. The timing of K-silicates (K-feldspar and possibly illite), relative to other diagenetic phases, is difficult to determine in cases where it occurs as cements within quartz arenites. However, some of it is clearly coeval with abundant iron sulfide mineralization in vertical fractures observed in the northern part of the study area (
Figure 8E).
New evidence (
Figure 9) suggests that at least some of the sulfate minerals precipitated during the MVT stage of mineralization. Sr-barite, barite, and celestine were observed with fluorite, sphalerite, and pyrite in some samples, likely forming during the waning stages of mineralization. Other late-stage minerals include beef-vein and intergranular gypsum, as well as late-stage calcite. Conditions of calcite mineralization are not well known, but at least some late calcite in Silurian rocks contains all-liquid inclusions, suggesting a lower temperature of entrapment [
23]. Strontianite was also found in the region as a relatively late mineral, but its precise location in the sequence is not well-constrained.
The order of mineralization shown in
Figure 12 should be used with caution. As a regionally derived diagram, local or stratigraphic variations may not be expressed fully. Many minerals are also found as isolated occurrences or with only one other mineral. Nevertheless, the diagram presents the only complete synopsis of the paragenetic sequence of mineralization in the study area.
3.4. ICP-AES Analytical Results and Discussion
Analytical results for all 310 samples are presented in Online
Supplementary Materials Tables S1 and S2. The main purpose for analyzing the whole-rock chemistry of these strata was to look for regional and stratigraphic trends in the character of the mineralization, with a focus on heavy metals.
Table 3 presents descriptive statistics for selected metals in all samples in the study that were designated as “Matrix” and “Enriched.”
Table 4 illustrates the typical (median) whole-rock concentrations for selected metals in representative “matrix” samples of bedrock from the region. Averages were not calculated because the data were heavily skewed right, as is common when measuring concentrations. Metals concentrations varied widely, from below detection limits to far above the median concentrations for the host rocks, which was consistent with observations of hand specimens and thin sections.
Based on field observations and elemental analysis, three specific stratigraphic horizons showed the most significant mineralization. These were the top of the Ancell Group, the Ordovician-Silurian contact, and, to a lesser extent, the Sinnipee Group dolostone. All three of these zones typically contained elevated metals concentrations, especially in “enriched” samples from the SCH, or along mineralized bedding planes, faults, or fractures.
Figure 13 shows box-plots (on the log
10 scale) for selected metals from
Table 3. Elevated metals occurred in several Cambrian and Ordovician units, but the Silurian dolostones generally exhibited lower concentrations. Additional box-plots for these and other metals are presented in
Supplementary Materials Figure S3. It is important to note that all stratigraphic units show some potential for locally elevated metals concentrations, including the Prairie du Chien Group and the Cambrian sandstones, which serve as an important regional aquifer system along with the Ancell Group [
56,
57].
Table 5 presents Spearman’s
correlations, estimated for a subset of log
10-transformed metals concentrations relevant to the study.
Figure 14 shows scatter plots for selected metals concentrations, again on the log
10 scale. While most pairs of elements in
Table 5 show moderate correlations, the highest values occurred for Ni
vs. Fe, Ni
vs. Co, Pb
vs. Fe, Co
vs. Fe, and Cr
vs. V. This result is consistent with the SEM-EDS observations of As-, Ni-, and Co-bearing sulfide minerals, suggesting that they are likely related to the same mineralization event.
Strong correlations for Ni
vs. Fe (
= 0.744), Ni
vs. Co (
= 0.718), and Co
vs. Fe (
= 0.612) suggest that these three elements are related to the same mineralization event (
Figure 14). Ni and Co are well correlated in all sample subsets (
Table 5), suggesting that these elements are more closely related. They are likely principally occurring as isomorphic substitutions in pyrite, or as separate Ni-Co-enriched sulfide minerals. This interpretation is consistent with the discovery of discrete, but microscopic, Ni-Co-Fe sulfide minerals vaesite and bravoite in the study area (
Figure 10 and
Figure 11). Correlations between Ni, Co, and Fe are higher than correlations for Ni and As (
= 0.437) or Co and As (
= 0.588). This is consistent with mineralogical observations in which As is typically in separate sulfide mineral phases from Ni and Co (
Figure 10). This is an important observation suggesting that water quality evaluations for wells in the study area should probably include analyses for Ni and Co more regularly, instead of only As.
Smaller correlations between Zn and Fe and between Cu and Fe are most likely due to the fact that those metals also precipitate as separate sulfide mineral phases, such as sphalerite and chalcopyrite, and are independent of the presence of pyrite or marcasite in any particular sample. The same would be expected between Pb and Fe, but there is a moderate correlation between these two elements ( = 0.676 for enriched samples). Because galena is far less commonly observed in the study area than sphalerite, chalcopyrite, pyrite, or marcasite, the correlation between Pb and Fe may represent isomorphic substitution of Pb in pyrite or marcasite rather than the discrete presence of galena.
The correlation between K and B (
= 0.694 for enriched samples) was unexpected, and is not well understood because boron could not be detected using SEM-EDS techniques. However, it is possible that B is substituting in the structure of authigenic feldspar or illite (e.g.,
Figure 8E), both of which are present in the region [
23]. These minerals could be the source of elevated boron levels reported for the Cambrian-Ordovician aquifer across the study area [
58].
Correlations between Ni and As estimated separately by stratigraphic units (
Supplementary Materials Table S2D) showed samples from the Maquoketa-Neda, the Ancell Group, and the Cambrian Sandstones were well correlated (
= 0.762, 0.722, and 0.797, respectively). The plot of Ni
vs. As (
Figure 14C) showed that some samples contained elevated Ni concentrations relative to As. These are best represented by some Ancell Group samples from three Oconto County quarries (Duame Quarry, Chase Quarry, and Montevideo Quarry) and one Sinnipee Group sample from the Vulcan Quarry in Winnebago County. Elevated nickel relative to arsenic is consistent with the existence of Ni-bearing minerals such as vaesite and bravoite that were observed in Oconto County (
Figure 10 and
Figure 11). Ni and As in the Silurian units appeared moderately correlated (
= 0.501), while in the remaining units (Sinnipee and Prairie du Chien groups), they did not correlate as strongly (
= 0.381 and −0.158, respectively).
Relatively strong correlations between Ni and Co persisted even for some finer stratigraphic divisions, such as individual formations within the Sinnipee Group. Specifically, for the Galena and Platteville formations,
was 0.694 and 0.657, respectively (
Figure 14D). As and Co were less strongly correlated for the Galena and Platteville formations, yielding
values of 0.572 and 0.385, respectively. Again, this is consistent with the observation of Ni and Co-bearing mineral phases as described above.
A moderate correlation (
= 0.541) existed for V and Cr in all samples over all stratigraphic intervals (
Figure 14E), but this correlation improved notably for the enriched sample subset (
= 0.769) (
Table 5). However, for a subset of samples from the mineralized zone near the Ordovician-Silurian contact in central Brown County (17 samples along a small portion of the Niagara Escarpment), a particularly high correlation between V and Cr was observed (
= 0.940) (
Table 5;
Figure 14F). In these samples, a slight enrichment of V, Sc, Tl, and La was also observed. The precise cause is unknown, but it might relate to the presence of oolitic hematite of the Neda Formation along this part of the escarpment. Although there is a general affinity of V for iron-bearing rocks, most of the samples with high V concentrations are dolostone samples from near the contact. Another stratigraphic difference noted is that iron sulfide-rich samples from the Maquoketa-Neda contact and the Silurian dolostones just above this contact are depleted in Ni, relative to samples from the Ancell and Sinnipee groups (
Figure 14A). Although depleted in Ni, they are slightly enriched in V, Sc, Tl, and La relative to the Ancell and Sinnipee Group samples (
Table S1). These differences are likely the result of diagenesis by hydrothermal groundwater. The Maquoketa Shale (
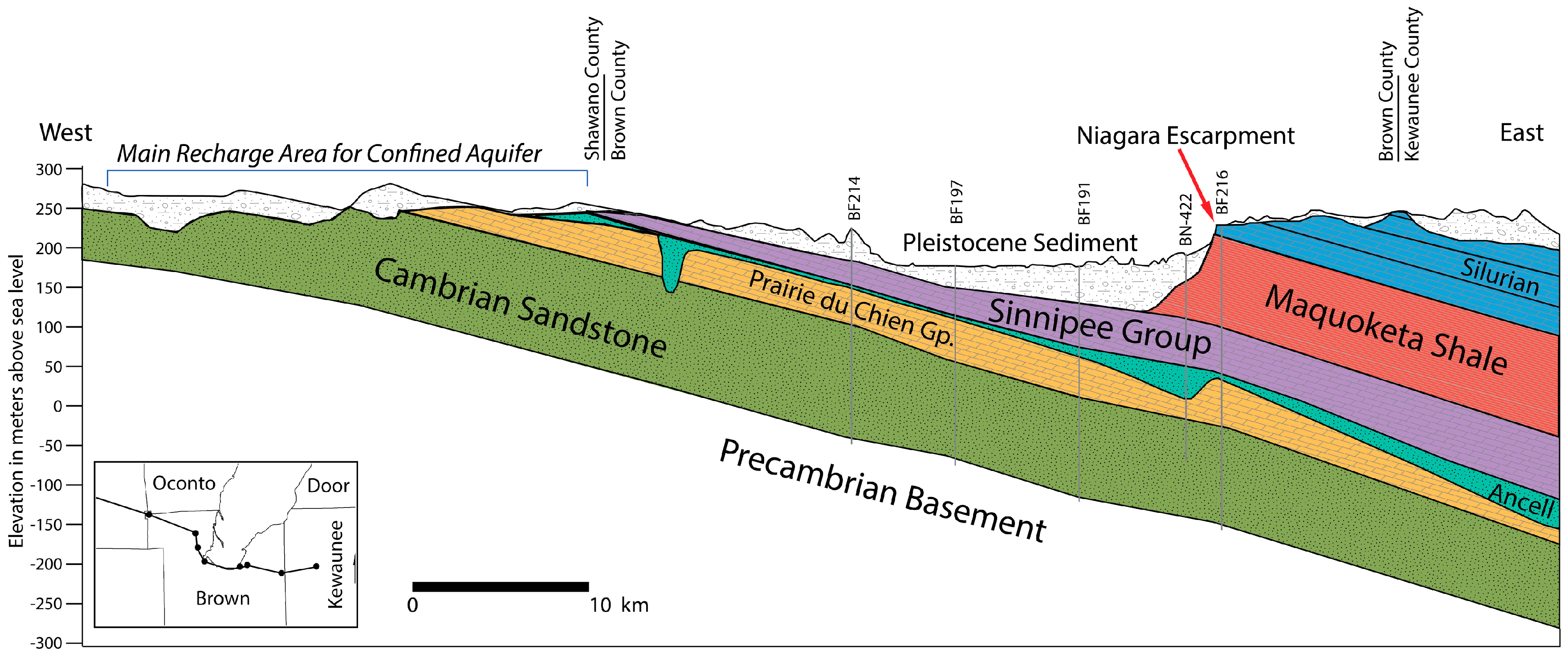
Figure 2 and
Figure 4) is a regional confining unit that likely isolated the Silurian dolostone units above from the Ordovician sandstones and carbonates below during mineralization. This local anomaly occurs near the only exposure of the Neda Ironstone, which suggests that mineralizing groundwater may have scavenged certain elements locally from the Neda Ironstone instead of being supplied by groundwater migrating through other strata.
Cu concentrations were highest in Oconto County (Ancell Group) and near the Ordovician-Silurian contact in central Brown County (Maquoketa-Neda units). This is consistent with the identification of chalcopyrite in field specimens from Oconto County Prairie du Chien Group samples.
Tuttle
et al. [
59] demonstrated that redistribution of minor and trace elements can occur in rocks with sulfide mineralization due to weathering processes and groundwater flow. While it is likely that remobilization of trace elements may have occurred in the study area, a systematic assessment of this was not performed. Extensive Late Pleistocene glaciation has removed bedrock-derived soil horizons, and most soils have developed on transported materials. In most cases, bedrock samples were obtained from relatively fresh host rocks in quarries, drill cores/cuttings, and excavated road cuts to keep sample weathering effects to a minimum. The most likely bedrock units to have been affected by weathering are the Silurian dolostones (
Figure 2), which show extensive karst development [
25] and relatively low trace element concentrations (
Table 4).