Cretaceous Ichthyosaurs: Dwindling Diversity, or the Empire Strikes Back?
Abstract
:1. Introduction
2. Results and Discussion
2.1. Ichthyosaur Genera in the Cretaceous
2.1.1. Platypterygius
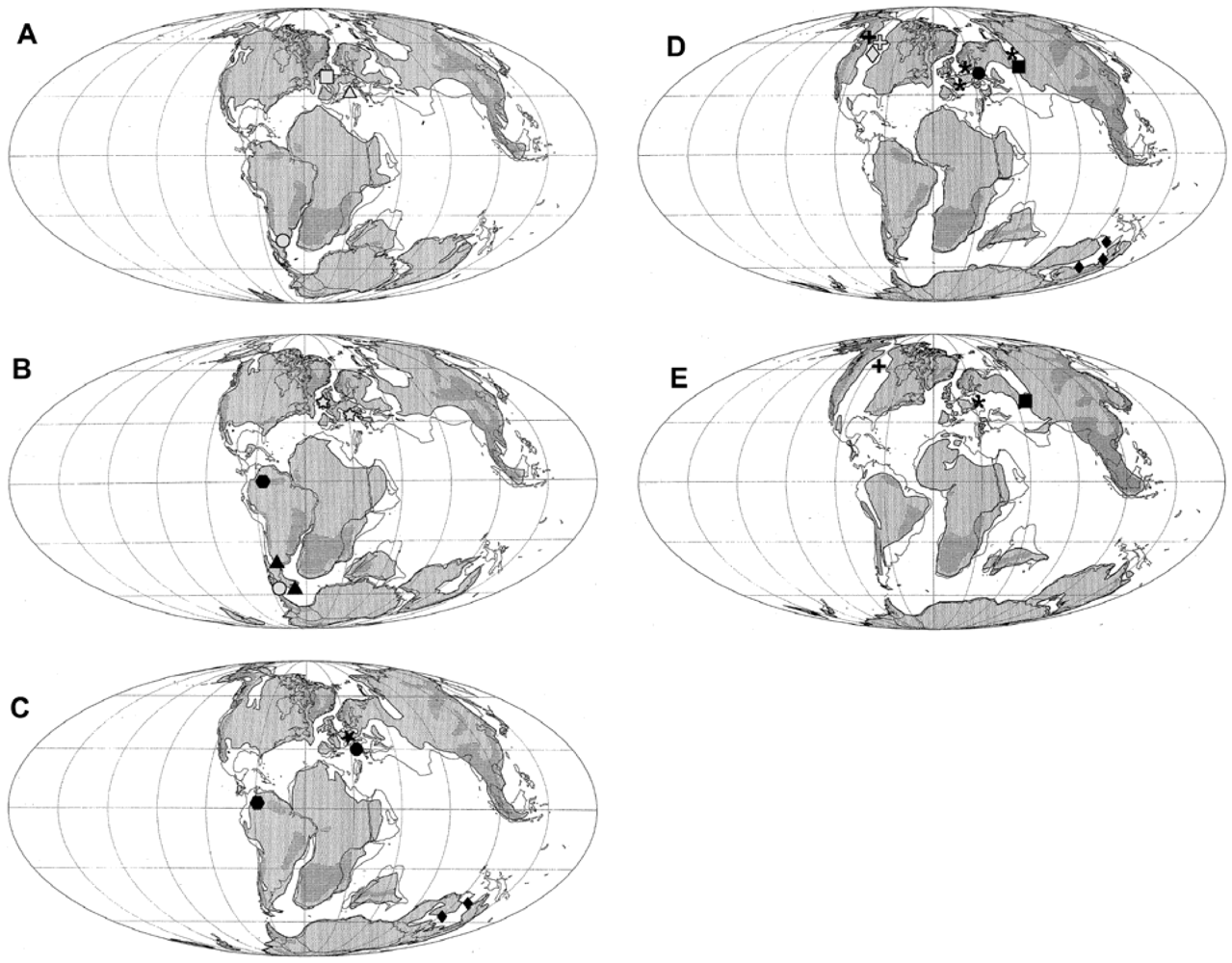
2.1.2. Maiaspondylus
2.1.3. Athabascasaurus
2.1.4. Sveltonectes
2.1.5. Acamptonectes
2.1.6. Ichthyosaurs Crossing the Jurassic—Cretaceous Boundary
2.2. Diversity and Evolution of Cretaceous Ichthyosaurs

3. Conclusions
Acknowledgments
References
- Maisch, M.W.; Matzke, A.T. The Ichthyosauria. Stuttg. Beitr. Naturkunde Ser. B (Geolog. Paläontol.) 2000, 298, 1–159, in German. [Google Scholar]
- McGowan, C.; Motani, R. Ichthyopterygia. Handbook of Paleoherpetology; Sues, H.D., Ed.; Verlag Dr. Friedrich Pfeil: Munich, Germany, 2003; Volume 8, p. 175. [Google Scholar]
- Sander, P.M. Ichthyosauria: Their diversity, distribution, and phylogeny. Paläontol. Z. 2000, 74, 1–35. [Google Scholar]
- Mazin, J.M.; Bardet, N.; Godefroit, P. The European Ichthyopterygia: A re-assessment. J. Vertebr. Paleontol. 1994, 14, 36–37. [Google Scholar]
- Bardet, N. Dental cross-sections in Cretaceous Ichthyopterygia: Systematic implications. Geobios-LYON 1990, 23, 169–172. [Google Scholar] [CrossRef]
- Bardet, N. Stratigraphic evidence for the extinction of ichthyosaurs. Terra Nova 1992, 4, 649–656. [Google Scholar] [CrossRef]
- Broili, F. Ein neuer Ichthyosaurus aus der norddeutschen Kreide. Palaeontogr. 1907, 54, 139–152, in German. [Google Scholar]
- Maxwell, E.E.; Caldwell, M.W. A new genus of ichthyosaur from the Lower Cretaceous of western Canada. Palaeontology 2006, 49, 1043–1052. [Google Scholar] [CrossRef]
- Druckenmiller, P.S.; Maxwell, E.E. A new Lower Cretaceous (lower Albian) ichthyosaur genus from the Clearwater Formation, Alberta, Canada. Can. J. Earth Sci. 2010, 47, 1037–1053. [Google Scholar] [CrossRef]
- Fischer, V.; Masure, E.; Arkhangelsky, M.S.; Godefroit, P. A new Barremian (Early Cretaceous) ichthyosaur from western Russia. J. Vertebr. Paleontol. 2011, 31, 1010–1025. [Google Scholar] [CrossRef]
- Fischer, V.; Maisch, M.W.; Naisch, D.; Kosma, R.; Liston, J.; Joger, U.; Krüger, F.J.; Pardo Pérez, J.; Tainsh, J.; Appleby, R.M. New ophthalmosaurid ichthyosaurs from the European Lower Cretaceous demonstrate extensive ichthyosaur survival across the Jurassic-Cretaceous boundary. PloS ONE 2012, 7, 1–23. [Google Scholar]
- Fischer, V. New ophthalmosaurids from Europe and Russia broaden the biodiversity of Early Cretaceous ichthyosaurs. J. Vertebr. Paleontol. Suppl. 2011, 31, 110. [Google Scholar]
- McGowan, C. The systematics of Cretaceous ichthyosaurs with particular reference to the material from North America. Contrib. Geol. Univ. Wyo. 1972, 11, 9–29. [Google Scholar]
- McGowan, C. Dinosaurs, Spitfires, and Seadragons, 1st ed; Harvard University Press: Cambridge, MA, USA, 1991; p. 365. [Google Scholar]
- Fischer, V. New data on the ichthyosaur Platypterygius hercynicus and its implications for the validity of the genus. Acta Palaeontol. Pol. 2012, 57, 123–134. [Google Scholar] [CrossRef]
- Kolb, C.; Sander, P.M. Redescription of the ichthyosaur Platypterygius hercynicus (Kuhn 1946) from the Lower Cretaceous of Salzgitter (Lower Saxony, Germany). Palaeontogr. Abt. A. 2009, 288, 151–192. [Google Scholar]
- Arkhangelsky, M.S.; Averianov, A.O.; Pervushov, E.M.; Tarnikow, V.Y.; Zozyrev, N.Y. On ichthyosaur remains from the Cretaceous of the Voronezh Region. Palaeontol. J. 2008, 42, 287–291. [Google Scholar] [CrossRef]
- Von Huene, F. Beitrag zur Kenntnis mariner mesozoischer Wirbeltiere in Argentina. Zent. Mineral. Geolog. Paläontol. B 1927, 1927, 22–29, in German. [Google Scholar]
- Fernández, M.; Aguirre-Urreta, M.B. Revision of Platypterygius hauthali von Huene, 1927 (Ichthyosauria: Ophthalmosauridae) from the Early Cretaceous of Patagonia, Argentin. J. Vertebr. Paleontol. 2005, 25, 583–587. [Google Scholar] [CrossRef]
- Pardo Pérez, J.; Frey, E.; Stinnesbeck, W.; Salazar, C.; Leppe, M. Life and death on the Torres del Pain ichthyosaurs, southern Chile. In Proceedings of 7th Annual Meeting of the European Association of Vertebrate Palaeontologists, Berlin, Germany, 20-24 July 2009; p. 55.
- Pardo Pérez, J.M.; Frey, E.; Stinnesbeck, W.; Rivas, L. Early Cretaceous ichthyosaurs from the Tyndall Glacier in Torres Del Paine National Park, southernmost Chile. J. Vertebr. Paleontol. Suppl. 2011, 31, 171. [Google Scholar]
- Pardo-Pérez, J.; Frey, E.; Stinnesbeck, W.; Fernánez, M.S.; Rivas, L.; Salazar, C.; Leppe, M. An ichthyosaurian forefin from the Lower Cretaceous Zapata Formation of southern Chile: Implicaitons for morphological variability within Platypterygius. Palaeobio. Palaeoenv. 2012; in submission. [Google Scholar]
- Maxwell, E.E.; Kear, B.P. Postcranial anatomy of Platypterygius americanus (Reptilia: Ichthyosauria) from the Cretaceous of Wyoming. J. Vertebr. Paleontol. 2010, 30, 1059–1068. [Google Scholar] [CrossRef]
- McCoy, F. On the occurrence of Ichthyosaurus and Plesiosaurus in Australia. J. Nat. Hist. Third Ser. 1867, 19, 355–356. [Google Scholar]
- McCoy, F. On the discovery of Enaliosauria and other Cretaceous fossils in Australia. Trans. Proc. Rol. Soc. Vic. 1867, 8, 41–42. [Google Scholar]
- Zammit, M. A review of Australasian ichthyosaurs. Alcheringa 2010, 34, 281–292. [Google Scholar]
- Etheridge, R. On additional evidence of the genus Ichthyosaurus in the Mesozoic rocks (“Rolling Downs Formation”) of north-eastern Australia. P. Linn. Soc. N. S. W. 1888, 2, 405–409. [Google Scholar]
- Wade, M. A review of the Australian Cretaceous longipinnate ichthyosaur Platypterygius (Ichthyosauria, Ichthyopterygia). Mem. Qld. Mus. 1990, 28, 115–137. [Google Scholar]
- Kear, B.P. Cretaceous marine reptiles of Australia: A review of taxonomy and distribution. Cretaceous Res. 2003, 24, 277–303. [Google Scholar] [CrossRef]
- Smith, A.G.; Smith, D.G.; Funnell, B.M. Atlas of Mesozoic and Cenozoic Coastlines; Cambridge University Press: Cambridge, UK, 1994; p. 99. [Google Scholar]
- Carter, J. Notice of the jaws of an Ichthyosaurus from the chalk in the neighbourhood of Cambridge. Rep. Br. Assoc. Advanc. Sci. 1845, 1845, 60. [Google Scholar]
- Carter, J. On the occurrence of a new species of Ichthyosaurus in the chalk. Lond. Geol. J. Rec. Discov. Br. Foreign Paleontol. 1846, 1, 7–9. [Google Scholar]
- Owen, R. A monograph on the fossil Reptilia of the Cretaceous formations. Palaeontogr. Soc. Monogr. 1851, 5, 1–118. [Google Scholar]
- Kiprijanoff, W. Studien über die fossilen Reptilien Russlands. Theil 1. Gattung Ichthyosaurus Konig aus dem Sewerischen Sandstein oder Kreide-Gruppe. Mém. Acad. Imp. Sci. St.-Pétersbg. 7e Sér. 1881, 28, 1–103, in German. [Google Scholar]
- Storrs, G.W.; Arkhangelsky, M.S.; Efimov, V.M. Mesozoic marine reptiles of Russia and other former Soviet Republics. In The Age of Dinosaurs in Russia and Mongolia; Benton, M.J., Shishkin, M.A., Unwin, D.M., Kurochkin, E.N., Eds.; Cambridge University Press: Cambridge, UK, 2000; pp. 187–210. [Google Scholar]
- Diedrich, C. New ichthyosaur remains of Platypterygius cf. campylodon (Carter 1846) (Ichthyopterygia, Reptilia) from the Cenomanian of NW Germany. Munstersche Forschungen Geol. Palaontol. 2002, 93, 97–108. [Google Scholar]
- Nace, R.L. A new ichthyosaur from the Upper Cretaceous Mowry formation of Wyoming. Am. J. Sci. 1939, 237, 673–686. [Google Scholar] [CrossRef]
- Nace, R.L. A new ichthyosaur from the Late Cretaceous, Northeast Wyoming. Am. J. Sci. 1941, 239, 908–914. [Google Scholar] [CrossRef]
- Kuhn, O. Ein Skelett von Ichthyosaurus (Platypterygius) hercynicus n. sp. aus dem Aptium von Gitter. Ber. Naturforschenden Ges. Bamb. 1946, 29, 69–82, in German. [Google Scholar]
- Páramo, M.E. Platypterygius sachicarum (Reptilia, Ichthyosauria) neuva especie del Cretacico de Colombia. Rev. Ingeominas 1997, 6, 1–12, in Spanish. [Google Scholar]
- Lydekker, R. Indian Pre-Tertiary vertebrate fossil Reptilia and Batrachia. Mem. Geolog. Surv. India 1879, 1, 1–36. [Google Scholar]
- Underwood, C.J.; Goswami, A.; Prasad, G.V.R.; Verma, O.; Flyn, J.J. Marine vertebrates from the ‘middle’ Cretaceous (early Cenomanian) of south India. J. Vertebr. Paleontol. 2011, 31, 539–552. [Google Scholar] [CrossRef]
- Sirotti, A.; Papazzoni, C. On the Cretaceous ichthyosaur remains from the Northern Apennines (Italy). Boll. Soci. Paleontol. Ital. 2002, 41, 237–248. [Google Scholar]
- Sachs, S.; Grant-Mackie, J.A. An ichthyosaur fragment from the Cretaceous of Northland, New Zealand. J. Royal Soc. New Zealand 2003, 33, 307–314. [Google Scholar] [CrossRef]
- Bardet, N.; Wellnhofer, P.; Herm, D. Discovery of ichthyosaur remains (Reptilia) in the Upper Cenomanian of Bavaria. Mitt. Bayer. Staatssamml. Paläontol. Hist. Geol. 1994, 34, 213–220. [Google Scholar]
- Maxwell, E.E.; Caldwell, M.W. Evidence for a second species of the ichthyosaur Platypterygius in North America: A new record from the Loon River Formation (Lower Cretaceous) of Northwestern Canada. Can. J. Earth Sci. 2006, 43, 1291–1295. [Google Scholar] [CrossRef]
- Adams, T.L.; Fiorillo, A. Platypterygius Huene, 1922 (Ichthyosauria, Ophthalmosauridae) from the Late Cretaceous of Texas, USA. Palaeontol. Electron. 2010, 14, 12. [Google Scholar]
- Maxwell, E.E.; Kear, B.P. Postcranial anatomy of Platypterygius americanus (Reptilia: Ichthyosauria) from the Cretaceous of Wyoming. J. Vertebr. Paleontol. 2010, 30, 1059–1068. [Google Scholar] [CrossRef]
- Zammit, M.; Norris, R.M.; Kear, B.P. The Australian Cretaceous ichthyosaur Platypterygius australis: A description and review of postcranial remains. J. Vertebr. Paleontol. 2010, 30, 1726–1735. [Google Scholar] [CrossRef]
- Choo, B. Cretaceous ichthyosaurs from Western Australia. Rec. West. Aust. Mus. Suppl. 1999, 57, 207–218. [Google Scholar]
- Kear, B.P. Biogeographic and biostratigraphic implications of Australian Mesozoic marine reptiles. Aust. Biologist 2004, 17, 4–22. [Google Scholar]
- Lydekker, R. Catalogue of the fossil Reptilia and Amphibia in the British Museum (Natural History). Part II. Containing the Orders Ichthyopterygia and Sauropterygia; Trustees of the British Museum: London, UK, 1889; p. 307. [Google Scholar]
- McGowan, C. Further evidence for the wide geographical distribution of ichthyosaur taxa (Reptilia: Ichthyosauria). J. Paleontol. 1978, 52, 1155–1162. [Google Scholar]
- Spalletti, L.; Gasparini, Z.; Veiga, G.; Schwarts, E.; Fernández, M.; Matheos, S. Facies anóxicas, procesos deposicionales y herpetofauna de la rampa marina titoniano-berriasisana en la Cuenca Neuqina (Yesera del Tromen), Neuquén, Argentina. Rev. Geológ. Chile 1999, 26, 109–123, in Spanish. [Google Scholar]
- Fischer, V.; Clément, A.; Guiomar, M.; Godefroit, P. The first definite record of a Valanginian ichthyosaur and its implications on the evolution of post-Liassic Ichthyosauria. Cretaceous Res. 2011, 32, 155–163. [Google Scholar] [CrossRef]
- Scheyer, T.M.; Moser, M. Survival of the thinnest: rediscovery of Bauer’s (1898) ichthyosaur tooth sections from Upper Jurassic lithographic limestone quarries, south Germany. Swiss J. Geosci. 2011, 104, 147–157. [Google Scholar] [CrossRef]
- Bardet, N.; Fernández, M. A new ichthyosaur from the upper Jurassic lithographic limestones of Bavaria. J. Paleontol. 2000, 74, 503–511. [Google Scholar] [CrossRef]
- Ensom, P.C.; Clements, R.G.; Feist-Brukhardt, S.; Milner, A.R.; Chitolie, J.; Jeffery, P.A.; Jones, C. The age and identity of an ichthyosaur reputedly from the Purbeck Limestone Group, Lower Cretaceous, Dorset, southern England. Cretaceous Res. 2009, 30, 699–709. [Google Scholar] [CrossRef]
- Massare, J.A. Tooth morphology and prey preference of Mesozoic marine reptiles. J. Vertebr. Paleontol. 1987, 7, 121–137. [Google Scholar] [CrossRef]
- Massare, J.A. Faunas, behavior, and evolution. In Ancient Marine Reptiles, 2nd; Callaway, J.M., Nicholls, E.L., Eds.; Academic Press: San Diego, CA, USA, 1997; pp. 401–421. [Google Scholar]
- Gilmore, C.W. Discovery of teeth in Baptanodon, an ichthyosaurian from the Jurassic of Wyoming. Science 1902, 16, 913–914. [Google Scholar]
- Buchy, M.C. First record of Ophthalmosaurus (Reptilia: Ichthyosauria) from the Tithonian (Upper Jurassic) of Mexico. J. Paleontol. 2010, 84, 149–155. [Google Scholar] [CrossRef]
- Kirton, A.J. A Review of British Upper Jurassic Ichthyosaurs. Ph.D. Dissertation, University of Newcastle-upon-Tyne, Newcastle-upon-Tyne, Tyne, Wear, UK, 1981. [Google Scholar]
- Fernández, M.S. A new ichthyosaur from the Tithonian (Late Jurassic) of the Neuquén Basin, Northwestern Patagonia, Argentina. J. Paleontol. 1997, 71, 479–484. [Google Scholar]
- Fernández, M. Nuevo material de Caypullisaurus bonapartei Fernández (Reptilia: Ichthyosauridae) del Jurásico superior de la Cuenca Neuquina, Argentina. Ameghiniana 1998, 35, 21–24, in Spanish. [Google Scholar]
- Kear, B.P. Cranial morphology of Platypterygius longmani Wade, 1990 (Reptilia: Ichthyosauria) from the Lower Cretaceous of Australia. Zool. J. Linn. Soc-Lond. 2005, 145, 583–622. [Google Scholar] [CrossRef]
- Kear, B.P.; Boles, W.E.; Smith, E.T. Unusual gut contents in a Cretaceous ichthyosaur. Proc. Roy. Soc. Lond. Biol. Lett. Suppl. 2003, 270, 206–208. [Google Scholar]
- Kear, B.P.; Barrett, P. Reassessment of the English Cretaceous ichthyosaur Platypterygius campylodon. In Proceedings of Symposium of Vertebrate Palaeontology and Comparative Anatomy, Glasgow, UK, August–September 2007; p. 38.
- Bennett, S.P.; Barrett, P.M.; Collinson, M.E.; Moore-Fay, S.; Davis, P.G.; Palmer, C.P. A new Specimen of Ichthyosaurus communis from Dorset, UK, and its bearing on the stratigraphical range of the species. P. Geologist. Assoc. 2012, 123, 146–154. [Google Scholar] [CrossRef]
- Lawrence, J.D. A Total Evidence Analysis of the Evolutionary History of the Thunnosaur Ichthyosaurs. M.Sc. Thesis, Graduate College of Bowling Green State University, Bowling Green, OH, USA, 2008. [Google Scholar]
- Gasparini, Z.; Fernández, M. 1997. Tithonian marine reptiles of the eastern Pacific. In Ancient Marine Reptiles, 2nd Edition; Calloway, J.M., Nichols, E.L., Eds.; Academic Press: San Diego, CA, USA, 1997; pp. 223–253. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zammit, M. Cretaceous Ichthyosaurs: Dwindling Diversity, or the Empire Strikes Back? Geosciences 2012, 2, 11-24. https://doi.org/10.3390/geosciences2020011
Zammit M. Cretaceous Ichthyosaurs: Dwindling Diversity, or the Empire Strikes Back? Geosciences. 2012; 2(2):11-24. https://doi.org/10.3390/geosciences2020011
Chicago/Turabian StyleZammit, Maria. 2012. "Cretaceous Ichthyosaurs: Dwindling Diversity, or the Empire Strikes Back?" Geosciences 2, no. 2: 11-24. https://doi.org/10.3390/geosciences2020011



